Abstract
Introduction:
The present report celebrates the benchmarking of 100,000 MiniMed™ 780G system users in Europe, Middle East, and Africa (EMEA) and summarizes the major insights into the usability and outcomes of this system.
Methods:
Carelink Personal data (August 2020–August 2023) of users living in EMEA were analyzed. Continuous glucose monitoring-based endpoints were aggregated for (1) the full cohort and (2) a 12-month longitudinal cohort. Subanalyses were done for users on optimal settings (those spending ≥95% of time with glucose target of 100 mg/dL, and ≥95% of time with active insulin time of 2 h), for self-reported age groups (≤15 and ≥56 years) and for various countries/regions.
Results:
Data from 101,629 users (34 countries) were analyzed. Mean time in range (TIR) was 72.3%, glucose management indicator (GMI) was 7%, time below 70 mg/dL (TBR70) was 2.0% and time below 54 mg/dL (TBR54) was 0.4%. In terms of international targets, 59.6% of users achieved a GMI <7%, 62.5% a TIR >70%, 88.4% a TBR70 < 4%, and 90.0% a TBR54 < 1%. Data improved impressively in optimal setting users (TIR = 78.8%, and users reaching TIR >70% = 86.3%) while safety remained (TBR70 = 2.2% and TBR54 = 0.4%). Data showed consistency across self-reported age groups and geographies. In the longitudinal cohort, TIR reached 75.5% in the first month and remained 73.3% or higher over the 12-month period.
Conclusion:
Over 100,000 users of the MiniMed™ 780G system have demonstrated consistency in achieving target control of glycemia.
Introduction
The wealth of information acquired using medical device data management systems, electronic medical records, and health insurance claims data has the capacity to enrich the knowledge of a medical intervention beyond the limitation of randomized controlled trials (RCTs). It can provide additional source knowledge that can drive access of innovative devices and drugs for patients. 1 However, the challenge of the regulatory bodies and the medical establishments to accept evidence provided through real-world data (RWD) is understandable; terms such as real-world evidence (RWE) and RWD, for instance, have been loosely used to provide clinical evidence from as few as seven people 2 and with significant population biases. 3 A literature review, conducted by Ioannidis et al., 4 demonstrated a high incongruity rate between outcomes based on RCTs and nonrandomized RWD studies. To build confidence in RWE generated from the analysis of RWD, several academic societies 1,5 –8 and regulatory bodies (including FDA and EMA) 9,10 have provided guidance in terms of data structure, data quality, (external) validity, consistency in data collection, extraction and processing, transparency, (statistical) methodologies, bias minimization, and databases being fit for purpose (collectively further referred to as the “principles of real-world evidence”).
The introduction of the first commercial automated insulin delivery system in 2016, the MiniMed™ 670G system, provided the opportunity to collect and analyze unbiased RWD in a robust and consistent way (following the above principles). It has provided real-world outcome measures and has assisted health care providers with predictor tools to assist in obtaining the best clinical results. 11 When the MiniMed™ 780G system became available, with its advanced hybrid closed loop algorithm (AHCL), the same process could be used to demonstrate the added value of AHCL in achieving glycemic control. It also gave additional insight into how to further optimize the device settings and provide guidance for user–device interaction to provide for best outcomes. 12,13
The present report celebrates the benchmarking of 100,000 users in Europe, Middle East, and Africa (EMEA) and summarizes the major insight into the usability and outcomes of the MiniMed™ 780G system.
Methods
Design
MiniMed™ 780G system data, uploaded to CareLink Personal software from August 2020 to August 2023, were analyzed. Continuous glucose monitoring (CGM)-based endpoints were analyzed for two cohorts of users. In cohort 1 (the post-AHCL cohort), the glycemic outcomes were assessed after AHCL was initiated for the first time. Per technical requirements, AHCL can only be initiated after a minimum 48 h days of open loop use. During this time, the system learns the insulin needs of the user and is active in manual mode (with basal rates prescribed by the health care provider). In cohort 2 (the longitudinal cohort), the sustainability of glycemic outcomes for each month was evaluated, over a 12-month period.
