Abstract
Background:
During MiniMed™ advanced hybrid closed-loop (AHCL) use by adolescents and adults in the pivotal trial, glycated hemoglobin (A1C) was significantly reduced, time spent in range (TIR) was significantly increased, and there were no episodes of severe hypoglycemia or diabetic ketoacidosis (DKA). The present study investigated the same primary safety and effectiveness endpoints during AHCL use by a younger cohort with type 1 diabetes (T1D).
Methods:
An intention-to-treat population (N = 160, aged 7–17 years) with T1D was enrolled in a single-arm study at 13 investigational centers. There was a run-in period (∼25 days) using HCL or sensor-augmented pump with/without predictive low-glucose management, followed by a 3-month study period with AHCL activated at two glucose targets (GTs; 100 and 120 mg/dL) for ∼45 days each. The mean ± standard deviation values of A1C, TIR, mean sensor glucose (SG), coefficient of variation (CV) of SG, time at SG ranges, and insulin delivered between run-in and study were analyzed (Wilcoxon signed-rank test or t-test).
Results:
Compared with baseline, AHCL use was associated with reduced A1C from 7.9 ± 0.9% (N = 160) to 7.4 ± 0.7% (N = 136) (P < 0.001) and overall TIR increased from the run-in 59.4 ± 11.8% to 70.3 ± 6.5% by end of study (P < 0.001), without change in CV, time spent below range (TBR) <70 mg/dL, or TBR <54 mg/dL. Relative to longer active insulin time (AIT) settings (N = 52), an AIT of 2 h (N = 19) with the 100 mg/dL GT increased mean TIR to 73.4%, reduced TBR <70 mg/dL from 3.5% to 2.2%, and reduced time spent above range (TAR) >180 mg/dL from 28.7% to 24.4%. During AHCL use, there was no severe hypoglycemia or DKA.
Conclusions:
In children and adolescents with T1D, MiniMed AHCL system use was safe, A1C was lower, and TIR was increased. The lowest GT and shortest AIT were associated with the highest TIR and lowest TBR and TAR, all of which met consensus-recommended glycemic targets.
ClinicalTrials.gov ID:
NCT03959423.
Introduction
Type 1
Since that 2019 publication, there has been a rapid evolution in diabetes management technology for T1D. A recent T1DX Quality Improvement collaborative analysis of 28,019 individuals (more than half of whom were ≤18 years of age) investigated glycemic outcomes when hybrid closed-loop (HCL), CGM + pump, or CGM + multiple daily injections therapy was used, from 2019 to 2021. This cross-sectional study demonstrated that HCL use resulted in the lowest A1C (median [interquartile range]: 7.5% [1.6%] vs. 8.0% [2.0%] and 8.2% [2.4%], respectively) and greatest time spent in target range (TIR) (mean ± standard deviation [SD]: 60 ± 17% vs. 52 ± 20% and 50 ± 21%, respectively). 5 The time spent below range (TBR, <70 mg/dL) and the time spent above range (TAR, >180 mg/dL) were significantly lower for HCL users, as well. These overall results, and findings from other retrospective data analyses, 6,7 strongly support the use of automated insulin delivery (AID) in children and adolescents with T1D, to reach current ADA-recommended glycemic goals.
Since the availability of the first AID system for pediatric T1D, 8 HCL algorithms that adjust insulin delivery to a specific sensor glucose (SG) range or target have advanced to provide automatic correction insulin boluses that further improve glycemia. Use of these advanced hybrid closed-loop (AHCL) therapies in pediatric randomized controlled trials (RCTs) 9 –14 and pediatric real-world AHCL performance analyses 15 –18 has shown significant improvement in A1C (or glucose management indicator [GMI]), TIR, and/or TAR, with either reductions or no change in TBR.
Compared with the predecessor MiniMed 670G system, the MiniMed AHCL algorithm can automatically deliver a correction bolus up to every 5 min, when appropriate, based on glucose target (GT) and reduces closed-loop exits and system alarms that, all together, help to improve device use. 19 The addition of basal GT settings that include 100, 110, 120 mg/dL, and the temporary 150 mg/dL target, alongside multiple active insulin time (AIT) settings, provides additional means by which glycemic needs can be managed. MiniMed AHCL system use during the pivotal trial in adolescents aged 14–21 years demonstrated significantly reduced A1C (from 7.6 ± 0.8% to 7.1 ± 0.6%, P < 0.001), increased TIR (from 62.4 ± 9.9% to 72.7 ± 5.6%, P < 0.001), and lowered TBR (from 3.3 ± 2.7% to 2.4 ± 1.8%, P = 0.021), compared with baseline run-in. 9
In addition, exploratory analysis of those using the lowest GT of 100 mg/dL with the shortest AIT of 2 h resulted in a TIR of 78.8% and TBR of 2.6%. The current study investigated safety and glycemic outcomes during MiniMed AHCL system use by a younger cohort aged 7–17 years, and assessed the impact of GT and AIT settings on TIR, TBR, and TAR.
