Abstract
The concept of maintaining blood glucose levels within the 70–180 mg/dL range, known as time-in-range, has raised questions regarding its representation of true physiological euglycemia. Some have speculated that focusing on the time spent within the 70–140 mg/dL range, introduced as time in tight range (TITR) through the International Consensus statement, could serve as a more precise metric for assessing normoglycemia in individuals with type 1 diabetes. This article delves into the current status of TITR as an emerging marker and explores how advanced hybrid closed-loop systems may offer a promising avenue for achieving this higher level of glycemic control.
There has been significant improvement in blood glucose control in people with type 1 diabetes (PwT1D) with the advancements in diabetes technologies and the introduction of ultrafast acting insulins in recent years. 1 Advanced hybrid closed-loop (AHCL) systems are becoming the new standard of care in type 1 diabetes (T1D) management and they have been shown to be superior to multiple daily injections (MDI) with or without continuous glucose monitoring (CGM) and sensor augmented insulin pump (SAP) therapy in several clinical trials in the past decade. 2 –4 In addition, early initiation of diabetes technologies (CGM and AHCL) has shown to significantly decrease A1c and improve CGM metrics for several consecutive years. 5 –7
One of the major challenges in T1D management is the control of postprandial blood glucose. 8 There has been a significant improvement in postprandial blood glucose with the AHCL systems with auto bolus/correct feature, in parallel to the improvements in all-day time-in-range (TIR, 70–180 mg/dL, a concept for the first time introduced in the AWARD-4 substudy). 9 –11 Despite the recommendations of bolusing about 10–15 min before meals, it is common for people with T1D to miss a bolus completely or deliver it at the same time of eating or after a meal. 8,12,13 The Medtronic MiniMed™ 780G AHCL system is an automated insulin delivery system, based on an external physiological insulin delivery controller, including insulin feedback and a supervision/adaptation module, which delivers auto-basal and auto-correction boluses every 5 min. 14 This algorithm has demonstrated to provide high TIR, reaching up to an average of 79% in real-world users what consistently use recommended settings (which is defined as spending ≥95% with a glucose target of 100 mg/dL and ≥95% of the time with an active insulin time of 2 h). 10 The ability of the algorithm to partially compensate for user inaccuracies was demonstrated in a recent study with the MiniMed™ 780G system, including 34 adolescents with T1D. 15 In this study, it was assessed if a simplified meal announcement (giving a preset of three personalized fixed carbohydrate amounts) was comparable with precise carbohydrate counting. 15 Within 12 weeks, 70% of participants in the simplified meal announcement group reached an A1c of <7%, 67% a TIR of >70%, and 82% a time <70 mg/dL (TBR70) of <4%. 15 As expected, the precise carbohydrate counting group achieved better outcomes (TIR of 80.3% ± 7.4% vs. 73.5% ± 6.7%) compared with the simplified meal announcement group; however, the achievement of the people in the simplified meal announcement group was noteworthy and still exceeded the current International Consensus targets. 15 Another AHCL system that can deliver auto-correction bolus is Tandem t:slim X2 with Control IQ technology. If glucose values are predicted to be >180 mg/dL, Control IQ technology calculates a correction bolus using the personal profile settings and a target of 110 mg/dL and delivers 60% of that value. 14,16 This can only occur once per hour in normal settings (excluding sleep mode). 14,16 A study by Akturk et al. evaluated Tandem t:slim X2 with Control IQ technology in 30 adults and compared users with minimal or no user-initiated boluses (auto >90%), to users with intermediate (auto 50%–90%) and to users with high (auto 10%–49%) bolusing behaviors (10 participants were allocated to each group, and matching was done by age, gender, and diabetes duration). 17 As also expected, adults who bolus with meals achieved the highest TIR compared with adults who do not bolus with meals; in 12 months, TIR increased compared with baseline by 22.9% in the auto 10%–49% group, 29.8% in the auto 50%–90% group, and 19.3% in the auto >90% group. However, in this study it was also shown that adults who do not bolus enough with meals can also improve TIR significantly. 17
