Abstract
Background:
While evidence supports glycemic control benefits for individuals with type 1 diabetes mellitus (T1DM) using hybrid closed-loop (HCL) systems, HCL automated insulin delivery therapy in China has not been assessed. This study evaluated safety events and effectiveness during HCL system use by Chinese adolescents and adults with T1DM.
Methods:
Sixty-two participants (n = 12 adolescents with a mean ± standard deviation [SD] of 15.5 ± 1.1 years and n = 50 adults [mean ± SD of 37.6 ± 11.1 years]) with T1DM and baseline A1C of 7.1% ± 1.0% underwent a run-in period (∼2 weeks) using open-loop Manual Mode (sensor-augmented pump) insulin delivery with the MiniMed™ 770G system with the Guardian™ Sensor (3) glucose sensor, followed by a study period (4 weeks) with HCL Auto Mode enabled. Analyses compared continuous glucose monitoring data and insulin delivered during the run-in versus study period (Wilcoxon signed-rank test or t-test). Safety events included rates of severe hypoglycemia and diabetic ketoacidosis (DKA).
Results:
Compared to baseline run-in, overall Auto Mode use increased time in range (TIR, 70–180 mg/dL) from 75.3% to 80.9% (P < 0.001) and reduced time below range (TBR, <70 mg/dL) from 4.7% to 2.2% (P < 0.001). Subgroup analysis demonstrated that participants (n = 29) with baseline A1C <7.0% had TBR that reduced from 5.6% to 2.0%, while participants (n = 21) with baseline A1C ≥7.5% had time above range (TAR, >180 mg/dL) that reduced from 31.6% to 20.8%. Auto Mode use also increased the percentage achieving combined recommendations for time at sensor glucose ranges (i.e., TIR of >70%, TBR of <4% and TAR of <25%) from 24.2% at baseline to 77.4% at study end. Total daily insulin dose reduced from 42.8 ± 19.8 to 40.7 ± 18.9 U (P = 0.013). There were no severe hypoglycemic, DKA, or serious adverse events.
Conclusions:
Chinese adolescents and adults, some of whom met target A1C at baseline, safely achieved significantly improved glycemia with 1 month of MiniMed 770G system use when compared to open-loop insulin delivery.
Introduction
Since the Diabetes Control and Complications Trial, 1,2 intensive insulin therapy that optimizes glycemic control, as measured by A1C, has served as a primary objective for reducing the risk of acute to long-term type 1 diabetes mellitus (T1DM)-related complications (e.g., hyperglycemia, diabetic ketoacidosis [DKA], cardiovascular disease, nephropathy, and retinopathy). Over time, intensive insulin therapy has included multiple daily injections (MDI) (i.e., ≥3 injections/day of basal and prandial insulin) or continuous subcutaneous insulin infusion (CSII). More recently, recommended goals 3,4 for time spent in target range (TIR, 70–180 mg/dL) and time spent below level 1 hypoglycemia (TBR, <70 mg/dL) 5 have evolved to become important clinical outcomes, as studies report or validate their relationship with A1C 6,7 and severe hypoglycemia risk 8 or T1DM-related complications. 6,9 Today, the assessment of individual achievements toward the recommended target A1C of <7.0%, 10 –12 and recommended TIR of >70% and TBR <70 mg/dL of <4% 3,4 provide the bases for managing T1DM.
Although T1DM management has included continuous glucose monitoring (CGM) use with either MDI or CSII therapy, 13 –15 many individuals still fall short of the aforementioned glycemic target goals. This is evidenced by the US T1D Exchange, 16 Swedish National Diabetes Register, 17 German/Austrian Prospective Diabetes Follow-up Registry, 18 and Guangdong T1DM Translational Medicine Study 19 that reports average rates of only 20%–56% achieving A1C targets. Thus, further improvement is needed.