Eligibility
Users who provided consent for their data to be aggregated and who resided in those EMEA countries where local data privacy regulation permitted data aggregation were included. In addition, users also needed to have a minimum of 10 days of sensor glucose (SG) data in each period evaluated; for cohort 1, this referred to the post-AHCL initiation period, for cohort 2, this referred to each month after AHCL initiation. This requirement (i.e., the minimum of 10 days of SG data) was based on a previous publication validating a similar duration of CGM metrics to estimate or determine long-term glycemic control. 14 All data available post-AHCL initiation were included, whether the system was in AHCL control or in open loop (e.g., after an AHCL exit triggered by either the system or the user).
Endpoints
Glycemic outcomes, including the mean percentage of time spent in 70–180 mg/dL (3.9–10.0 mmol/L) time in range (TIR), at <54 mg/dL (<3.0 mmol/L) (TBR54 [time below range]), at <70 mg/dL (<3.9 mmol/L) (TBR70), at >180 mg/dL (>10.0 mmol/L) (TAR180 [time above range]), and at >250 mg/dL (>13.9 mmol/L) (TAR250), were calculated. The mean SG, standard deviations hereof (SD), and glucose management indicator (GMI) were also assessed, as well as the time the system was used, the percentage of time spent in AHCL, and the percentage of time spent in certain system settings.
Subanalyses
Apart from analyses based on the two cohorts, several subanalyses were performed. The first subanalysis was based on system settings use. The percentage of time that 100 mg/dL (5.6 mmol/L), 110 mg/dL (6.1 mmol/L), or 120 mg/dL (6.7 mmol/L) was used as glucose target (GT) was calculated per user, as well as the percentage of time where 2, 2–3, 3–4, or 4 or more hours was set as active insulin time (AIT). The glycemic outcomes were then analyzed for users who used optimal settings (i.e., a GT = 100 mg/dL of at least 95% of the time as well as an AIT = 2 h of at least 95% of the time).
Second, subanalyses per age group were also conducted. Age was self-reported by users within the CareLink Personal software, where they could select between five age groups (i.e., ≤15, 16–28, 29–42, 43–55 and >56 years), or decide not to provide this information. Third, subanalyses per region/country were done.
Statistics
Means and SDs were used for continuous variables, number of users, and proportions for categorical variables. Analyses were conducted in SAS version 9.4.
Results
Post-AHCL cohort (cohort 1)
Data from 101,629 users (from 34 countries) were analyzed. The mean duration of the observation period post-AHCL initiation was 287.0 days. The mean TIR was 72.3% (Fig. 1) and increased to 78.8% (Fig. 2) when optimal settings were consistently used (N = 6531). The mean GMI was 7%, and mean TBR70 and TBR54 were low with 2.0% and 0.4%, respectively. In terms of international targets, there were 59.6% of users who achieved a GMI <7%, 62.5% who achieved a TIR >70%, 88.4% who achieved a TBR70 < 4%, and 90.0% who achieved a TBR54 < 1% (Fig. 3). Data improved in the cohort of optimal setting users. Of those users consistently using a GT = 100 mg/dL and an AIT = 2 h, 85.0% of users achieved a GMI <7%, 86.3% achieved a TIR >70%, 87.0% achieved a TBR70 < 4%, and 90.5% achieved a TBR54 < 1% (Fig. 4).