Methods
This prospective, single-arm, nonrandomized trial conducted at 13 investigational centers throughout the United States enrolled children and adolescents 7 to 17 years of age from June 7, 2019, to March 17, 2021. Investigators and investigational staff were trained to the protocol and internal review board approvals were obtained before study start. Informed consent and participant assent were obtained and documented as per the U.S. Food and Drug Administration regulations, and all research efforts complied with the principles of the Declaration of Helsinki.
Criteria for study participation included a T1D diagnosis of ≥1 year for participants up to 13 years of age and ≥2 years for participants 14 to 17 years of age; an A1C <10%; a total daily insulin dose (TDD) of ≥8 U; and >6 months of insulin pump therapy use with or without CGM experience, at screening. Study exclusion criteria included a history of one or more episodes of severe hypoglycemia, diabetic ketoacidosis (DKA), hospitalization or a visit to the emergency room resulting in a primary diagnosis of uncontrolled diabetes in the 6 months before screening, or hypoglycemia unawareness, as measured by the Gold questionnaire, 20 at screening. A complete list of study inclusion and exclusion criteria is listed in the Supplementary Data.
Visit schedule and procedures
The trial comprised a total of 18 visits (phone or clinic), four of which were during the run-in period and 14 during the study period. Informed consent and blood work for A1C were obtained at Visit 1 (the time of enrollment), while Visit 2 consisted of device training and device start, if participants had prior sensor-augmented pump (SAP) experience. The investigational study device consisted of the MiniMed 670G version 4.0 AHCL with the Guardian™ Sensor (3) glucose sensor, Guardian Link (3) transmitter, and CONTOUR®NEXT LINK 2.4 blood glucose meter (Ascensia Diabetes Care, Piscataway, NJ, USA). Participants who had not previously used SAP were instructed to start CGM at Visit 3 and all participants were requested to start 2 weeks of sensor wear at this same visit. During the final run-in visit, study participants had to demonstrate sensor wear compliance before moving on to the study period.
At Visit 5, AHCL was activated. Follow-up visits, thereafter, included device upload, review of device settings, and review of adverse events, if any.
The run-in period (Visits 1 to 4) involved device use with or without predictive low-glucose management or HCL (Auto Basal only), without Auto Correction turned on. The study period (Visits 5 to 18) consisted of ∼90 days in which the Auto Correction and Auto Basal were activated for the entire time. Participants were trained on study devices and advised to remain in automation for as much time as possible. During the study period, participants were instructed to use the 100 mg/dL GT and 120 mg/dL GT for half of the study period duration. Study investigators were able to start participants at either GT (without randomization) and participants were trained to change to the other GT after 45 days (±5 days) into the study period. Throughout the study, insulin-to-carbohydrate ratio (ICR), insulin sensitivity, and AIT adjustments could be made at the discretion of study investigators.
Safety
Safety events were collected from enrollment through the end of the study. Study investigators were instructed to monitor participants throughout the course of the study for the occurrence of any adverse event. A1C was the primary safety endpoint and, also, served as a clinical effectiveness endpoint (see the Statistical Analyses section). Additional safety endpoints were presented as descriptive summaries (i.e., number of events) and included the incidence of serious adverse events (SAEs), serious adverse device effects (SADEs), unanticipated adverse device effects (UADEs), severe hypoglycemia, and DKA. Severe hypoglycemia was defined as an event requiring the active assistance of another individual to administer carbohydrate, glucagon, or other resuscitative actions due to altered participant consciousness. DKA was defined as a blood glucose (BG) meter reading >250 mg/dL, arterial pH <7.3, bicarbonate <15 mEq/L, and moderate ketonuria or ketonemia requiring treatment in a medical facility.
Statistical analyses
A sensitivity analysis conducted on the per protocol population of 129 participants was shown to support the results from a primary data analysis using an intention-to-treat (ITT) population of N = 160 (i.e., individuals who entered the study phase and used AHCL). Thus, analyses of changes in outcomes, descriptive comparisons, and associations were based on the ITT population.
The overall mean difference of change in A1C from baseline to end of study was analyzed to show simple superiority in A1C reduction. The primary effectiveness endpoint was the overall (24-h day) mean change in percentage of time spent in target range (70–180 mg/dL) from baseline to the end of study. The secondary effectiveness endpoints were the overall mean change in percentage of TBR (<70 mg/dL) and TAR (>180 mg/dL).
Additional analyses included change in mean SG, coefficient of variation (CV) of SG, and percentage of time spent at <54 and at >250 mg/dL, TDD, total basal insulin (Auto Basal + programmed open-loop basal), total bolus insulin, Auto Correction insulin, user-initiated insulin, ICR, and daily carbohydrate. Analyses were also determined for the daytime period (06:00 AM to 11:59 AM) and nighttime period (12:00 AM to 05:59 AM).
The present study included exploratory descriptive analyses of the proportion of participants who reached/achieved specific consensus-recommended targets for A1C (<7.0%), TIR (>70%), TBR (<4%), TAR (<25%), and the GMI (<7.0%) during baseline compared with the end of study. In addition, TIR, TBR <70 mg/dL, and TAR >180 mg/dL were determined, when participants used each GT (100 and 120 mg/dL) with a specific AIT setting. System interactions (i.e., closed-loop exits and user-initiated boluses) during baseline versus study were also assessed.