The MiniMed™ 670G hybrid closed-loop (HCL) system (Medtronic, Northridge, CA, USA) integrates CSII, CGM, and a proportional integrated derivative algorithm with insulin feedback that automatically adjusts basal insulin delivery every 5 min. This therapy has been available in several countries including the United States, Canada, Europe, and Australia for the last few years. Although early single-center system use reported challenges (e.g., frequent Auto Mode exits and calibration requests) that were associated with discontinued use of the MiniMed 670G system by youth with T1DM, 20 –22 previous observational studies and randomized controlled trials (RCTs) that included adolescents and adults with T1DM have shown that the MiniMed 670G HCL system can increase TIR, reduce TAR without increasing the risk of hypoglycemia, and improve overall glycemic control (i.e., A1C or glucose management indicator). 23 –27 Analyses of real-world MiniMed 670G system use across multiple countries have supported these findings. 28,29 A more recent meta-analysis of several HCL system RCTs (with either a MDI, CSII or SAP comparator) provides further confirmation of the safety and effectiveness of HCL therapy for T1DM. 30
Although there is ample clinical and real-world evidence supporting MiniMed 670G HCL system benefits, 24 –29,31,32 the therapy has not been assessed in individuals from mainland China, and there is no HCL system therapy currently approved in China. The reason for investigating MiniMed 770G system outcomes in China is a requirement of the China Food and Drug Administration (CFDA) for device registration because HCL trials have been conducted primarily in Caucasian subjects. The CFDA wants proof that the outcomes seen in Chinese subjects are similar to those seen in Caucasian subjects. As such, this study is the first to evaluate safety events and performance during use of the MiniMed 770G system (i.e., the MiniMed 670G system with Bluetooth™ [Bluetooth SIG, Inc.] functionality) by Chinese adolescents and adults with T1DM.
Methods
This prospective, single-arm, multicenter, nonrandomized trial, conducted from October 31, 2020, to June 22, 2021, at four investigational centers throughout China, enrolled individuals aged 14–75 years. Investigators and investigational staff were trained to the protocol and ethics committee approvals were obtained before study start. Informed consent was obtained and documented per the specifications of the China National Medical Products Administration (NMPA) and in accordance with Order No. 25. All research efforts complied with the principles of the Declaration of Helsinki.
Criteria for study participation included T1DM diagnosis for 2 years or greater; an A1C <10%, as processed by an investigational center lab or contracted local lab; a minimum daily insulin requirement (total daily dose [TDD]) of ≥8 U; and use of insulin pump therapy greater than 6 months before screening. Study exclusion criteria included a history of hospitalization or visit to the emergency room resulting in a primary diagnosis of uncontrolled diabetes within 6 months before screening; an episode of DKA within 6 months before screening; and a history of two or more episodes of severe hypoglycemia that resulted in a coma, seizures, or required medical assistance within 6 months before screening.
Study devices
After consent was obtained, all study participants underwent device training on the MiniMed 770G HCL system (MiniMed 770G insulin pump [MMT-1883] with the Guardian™ Sensor [3] glucose sensor and Guardian™ Link [3] Transmitter), before study start. They were instructed to check their blood glucose (BG) using the Accu-Chek® Guide Link BG meter (Roche Diabetes Care, Inc., Indianapolis, IN, USA) if they were experiencing severe hypoglycemia, severe hyperglycemia, DKA, or received a low sensor glucose (SG) alert on the pump. If the BG meter reading was >300 mg/dL, participants were also instructed to check blood ketones using a FreeStyle Optium Neo ketone meter (Abbott Diabetes Care, Alameda, CA, USA).
There was a baseline run-in period (∼2 weeks) of open-loop Manual Mode treatment (i.e., MiniMed 770G SAP function without predictive low glucose management) followed by a study period (4 weeks) with Auto Mode turned on. During the study period, participants were instructed to upload pump system data to CareLink™ Clinical software daily for the first week of Auto Mode activation, then at least weekly thereafter. The HCL basal rate target was factory set at 120 mg/dL, with the correction target set at 150 mg/dL. The temporary target setting (150 mg/dL) could be used when a participant exercised. Fixed alerts included (1) SG <50 mg/dL, (2) SG >300 mg/dL for 1 h, and (3) SG >250 mg/dL for 3 h. High and low SG alert limits were recommended to be set at 300 and 70 mg/dL, respectively.
The study staff set the open-loop Manual Mode active insulin time, carbohydrate-to-insulin ratio(s), basal rates, glucose targets, and sensitivity factors. Although only active insulin time and carbohydrate-to-insulin ratio settings were carried over when Auto Mode was activated, both could be adjusted throughout the study. During Auto Mode, user-initiated insulin boluses included carbohydrate announcements or correction boluses.
