Abstract
Objective:
The article provides practical guidance for (1) interpreting and confirming islet autoantibody screening results for type 1 diabetes (T1D) and (2) follow-up of individuals with early stages of T1D with the goal of ensuring medical safety and providing patients and their families with an assessment of risk for progression to a clinical diagnosis of T1D.
Research Design and Methods:
We used an explicit a priori methodology to identify areas of agreement and disagreement in how to manage patients with early T1D. We used a modified Delphi method, which is a systematic, iterative approach to identifying consensus. We developed a list of topic questions, ranked them by importance, and developed consensus statements based on available evidence and expert opinion around each of the 30 topic questions consistently ranked as being most important.
Results:
Consensus statements for screening and monitoring are supported with figures proposing an algorithm for confirmation of T1D diagnosis and management of early T1D until clinical diagnosis.
Conclusions:
Disseminating and increasing knowledge related to how to interpret T1D screening tests, confirm early T1D diagnosis and monitor for medical safety and clinical disease risk prediction is critically important as there are currently no clinical recommendations. Published guidance will promote better management of T1D screening-detected individuals.
Introduction
For nearly 30
Screening for all four major IA has been available commercially, outside of a clinical research setting, since 2017. It is now also available directly to consumers through programs such as T1Detect (
On November 17, 2022, the Food and Drug Administration (FDA) approved teplizumab as the first drug to delay progression to Stage 3 T1D in adults and children 8 years of age and older with Stage 2 T1D. 7 –9 This approval of a treatment option for people identified with presymptomatic T1D through screening now completes the World Health Organization's criteria for establishing a screening program. 10
In the current environment, there is an urgent need for published guidance on how to diagnose and manage presymptomatic stage 1 and stage 2 T1D in a clinical setting. Screening for early stages of T1D will likely increase now that a clinical treatment is available. Both endocrinologists and primary care providers will now be following and managing presymptomatic T1D, before stage 3 diagnosis. The goal of this article is to disseminate evidence-based recommendations supplemented with the extensive experience at our Center to provide recommendations for early T1D diagnosis and management.
Methods
Given the lack of evidence in some of the areas, a modified Delphi method was used to develop consensus statements. The Delphi method is a process used to gain consensus in a topic when evidence-based guidelines are needed, but there are limited data or evidence available. This method has been used across a wide variety of medical subspecialties and recently was used to develop numerous practice guidelines related to managing COVID-19. There are no standardized assessments for adherence to the Delphi method; however, key elements include “anonymity, iteration, controlled feedback, and statistical stability of consensus.” 11 These key elements help protect the process and product from influence of dominant individuals and group pressure to conform while drawing from the knowledge of multiple experts. 11,12
First, we assembled a group of (n = 9) local experts who have extensive experience in management of patients with early T1D in the setting of large clinical research studies (DAISY, TEDDY, TrialNet, ASK). All panel members were surveyed to generate topic questions. 13 Through an interactive discussion, 50 questions were identified as important to answer to optimize early T1D identification and management. An extensive literature search on early T1D diagnosis and management was performed and reviewed by the lead authors in conjunction with the University Health Sciences librarian; this is a common and highly accepted technique for generating initial content. Each author independently ranked 10 questions in order of most to least importance and submitted these to the lead author to preserve anonymity. Questions ranked by two or more individuals were included as questions requiring development of a consensus statement. 14 Statements (n = 30) were then developed collaboratively and iteratively.
Once consensus was reached regarding the statements to be included, they were separated into five main areas: (1) Interpretation of Screening and Confirmation Results, (2) Monitoring for Medical Safety (prevention of diabetic ketoacidosis (DKA)) and Risk of Progression to a Clinical Diagnosis of T1D, (3) Education, (4) Early Treatment, and (5) Psychological Aspects of Screening and Monitoring for T1D.
Interpretation of screening and confirmation results
People with a family history of T1D and/or a personal or family history of autoimmunity should be screened for T1D by measuring all four major T1D-associated IA. Screening for T1D can be considered for any child through a clinical research program.
Screening for T1D-associated IA is informative at any age during childhood; the optimal ages to start screening is in the second year of a child's life.
Following a negative initial screen, screening should be repeated to improve the sensitivity of detecting T1D. The optimal age to rescreen is around 6 years of life and, if negative, 9–11 years of life.
