Abstract
Differences in the effectiveness of real-time continuous glucose monitoring (rtCGM) and intermittently scanned continuous glucose monitoring (isCGM) in type 1 diabetes (T1D) are reported. The impact on percent time in range of switching from an isCGM with glucose threshold-based optional alerts only (FreeStyle Libre 2 [FSL2]) to an rtCGM (Dexcom G7) with an urgent low soon predictive alert was assessed, alongside other secondary outcomes including hemoglobin A1c (HbA1c) and other continuous glucose monitoring metrics. Adults with T1D using FSL2 were switched to Dexcom G7 for 12 weeks. HbA1c and continuous glucose data during FSL2 and Dexcom G7 use were compared. Data from 29 participants (aged 44.8 ± 16.5 years, 12 male and 17 female) were analyzed. After switching to rtCGM, participants spent less time in hypoglycemia below 3.9 mmol/L (70 mg/dL) (3.0% [1.0%, 5.0%] vs. 2.0% [1.0%, 3.0%], P = 0.006) and had higher percentage achievement of time below 3.9 mmol/L (70 mg/dL) of <4% (55.2% vs. 82.8%, P = 0.005). Coefficient of variation was lower (39.3 ± 6.6% vs. 37.2 ± 5.6%, P = 0.008). In conclusion, adults with T1D who switched from isCGM to rtCGM may benefit from reduced exposure to hypoglycemia and glycemic variability.
Introduction
Optimizing glycemia reduces the risk of micro- and macrovascular complications but can be challenging for people living with type 1 diabetes (T1D) due to hypoglycemia. 1,2 Continuous glucose monitoring (CGM) devices measure interstitial glucose and can provide real-time glucose measurements, with glucose trend arrows and alerts/alarms to facilitate self-management. CGM may be implemented as intermittently scanned continuous glucose monitoring (isCGM) and real-time continuous glucose monitoring (rtCGM). 3,4 The National Institute for Health and Care Excellence recommends offering adults with T1D a choice of isCGM or rtCGM based on their individual preferences, needs, characteristics, and the functionality of the devices available. 5
Previous studies demonstrated benefits of rtCGM over the first-generation isCGM that did not have alerts in various groups of people with T1D, 6 –9 including reduction in time spent in hypoglycemia 10 and glycemic variability. 11 The second generation of isCGM (FreeStyle Libre 2 [FSL2]; Abbott Diabetes Care, Alameda, CA, USA) has optional threshold-based low and high glucose alarm functions and a scan-dependent message of “glucose going low” when glucose is projected to be lower than 3.9 mmol (70 mg/dL) within 15 min. However, at the time of this study, it still requires scanning to show the current glucose value and to provide the message notification. 4 The Dexcom G7 system (Dexcom, San Diego, CA, USA) is an rtCGM which also has optional alerts, but in addition to the threshold-based glucose alerts, it provides a predictive urgent low soon alert (i.e., when glucose is predicted to reach 3.1 mmol/L [55 mg/dL] within the next 20 min).
Data comparing CGM devices with or without predictive low glucose alerts are lacking. Therefore, we aimed to explore the benefits of switching from a second-generation isCGM to an rtCGM with a predictive low glucose alert on glycemic control and continuous glucose metrics.
Methods
This prospective single-arm data collection was conducted from February to June 2023 as part of a Service Evaluation project (registration number 718) for adults with T1D attending the diabetes services across two hospitals (Charing Cross Hospital and St Mary's Hospital) at Imperial College London NHS Trust. Imperial College London NHS Trust is a specialist diabetes referral center for northwest London, providing services to a diverse urban population. Access to CGM technologies was in line with NHS England London technology commissioning recommendations, which reflect the National Institute of Clinical Excellence (NG17, NG18, and TA151) and NHS England National guidance. NHS Research Ethics Committee review was not required.
