Abstract
Introduction:
To evaluate hybrid closed-loop with ultra-rapid insulin lispro (Lyumjev) compared with hybrid closed-loop with standard insulin lispro in adults with type 1 diabetes.
Materials and Methods:
In a single-center, double-blind, randomized, crossover study, 28 adults with type 1 diabetes (mean ± standard deviation [SD]: age 44.5 ± 10.7 years, glycated hemoglobin (HbA1c) 7.1 ± 0.9% [54 ± 10 mmol/mol]) underwent two 8-week periods comparing hybrid closed-loop with ultra-rapid insulin lispro and hybrid closed-loop with standard insulin lispro in random order. The same CamAPS FX closed-loop algorithm was used in both periods.
Results:
In an intention-to-treat analysis, the proportion of time sensor glucose was in target range (3.9–10 mmol/L [70–180 mg/dL]; primary endpoint) was greater with ultra-rapid lispro compared with standard insulin lispro (mean ± SD: 78.7 ± 9.8% vs. 76.2 ± 9.6%; mean difference 2.5 percentage points [95% confidence interval 0.8 to 4.2]; P = 0.005). Mean sensor glucose was lower with ultra-rapid lispro compared with standard insulin lispro (7.9 ± 0.8 mmol/L [142 ± 14 mg/dL] vs. 8.1 ± 0.9 mmol/L [146 ± 16 mg/dL]; P = 0.048). The proportion of time with sensor glucose <3.9 mmol/L [70 mg/dL] was similar between interventions (median [interquartile range] ultra-rapid lispro 2.3% [1.3%–2.7%] vs. standard insulin lispro 2.1% [1.4%–3.3%]; P = 0.33). No severe hypoglycemia or ketoacidosis occurred.
Conclusions:
The use of ultra-rapid lispro with CamAPS FX hybrid closed-loop increases time in range and reduces mean glucose with no difference in hypoglycemia compared with standard insulin lispro in adults with type 1 diabetes.
ClinicalTrials.gov:
Trial registration number NCT05257460.
Introduction
Hybrid closed-loop systems, combining an insulin pump, a continuous glucose monitor (CGM) and a control algorithm, improve glycemic outcomes for people living with type 1 diabetes. 1 –3 Rapid-acting insulin analogues are used to good effect in closed-loop systems 1 but limit closed-loop efficacy due to relatively slow absorption compared to physiological insulin secretion. 4,5
Outside closed-loop use, novel ultra-rapid acting insulin analogues have been shown to reduce postprandial hyperglycemia without increasing hypoglycemia risk. 6 –10 When used with hybrid closed-loop systems, the findings have been inconsistent. Randomized clinical studies of the Medtronic hybrid closed-loop system have demonstrated greater time in range and reduction in hypoglycemia (<3.9 mmol/L [70 mg/dL]) with faster acting insulin aspart when compared to standard insulin aspart. 11,12 In a study utilizing the CamAPS FX closed-loop system, we found that faster aspart reduced hypoglycemia (<3.9 mmol/L [70 mg/dL] and <3.0 mmol/L [54 mg/dL]) versus standard insulin aspart with comparable time in range and mean glucose. 13
Ultra-rapid lispro (Lyumjev, Eli Lilly, Indianapolis, IN, USA) combines standard insulin lispro with treprostinil and sodium citrate excipients which facilitate faster insulin absorption, earlier glucose-lowering, and shorter duration of action. In a randomized study of the Medtronic 670G hybrid closed-loop system, ultra-rapid lispro significantly reduced time spent below 3.9 mmol/L [70 mg/dL]; however, no significant difference was seen in time in range. 14
This study aimed to expand the limited evidence and evaluate the efficacy of ultra-rapid lispro compared with standard insulin lispro in the CamAPS FX hybrid closed-loop system in adults with type 1 diabetes.