When the initial screening test is positive, a health care provider (HCP) should have a verbal conversation with the screened individual and the parent/caregiver using simple language to convey the need for a confirmation test, evaluate for symptoms of hyperglycemia, and briefly explain islet autoimmunity and T1D.
A random blood glucose and hemoglobin A1c should be obtained after a positive screen for T1D-associated IA to ensure medical safety (prevention of DKA) and assist in developing a follow-up plan.
A positive screen for one or more IA should be confirmed by measuring all four IAs (to insulin, GAD, IA-2, and ZnT8A) from a separate serum sample in a Clinical Laboratory Improvement Amendments (CLIA)/College of American Pathologists (CAP) certified reference laboratory with an assay that ideally has a high specificity and positive predictive value (PPV).
When a confirmation test is positive, the person screened and the parent/caregiver should have a verbal conversation with a physician, advanced practice provider, or staff member with specialized training and knowledge in early diagnosis of T1D to discuss risk for progression to clinical T1D, frequency of monitoring needed for medical safety and options for early intervention.
Based on IA status alone, the 10-year risk for clinical T1D in a child with confirmed positive multiple IAs is at least ∼70% and approaches a lifetime risk of 100%. Confirmed positive result for a single T1D-associated IA confers a 15%–50% 10-year risk for clinical T1D, depending on the specificity and PPV of the test.
Younger age predicts higher risk of progression to clinical T1D in children with one or more positive IA. 15
A positive screening test for multiple IA is rarely followed by a negative confirmation test. In such case, the HCP should assess for and provide education on symptoms of hyperglycemia and recommend repeat measurement of IA in a CLIA/CAP certified reference laboratory in 1 year.
Approximately a third of positive screening tests for a single IA are followed by a negative confirmation test. In such case, the HCP may recommend repeat measurement of IA in a CLIA/CAP-certified reference laboratory in 1 year.
Until the age of 6 months, IA measured in serum reflects maternal IA status. Up to the age of 1 year, the adaptive immune system is still maturing, and IA may be absent in infants that develop clinical T1D. Despite this, studies have shown that IA develops at a young age with the incidence of islet autoimmunity peaking as early as 9 months old. 16 –18 A recent analysis combined data from prospective birth cohorts in Finland (DIPP), Germany (BABYDIAB), Sweden (DiPiS), and the United States (DAISY and DEW-IT) into the T1D Intelligence (T1DI) cohort to determine what ages are optimal for screening IA if limited to two time points. Screening at 2 and 6 years of age yielded the highest sensitivity (82%, 95% confidence interval [CI] 79%–86%) and PPV (79%, 95% CI 75%–80%) for predicting T1D development by age 15 years.
This analysis also confirmed that IA usually appears before the age of 6 years in children who develop T1D. A similar analysis in TEDDY also indicated that screening for IA at ages of 2 and 5–7 years optimized sensitivity and PPV for clinical T1D development. 19 A more recent T1DI analysis suggests that an additional screening for IA at the age of 10 years can optimize detection of T1D cases that are diagnosed before adulthood. 20 At pediatric practices in the United States, children have well-child visits where vaccinations are administered and hemoglobin and or lead levels may be checked based on American Academy of Pediatrics recommendations at the ages of 1–2 years, 4–6 years, and 11–13 years. Therefore, screening for T1D risk during these time intervals may improve feasibility while also aligning closely with T1D1 and TEDDY data.
Evaluation of an IA screening detected persons is important to ensure that they do not have clinical disease at the time of the positive screen. Following American Diabetes Association recommendations to diagnose T1D, a blood glucose and hemoglobin HbA1c are two tools that can be used to easily assess a patient's glycemic status in clinic along with assessment for symptoms of hyperglycemia.
As with any screening test, it is also important to confirm the IA results by repeating the IA test (Fig. 1). The IA assays used to establish the risk for progression to clinical T1D based on single or multiple IA status are validated assays that have been continuously optimized through the Islet Autoantibody Standardization Program. Based on birth cohort studies using these assays, we know that when a child develops two or more IA, their 5-year risk of clinical T1D is 44% (95% CI 39%–58%), 10-year risk is 70% (65%–74%), and lifetime risk approaches 100%. 4 It is important to note that these risk estimates are from the time of seroconversion and that the time that has already lapsed from seroconversion is often unknown in IA screening detected individuals.