Visit schedule and procedures
The trial comprised a total of 12 visits, 3 of which were during the run-in period and 9 during the study period. Informed consent/assent was obtained at Visit 1. Eligibility criteria confirmation and study device training occurred at Visit 2. Participants who did not use insulin pump therapy with CGM before screening were instructed to start the run-in period with CGM during Visit 3. All participants began the study period with Auto Mode enabled at Visit 4. Follow-up visits 5 to 9 (office or telephone) comprised pump system uploads and pump setting adjustments as needed. Follow-up visits 10 and 11 (office) comprised pump system upload, review of CareLink Clinical Reports, and inquiry about whether there were any adverse events experienced.
Safety endpoints
Safety events were collected from enrollment through the end of the study period. Investigators were instructed to monitor participants throughout the course of the study for the occurrence of any adverse event. These were presented as descriptive summary and included the incidence rate of serious adverse events (SAEs), serious adverse device effects (SADEs), unanticipated adverse device effects (UADEs), severe hypoglycemia, severe hyperglycemia, and DKA. Severe hypoglycemia was defined as an event requiring the assistance of another individual to actively administer carbohydrate, glucagon, or other resuscitative actions due to altered consciousness. Severe hyperglycemia was defined as hyperglycemia (BG >300 mg/dL) with blood ketones >1.5 mmol/L, moderate or large urine ketones, or symptoms of nausea, vomiting or abdominal pain. DKA was defined as a BG meter reading >250 mg/dL, arterial pH <7.3, bicarbonate <15 mEq/L, and moderate ketonuria or ketonemia requiring treatment in a medical facility.
Glycemic outcomes and analyses
The primary effectiveness endpoint was the mean change in percentage of TIR (70–180 mg/dL) between the run-in period and study period, which was analyzed with simple superiority test (Wilcoxon signed-rank test or paired t-test). The secondary effectiveness endpoints included percentage of TBR <54, <60, and <70 mg/dL; percentage of TAR >180, >250, and >350 mg/dL; standard deviation (SD) of SG; coefficient of variation (CV) of SG; TDD and weight, from baseline to end of study. For the secondary effectiveness endpoints, endpoint data were averaged per participant and compared between the run-in and study periods (Wilcoxon signed-rank test or paired t-test). The TDD and weight were analyzed by comparing the averaged baseline run-in period values to the end of study values (average of the 2 weeks before study exit). An exploratory analysis included summary of the aforementioned glycemic metrics stratified by baseline A1C ranges (i.e., ≤6.0%, >6.0% to <7.0%, 7.0% to <7.5%, 7.5% to 8.0%, >8.0%).
The percentage of sensor use was calculated using the total number of sensor data points available divided by the total number of expected sensor data points based on the number of days for each subject that participated in the study period. Auto Mode usage during the study period was calculated as the total time each participant spent in Auto Mode divided by the total time that each participated in the study period. Analyses of the distribution of participants achieving a TIR of >70%, in addition to other recommended goals for time at SG ranges (i.e., TBR <54 mg/dL of <1%, TBR <70 mg/dL of <4%, and TAR >180 mg/dL of <25%) 3,4 underwent descriptive analysis.
Sample size estimation was based on previously published data, 24,31 where the estimated difference of TIR from run-in period to study period was expected to be 5.3%. A sample size of 50 would yield >80% power to detect simple superiority using a one-sided t-test with a SD of 13.0% at the significance level (alpha) of 0.025. Therefore, ∼75 enrolled participants were estimated to ensure that at least 50 completed the study.
Results
Participant disposition
The study enrolled 74 individuals aged 14–75 years at four investigational centers (hospitals) in China. A total of 65 participants entered the run-in period, 62 participants (the intention to treat [ITT] population) entered the study phase, and 60 completed the study (Fig. 1). The ITT population, whose demographics are provided in Table 1, included 12 adolescents (aged 14–17 years) and 50 adults (aged ≥18 years).