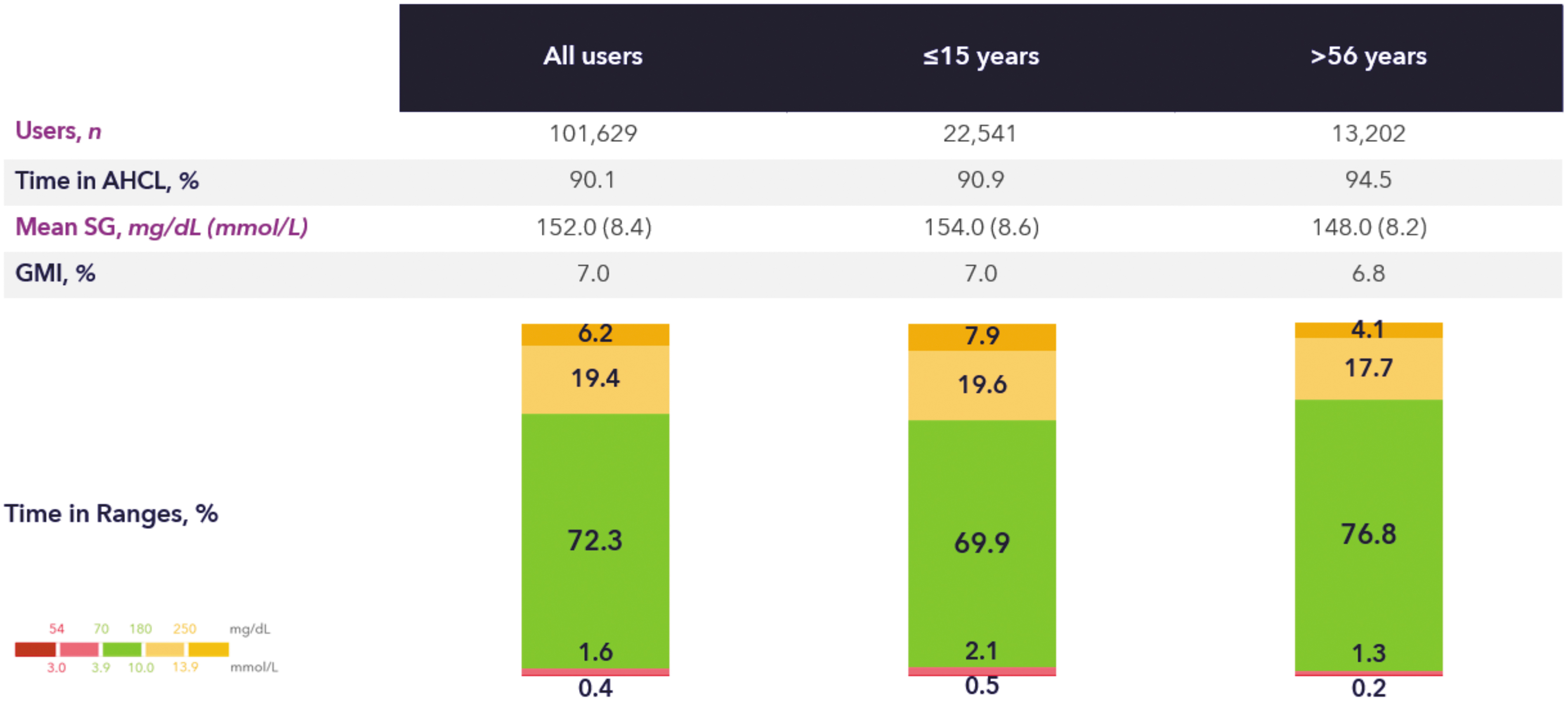
Glycemic control of real-world MiniMed™ 780G system users. Age groups are based on self-reported age in Carelink Personal. AHCL, advanced hybrid closed loop; GMI, glucose management indicator; SG, sensor glucose.