Values from comparisons were averaged per study participant and compared between baseline or run-in and the study period using a Wilcoxon signed-rank test or paired t-test (significance level of 0.05, two-sided). Univariate associations between TIR, TBR <70 mg/dL, TAR <180 mg/dL, and AIT settings were determined using Spearman's rank-order correlation method and were reported as the correlation with 95% confidence interval. Analyses were performed using SAS™ 9.4 (SAS Institute, Cary, NC, USA).
Results
Study participants and disposition
The demographics, baseline characteristics, and therapies used before study start, of participants (N = 160, 11.3 ± 2.5 years of age) who entered the study period and activated AHCL, are listed in Table 1. Participant disposition from study enrollment to study completion is shown in Figure 1. Withdrawals during the run-in were due to participant dissatisfaction with the system (N = 4) and an adverse event (N = 1), both of which were not related to the study device. The study period withdrawals (N = 10) were primarily due to preference for a previous device or system, participant nonadherence to study activities, or loss to follow up. A total of 151 participants completed the study.
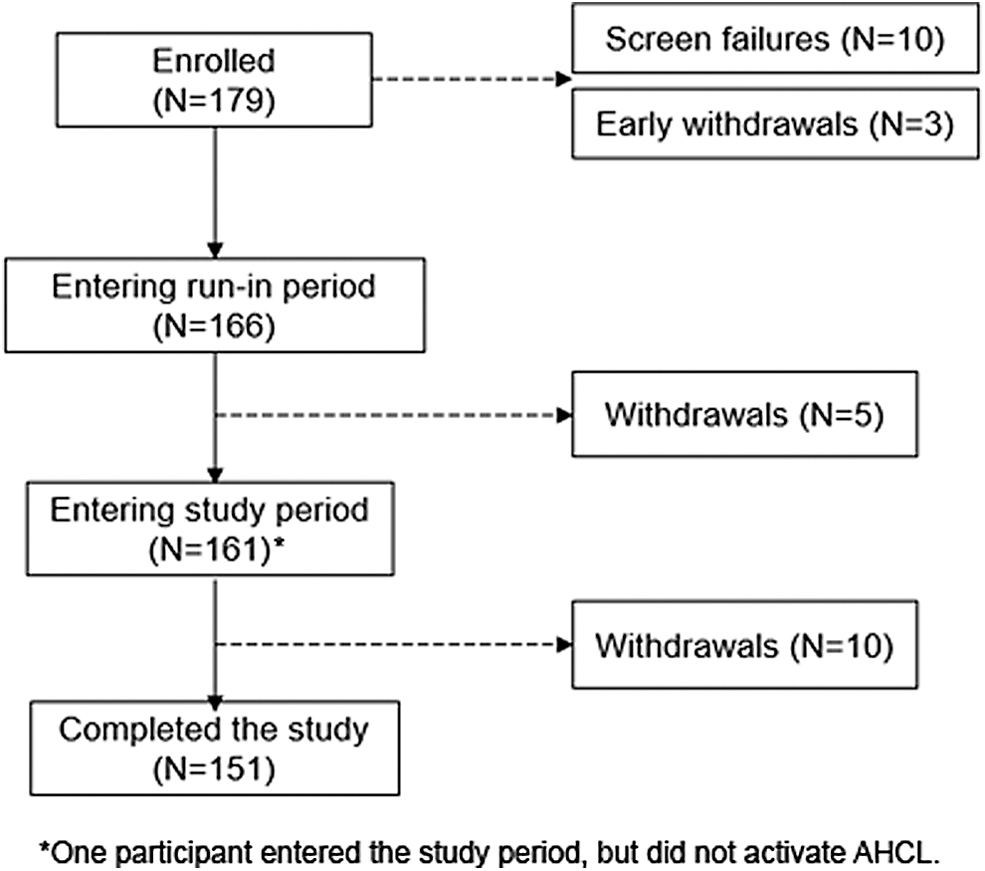
Participant disposition. AHCL, advanced hybrid closed loop.
Demographics and Baseline Characteristics
Data are shown as mean ± SD or minimum–maximum, except where noted.
Estimated error of 0.1 based on the Centers for Disease Control program for growth chart calculation.
Self-reported.
Participant did not designate that continuous glucose monitoring was used.
BMI, body mass index; CSII, continuous subcutaneous insulin infusion; HCL, hybrid closed loop; SAP, sensor-augmented pump; SD, standard deviation.
Glycemic outcomes
Compared with baseline, A1C was significantly reduced from a mean ± SD of 7.9 ± 0.9% (N = 160) to 7.4 ± 0.7% (N = 136), at the end of study. CGM metrics for the 24-h day, daytime, and nighttime periods (Table 2) show that when AHCL was activated, TIR was significantly increased for every period. Factors underlying increased TIR were the statistically significant reductions in TAR (both levels 1 and 2 hyperglycemia) accompanied by the decreased mean SG (Table 2). There were no significant changes in CV of SG and TBR (either level 1 or level 2 hypoglycemia), except for the nighttime period where the CV of SG was reduced from 34.2 ± 5.3% to 32.9 ± 5.2% (P = 0.003).
Glycemic Outcomes During the Run-In and Study Periods
Values are shown as mean ± SD. During run-in, AHCL was inadvertently, temporarily, activated by seven participants. The P-values indicate difference in comparison with baseline run-in.