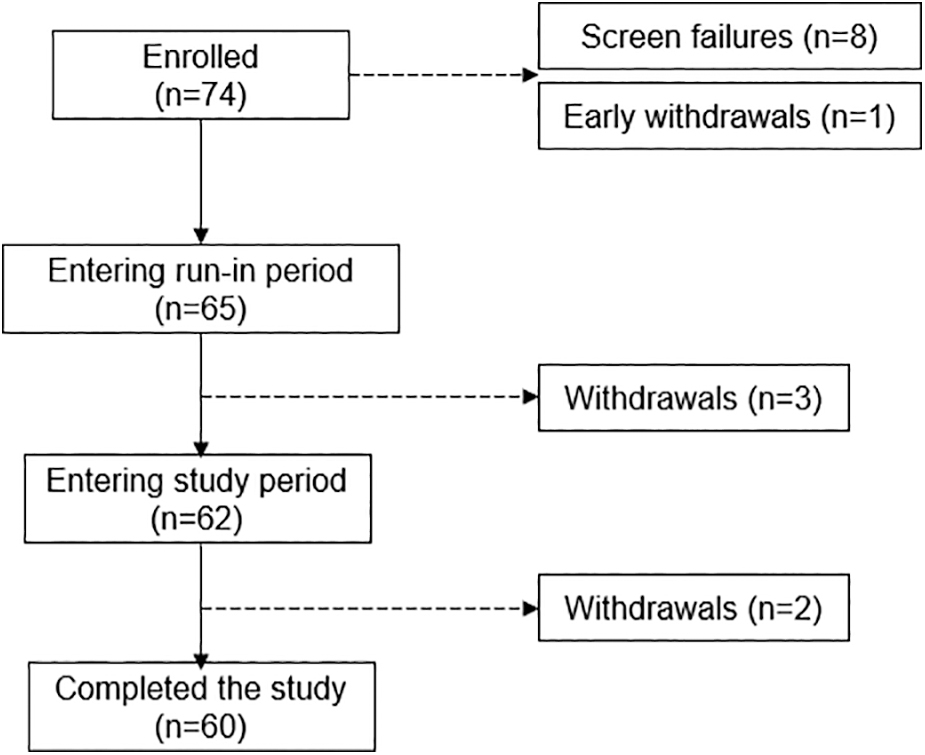
Participant disposition. The number of participants enrolled was n = 74 and the ITT population included n = 62 participants (12 adolescents [14–17 years] and 50 adults [≥18 years]) entering the study period. ITT, intention to treat.
Baseline Characteristics of the Intention to Treat Study Population
ITT, intention to treat; BMI, body mass index; SD, standard deviation.
Safety endpoints
Throughout the study, there were no reported SAEs, SADEs, UADEs, DKA, or severe hypoglycemic events. There were also no reported device deficiencies. There were three severe hyperglycemic events, two of which were related to a study device. One event occurred in an adult during the run-in period and was due to the infusion set cannula becoming disconnected from the participant. The other event occurred in an adult during the study period and was due to a possible infusion set occlusion.
Glycemic outcomes
Participants spent a mean of 93.5% of their time in Auto Mode and used CGM for a mean of 94.1% of the time. Table 2 shows overall group glycemic outcomes, insulin delivered, and weight during the run-in period and Auto Mode-enabled study period. A clinically significant increase in mean TIR to 80.9% (Δ5.6%, P < 0.001, Wilcoxon signed-rank test) and a clinically significant decrease in mean TBR <70 mg/dL to 2.2% (Δ −2.5%, P < 0.001) were observed, with Auto Mode. There were also statistically significant reductions in SD of SG (−4.9 mg/dL), CV of SG (−3.7%) and TDD (−2.1 U), with reduced TDD and no change in weight, when compared with the run-in period (Table 2). The proportion of the overall group achieving TIR of >70% increased from a high 74% (n = 46) at baseline to 95% (n = 59) at end of study, while the proportion meeting all recommended goals for time spent at SG ranges increased from 24.2% (n = 15) at baseline to 77.4% (n = 48).
Glycemic Outcomes, Insulin Delivered, and Weight During the Run-In and Study Period
Data are shown as mean ± SD (median, range). Change is from baseline to end of study.
Wilcoxon signed-rank test.
Weight was not captured at the end of study, for two participants.
CV, coefficient of variation; SD, standard deviation; SG, sensor glucose; TDD, total daily insulin dose.