Materials and Methods
Study participants and study design
Adults aged 18 years and above with type 1 diabetes on insulin pump for at least 6 months and glycated hemoglobin of 10% or less (≤86 mmol/mol) were recruited. Exclusion criteria included more than one episode of severe hypoglycemia in the preceding 12 months, a total daily insulin dose of 2.0 U/(kg·d) or higher and pregnancy. Eligible adults were recruited from diabetes clinics at Addenbrooke's Hospital (Cambridge, UK). Ethical approvals were obtained from an independent research ethics committee. Participants signed informed consent before any study-related activity. Trial registration NCT05257460.
The study adopted a double-blind, single-center randomized, two-period crossover design comparing hybrid closed-loop insulin delivery using ultra-rapid insulin lispro (Lyumjev) and hybrid closed-loop using standard insulin lispro. A 2-to-4-week run-in period during which the participants used the study insulin pump (Dana Diabecare RS or Dana I; Sooil, Seoul, South Korea) and CGM (Dexcom G6; Dexcom, San Diego, CA, USA) in open-loop preceded the intervention period.
At the initial visit, blood samples were taken for analysis of HbA1c using an International Federation of Clinical Chemistry and Laboratory Medicine-aligned method and following National Glycohemoglobin Standardization Program (NGSP) standards. Participants received training on the use of the study insulin pump and the study CGM system. At the end of the run-in period, appropriate use of the devices was assessed.
Participants were randomly assigned to receive either 8 weeks of hybrid closed-loop with standard insulin lispro followed by hybrid closed-loop with ultra-rapid insulin lispro or vice versa. Permuted block randomization was applied. The assignment was blinded to study participants and study personnel.
At the start of the first closed-loop period, participants attended training on the CamAPS FX hybrid closed-loop app (CamDiab, Cambridge, UK) and competency was assessed. At the start of each study period, participants were provided with blinded insulin vials, and thereafter participants continued the study interventions, each lasting 8 weeks. There was no washout period separating the two interventions. No restrictions were imposed on food intake, physical activity, or travel. Participants were advised to bolus 15 min before eating and adjust as required in both treatment periods, as per standard clinical practice. All participants were provided with a 24-h telephone helpline to contact the study team for study-related support.
Psychosocial metrics were assessed using the Hypoglycemia Confidence Scale, 15 Problem Areas In Diabetes (PAID) questionnaire, 16 and INSPIRE measure (Insulin delivery Systems: Perceptions, Ideas, Reflections and Expectations). 17
Hybrid closed-loop system
The CamAPS FX interoperable app resided on an unlocked Android phone (Galaxy S8, Samsung, South Korea) and used the Cambridge adaptive model predictive control algorithm to direct insulin delivery by insulin pump based on sensor glucose levels. The app includes a bolus calculator for app-initiated meal bolus insulin delivery. Every 8 to 12 min, the adaptive control algorithm calculates the insulin infusion rate, which is communicated wirelessly to the insulin pump. The control algorithm is initialized using participant weight and total daily insulin dose and gradually adapts its insulin dosing based on glucose patterns. The algorithm adapts to duration of insulin action to optimize compatibility with faster acting insulins. The default glucose target is 5.8 mmol/L [105 mg/dL] and can be adjusted by participants as required between 4.4 and 11.0 mmol/L [79 and 198 mg/dL]. 2 The same algorithm was used in both study periods.
Study endpoints
The primary endpoint was the proportion of time when glucose was in the target range between 3.9 mmol/L [70 mg/dL] and 10.0 mmol/L [180 mg/dL] during the study periods as recorded by sensor glucose measurements. Secondary endpoints included mean sensor glucose, glucose variability measured by the standard deviation (SD) and coefficient of variation (CV), time spent at glucose concentrations of <3.9 mmol/L [70 mg/dL], <3.0 mmol/L [54 mg/dL], >10.0 mmol/L [180 mg/dL] and >16.7 mmol/L [300 mg/dL], and insulin delivery (total, basal and bolus amounts). Endpoints were calculated over the whole of each study period and during daytime and night-time periods; daytime was classified as 06:00 to 23:59 and night-time as midnight to 05:59.