Sensor-augmented pump with/without predictive low-glucose management or Auto Basal use.
Auto Basal and Auto Correction use.
N = 136.
Wilcoxon signed-rank test.
AHCL, advanced hybrid closed loop; CV, coefficient of variation; SG, sensor glucose; TAR, time spent above target range; TBR, time spent below target range; TIR, time spent in target range.
Figure 2 shows SG profiles throughout the 24-h day and that the difference in SG for the AHCL-activated study period, compared with the run-in period, was most apparent in the 75th percentile. The lowered median of SG was most notable from midnight to early afternoon. Another striking difference between SG profiles was the lowered and narrowed interquartile range for the AHCL-activated study period that was observed from midnight to ∼9 AM.
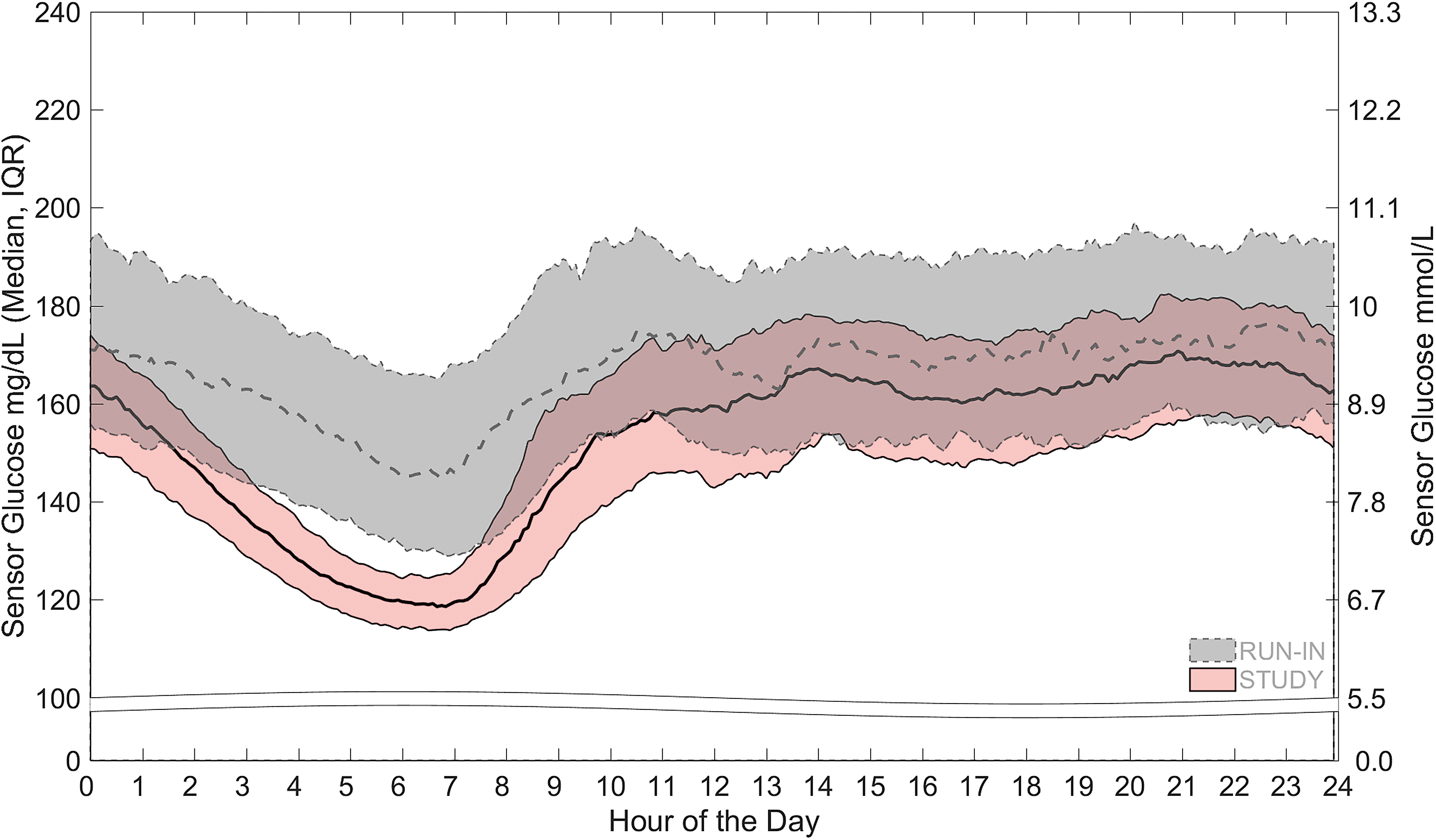
The median and IQR of sensor glucose throughout the 24-h day are shown. The gray band with dotted line represents the run-in period sensor glucose and the pink band with solid line represents the study period sensor glucose. IQR, interquartile range.
The proportion of participants achieving the consensus-recommended A1C of <7.0% and CGM targets (GMI of <7%, TIR of >70%, TBR <70 mg/dL of <4%, and TAR >180 mg/dL) were increased with AHCL versus run-in therapies. The A1C target was reached by 25.7% (35/136) of those using AHCL versus 15.6% (25/160) at baseline. Figure 3 shows that, compared with run-in, the percentage achieving the GMI target was doubled. The proportions achieving the individual targets for TIR and TAR, in addition to the combined targets, were more than doubled or tripled (Fig. 3).