These advancements in diabetes technologies and therapeutics brought a possibility of tighter control. Therefore, new targets and metrics in CGM may be warranted. Historically, A1c has been the surrogate marker for long-term diabetes complications and its relationship with the diabetic complications has been well recognized. However, due to the limitations of A1c and the advances in CGM technology, new targets in T1D management have been recommended. 18,19 Unlike A1c measurement, CGM can help for pattern recognition for hypo- and hyperglycemia, can show glycemic variability and can guide users for lifestyle changes, prevention of hypoglycemia, and improvement in postprandial hyperglycemia. 20 –22 Standardized CGM metrics have been established by the International Consensus. 18,19
TIR, defined as the percentage of time spent between 70 and 180 mg/dL on CGM, is the mainstay of CGM metrics. 18 Each incremental 5% increase in TIR is associated with clinically significant benefits for individuals with T1D. 18 The International Consensus recommends having >70% TIR per day for people with T1D; however, individualizing the CGM metrics to meet the goals of each person with diabetes may be necessary. 18 Achieving a TIR >70% is associated with A1c target of ∼7%, 23 as per the American Diabetes Association and European Association for the Study of Diabetes recommendations. 18,24 TIR was also suggested as a primary endpoint in diabetes clinical trials by the International Consensus. 25 However, in the recently published “Diabetes Mellitus: Efficacy Endpoints for Clinical Trials Investigating Antidiabetic Drugs and Biological Products Guidance for Industry” draft guidance, FDA stated that TIR is not acceptable as the primary endpoint for glycemic-control indication, citing that TIR is a biomarker that has not been established as a surrogate for a clinical outcome. 26 In contrast, in the same draft guidance, FDA mentioned that CGM-based metrics will be considered with efficacy demonstrated by change in A1c or an appropriate hypoglycemia endpoint, provided that the performance characteristics of CGM devices, data collection, and analyses are adequate. 26 The FDA encouraged a broad input from the professional and lay communities on their draft guidance.
There are, however, many retrospective studies that show an association between TIR and microvascular (retinopathy, nephropathy, and neuropathy) and macrovascular (cardiovascular disease) diabetic complications. 27 –38 In a post hoc analysis from the Diabetes Control and Complications Trial (DCCT), for instance, TIR is derived from 7-point self-monitoring of blood glucose. It shows that every 10% decrease in TIR is associated with a 64% increased risk in retinopathy progression and a 40% increased risk in microalbuminuria in PwT1D. 28 In a post hoc analysis from the DEVOTE trial, TIR is derived from 8-point glucose profiles and the TIR is significantly negatively associated with time to first major adverse cardiovascular event, severe hypoglycemic episode, or microvascular event in people with type 2 diabetes (PwT2D). 39 In 3262 PwT2D, it is also shown that those with more advanced diabetic retinopathy (DR) have significantly less TIR. 29 The prevalence of DR based on severity decreases with ascending TIR quartiles and the severity of DR is inversely correlated with TIR quartiles. In another study with 866 PwT2D, albuminuria risk decreases 6% with 10% increase in TIR. 32 In parallel, Jin et al. showed that all-day TIR and nocturnal TIR are associated with the presence of albuminuria, independent of A1c. 40 In 2215 PwT2D, Lu et al. show that each 10% increase in TIR is associated with 6.4% lower risk of abnormal carotid intima media thickness. 31 Although none of these and other TIR studies come close to the rigor and breadth of the DCCT, collectively they indicate that the association of TIR with chronic complications is likely comparable with that of HbA1c. 37 Nevertheless, long-term prospective randomized controlled studies are suggested to support the relationship between TIR and long-term micro- or macrovascular complications of diabetes.