Statistical analysis
This was an exploratory analysis aiming for 24 participants completing the study. The statistical analysis plan was agreed by the investigators in advance. The sample was chosen to match the sample size of a study in adults comparing faster insulin aspart to standard insulin aspart using the CamAPS FX closed-loop algorithm. 13 All analyses were performed on an intention-to-treat basis. Mean ± SD or summary statistics appropriate to the distribution were calculated for the primary and secondary outcomes over the 8-week period by treatment intervention. The treatment interventions were compared using a linear mixed model adjusting for period as a fixed effect. The analysis datasets were three records per subject (one for baseline and one for each period). Inclusion of the prerandomization baseline value as a third observation for each subject in the model gave a variance reduction analogous to adjusting for it as a covariate. The model accounted for correlated data from the same subject. Analyses of secondary endpoints were considered exploratory; thus, no formal corrections for multiple comparisons were performed.
Endpoints from participants with a minimum of 48 h of sensor data in at least one study period were analyzed. A 95% confidence interval was reported for the difference between interventions, and P-values <0.05 were considered significant. Nonnormally distributed data were winsorized. Missing data were not imputed for the primary analysis. Outcomes were calculated using GStat software, version 2.3 (University of Cambridge, Cambridge, UK), and statistical analyses carried out using SPSS Statistics software, version 28 (IBM Software, Hampshire, UK).
Results
From January 2022 to August 2022, 28 participants were recruited (18 males, mean ± SD age 44.5 ± 10.7 years, HbA1c 7.1 ± 0.9% [54 ± 10 mmol/mol], and total daily insulin 39.7 U/day) (Table 1). One participant withdrew before randomization due to COVID-19. Twenty-seven participants were randomized, and 14 participants were allocated to hybrid closed-loop with standard insulin lispro first. One participant from this group withdrew during the first study period due to device issues. Twenty-six participants completed both study periods. All randomized participants had at least 48 h of sensor data in at least one study period.
Characteristics of Study Participants at Baseline
Data are presented as mean ± SD or median (Q1, Q3) unless otherwise indicated. Glucose data are based on sensor glucose measurements during the run-in period.
BMI, body mass index; CGM, continuous glucose monitoring; CV, coefficient of variation; SD, standard deviation.
Primary and secondary endpoints calculated using data from all randomized participants are presented in Table 2. Hybrid closed-loop with ultra-rapid lispro demonstrated superiority to hybrid closed-loop with standard insulin lispro for the primary endpoint, the proportion of time sensor glucose was in target range between 3.9 mmol/L [70 mg/dL] and 10.0 mmol/L [180 mg/dL] (ultra-rapid insulin lispro vs. standard insulin lispro; 78.7 ± 9.8% vs. 76.2 ± 9.6%; P = 0.005 with a mean difference of 2.52 percentage points [95% confidence interval {CI} 0.82 to 4.23]). Figure 1 shows 24-h sensor glucose profiles and individual participant's percentage of time spent in target glucose by treatment period.