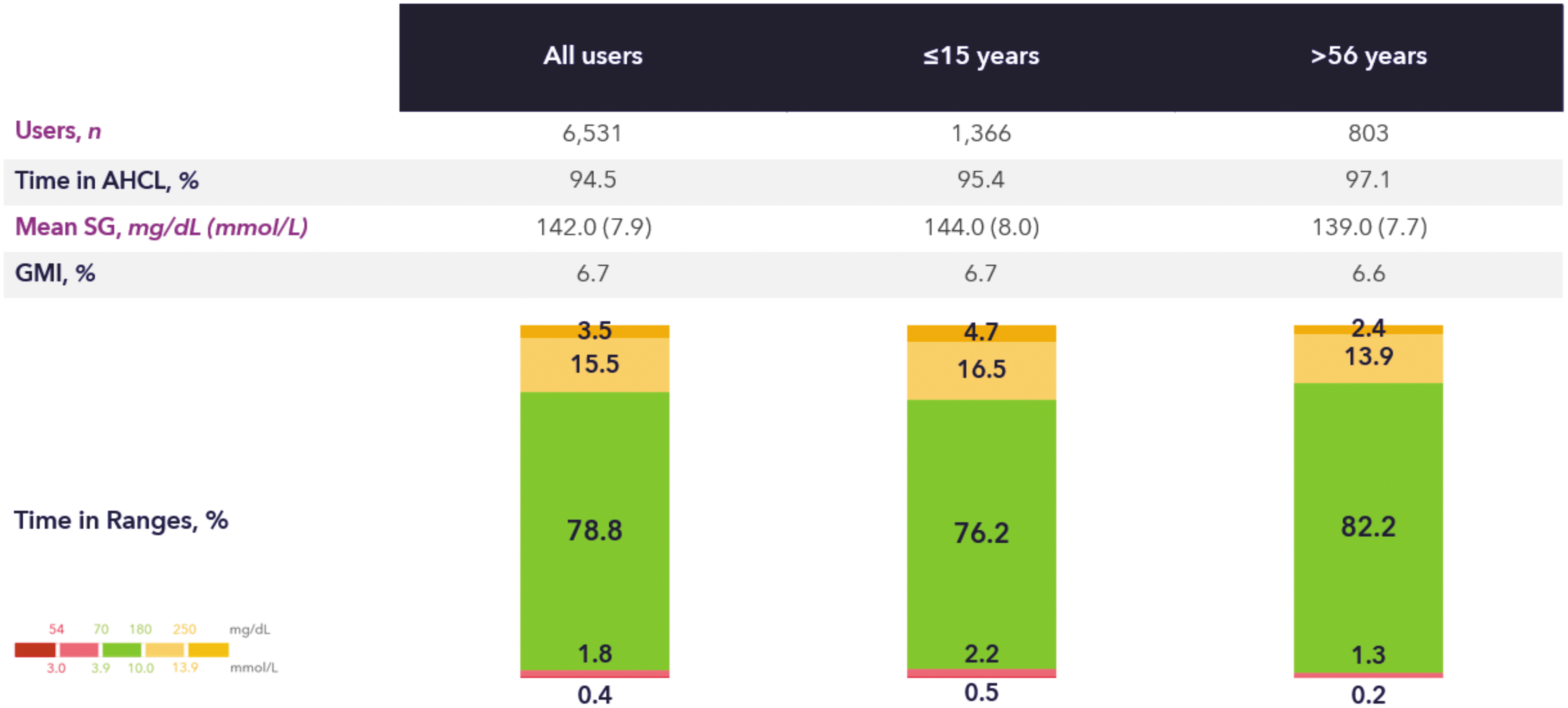
Glycemic control of real-world MiniMed™ 780G system users on optimal settings. Age groups are based on self-reported age in Carelink Personal. Optimal settings are defined as a glucose target of 100 mg/dL for at least 95% of the time as well as an active insulin time of 2 h for at least 95% of the time.