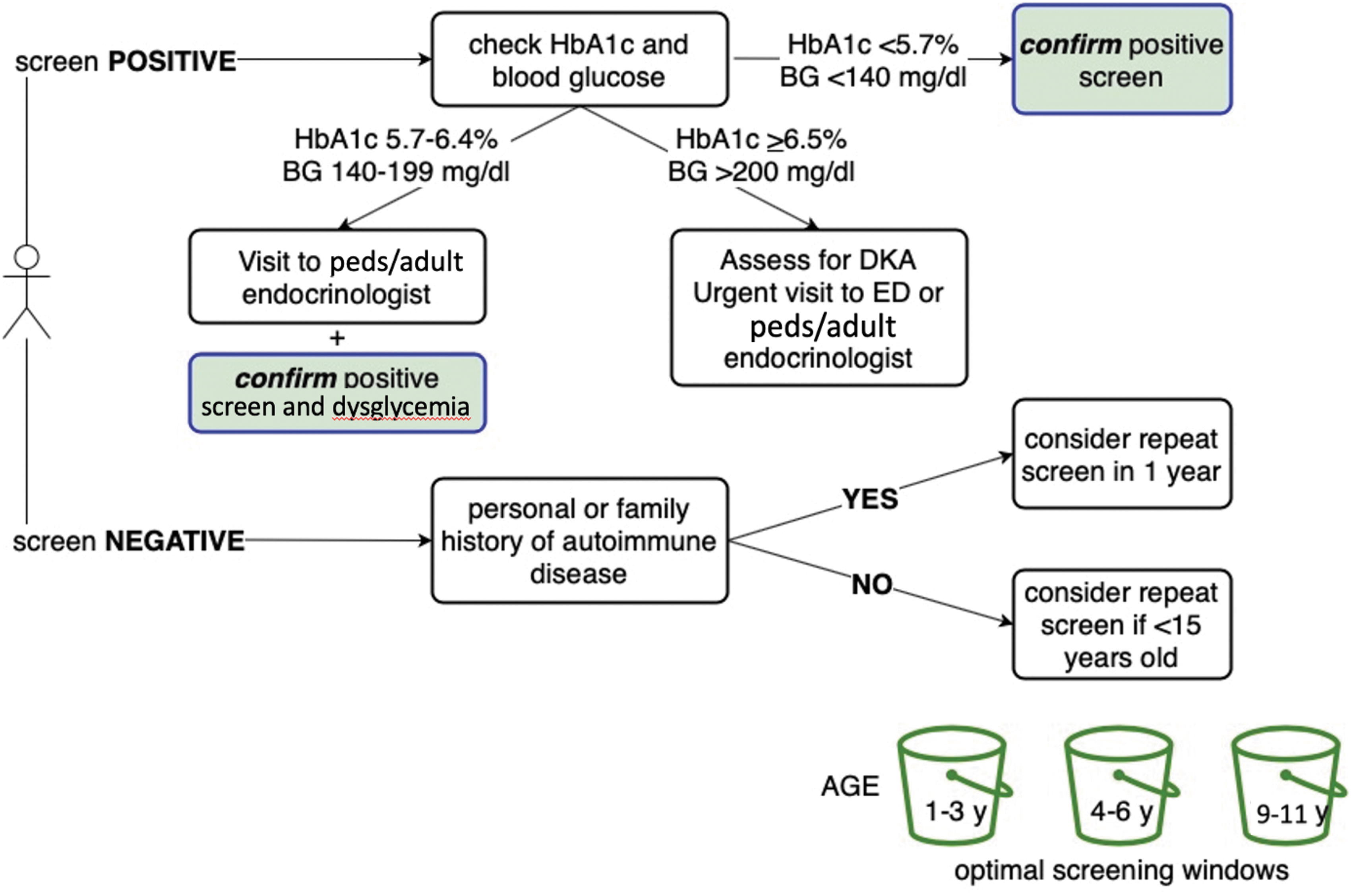
Algorithm for management of a person screened for T1D. Confirmation of a positive screening test by measuring all four biochemical IA (GADA, IA-2A, mIAA, and ZnT8A) and excluding acute hyperglycemia are critical. IA, islet autoantibodies; T1D, type 1 diabetes.