The glycemic metrics and percentage of time spent at different SG ranges during the run-in and study periods were also stratified by baseline A1C ranges (≤6.0%, >6.0% to <7.0%, 7.0% to <7.5%, 7.5% to 8.0% and >8.0%) (Fig. 2). In participants (n = 13) with an A1C ≤6.0% at baseline, a nominal increase in TIR was observed, although the mean TIR was 87% by the end of the study. However, their time spent below 54 and 70 mg/dL was reduced by −0.5% ± 0.6% and −3.5% ± 3.0%, respectively. In contrast and in comparison, to baseline, the greatest change in TIR (16.1% ± 13.5%) was observed in participants (n = 10) with a baseline A1C >8.0% (Fig. 2).
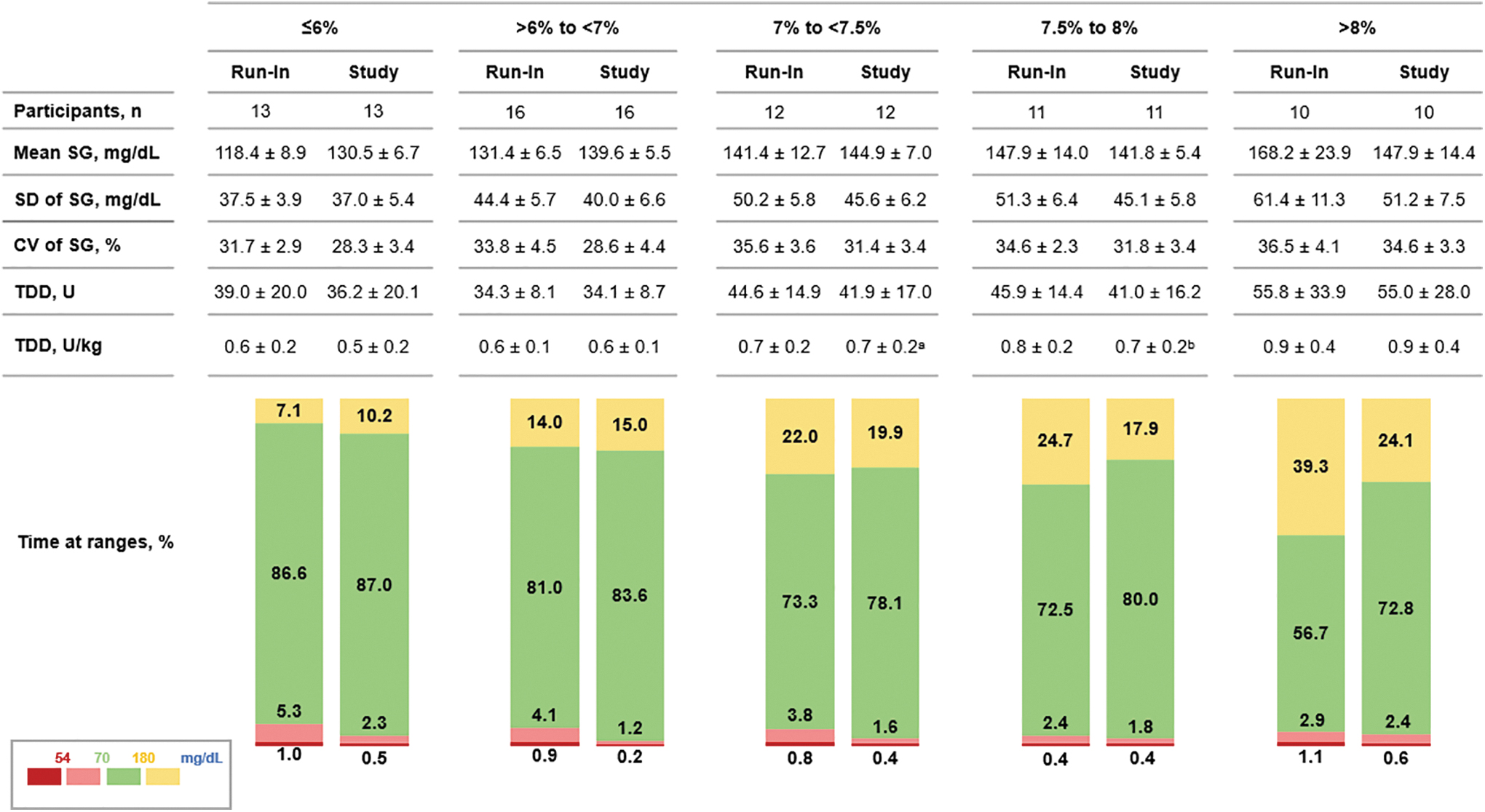
Glycemic outcomes and insulin delivered during the run-in and study period stratified by baseline A1C. Mean SG, glucose variability, insulin delivered, and percentage of time spent at SG ranges are displayed and stratified by baseline A1C. Data are shown as mean ± SD or mean. a n = 11; b n = 10. CV, coefficient of variation; SD, standard deviation; SG, sensor glucose.