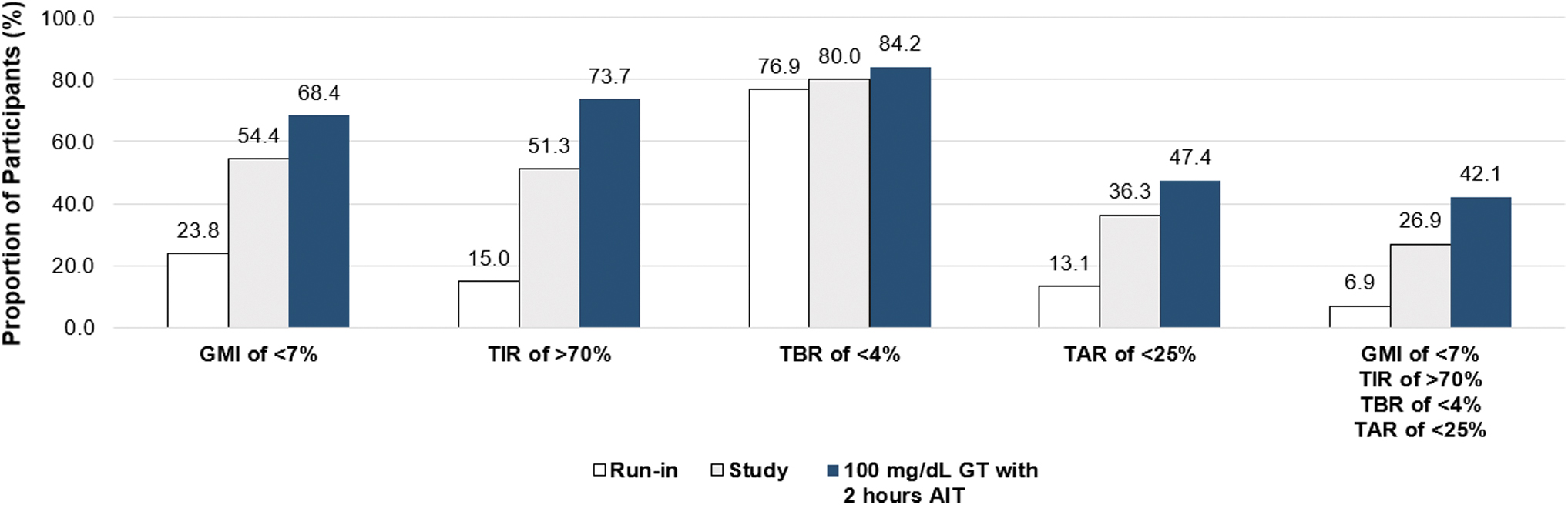
The percentage of participants who achieved consensus-recommended targets for GMI (<7.0%), TIR (>70%), TBR (<4%), TAR (<25%), and the combined targets is shown during the run-in period, overall study period, and study-period use of the 100 mg/dL GT with the 2-h AIT setting. AIT, active insulin time; GMI, glucose management indicator; GT, glucose target; TAR, time spent above target range; TBR, time spent below target range; TIR, time spent in target range.
The impact of AIT setting on time spent at SG ranges when participants used the 100 mg/dL GT and 120 mg/dL GT was explored, and the results are shown in Table 3. Compared with AIT settings ranging from 3 h up to, and including, 4 h (N = 52), the shortest AIT setting of 2 h with the 100 mg/dL GT (N = 19) resulted in a mean TIR of 73.4% versus 67.8% (Spearman correlation, R = −0.29, [−0.42 to −0.14]). Use of the shortest AIT with the 100 mg/dL GT also resulted in a reduced TBR <70 mg/dL (from 3.5% to 2.2%) and TAR >180 mg/dL (from 28.7% to 24.4%) (Table 3).
Glycemic Outcomes During Use of the 100 and 120 mg/dL Glucose Target with Active Insulin Time Settings
Data are shown as mean or correlation (95% confidence interval).
CGM, continuous glucose monitoring; GMI, glucose management indicator.
When participants used the higher 120 mg/dL GT with the shortest AIT setting (N = 26) versus the higher AIT settings (N = 53), mean TIR was 71.0% versus 67.4% (Spearman correlation, R = −0.21, [−0.34 to −0.07]). While mean TBR <70 mg/dL was minimally reduced, mean TAR >180 mg/dL was increased (Table 3). The proportion of participants achieving consensus-recommended CGM targets at the aforementioned GT and AIT settings is, also, listed in Table 3. For visual comparison, the proportion achieving CGM targets when using the 100 mg/dL GT with the 2-h AIT is shown alongside those who reached the same targets during run-in and the overall study (Fig. 3).
Insulin and system use
The insulin delivered, ICR, and daily carbohydrate entered during the 24-h day of the run-in and study periods are listed in Table 4. Over the 3-month study period with AHCL, there was a significant increase in TDD that was due to a significant increase in total bolus insulin (Table 4). When factored as units per kilogram of body weight, the increases in both TDD (1.0 ± 0.2 vs. 0.9 ± 0.2 U/kg, P < 0.001) and total bolus insulin (0.6 ± 0.2 vs. 0.5 ± 0.2 U/kg, P < 0.001) were apparent.