Also, the 15-year risk of progression to clinical T1D is dependent upon how multiple IA is defined. The risk for a child with two IA that are not present at the same time and do not persist over time is only 18% compared to a clinical T1D risk of 88% in those who have two or more IA present at the same visit that persist at the subsequent visit. 21
The risk estimates, based on IA status provided above, are based on positive results confirmed by testing a separate serum sample, generally obtained within 3–6 months after the initial positive test. Because IA assay methodology, sensitivity, specificity, and predictive values vary among laboratories, we expect there to be nonconcordance at times between screening and confirmation. When a positive screening test is followed by a negative confirmation test, measurement of T1D-associated IA should be repeated. If the initial positive screen was multiple IA positive, testing should be repeated in 1 year. If the initial positive screen was a single IA, testing can be repeated per routine recommended intervals, that is, at 4–6 years or 9–11 years. IA can be retested sooner if there is concern for symptoms of hyperglycemia and evidence of dysglycemia.
When a confirmation test is positive, the person screened should have a verbal conversation with a physician, advanced practice provider, nurse, certified diabetes educator (CDCES), or staff member with specialized training and knowledge in early diagnosis of T1D to discuss risk for progression to clinical T1D, frequency of monitoring needed for medical safety, and options for early intervention. Adequate follow-up is essential to reduce the risk of DKA at the onset of clinic T1Dx. 22 Once people enter stage 2 T1D, they require more intensive follow-up as the risk of progression from stage 2 to stage 3 T1D is 50%–60% in 2 years, 75% in age younger than 5 years, and again approaching 100% over a lifetime. 23
Monitoring for medical safety and risk of progression to a clinical diagnosis of T1D
Frequency of medical monitoring for progression to clinical T1D in an individual with IA should be dependent on the number of autoantibodies present, glycemic status, and age.
Patients and their family members should be taught signs and symptoms of diabetes as well as home blood glucose testing during illness and periodically after their main meals to ensure medical safety. Frequency of blood glucose testing is based on age and metabolic status.
HbA1c, oral glucose tolerance test (OGTT), and/or continuous glucose monitoring (CGM) every 3–6 months may be used by medical staff to assess for progression and staging of T1D.
Patients with multiple IA or evidence of dysglycemia should be given directions on when to call their HCP.
Intermittent CGM is emerging as a clinical tool that may be used for both medical safety and monitoring for progression of T1D.
A person with IA who has normal glycemic status can be managed by a HCP or referred for consultation with an endocrinologist at the family's request.
A person with IA who has dysglycemia, per American Diabetes Association (ADA) criteria, can be nonurgently referred to an endocrinologist for management or consultation regarding early treatment options.
A person with IA who meets any of the ADA criteria for clinical T1D should be managed by an endocrinologist. The HCP and endocrinologist should have a verbal conversation before the referral being placed as urgent treatment is needed to avoid DKA.
Evaluation of glycemic status, using OGTT results and metabolic risk scores, can further refine risk estimation.
Once an individual is confirmed to have two or more persistent IA, it is important to develop a follow-up plan that will prevent DKA at clinical T1D onset, allow for access to early treatment options through clinic (e.g., Tzield) or research and offer education as patients progress through the stages of T1D, and eventually to insulin therapy. This follow-up plan can vary based on IA status, metabolic status, and age. The simplest and least expensive tools to help keep children with early T1D out of DKA are educating on signs/symptoms of hyperglycemia and home blood glucose testing (Fig. 2).