Adults with T1D aged ≥18 years on multiple daily insulin injections (MDI) or continuous subcutaneous insulin infusion who were using a second-generation isCGM (FSL2) for at least 4 weeks were invited to switch to rtCGM (Dexcom G7) for 12 weeks. Subjects were excluded if there was a concurrent plan to change insulin regimen or insulin type, had end-stage renal failure, or were pregnant or breastfeeding. People using Dexcom G6 were not included in this study. Included participants received education about the rtCGM device including the real-time glucose data display, trend arrow, alert function, and sensor application. All alert settings were discussed and set according to the preferences and needs of each participant except for the fixed urgent low alert on Dexcom G7 that is set at 3.1 mmol/L (55 mg/dL). rtCGM was used non-adjunctively with a smartphone or receiver. Participants were advised to test capillary blood glucose if their symptoms did not match with sensor glucose reading. Participants received the usual diabetes care and diabetes education by their multidisciplinary team.
Baseline characteristics including age, sex, diabetes duration, and insulin treatment regimen were obtained from the electronic health records (Cerner Corporation). Socioeconomic status was assessed by the Index of Multiple Deprivation, the English Indices of Deprivation 2019. 12 The deprivation deciles were divided into quintiles, as in the National Diabetes Audits. Hemoglobin A1c (HbA1c) level before start of rtCGM and at 12 weeks of rtCGM use was compared. CGM data from the last 28 days of isCGM and rtCGM were collected from web-based glucose monitoring platforms LibreView and Dexcom Clarity, respectively. Percent time spent in hypoglycemia (<3.9 mmol/L [70 mg/dL] and <3.0 mmol/L [54 mg/dL]), percent time in target range (3.9–10 mmol/L [70–180 mg/dL]), percent time spent in hyperglycemia (>10 mmol/L [180 mg/dL] and >13.9 mmol/L [250 mg/dL]), and percentage of coefficient of variation (%CV) were analyzed. The percentage of participants achieving glycemic targets based on the International Consensus 13 from the last 28 days of isCGM and rtCGM was compared. Participants were asked to report any serious adverse events and any issues with the device.
Normally distributed data are presented as mean (standard deviation [SD]); non-normally distributed data are presented as median (interquartile range [IQR]); categorical data are presented as number (percentage). HbA1c and CGM data from the last 28 days of isCGM and rtCGM were compared using paired sample t-test for normally distributed data, the Wilcoxon test for non-normally distributed data, and the McNemar test for categorical data. Statistical analyses were performed using SPSS Statistics version 20 (SPSS, Inc., Chicago, IL, USA).
Results
Thirty people were recruited for the service evaluation project switching from FSL2 to Dexcom G7. One participant decided to revert to FSL2 after one Dexcom sensor use because of the concern of the difference between sensor and capillary blood glucose value. Therefore, data from 29 participants (12 male and 17 female) were analyzed. The mean age of participants was 44.8 ± 16.5 years, and the mean body mass index was 26.9 ± 5.1 kg/m2. The median duration of diabetes was 21 (12, 35) years, and 22 (75.9%) participants used MDI therapy. The estimated median basal insulin dose was 0.31 (0.19, 0.35) U/(kg·d), and the estimated median bolus insulin dose was 0.26 (0.17, 0.41) U/(kg·d). The baseline mean HbA1c was 60 ± 18 mmol/mol (7.6 ± 3.8%). Participants had used a second-generation isCGM for mean ± SD of 11 ± 5 months (median 11.0 [IQR 6.4, 14.8] months), with a median number of sensor scans per day of 9 (5, 13). One participant (3.4%) had an episode of severe hypoglycemia within the previous 12 months. Three participants (10.3%) were in the most deprived quintile; 13 participants (44.8%) were in the second most deprived quintile; and 1 participant (3.4%) was in the least deprived quintile.