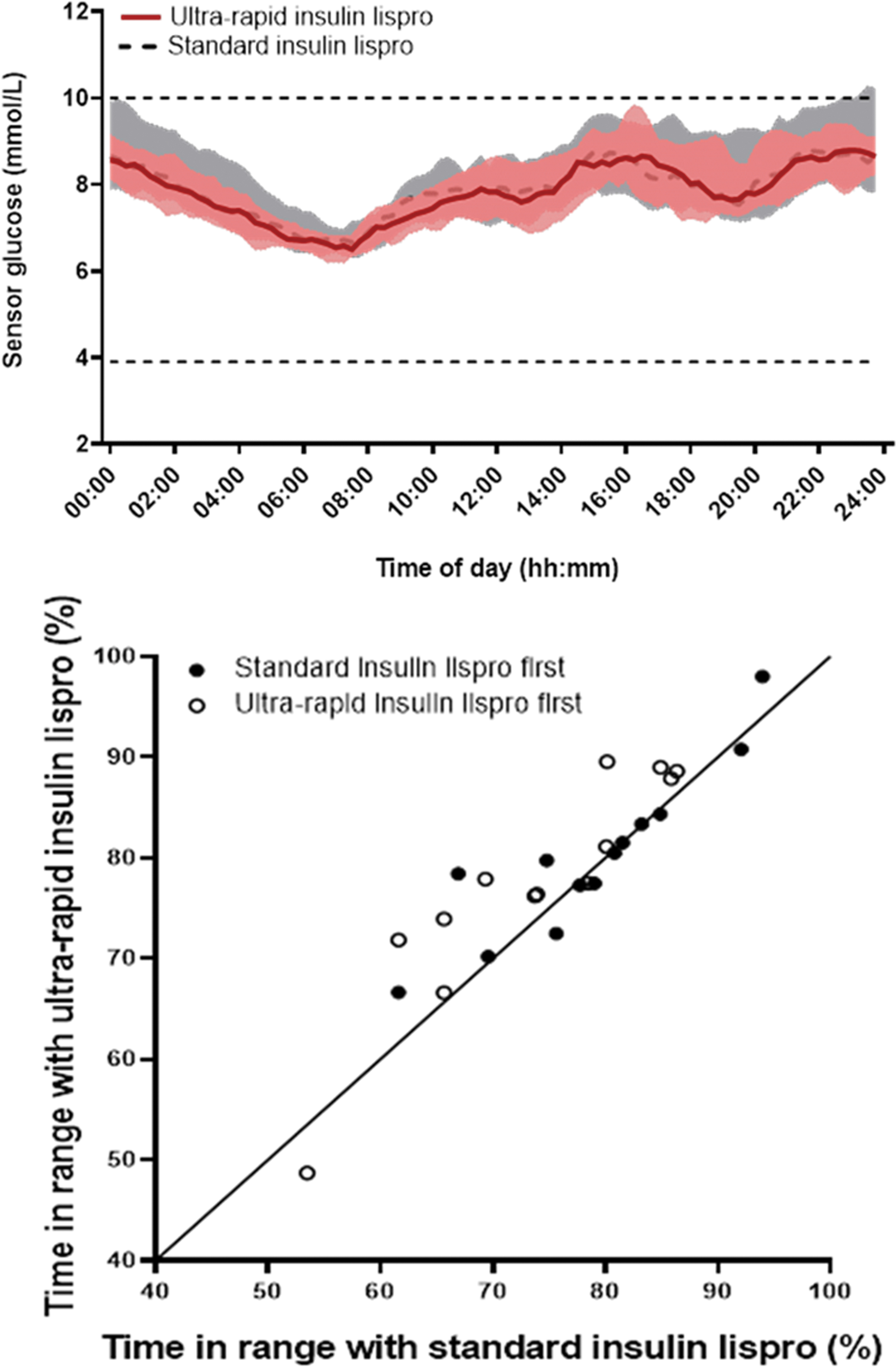
Top panel—Sensor glucose levels (median, interquartile range) during closed-loop with ultra-rapid insulin lispro (n = 26; solid red line and red shaded area) and during closed-loop with standard insulin lispro (n = 27; dashed black line and gray shaded area). Dashed horizontal lines indicate the target glucose range between 3.9 and 10.0 mmol/L. Bottom panel—Percentage of time spent in target glucose range using ultra-rapid insulin lispro compared with standard insulin lispro (n = 26).
Glucose Control and Insulin Delivery Over 8 Weeks of Closed-Loop with Faster-Acting Insulin Lispro and Closed-Loop with Standard Insulin Lispro
Data are mean ± SD for normally distributed values, or median (IQR) for nonnormally distributed values. Transformation (winsorisation) was applied to highly skewed secondary endpoints before statistical analysis.