The TIR of 70–180 mg/dL, however, does not represent physiological euglycemia. A study with 153 adults and children without diabetes showed that, although the median TIR was 99.6%, the time spent in the range of 70–140 mg/dL was 96%. 41 Therefore, it is speculated that time spent in the 70–140 mg/dL range, introduced by the International Consensus statement as time in tight range (TITR), 18,19 can be a valuable metric to assess normoglycemia, and might be a more appropriate target for glycemia in T1D and T2D. In addition, although TIR and TITR are highly correlated (+0.94 Spearman partial correlation), 23 this correlation has shown to be nonlinear, 42 indicating that TITR possesses a unique value and is not mere a substitute for TIR. The new AHCL systems as described earlier, might be the therapy suited to provide this next level of glycemic control.
The International Consensus statement suggested to target >70% time per day for TITR for people with T1D, similar to the recommendation for TIR. 25 However, this was based on data from the SURPASS-3 trial in PwT2D. 43 Shah et al., however, demonstrated that the average TITR was much lower than the average TIR (38.9% ± 16.6% vs. 60.4% ± 18.1%) in adults without diabetes. 44 Similarly, ∼20% point difference was also noted between TIR and TITR in the four trials described by Beck et al. 23 To get insights in the achievable TITR with the MiniMed™ 780G system, we assessed TITR from a large cohort of real-world users of this system (N > 10,000 users) and correlated this with the glucose management indicator (GMI). 45 It is shown (Fig. 1) that the mean TITR achieved in >10,000 adult users was 48.8%, and in those consistently using the recommended settings it even increased to >57%. A target of ∼50% TITR is highly predictive of GMI <6.8% and target of ∼55% is highly predictive of GMI <6.5%. These targets can be achieved in the majority of users that are in concordance with the recommended setting of the MiniMed™ 780G system. Similarly, a study with 105 children and adolescents with T1D revealed that there was a nonlinear relation between TITR and A1c for 60 days (R 2 = 0.69) and that a TITR of 50% corresponded to an A1c of 6.5%. 46 A cross-sectional study from Italy with 854 children and adolescents with T1D using different forms of diabetes technologies showed that TITR was highest in the hybrid closed-loop therapy group (45% ± 11.2%), followed by the SAP group (36.2% ± 11%), the MDI+real-time CGM group (34.2% ± 12.3%), and the MDI+intermittently scanned CGM group (28% ± 10.6%). 