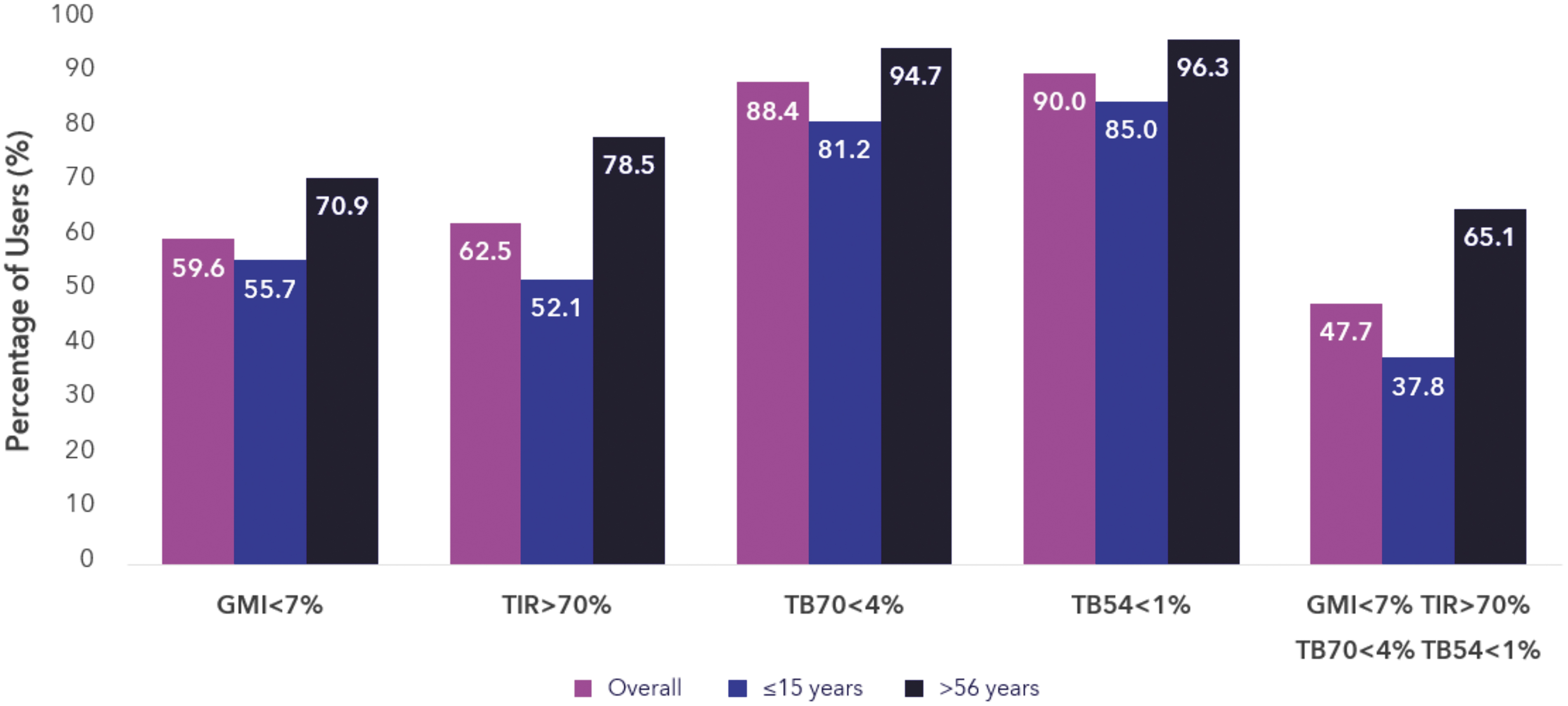
Percentage of real-world MiniMed™ 780G system users that reach international targets. Age groups are based on self-reported age in Carelink Personal. TBR54, time below range (<54 mg/dL); TBR70, time below range (<70 mg/dL); TIR, time in range (70–180 mg/dL).