Delivered Insulin, Insulin-to-Carbohydrate Ratio, and Daily Carbohydrate During the Run-In and Study Periods
Values are shown as mean ± SD. During run-in, AHCL was inadvertently, temporarily, activated by seven participants. P-values indicate difference in comparison with baseline run-in.
Sensor-augmented pump with/without predictive low-glucose management or Auto Basal use.
Auto Basal and Auto Correction use.
Wilcoxon signed-rank test.
ICR, insulin-to-carbohydrate ratio; TDD, total daily insulin dose.
The Auto Correction insulin bolus comprised a mean of 15.2% of the TDD and a mean of 25.2% of the total bolus insulin. This corresponded with a reduction in user-initiated insulin delivery from a mean of 25.2 U (59.2% of the TDD) during run-in to a mean of 20.5 U (45.9% of the TDD) for the study period. There was no change in total basal insulin delivery. While daily carbohydrate entry did not differ between the run-in and AHCL-activated study period, the ICR setting, number of daily user-initiated boluses, and amount of user-initiated insulin (in units) were significantly lower during the study period (Table 4).
The number of overall AHCL exits during the study period was low, where 1.0 ± 0.8 exits/week were initiated by the system and 0.4 ± 0.5 exits/week were initiated by participants. There were even fewer exits during nighttime, where 0.2 ± 0.3 exits/week were initiated by the system and 0.1 ± 0.1 exits/week were initiated by participants.
Safety endpoints
There was one SAE that involved severe hypoglycemia in a 14-year-old participant during the run-in period and one SAE that involved abdominal pain in a 7-year old during the study period. Both events were not related to the study device and both resolved without sequelae. There were no events involving DKA, SADEs, UADEs, or death.
Discussion
During 3-month MiniMed AHCL use by children and adolescents aged 7–17 years with T1D, there were no DKA or severe hypoglycemic events. In addition, a clinically significant improvement in A1C (a mean reduction of 0.5%) was observed, compared with baseline, and the overall TIR increased by a mean of 10.9% (2.6 h/day) by end of study. These safety and glycemic outcomes were similar to those reported in the older cohort aged 14–21 years (N = 39) who used the system for the same duration of time, where A1C was reduced by 0.5% (from 7.6 ± 0.8% to 7.1 ± 0.6%, P < 0.001) and overall TIR increased by 10.3% (from 62.4 ± 9.9% to 72.7 ± 5.6%, P < 0.001). 9
Important distinctions between the older adolescent cohort in Carlson et al. 9 and that in the present study were that baseline TBR <70 mg/dL and TBR <54 mg/dL in the younger participants were lower (mean of 2.7% vs. 3.3% and mean of 0.7% vs. 0.9%, respectively) and did not change for the 24-h, daytime, or nighttime periods, during AHCL use. In contrast, AHCL reductions in TBR <70 mg/dL were observed for all time periods in the older cohort. Importantly, the younger cohort in the present study had a higher baseline TAR >180 mg/dL (mean of 38.0% vs. 34.3%) and TAR >250 mg/dL (mean of 12.1% vs. 9.1%), both of which were significantly reduced alongside mean SG, with AHCL use. Aligning with other pediatric AID publications, with either HCL 21,22 or AHCL, 9,13,14,23 glycemic metrics in the present study were notably improved for the nighttime period.
For some time, varied insulin pump settings, such as insulin sensitivity factor, ICR, and AIT (i.e., the duration of time a pump considers a previously administered dose to still have a glucose lowering effect), have been leveraged to optimize glycemic outcomes for youth with T1D. 24,25 The investigational MiniMed AHCL system and the approved MiniMed 780G system include modifiable GT, AIT, and ICR settings. Real-world analyses, 16,26 prospective observational studies, 7,9,27,28 and one RCT 12 have shown that pediatric use of the 100 mg/dL GT and 2-h AIT demonstrated greater likelihood of attaining consensus-recommended targets 4,29,30 for TIR, TBR, and TAR, in addition to A1C or GMI. Regression analyses 26,28 have also determined that use of those settings predicted greater TIR and reduced TAR, without impacting TBR.
This was evident in the present study, where AHCL significantly reduced TAR >180 mg/dL and TAR >250 mg/dL compared with run-in, but fell short of the consensus-recommended targets 4,29,30 of <25% and <5%, respectively, until application of the aforementioned settings. An exploratory analysis demonstrated a mean TIR that increased to 73.4% and a lowered mean TAR and TBR of 24.4% and 2.2%, respectively, when a small number of participants used the lowest GT and shortest AIT. These changes were likely due to the algorithm's more frequent and increased Auto Correction insulin delivery based on the 2-h AIT, in addition to an Auto Basal that targeted glucose to the lowest 100 mg/dL GT. It is also possible that the strengthened (i.e., lowered) ICR during the study period played a small role in glycemic changes.