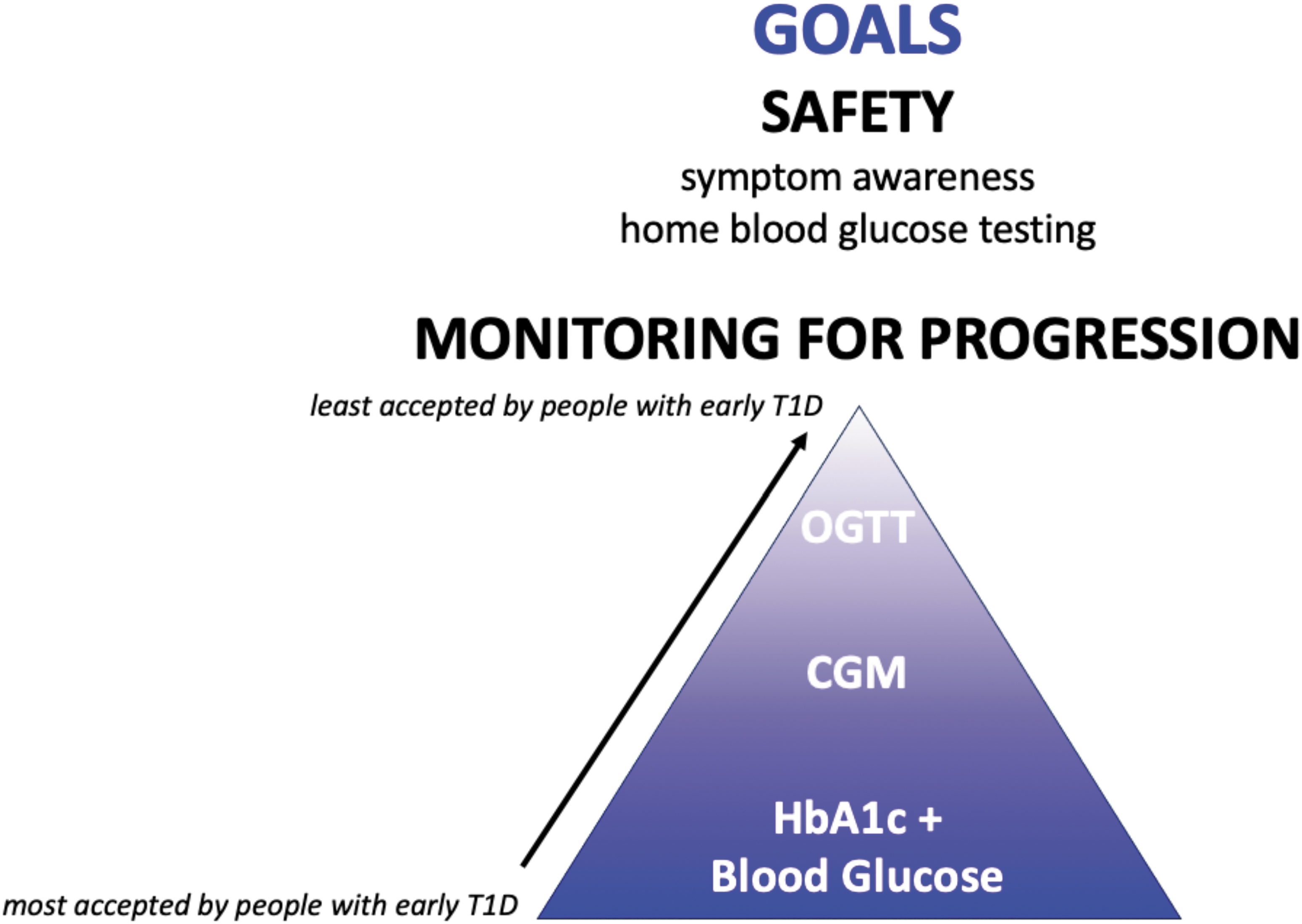
Medical safety (prevention of DKA) and monitoring for T1D progression (to allow for appropriate education and access to appropriate clinical and research treatment interventions) are key goals for management of people in early stages of T1D. DKA, diabetic ketoacidosis.
Home blood glucose testing is not an official criterion used to assess T1D progression, yet we find that home testing is key to ensuring successful monitoring. The frequency of home blood glucose testing recommended varies based on age and glycemic status. In general, patients tend to test less than what is recommended, but every patient should test with increased frequency when ill or if having any diabetes symptoms.
HbA1c and/or OGTT and/or CGM wear should be completed every 3–6 months. Children younger than the age of 6 and individuals with dysglycemia (stage 2 T1D) should use these monitoring tools every 3 months. Some older individuals and those with stage 1 T1D may also benefit from intermittent CGM wear every 3 months. HbA1c is a specific test for assessing T1D clinical diagnosis, yet it is not a sensitive indicator for diagnosis of early T1D in children. 24 HbA1c is important for staging per current ADA criteria. Rising HbA1c in the nondiabetic range may predict progression to clinical T1D. 25 –27
OGTTs with measurement of C-peptide at specified time points can also be used to calculate metabolic risk scores, which have the potential to help improve the prediction of how likely a person is to progress to clinical T1D. 28 –30 In the 6 months before clinical diagnosis, there is an early decrease in C-peptide and the peak to C-peptide occurs later during an OGTT. 31 OGTTs are most predictive of progression when C-peptide is incorporated, but the measurement of C-peptide in a clinical setting is not currently standard of care and will need to be evaluated for feasibility as OGTTs in general are time-consuming, require a peripheral IV placement, and generally accepted poorly by individuals being monitored in early T1D studies. 32 Despite this, OGTTs may remain important for obtaining insurance approval for early treatment options.