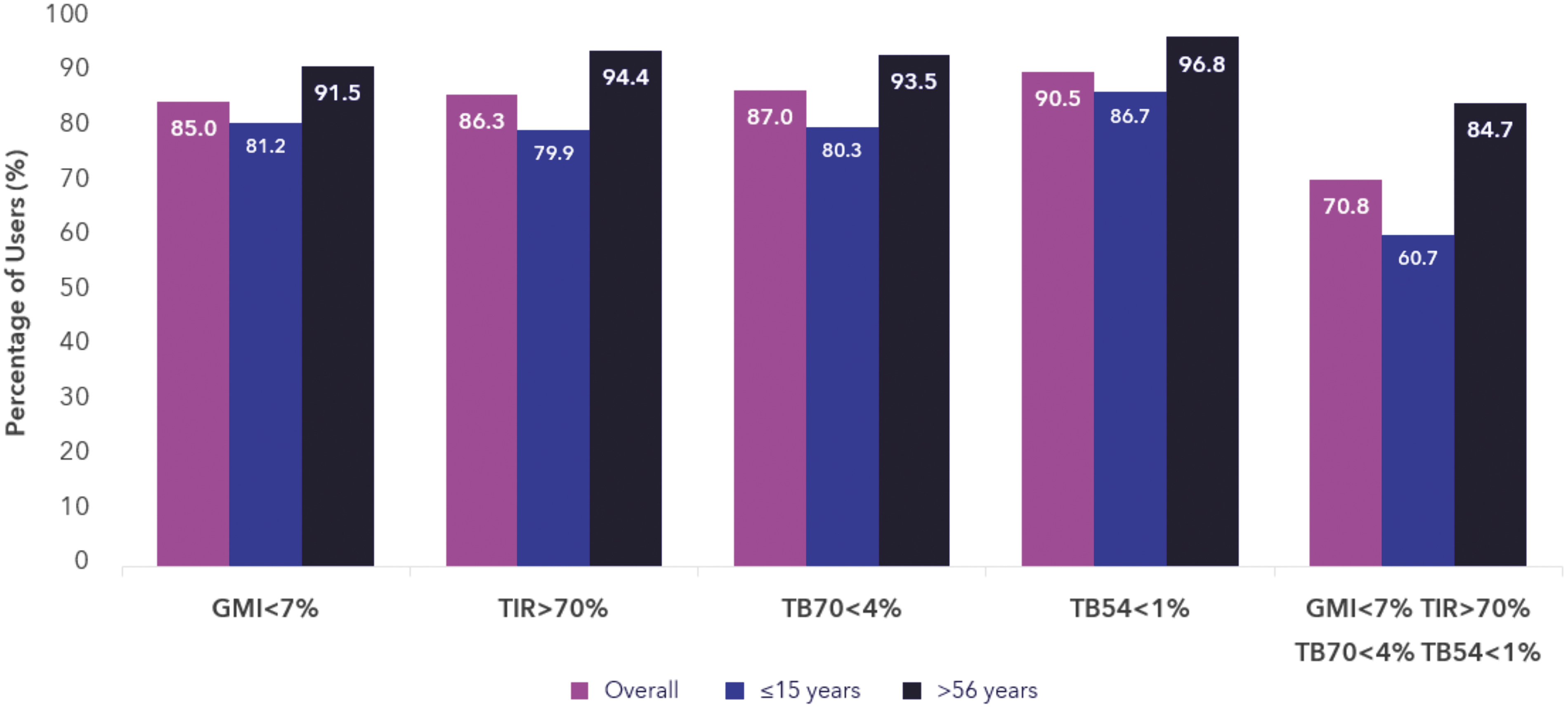
Percentage of real-world MiniMed™ 780G system users on optimal settings that reach international targets. Age groups are based on self-reported age in Carelink Personal. Optimal settings are defined as a glucose target of 100 mg/dL for at least 95% of the time as well as an active insulin time of 2 h for at least 95% of the time.
In terms of age groups, there were 22,541 (22%) users who reported to be aged 15 years or younger, and for whom the mean observation period was 326.0 days. There were 13,202 (13%) users who reported to be aged 56 years or older, and for whom the mean observation period was 289.0 days. Figures 1 to 4 also show the breakdown per age category. The mean TIR was 69.9% for ≤15 years, and 76.8% for ≥56 years. Also here, TIR increased when optimal settings were consistently used (for ≤15 years: 76.2%, for ≥56 years: 82.2%). For both age groups, the average TBR70 and TBR54 were low (for ≤15 years: 2.5% and 0.5%, respectively, and for ≥56 years: 1.5% and 0.2%, respectively). GMI was 7.0% for the ≤15 years group and 6.8% for the elderly.