47 Only 1.2% of participants were able to achieve a TITR >70%, whereas 4% achieved >60% and 17% achieved >50% TITR. 47 The ROC analysis determined that the optimal cutoff for TIR to discriminate the achievement TITR ≥50% was 71.9%. 47 An increase in TIR of 1% was associated with 1.58 × increased likelihood of achieving TITR >50%. 47 These reports from several independent cohorts seem to be congruent and provide for an acceptable goal of 50%–55% time spent in the tight range of 70–140 mg/dL.
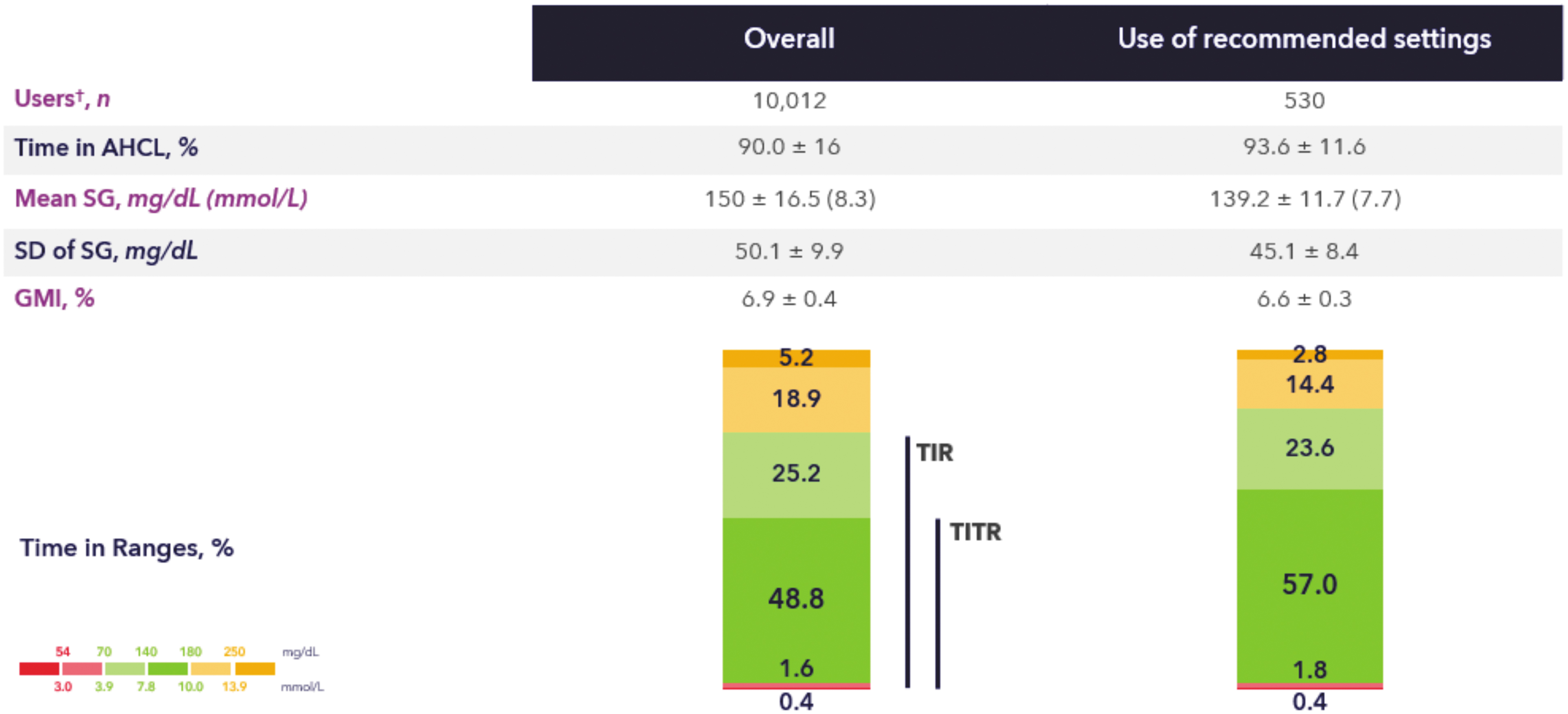
Times in ranges of real-world MiniMed™ 780G system users. Values are shown as mean ± standard deviation. The figure shows glycemic control post-AHCL (overall cohort and recommended settings cohort). For the recommended settings cohort individuals used GT = 100 mg/dL for ≥95% of the time as well as AIT = 2 h for ≥95% of the time. †Data extracted in December 2022 from users living in Europe, Middle East, or Africa, who have a minimum 10 days of SG data pre- and post-AHCL initiation, and who consented for their data to be used in research. AHCL, advanced hybrid closed-loop; GMI, glucose management indicator; SG, sensor glucose; TIR, time-in-range; TITR, time in tight range.
Because much evidence shows that TIR is associated with long-term complications of diabetes and due to the strong nonlinear correlation between TIR and TITR, it is expected that increase in TITR will be associated with the decrease in long-term complications. However, additional research is needed to assess whether TITR can be a good marker in the assessment of long-term diabetic complications. Nevertheless, we believe the timing is opportune for the International Consensus (group) to intensify conversations regarding the inclusion of TITR to the statement, 18 particularly for T1D, and to the preferred CGM metrics for clinical trials. 25
Footnotes
Authors' Contributions
H.K.A., T.B., and O.C. were responsible for the concept of the article. T.v.d.H. and O.C. wrote the article. All authors edited, reviewed, and approved the final version of the article. O.C. is the guarantor of this study and, as such, takes responsibility for the integrity and accuracy of the study.
Author Disclosure Statement
H.K.A. and T.B. have served as speakers for Medtronic events. J.C., A.A., T.v.d.H., and O.C. are employees of Medtronic.
Funding Information
The writing of this article was funded by Medtronic.