The median (IQR) duration use of rtCGM was 11.9 (11.6, 12.0) weeks. After switching to rtCGM, participants spent less time in hypoglycemia below 3.9 mmol/L (70 mg/dL) (3.0% [1.0%, 5.0%] vs. 2.0% [1.0%, 3.0%], P = 0.006) and had lower %CV (39.3 ± 6.6% vs. 37.2 ± 5.6%, P = 0.008) (Table 1). The median (IQR) number of hypoglycemia (<3.9 mmol/L [70 mg/dL]) events with duration ≥15 min/day was 0.62 (0.19, 1.00) versus 0.63 (0.31, 1.07), P = 0.212, with isCGM and rtCGM, respectively. Similarly, the median (IQR) number of clinically significant hypoglycemia events (<3.0 mmol/L [54 mg/dL]) for ≥15 min/day was 0.04 (0.00, 0.17) versus 0.07 (0.00, 0.19), P = 0.435, with isCGM and rtCGM, respectively.
Hemoglobin A1c and Ambulatory Glucose Profile Comparing Between Intermittently Scanned Continuous Glucose Monitoring and Real-Time Continuous Glucose Monitoring
Data are presented as mean ± SD and median (IQR).
HbA1c only available for 23 participants.
n = 28; one participant had no data of time below 3 mmol/L (54 mg/dL) and time above 13.9 mmol/L (250 mg/dL) during FSL2 use.
n = 27; two participants had no data of %CV use during FSL2 use.
%CV, percentage of coefficient of variation; CI, confidence interval; HbA1c, hemoglobin A1c; IQR, interquartile range; isCGM, intermittently scanned continuous glucose monitoring; rtCGM, real-time continuous glucose monitoring; SD, standard deviation.