In term of geographies, Supplementary Figure S1 shows a breakdown per regions/countries and Supplementary Figure S2 shows similar data but only for users of optimal settings. Data show consistency within the EMEA region for all endpoints.
Longitudinal cohort (cohort 2)
The sustainability of the MiniMed™ 780G system performance over 12 months (post-AHCL initiation) is presented in Figure 5. The 32,028 users from this cohort on average had a GMI of 6.8%, which was reached within the first month and that sustained throughout the year. Their mean TIR in the first month was 75.5% and remained 73.3% or higher over the 12 months. TBR70 and TBR54 remained below international targets in all months.
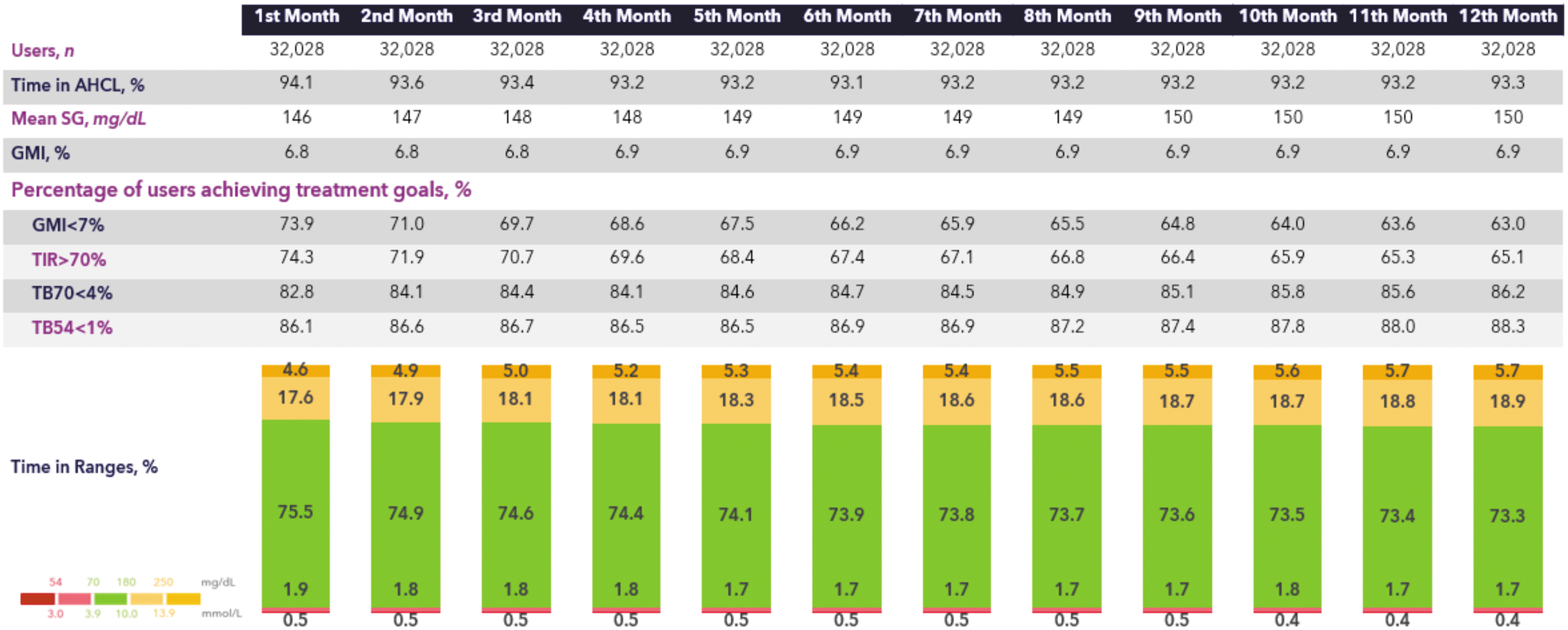
Glycemic control of real-world MiniMed™ 780G system users over 12 months.