The median and interquartile range (IQR) of SG values throughout the 24-h day of the run-in and study periods are shown in Figure 3. The IQR of SG for the Auto Mode-enabled study period appeared narrower than that during the run-in period, and even more so throughout the overnight and early morning hours. Figure 3 also shows three distinguishable increases in SG for both the run-in and study periods. One increase in SG was seen at 10:00 AM, where the median of SG for the run-in period was 142.4 mg/dL and the median of SG for the study period was 153.2 mg/dL. A second increase occurred at ∼4:00 PM, where the medians for the periods were 142.4 and 155.5 mg/dL, respectively. A third increase in median of SG was seen after ∼10:00 PM and was 142.7 mg/dL for the run-in period and 149.7 mg/dL for the study period. Despite these instances of increased SG, the mean ± SD (median, range) of SG levels during both the run-in and study periods were only 139.5 ± 20.8 mg/dL (135.8 mg/dL, 107.1–204.3 mg/dL) and 140.5 ± 9.7 mg/dL (139.8 mg/dL, 121.6–176.8 mg/dL), respectively.
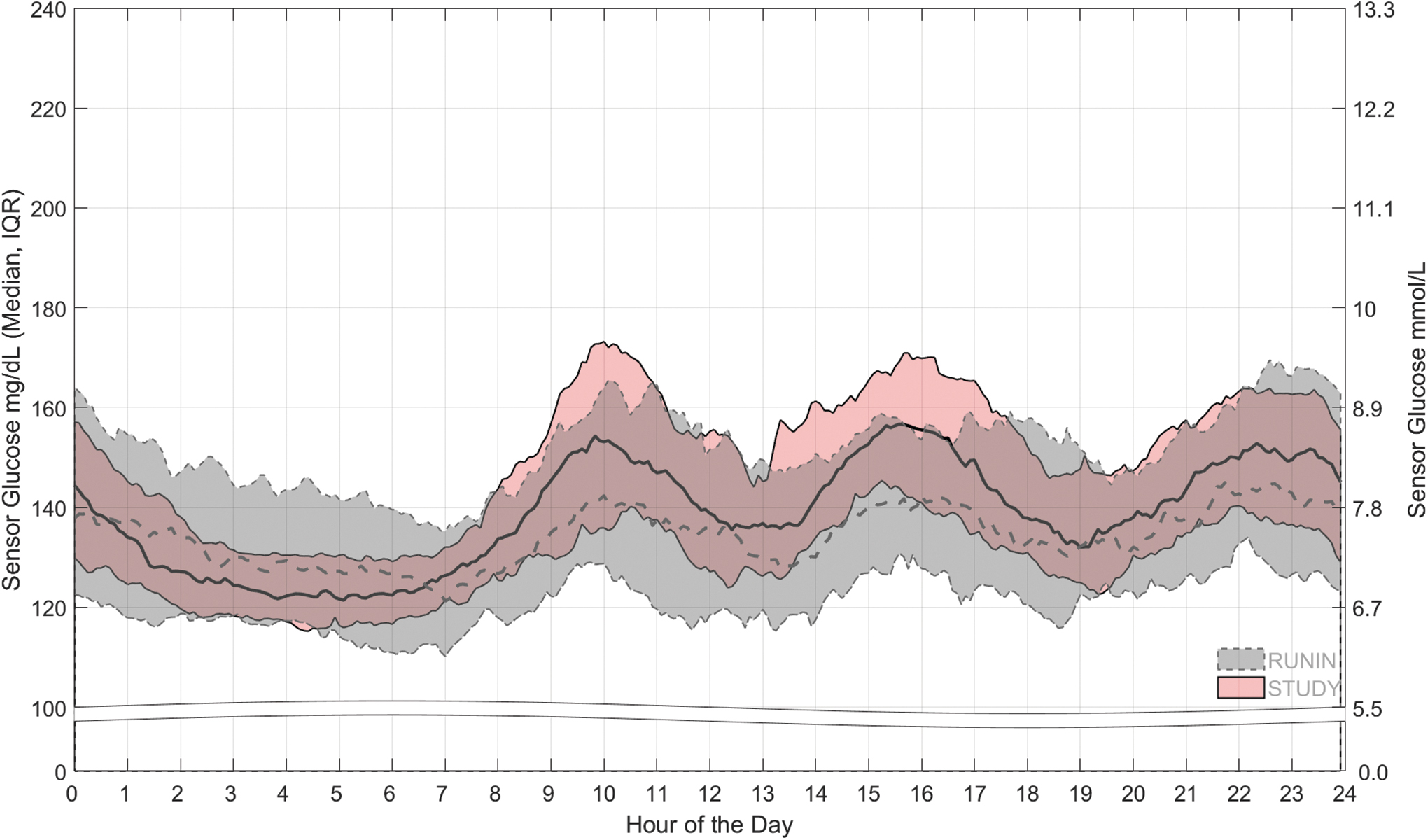
The 24-h day SG during the run-in and study periods. The median and IQR of SG over a 24-h day during the baseline open-loop run-in period (dashed line and gray band) and Auto Mode-enabled study period (solid line and pink band) are shown. IQR, interquartile range.
Discussion
In this study, there were no DKA or severe hypoglycemic events during MiniMed 770G system use in Chinese adolescents and adults with T1DM, some of whom had relatively well-controlled glycemia at study start. Auto Mode use in the overall group increased TIR from 75.3% to 80.9% (a mean [median, range] change of 5.6% [3.6%, −7.1% to 37.8%]), with significant reductions in all hypoglycemic ranges, demonstrating consistent efficacy. Although the mean change in TIR was comparable with that observed in the combined adult/adolescent population of the predicate HCL device pivotal trial, 24 it is impressive that participants in the current study managed to increase overall TIR from an already high 75.3% at baseline to 80.9%, while decreasing TBR. This overall improvement in TIR (5.6%) is equivalent to an ∼0.45% reduction in A1C. 7