The proportion of participants achieving CV <36% as per the International Consensus 13 was 24.1% in isCGM and 37.0% in rtCGM (P = 0.38). The proportion of participants achieving <4% time below 3.9 mmol/L (70 mg/dL) increased from 55.2% to 82.8% (P = 0.008), and participants who achieved <1% time below 3.0 mmol/L (54 mg/dL) increased from 58.6% to 89.3% (P = 0.008) after switching to rtCGM (Fig. 1). A greater proportion of rtCGM users achieved HbA1c <58 mmol/mol (7.5%), although this did not reach statistical significance (isCGM 39.1% vs. rtCGM 60.9%, P = 0.063).
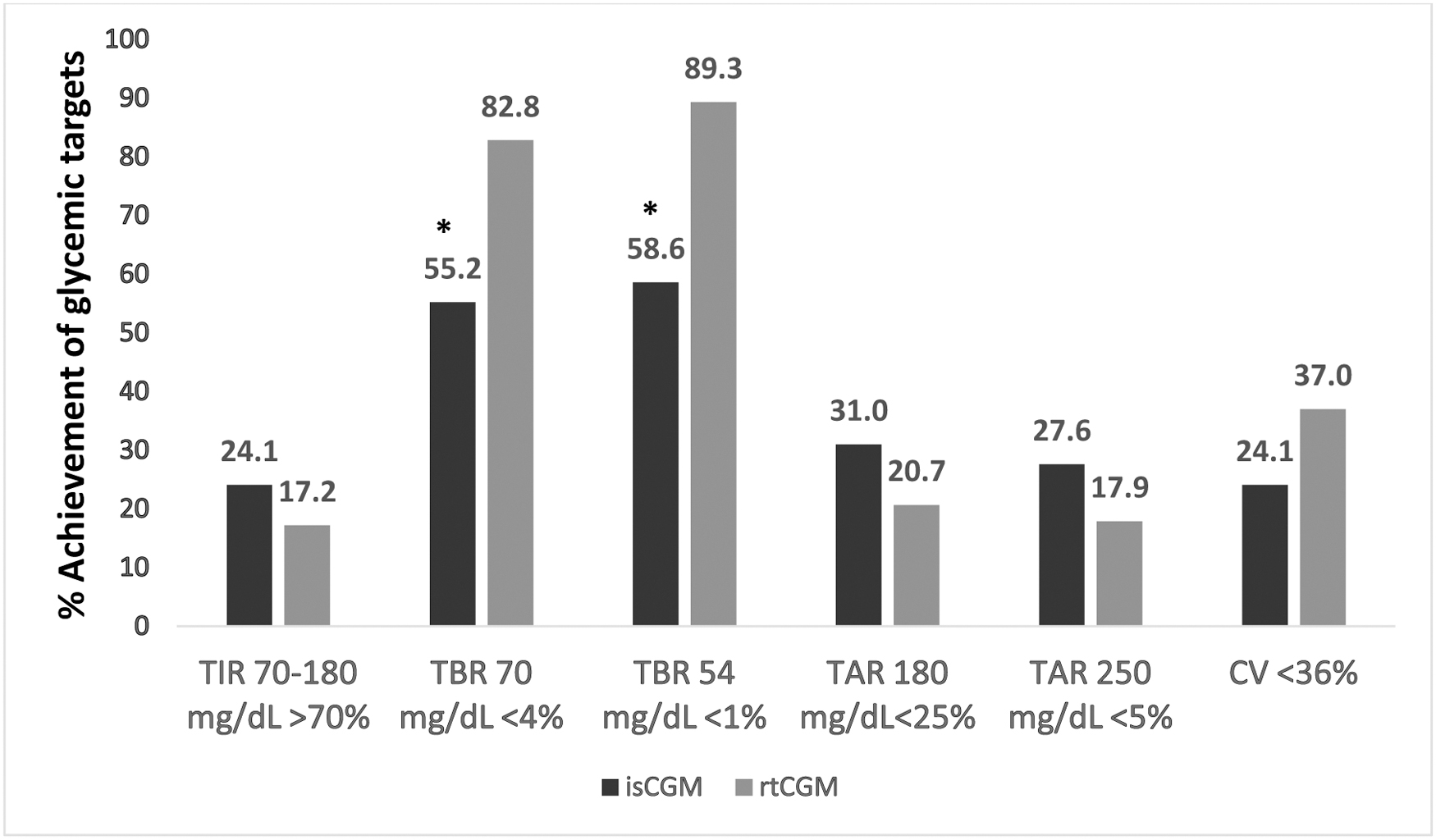
Percentage of participants achieving glycemic targets based on the International Consensus. *P < 0.05. CV, coefficient of variation; isCGM, intermittently scanned continuous glucose monitoring; rtCGM, real-time continuous glucose monitoring; TAR, time above range; TBR, time below range; TIR, time in range.
The number of participants who used the low alert increased from 50% with isCGM to 93.1% with rtCGM (P = 0.001), and the number of participants who used the high alert increased from 37% with isCGM to 82.8% with rtCGM (P < 0.001). During the rtCGM use, 89.7% of participants used the urgent low soon alert, 51.7% used the falling fast alert, and 31% used the rising fast alert. The completeness of data was 92% (76%, 99%) during isCGM use and 100% (96%, 100%) during rtCGM use. No adverse events were reported.