CGM is an emerging tool that may be useful for both medical safety and for staging and estimating risk of T1D progression. 33 –37 The psychological impact the CGM results/readings may have on the individual and parent/caregiver when worn unblinded must be considered. HCPs should be prepared to provide adequate support and guidance. The risk of progression to clinical T1D may be as great as 80% in 1-year if an individual has more than 10% of glucose levels >140 mg/dL over a 2-week period. 35
Education
Education about staging of T1D, symptoms of hyperglycemia, and risk of progression to clinical T1D must include both verbal and written communication and should be repeated at regular intervals.
Language and socioeconomic barriers should be addressed when providing education.
People with IA should be encouraged to adhere to a standard healthy lifestyle.
When a person has a confirmed positive IA, the HCP should place a note in the medical record with IA results, risk assessment, and monitoring plan.
Each patient with one or more confirmed IA should receive a medical information card or letter to share at the time of any encounter with a medical provider explaining his/her risk for T1D, symptoms raising concern for progression to clinical diabetes, and recommended laboratory assessment to evaluate for clinical diabetes.
Once an individual is confirmed positive for T1D-associated IA, it is important to inform the individual about the autoimmune process of T1D, the individual risk for T1D, and symptoms of hyperglycemia. For individuals at a high risk for T1D, they should be provided information on how to test blood glucose levels at home and the steps to take if they have an abnormal blood glucose result (see “Monitoring” section for further details). Education must include both verbal and written communication and should be repeated at regular intervals. It should be given in simple language and avoid scientific jargon, using visual aids when available. Families should be given the opportunity to ask questions and seek clarification or access further relevant resources. Experience from the DAISY, TEDDY, and ASK studies has shown that consistent touch points with individuals regarding this information assists with self-awareness and reporting of changes to a HCP.
Educating individuals on the symptoms of hyperglycemia is an important tool for clinical safety. Written materials are an effective way to communicate T1D symptoms and can be shared with caretakers. When reviewing symptoms of hyperglycemia with the individual and caretakers, it is important to acknowledge the effect denial can have on reporting of symptoms. HCPs should be aware that denial is common and that families often attribute symptoms to growth spurts, increased thirst due to activity or heat, and so on. Any time that a symptom occurs, reinforcement of a blood glucose check is imperative.
Individuals with one or more confirmed IA should also receive a medical information card or letter to share at the time of any encounter with a medical provider explaining the risk for T1D, symptoms raising concern for progression to clinical diabetes, and recommended laboratory assessment to evaluate for clinical diabetes. Any provider seeing patient with positive IA should have a low threshold for checking glucose through urinalysis or blood glucose measurement and/or a HbA1c.
Culturally appropriate care and education that factors in language and socioeconomic barriers are essential to improving access to health care and reducing health disparities. Education must be given in a patient's first language, using a translator if needed. The patient should always repeat key information learned back to the provider to assess education and knowledge. Health care teams should also inquire about other barriers, including physical or learning disabilities, medical comorbidities, or access to care issues (e.g., location, transportation) that may influence education, management, and treatment.
A person's background can often contribute to misconceptions about T1D, including the effect that diet, weight, and family history can have on risk of T1D development. Addressing these potential misconceptions up front may aid in working through any denial that comes with an early T1D diagnosis. Individuals with T1D-associated IA should be encouraged to adhere to a standard healthy lifestyle.
Early treatment
Individuals who test positive for T1D-associated antibodies should be referred to the TrialNet Clinical Research Network for opportunities to participate in clinical trials aimed at preventing the onset of clinical T1D in conjunction with referral to a HCP with specialized training and knowledge in early diagnosis of T1D.
Individuals should be assessed for eligibility and have an informed discussion about any clinical treatment options that are available such as teplizumab-mzwv (Tzield) for Stage 2 T1D.
Several immunotherapies have preserved beta cell function in patients with newly diagnosed T1D in research studies, including the Janssen-sponsored golimumab (T1GER study), the T1D TrialNet-sponsored rituximab, abatacept and low-dose antithymocyte globulin trials, and the Immune Tolerance Network-sponsored teplizumab and alefacept trials.