The extent of increased TIR and reduced TBR were easily discerned when glycemic metrics of participants were shown across baseline A1C ranges. Participants (n = 29) who had a target A1C (i.e., <7.0%) at baseline spent more time in hypoglycemia during run-in compared with that recommended by international consensus 3,4 and the American Diabetes Association. 10,11 However, Auto Mode significantly reduced TBR and further increased mean TIR (>80%) in this group. In marked contrast, individuals (n = 10) with the greatest baseline A1C (>8.0%) and highest TAR achieved the most increased TIR and reduced SG and glucose variability, with Auto Mode. These observed glycemic benefits align with those reported during the system pivotal trials in adolescents and adults, outside of China. 24,33,34
A recent Type 1 Diabetes Index modeling project has shed light on the global burden of T1DM. 35 It reports that China is one of the top 10 countries with the highest T1DM prevalence and that an estimated 32 years are lost to morbidity, disability, or early death due to T1DM. These findings are highly relevant as the T1DM rate in China (based on per 100,000 person-years) was shown to significantly increase from 2.72 (95% confidence interval [CI]: 2.51–2.93) in 2007 to 3.60 (95% CI: 3.38–3.78) in 2017 (P < 0.001). 36 The increased incidence was most significant in those ≥30 years of age (1.87–3.52, P < 0.05). While the T1DM incidence rate for those 15–29 years of age was 3.64 (95% CI: 3.03–4.37) in 2017, new cases are estimated to be 7.4% for this group and 73.8% for those ≥30 years of age by 2027. 36 Given these trending and notable increases in T1DM in China, it was important to assess the impact of HCL therapy, already shown to benefit glycemic outcomes of individuals with T1DM outside of China. 24 –29,31,33,34
An interesting finding in this study was the 24-h day tracing of median and IQR of SG during the run-in and study periods that displayed three elevations (likely, post-prandial increases in SG) in the morning, afternoon, and night. Throughout the day, the IQR of SG during Auto Mode appeared tighter compared with the open-loop IQR of SG, yet the elevations in SG at ∼10:00 AM and 4:00 PM were more apparent for the study period. This could indicate a difference in insulin-to-carb ratio or timing of carbohydrate announcement pre- versus post-Auto Mode initiation by some participants. Nevertheless, this study population had SG levels of only 139.5 ± 20.8 mg/dL during the run-in period and only 140.5 ± 9.7 mg/dL during the study period that averaged well within international consensus and American Diabetes Association (ADA) standards of medicine recommendations for adults (<154 mg/dL) 4 and adolescents (126–180 mg/dL). 10,11 In addition, a remarkable 95% versus 74% (at baseline) achieved TIR of >70%, with Auto Mode. Further, Auto Mode allowed 89% versus 52% at baseline to achieve a TBR of <4%.