Discussion
Our service evaluation suggests that switching from second-generation isCGM to rtCGM with a predictive low glucose alert in people with T1D reduced the time spent in hypoglycemia and reduced glucose variability (%CV). The number of episodes of hypoglycemia was not different, but the difference in frequency of glucose reporting (15 min sampling with isCGM, 5 min with rtCGM) makes this challenging to interpret. These results are in keeping with previous studies demonstrating similar benefits with rtCGM over a first-generation isCGM. 6 –9 Furthermore, they support the current guidance for preferential rtCGM use in people with impaired hypoglycemia awareness or those at highest risk. 5,14
The Dexcom G7 system has a fixed urgent low alert (set at 3.1 mmol/L [55 mg/dL]), as well as a predictive alert (urgent low soon) to warn of impending hypoglycemia, enabling individuals to take corrective action. Although FSL2 provides the option of low and high threshold-based alarms, only 50% of our participants chose to use this function, whereas ∼90% used the urgent low soon alert and low alert function with Dexcom G7. Previous studies have shown that participants who transitioned from rtCGM without a predictive alert (Dexcom G5) to rtCGM with a predictive alert (Dexcom G6) had lower frequency, duration, and severity of hypoglycemia. 15,16 Furthermore, real-world evidence of rtCGM (Dexcom G6) use showed that people with higher engagement of alert and notification features had reduced exposure to a sensor glucose level <3.0 mmol/L (54 mg/dL) than people with low engagement. 17
Besides the alert function, rtCGM demonstrates glucose data in real time. A qualitative study of people with diabetes using rtCGM emphasized the ease of access to continuous data and benefits of predictive information. 18 It is important to note that at time of writing this article, FSL2 has upgraded its software so that continuous data are directly sent to smartphone users, without the need to scan. 19 This update does not include any changes to the alarm settings for people using FSL2.
Despite a greater proportion of individuals achieving HbA1c <58 mmol/mol, no significant change was demonstrated in HbA1c and percent time spent in range (3.9–10.0 mmol/L [70–180 mg/dL]) after switching from isCGM to rtCGM. This finding is different from the previous ALERTT1 study, which was a 6-month randomized controlled trial switching from isCGM without alerts to rtCGM (Dexcom G6). 20 This is likely to be due to the shorter study duration and that our service evaluation was not powered to detect an HbA1c change, although the magnitude of HbA1c difference between rtCGM and isCGM in ALERTT1 and our service evaluation was very similar. The time spent in range with Dexcom G7 in our study (56.1 ± 18.8%) was comparable to other real-world studies. 21,22
Importantly, the clinical effects seen in our data were achieved with minimal education input and no additional clinic visits to usual care, indicating that sensor choice for adults with T1D can be implemented in the context of routine clinic visits. This is especially critical during consultations with people at highest risk of problematic hypoglycemia or with those not meeting the consensus targets for time spent below range.
The limitations of our data include the small sample size and short duration of follow-up. The comparison of CGM data was obtained from different sensors. However, both sensors have comparable accuracy with a mean absolute relative difference of <10%, 23,24 suggesting that any contribution of differential accuracy is minor. In addition, the consensus targets for glucose times in ranges are sensor-independent, meaning that cross comparison in clinical practice is common. Another limitation was the low number of low alert function usage during the isCGM phase. Information on why participants used or did not use alert and alarm functions of isCGM and rtCGM was not collected; however, alarm burden may be a reason.
In conclusion, adults with T1D who switched from isCGM to rtCGM may benefit from reduced exposure to hypoglycemia and glycemic variability. This is particularly relevant for individuals who are at high risk of hypoglycemia or impaired hypoglycemia awareness.
Footnotes
Acknowledgments
The authors gratefully acknowledge the participants who agreed to participate in this service evaluation project.
Authors' Contributions
N.O. and M.R. conceived and designed the study plan. L.P. and P.A. acquired, analyzed, and interpreted the data. L.P. drafted the article. All authors reviewed, edited, and approved the final version of the article. L.P., P.A., N.O., and M.R. are the guarantors of this study and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Author Disclosure Statement
N.O. has received honoraria for speaking and advisory board participation from Abbott Diabetes, Dexcom, Medtronic Diabetes, Tandem Diabetes, Sanofi, and Roche Diabetes and has received research funding from Medtronic Diabetes and Dexcom. M.R. has received honoraria for advisory board participation from Dexcom and Roche Diabetes.
Funding Information
The Dexcom G7 CGM systems in this service evaluation project were provided by Dexcom. P.A. is funded by the NIHR Grant No. 132960. Infrastructure support for this research was provided by the National Institute for Health and Care Research (NIHR) Imperial Biomedical Research Centre.