38
–46
However, clinical trials at stage 2 T1D have been completed only for teplizumab and golimumab and only teplizumab is currently approved for clinical use.
7
–9
Prevention trials to delay the onset or prevent symptomatic T1D are led in the US by the TrialNet Clinical Research Network (see
Current data do not support the use of any supplements to prevent or delay the onset of clinical T1D. Several studies have reported inconsistent findings regarding the association of vitamin D, omega-3 fatty acids, and probiotics with the development of islet autoimmunity and T1D.
47
–54
Randomized controlled trials (RCTs) to prevent or delay the onset of clinical T1D include the TrialNet Nutritional Intervention to Prevent T1D study with docosahexaenoic acid and the immune-tolerance with Alum-GAD (Diamyd) and Vitamin D3 to children with multiple IA (DiAPREV-IT2) trial (
Similarly, there have been inconsistent findings regarding the association of cow's milk or gluten and an increased risk of developing islet autoimmunity and/or T1D.
56
–59
Primary dietary modification trials to delay or prevent clinical T1D have failed to show efficacy so far, including the Trial to Reduce IDDM in the Genetically at Risk, evaluating the role of a hydrolyzed casein-based formula compared to cow's milk-based formula and the BABYDIET looking at a gluten-free diet in the first year of life.
60,61
The TEFA Family Prevention: Gluten-free Diet to Preserve Beta-cell Function (TEFA) trial is ongoing (
In addition, there has not been a RCT evaluating the effects of a low carbohydrate diet in preventing or delaying the clinical onset of T1D and dietary changes should only be adopted under medical guidance. 62 Physical activity may play a role in progression to T1D. In TEDDY, children aged 5–15 years with multiple IA who engaged in moderate to vigorous physical activity had a lower risk of progression to clinical T1D. 63
In summary, individuals who test positive for diabetes-associated IA should be referred to the TrialNet Clinical Research Network for opportunities to participate in clinical trials aimed at preventing the onset of clinical T1D in conjunction with referral to a HCP with specialized training and knowledge in early diagnosis of T1D. If eligible for an approved treatment option, the HCP should counsel the patient regarding eligibility, administration, adverse events, and follow-up. If the patient is not eligible for or not interested in early treatment, the patient should continue to have regular metabolic monitoring to prevent the onset of DKA.
Psychological aspects of screening and monitoring for T1D
Accurate understanding of one's risk of T1D is quite challenging for many to comprehend and efforts to improve risk perception accuracy should be continuous.
Anxiety is initially high but decreases over time in many parents of children who are at risk for T1D. However, anxiety tends to remain high in parents of children with multiple persistent IA who are at the highest risk for progression to clinical T1D. 64
An individual or family experiencing persistent anxiety or distress should be referred to a psychologist with expertise in health psychology and/or pediatric psychology.
Inaccurate risk perception is common among parents and caregivers of children at increased risk for T1D, especially among those from the general population who are less familiar with T1D than those who are first-degree relatives. 65 Clear communication about risk using a variety of methods is important for maximizing engagement in screening and monitoring programs for presymptomatic T1D. For example, messaging using specific numbers (e.g., “your child has a 50 in 100 chance of getting T1D”) appears to be a helpful tool when explaining risk to families. In the TEDDY study, risk underestimation was associated with increased risk for early withdrawal, and accurate risk perception was associated with adherence to important aspects of the monitoring protocol (i.e., OGTT). 32,66 Risk perception accuracy tends to naturally decline over time, but this effect can be mitigated by repeating information about risk throughout the course of participation in a screening or monitoring program. 65
Anxiety is common in parents of children at risk for developing T1D. In the TEDDY study, nearly 1/2 of mothers and 1/3 of fathers had elevated anxiety scores when initially informed of their child's increased genetic risk for developing T1D. 64 Anxiety is higher in mothers compared to fathers and in first-degree relatives compared to individuals from the general population who are not as familiar with T1D. Anxiety declines with repeat negative IA results. 64,67 In contrast, anxiety increases when notified of a child's positive IA results and remains high among parents of children with multiple IA. 64 In TEDDY, 57% of mothers and 44% of fathers had elevated anxiety 1 year after initial notification. Even 3 years later, 43% of mothers and 34% of fathers continued to report elevated anxiety. 64