Discussion
With data spanning over 3 years and from over 100,000 users, we can confidently conclude that the MiniMed™ 780G system has consistent outcomes for efficacy and safety (as pertains to glycemia) in diverse populations of users: diverse in terms of ages, gender, geographies, and preautomation glycemic control.
The robustness of the data presented in this study can be attributed to our strict adherence to the principles of RWE. 1,5 –10 We, for instance, predefined and validated definitions and parameter parsing, maintained a rigorous statistical plan and methodology, and ensured reproducibility. Most notably, we implemented measures to reduce selection bias. In this context, we primarily mitigated bias by employing technology that enables seamless automated data uploads (through Carelink Personal). In addition, the MiniMed™ pumps have a 3-month storage capacity, ensuring that interruptions lasting <3 months do not lead to missing data. Furthermore, we obtained permission from over 90% of users to utilize their data, and the extensive data set we collected has sufficient granularity and covers all endpoints as suggested per the international consensus statement. 14
The minimal bias in our data becomes evident through the consistent results as shown in this and previous reports, on data across various population sizes, 12,15 age groups, 12 and geographic locations. 12,15,16 It is worth noting that data collected through “user responses to requests” can introduce a selection bias favoring those willing to participate, potentially leading to different outcomes compared with “all real-world users” of the same device. Importantly, our data have remained consistent from in silico studies, 17 through RCTs, 18,19 and up to RWE studies.. 12,13,15,16,20 However, we do acknowledge that in some countries where the MiniMed™ 780G system is not (well) reimbursed, there may be an over-representation of users who can self-finance the device.
Evidence from the RWD of these 100,000 users provides additional support on the importance of utilizing optimal settings (i.e., GT = 100 mg/dL and AIT = 2 h). These as well as previously published findings on optimal setting 13,15 led us to conclude that most users can achieve best outcomes by employing these optimal settings, assuming there are no contraindications. These settings provide the optimal outcomes while maintaining the time below range goal.
Limitations of this study mainly pertain to the constraints of the data collected in CareLink Personal. Certain (sociodemographic) variables, such as gender, age, and diabetes type, are self-reported due to privacy regulations, and age information is grouped. Second, despite the strict adherence to the principles of RWE, the potential impact of missing data on outcomes remains uncertain. A third limitation is that HbA1c could not be utilized as an endpoint in this study, necessitating the use of GMI. Fourth, safety data are restricted to CGM readings, with no information on adverse events such as severe hypoglycemia and diabetic ketoacidosis. Finally, there is an absence of CGM data for the period preceding the initiation of the MiniMed™ 780G system, which impedes our ability to report on true improvements in glycemic control.
In conclusion, over 100,000 users of the MiniMed™ 780G system have demonstrated consistency in achieving target control of glycemia. The results provide reassurance in device performance. Hopefully these outstanding results will be transformed into a longer, healthier, and high-quality life for those living with diabetes.
Footnotes
Authors' Contributions
All authors were involved in the design of the study. A.A., T.v.d.H. and J.C. were involved in the conduct of the study. A.A. and J.C. performed the statistical analyses. All authors were involved in the interpretation of the results. T.v.d.H. and O.C. wrote the article. All authors edited, reviewed, and approved the final version of the article. O.C. is the guarantor of this study and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Data and Resource Availability Statement
The data sets generated during and/or analyzed in this study are available from the corresponding author upon reasonable request.
Author Disclosure Statement
P.C. has served as speaker for Medtronic events and member of Medtronic advisory boards. Other authors are employees of Medtronic.
Funding Information
This study was funded by Medtronic.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