Primary endpoint.
Based on linear mixed model adjusting for repeated participant measures, period as fixed effect. Baseline values from the run-in period were included in the model.
CI, confidence interval; IQR, interquartile range.
Mean glucose was significantly lower with ultra-rapid insulin lispro (ultra-rapid insulin lispro vs. standard insulin lispro; 7.9 ± 0.8 mmol/L [142 ± 14 mg/dL] vs. 8.1 ± 0.9 mmol/L [146 ± 16 mg/dL]; P = 0.048 with a mean difference of −0.17 mmol/L [95% CI −0.33 to 0.00] or −3.1 mg/dL [95% CI −5.9 to 0.0]) (Table 2). The time spent with the sensor glucose reading above 10 mmol/L [180 mg/dL] was significantly reduced with ultra-rapid lispro (ultra-rapid insulin lispro vs. standard insulin lispro; 19.2 ± 9.9% vs. 21.5 ± 10.1%; P = 0.011 with a mean difference of −2.38 percentage points [95% CI -4.18 to 0.59]) (Table 2). There was no significant difference in the proportion of time sensor glucose was <3.9 mmol/L [70 mg/dL] (median [interquartile range {IQR}] ultra-rapid insulin lispro vs. standard insulin lispro; 2.3% [1.3 to 2.7] vs. 2.1% [1.4 to 3.3]; P = 0.327), <3.0 mmol/L [54 mg/dL] (0.30% [0.15 to 0.46] vs. 0.30% [0.16 to 0.46]; P = 0.650), or >16.7 mmol/L [300 mg/dL] (0.8% [0.2 to 2.0] vs. 1.1% [0.3 to 3.5]; P = 0.083). There was no significant difference in measures of glucose variability (SD or CV of sensor glucose); ultra-rapid insulin lispro versus standard insulin lispro; SD: 2.8 ± 0.7 mmol/L [50 ± 13 mg/dL] versus 2.9 ± 0.6 mmol/L [52 ± 11 mg/dL]; P = 0.098, CV 34.7 ± 5.0% versus 35.5 ± 4.6%; P = 0.279.
Total daily insulin delivery was similar between interventions (median [IQR] ultra-rapid insulin lispro vs. standard insulin lispro; 41.7 U/day [32.6 to 54.7] vs. 42.8 U/day [31.3 to 53.4] P = 0.502). There was no difference in basal or bolus insulin delivery between study interventions (Table 2).
Closed-loop use and glucose sensor use were high (Table 2). Closed-loop was in use for a median of 96.8% [IQR 95.4 to 97.6] with ultra-rapid lispro and 96.5% [IQR 94.7 to 98.0] for standard insulin lispro, P = 0.714 (Table 2).
There was no evidence of a carryover effect between interventions when a period by treatment interaction term was included in the model (P = 0.282). Increased time in range and lower hyperglycemia with ultra-rapid insulin lispro compared to standard lispro was seen during both day time and night time (Table 3).
Daytime and Night-Time Glucose Control and Insulin Delivery During Hybrid Closed-Loop with Ultra-Rapid Insulin Lispro (Lyumjev) and Standard Insulin Lispro
Data are mean ± SD for normally distributed values, or median (IQR) for nonnormally distributed values. Glucose data are based on sensor glucose measurements.
No severe hypoglycemia or diabetic ketoacidosis occurred during the study. There was one nonstudy-related severe adverse event before randomization. Twenty-two other adverse events were reported; 2 occurred during run-in (all nonstudy related), 6 occurred during the standard insulin lispro period (all nonstudy related), and 14 occurred during the ultra-rapid lispro period (4 nonstudy related, 10 study-related). All participants recovered fully without clinical sequelae. The 10-study-related adverse events occurred during the ultra-rapid lispro period and were all due to stinging at the infusion site. In all cases, this was mild and did not result in any of the participants withdrawing from the study. There were 10 device deficiencies reported by seven participants in the study, eight were pump-related, one was sensor-related, and one was due to the smartphone.