The AHCL system's ability to help lower insulin-dosing burden, as observed in Carlson et al. 9 and other studies investigating MiniMed 780G system use, 12,18,31 was also observed in the present study. For example, and compared with run-in, there was a reduced amount of insulin and the number of insulin boluses delivered by study participants, while daily carbohydrate announcements remained unchanged. It is important to note, however, that Auto Correction insulin, in the present study, comprised a mean of 25% of total bolus, which suggests that the system attempted to make up for the increased TAR that, in turn, minimized the need for manual insulin corrections. In fact, a previous MiniMed 780G study postulated that a clinical “auto-correction bolus threshold” may prove helpful with identifying system users who may need help with either carbohydrate counting and ICR adjustments 18 or, as seen in the present study, changes to system settings.
The observed impact of MiniMed AHCL therapy settings on reaching consensus-recommended glycemic targets provides a level of reassurance for pediatric use. This is important for young T1D populations who face numerous challenges achieving and maintaining recommended glycemic targets, 3 and many who face considerable barriers to diabetes technology access. 32,33 Notwithstanding the varied complexities (e.g., socioeconomic status, clinical and family support, health insurance coverage, and social bias or discrimination) that influence pediatric diabetes technology adoption, suboptimal glycemia has been a key modifiable risk factor associated with diabetes-related complications. 34 Accordingly, glycemic benefits of AHCL therapy have spanned from those with suboptimal glycemia 7,13,35,36 to those who are “well-controlled” and who achieve further improvement in glycemia. 27,37
Findings from these AHCL investigations support clinically substantial benefits for many with T1D who experience a range of glycemia, and the basis for the recent strong recommendation for AID therapy use by the International Society for Pediatric and Adolescent Diabetes Clinical Practice Guidelines. 30
Limitations to the present study include the nonrandomized design that did not involve a control group and the minimal enrollment of youth with T1D from a diverse race/ethnicity or underserved socioeconomic status. Study inclusion also precluded a baseline A1C of >10% and the requirement of at least 6 months of insulin pump experience (with or without CGM use) before enrollment, which limit generalizability of results to a larger pediatric population with T1D. Strengths of the present study include the ITT population size (n = 160) that was three times as large as the older adolescent cohort (N = 39) using the MiniMed AHCL system for the same duration of time. This allowed assessment of the 100 mg/dL GT with the AIT of 2 h in a greater number of younger system users (N = 19 vs. N = 7), which clearly showed a positive impact on glycemic control.
Conclusions
The present study demonstrates safe use of the MiniMed AHCL system in youths 7–17 years of age and a clinically significant reduction in A1C versus baseline, with an increase in TIR (compared with run-in). Similar to the MiniMed AHCL pivotal trial results in the older adolescent and adult cohorts, and consistent with real-world data from pediatric and adult MiniMed 780G system users, specific system settings helped participants achieve consensus-recommended glycemic targets.
Footnotes
Acknowledgments
The authors gratefully thank the MiniMed AHCL system study participants and their families, in addition to the investigational staff managing the pivotal trial. They also wish to acknowledge Yuri Treminio, BA (Medtronic employee), for her medical writing contribution.
MiniMed AHCL System Study Group: Bruce W. Bode (Atlanta Diabetes Associates, Atlanta, Georgia), Ron L. Brazg (Rainier Clinical Research Center, Renton, Washington), Bruce A. Buckingham (Stanford University School of Medicine, Stanford, California), Anders L. Carlson (International Diabetes Center, HealthPartners Institute, Minneapolis, Minnesota), Luis Casaubon (Texas Diabetes and Endocrinology, Austin, Texas), Xiaoxiao Chen, (Medtronic), Mark P. Christiansen (Diablo Clinical Research Center, Walnut Creek, California), Toni L. Cordero (Medtronic), Gregory P. Forlenza (Barbara Davis Center of Childhood Diabetes, Aurora, Colorado), Satish K. Garg (Barbara Davis Center of Diabetes, Aurora, Colorado), Benyamin Grosman (Medtronic), Richard A.M. Jonkers (Medtronic), Kevin B. Kaiserman (SoCal Diabetes, Torrance, California), Mark S. Kipnes (Diabetes and Glandular Disease Clinic, San Antonio, Texas), Kashif Latif (AM Diabetes and Endocrinology Center, Bartlett, Tennessee), Scott W. Lee (Loma Linda University, Loma Linda, California), David R. Liljenquist (Rocky Mountain Diabetes and Osteoporosis Center, Idaho Falls, Idaho), Louis J. Lintereur (Medtronic), Margaret Liu (Medtronic), Jennifer McVean (Medtronic), Neha Parikh (Medtronic), Fen Peng (Medtronic), Catherine Pihoker (University of Washington, Seattle, Washington), Athena Philis-Tsimikas (Scripps Whittier Diabetes Institute, La Jolla, California), Rodica Pop-Busui (University of Michigan, Division of Metabolism, Endocrinology and Diabetes, Ann Arbor, Michigan), John H. Chip Reed (Endocrine Research Solutions, Inc., Roswell, Georgia), Andrew S. Rhinehart (Medtronic), Anirban Roy (Medtronic), Jennifer L. Sherr (Yale University School of Medicine, New Haven, Connecticut), John Shin (Medtronic), Dorothy I. Shulman (University of South Florida, Pediatric Diabetes and Endocrinology, Tampa, Florida), Kamalpreet Singh (Texas Diabetes and Endocrinology, Round Rock, Texas), Ronald H. Slover (Barbara Davis Center of Childhood Diabetes, Aurora, Colorado), James R. Thrasher (Arkansas Diabetes and Endocrinology Center, Little Rock, Arkansas), Melissa Vella (Medtronic), Robert A. Vigersky (Medtronic), and Di Wu (Medtronic).