Maintaining blood glucose levels within the normal or near-normal range and minimizing variations or excursions is important for long-term outcomes with T1DM. While glycated hemoglobin level has been associated with the risk of developing long-term complications from diabetes, 37 –39 publications evaluating the relationship between TIR and diabetes complications have increased. 40 While data from RCTs are limited, findings from observational or retrospective studies, including Chinese individuals with insulin-requiring type 2 diabetes, demonstrated a lower prevalence of retinopathy, 41 diabetic peripheral neuropathy, 42 and abnormal carotid intima-media thickness with high TIR.
Similarly important is that increased glucose variability (i.e., CV of SG) and greater time spent below normal range have been shown to increase the risk of hypoglycemia 41 and future severe hypoglycemic events, 42 respectively. Thus, continued HCL system use by Chinese participants in this study who achieved higher TIR and lower TBR, alongside reduced CV of SG, may help them experience fewer hypoglycemic events and a reduced risk of long-term diabetes-related complications.
The strengths of this study are that safety events and effectiveness of the MiniMed 770G HCL system were evaluated, for the first time, in a sample of Chinese individuals with T1DM. In addition, the sample included both adolescents and adults, with an average diabetes duration of 15.0 years. Regarding limitations, the study had a single-arm and nonrandomized design and was not powered for a separate analysis of the adolescent and adult participant outcomes. While inclusion criteria were open to all majority and minority groups, this study was not powered to investigate potential difference in outcomes among the various ethnic minorities. It is also important to note that inclusion of multiple ethnic minorities was not required by CFDA for registration and use of the device. Another limitation is that the 1-month study period duration limits generalization of findings to a greater population of individuals with T1D and longer HCL system use. Nevertheless, results show the early benefits of the HCL system on glycemic outcomes irrespective of baseline glycemic control (i.e., A1C ≤6.0% to A1C >8.0%). Additional MiniMed 770G system studies may provide more insight on participant insulin dosing behaviors after Auto Mode initiation or factors that underlie improved time spent at recommended SG ranges.
Conclusions
In summary, this study confirmed that 1-month use of Auto Mode was associated with improved glycemia in Chinese adolescents and adults with T1DM. This is the first study during which >95% of participants managed to achieve a mean TIR >70%, which help confirm versatility of this therapy. Nevertheless, long-term follow-up of MiniMed 770G system use in Chinese individuals with T1DM will be important to confirm the sustainability of these outcomes.
Footnotes
Acknowledgments
The authors gratefully thank the research staff and study participants for their time and dedication to the clinical trial in addition to the contribution and support of Thomas Troub (Medtronic).
Authors' Contributions
All principal investigator authors contributed substantially to the acquisition and interpretation of data, critical review of the article, and agreed to its finalized and submitted version. All Medtronic authors contributed substantially to the study design development, data analyses, and/or interpretation of data, and critical review of the article to its finalized and submitted version.
Author Disclosure Statement
The investigator authors received research support from Medtronic to conduct the study. J.H., X.Z., X.C., Y.T., S.W.L., J.S., A.S.R., and R.A.V. are or were Medtronic employees during development of this work. Toni L. Cordero, PhD, a Medtronic employee, helped with manuscript development and submission.
Funding Information
This work was funded by Medtronic Diabetes.