There were no differences in psychosocial outcomes as measured by the Hypoglycemia Confidence Scale, Problem Areas in Diabetes questionnaire, and INSPIRE measure (results not shown).
Discussion
This study demonstrated that ultra-rapid lispro with CamAPS FX hybrid closed-loop significantly increased time in range by 2.5 percentage points (36 min/day) when compared to standard insulin lispro. Additionally, significant reductions in time above range and mean glucose were observed. Use of ultra-rapid lispro may offer additional benefits without increased risk of hypoglycemia.
Time in range is a clinically significant glucose metric 18 endorsed by international consensus and associated with a meaningful reduction in microvascular and macrovascular complication risk and HbA1c. 19,20 Although previous guidelines suggest a 5 percentage point change in time in range (TIR) is clinically significant, this is in relation to a technology change rather than an insulin change within existing technology. 18 We suggest that a 2.5 percentage point improvement is a significant clinical benefit, as changing insulin involves no additional user burden or training.
In contrast, a shorter 4-week study of the hybrid closed-loop system Medtronic 670G comparing ultra-rapid lispro with standard insulin lispro found no difference in time in range, with a small but significant increase in mean glucose and significant reduction in time spent below 3.9 mmol/L [70 mg/dL]. 14 The differences between the studies may be attributable to the hybrid closed-loop control algorithms. The CamAPS FX algorithm adapts to the duration of insulin action, day-to-day prandial and diurnal glucose patterns, 1 optimizing compatibility with faster-acting insulin profiles. Bolus dose timing may also be a contributing factor. In this study, participants were advised to bolus 15 min before meals and adjust as necessary. In the 670G hybrid closed-loop study, bolus doses were administered 0 to 2 min before food; time in range was no different; however, a reduction in time spent in hypoglycemia was seen 14 Further studies are warranted to examine the optimal timing of faster-acting insulin bolus dosing in the context of hybrid closed-loop systems. 4
Time spent in hypoglycemia below 3.9 mmol/L [70 mg/dL] or 3.0 mmol/L [54 mg/dL] and total daily dose of insulin between ultra-rapid lispro and standard insulin lispro were comparable. Increase in time in range did not come at the expense of increased hypoglycemia risk or excess insulin use. Previous hybrid closed-loop studies have demonstrated small but significant hypoglycemia reduction with faster aspart in comparison to standard insulin aspart, 11 –13 and with ultra-rapid lispro versus standard lispro. 14 Time spent in hypoglycemia in this study was low for both ultra-rapid lispro and standard lispro, and within the ranges recommended by international consensus guidance on time in range 21 during closed-loop study periods.
The CamAPS FX hybrid closed-loop system performed well with an increase in time in target glucose range from 63.2% at baseline to 78.7% for ultra-rapid lispro and 76.2% for standard insulin lispro, consistent with previous CamAPS FX studies. 13 Time spent in hypoglycemia and total daily insulin were comparable between baseline and closed-loop periods. High use of closed-loop and CGM during the study (median >96%) demonstrates the usability of the CamAPS FX hybrid closed-loop system, an important factor in realizing the benefits of advanced diabetes technology. 22
Ultra-rapid lispro was acceptable despite 10 participants reporting stinging at infusion site, all of whom completed the study. Infusion site discomfort is a recognized side-effect of ultra-rapid lispro, seen in similar studies, 14 likely due to the treprostinil and citrate additives. No participant had previous experience of ultra-rapid lispro (Table 1) and participants and investigators remained blind to the insulin throughout the study.