In the ASK study, which screens for T1D-associated IA in the general population, 74.4% of the parents with an IA-positive child had elevated anxiety (unpublished data). Assessment of the child's experience in screening and monitoring programs, and anxiety about developing T1D has been extremely limited thus far. The only published data on this topic suggests that children may respond similarly to adults with an increase in anxiety after learning that they are IA positive that tends to decrease over time. 68 However, the overall child experience, including which aspects of screening and monitoring are most distressing, are different from parents and adult participants. 69
TEDDY study data are potentially biased because it included parents who consented to genetic screening for T1D and participation in a longitudinal study monitoring for development of T1D. Less than half (41%) of the parents eligible enrolled in TEDDY and there was a high percentage of “passive refusers” (i.e., the parents never responded to the TEDDY team about participation). 70 For those who enrolled, routine monitoring for T1D may have increased parents' sense of control in a situation that was neither preventable nor predictable. In contrast, parents may choose to cope with uncertainty of developing T1D by declining participation in studies or disengaging. For example, mothers in TEDDY who had high anxiety and accurate risk perception about their child developing T1D were more likely to withdraw in the first year. 64
T1D is threatening, unpredictable, and uncontrollable. As such, some participants may respond with avoidance and others may respond with high levels of anxiety and distress. Testing for IA should be performed in centers that can provide appropriate counseling and support or have a referral plan in place. 64 Positive screening results can leave families with a sense of uncertainty and lack of control, both factors that are associated with anxiety. Therefore, it is imperative that screening programs be conducted at centers prepared to provide psychological support.
Screening outside of large centers with resources to support and monitor those who are at risk for T1D will be increasingly common. Considering the high rates of anxiety in this population, HCPs should have a low threshold for referring patients to behavioral health specialists, especially given that wait-lists for such services are often long. Clinical health psychologists, pediatric psychologists, or other behavioral health specialists with training in health psychology are ideal referrals for this population given their expertise in working with families coping with medical illnesses.
Resources to use until a patient can be seen by a behavioral health specialist include self-help books such as What to Do When You Worry Too Much: A Kid's Guide to Overcoming Anxiety by Dawn Huebner, PhD for ages 6–12 years and Stuff That Sucks: A Teen's Guide to Accepting What You Can't Change and Committing to What you Can by Ben Sedley, PhD for ages 13-adult.
The decision to not monitor for signs and symptoms of T1D is an individual parent's decision which may be frustrating for medical professionals, especially given that the child will still be at risk for DKA at diagnosis. However, at this time, lack of monitoring coupled with the knowledge of risk for developing T1D, does not constitute medical neglect. A referral to a mental health professional may be helpful to assist the family with adjusting to the fact that their child is at high risk for developing T1D.
Conclusion
Screening for early T1D affords the benefit of reducing DKA at diagnosis and the associated acute and chronic morbidity that can follow. Screening also opens the door for enrollment into T1D prevention trials and access to available early treatment options such as teplizumab mzwv (Tzield). General population screening programs are already in place and continuing to expand. As screening and follow-up of IA-detected individuals transition from research to clinical practice, we will gain more experience as a field, and consensus statements will evolve. With increased evidence related to best practices for screening and monitoring, clinical practice guidelines will need to be developed and updated. Having international experts who represent experience outside of a single center will strengthen recommendations. In the short term, a consistent message for how to diagnose and manage individuals with early T1D is important for families and HCPs during his exciting paradigm shift in the identification and treatment of early T1D.
Footnotes
Authors' Contributions
K.M.W.S.: conceptualization, methodology, writing—original draft, writing—review and editing, visualization, and supervision. B.I.F.: conceptualization, writing—review and editing, and visualization. H.K. O.: conceptualization, writing—original draft, and writing—review and editing. A.K.S.: conceptualization, writing—original draft, and writing—review and editing. K.B.: conceptualization, writing—original draft, and writing—review and editing. C.G.R.: conceptualization, writing—original draft, and writing—review and editing. A.G.G.: writing—review and editing. M.J.R.: conceptualization, writing—review and editing, supervision, and funding acquisition.
Author Disclosure Statement
B.I.F., M.J.R., K.M.W.S., and A.K.S. have participated in medical advisory boards and/or acted as consultants for Provention Bio. K.M.W.S. research funding from Provention Bio.
Funding Information
This work was supported by a grant (R-2109-04817) from The Leona M. and Harry B. Helmsley Charitable Trust.