Authors' Contributions
The principal investigator authors (C.P., D.I.S., G.P.F., K.B.K., J.L.S., J.R.T., B.A.B., M.S.K., B.W.B., A.L.C., K.L., D.R.L., and R.H.S.) contributed substantially to the acquisition and interpretation of data and the critical review of the article to its finalized version. The current and prior Medtronic employee authors (S.W.L., Z.D., F.N., J.S., R.A.M.J., A.R., B.G., M.V., T.L.C., J.M., A.S.R., and R.A.V.) provided substantial contribution to the study design, data analyses and/or interpretation of data, and critically reviewed the article to its finalized version.
Author Disclosure Statement
C.P. has no competing financial interests to disclose. D.I.S. serves as an advisory board member for Medtronic. G.P.F. has conducted research supported by Medtronic, Dexcom, Abbott Diabetes Care, Tandem Diabetes Care, Insulet Corporation, Beta Bionics, and Lilly, and has served as a speaker/consultant/advisory board member for Medtronic, Dexcom, Abbott Diabetes Care, Tandem Diabetes Care, Insulet Corporation, Beta Bionics, and Lilly. K.B.K. is an employee of MannKind Corporation and has received research support from Medtronic. J.L.S. serves or has served on advisory panels for Bigfoot Biomedical, Cecelia Health, Insulet Corporation, Medtronic, StartUp Health Diabetes Moonshot, and Vertex Pharmaceuticals; has served as a consultant to Abbott Diabetes Care, Bigfoot Biomedical, Insulet Corporation, Medtronic, and Zealand Pharma. The Yale School of Medicine has received research support for J.L.S. from Abbott Diabetes Care, JAEB Center for Health Research, the JDRF, Insulet Corporation, Medtronic, the National Institutes of Health and ProventionBio.
J.R.T. has served on the advisory board and as a data safety monitoring board member to Medtronic and provided editorial support to Boehringer Ingelheim. J.R.T. is President of the Arkansas Diabetes and Endocrinology Center and Medical Investigations, Inc. and has received research support and speaking fees from Medtronic and Lilly, research support from Novo Nordisk and Inversago Pharma, and speaking fees from Bayer. B.A.B. has served as an advisory board member for Medtronic, Novo Nordisk, and Lilly, and conducted studies sponsored by Medtronic, Tandem Diabetes Care, Insulet Corporation, the JDRF, and National Institutes of Health.
M.S.K. has served as an advisory board member to Quest Diagnostics and Corcept Therapeutics and received research support from Abbott Diabetes Care, Aeterna Zentaris, Allergan Sales, LLC., Amgen, Ascendis Pharma, AstraZeneca, 89Bio, Inc., Biolinq, Inc., Corcept Therapeutics, Dexcom, Lily, Giliad Sciences, Inc., Inventiva BioPharma, Ionis Pharmaceuticals, Insulet Corporation, Kowa Research Institute, Inc., Lumos Pharma, Mannkind Corporation, Medtronic, Metacrine, Inc., NGM Biopharmaceuticals, Pfizer, Reata Pharmaceuticals, Regenacy Pharmaceuticals, Inc., Sagimet Biosciences, Senseonics, Inc., Tandem Diabetes Care, Tolerion, Inc., Vertex Pharmaceuticals, Inc., Zydus Discovery, and Zydus Therapeutics, Inc.
B.W.B. has served as speaker for Abbott Diabetes Care, Insulet Corporation, Lilly, Boehringer Ingelheim, Medtronic, Mannkind Corporation, Novo Nordisk, ProventionBio, Sanofi and Xeris Pharmaceuticals, Inc.; received grant or research support from Abvance Therapeutics, Abbott Diabetes Care, Dexcom, Diasome, Dompé, Insulet Corporation, Lilly, Mannkind Corporation, Medtronic, Novo Nordisk, ProventionBio, and Senseonics; and served as a consultant for Abvance Therapeutics, Ascensia Diabetes Care, Medtronic, Mannkind Corporation, and Novo Nordisk. A.L.C. has served as a consultant for Novo Nordisk and Mannkind Corporation and received research support from Medtronic, Novo Nordisk, Lilly, Dexcom, Insulet Corporation, and Sanofi and Abbott Diabetes Care. K.L. has no competing finances to disclose. D.R.L. has conducted research supported by Abbott Diabetes Care, Dexcom, Biolinq Inc., Tandem Diabetes Care, Sanvita, Provention, Senseonics, and TrialNet. S.W.L., Z.D., F.N., J.S., R.A.M.J., A.R., B.G., M.V., T.L.C., J.M., A.S.R., and R.A.V. are or were employees of Medtronic.
Funding Information
Medtronic funded this study.
Supplementary Material
Supplementary Data
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