The strengths of our study include the double-blind, randomized, crossover design with each participant acting as their own control, with data collected over a longer duration than similar studies evaluating ultra-rapid lispro. Participants used the study insulin pump and glucose sensor during the run-in period (in open-loop) and intervention periods, so that any differences between the closed-loop periods could be attributed to the insulin rather than the component hardware. Limitations include a small study population with good baseline glycemic control (mean HbA1c 7.1%). This limits generalizability and may have contributed to the significant but small improvement in time in range. The study participants were solely of white ethnicity, also limiting generalizability. The group randomized to receive ultra-rapid lispro first had higher time in range at baseline, but this was unlikely to have impacted on study outcomes due to the cross-over study design with each participant acting as their own control and lack of period effect for primary outcome.
It would be interesting to analyze the effect of ultra-rapid insulin lispro specifically in the postprandial period. Future studies evaluating closed-loop and ultra-rapid lispro in populations with suboptimal glucose levels at baseline are warranted as increased time in range would be of significant clinical benefit. Ultra-rapid lispro use in fully automated closed-loop systems warrants investigation as the glucodynamic and pharmacokinetic properties of this faster-acting insulin may result in additional benefit.
Conclusion
In conclusion, the CamAPS FX hybrid closed-loop system with ultra-rapid insulin lispro increases time in range and reduces mean glucose, without compromise of hypoglycemic risk. This advanced therapy is safe and effective in adults with type 1 diabetes.
Footnotes
Acknowledgments
We are grateful to all study participants for their contribution, time, and support. We acknowledge administrative support from Josephine Hayes, Alina Cezar, and Matthew Haydock at the University of Cambridge. We acknowledge assistance from NIHR/Wellcome Trust Clinical Research Facility, Cambridge Clinical Research Centre staff Carol Keel, Ranalie De Jesus, Sherly Jose, Samuel King, and Helen Celestino.
Authors' Contributions
C.K.B. and R.H. codesigned the study. M.N., R.L., C.K.B., H.A., and S.H. were responsible for screening and enrolment of participants, arranged informed consent from the participants, and provided patient care. J.W. and J.M.A. supported study monitoring and randomization. R.H. designed and implemented the glucose controller. R.L., C.K.B., M.E.W., and R.H. undertook data analysis. M.N., R.L., C.K.B., and R.H. contributed to the interpretation of the results. M.N. wrote the report. All authors critically reviewed the manuscript. M.N., R.L., C.K.B., and R.H. had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data and analyses and for the adherence of the trial to the protocol.
Author Disclosure Statement
M.N. has received travel grant support from Sanofi, Janssen, and Eli Lilly and was previously chair of the Young Diabetologists' and Endocrinologists' Forum in the United Kingdom, which uses unrestricted sponsorship from industry partners to deliver educational programs for health care professionals. C.K.B. has received consultancy fees from CamDiab and speaker honoraria from Ypsomed. S.H. reports speaker and advisory board fees from Dexcom, Medtronic, Sanofi, and Ypsomed, being director at ASK Diabetes Ltd. and receiving consulting/training fees from CamDiab. J.W. reports receiving speaker honoraria from Ypsomed and Novo Nordisk. J.M.A. reports training fees from CamDiab. M.E.W. reports receiving license fees from Bbraun, patents related to closed-loop, and being a consultant at CamDiab. M.L.E. has been a member of advisory panels and/or received speaker fees from NovoNordisk, Eli Lilly, Abbott Diabetes Care, Medtronic, Ypsomed, Pila Pharma, and Zucara. The University of Cambridge has received salary support for M.L.E. through the National Health Service in the East of England through the Clinical Academic Reserve. R.H. reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license fees from Bbraun, patents related to closed-loop, and being director at CamDiab. H.A. is a consultant at CamDiab. R.L. declares no duality of interest associated with this manuscript.
Funding Information
Dexcom supplied discounted CGM devices and sensors for the study; company representatives had no role in the study conduct. Supported by National Institute for Health and Care Research Cambridge Biomedical Research Centre. The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The views expressed are those of the author(s) and not necessarily those of the NIHR, the Department of Health and Social Care, or other funders.
