Abstract
Objective:
We aimed to assess whether percentage of time spent in hypoglycemia during closed-loop insulin delivery differs by age group and time of day.
Methods:
We retrospectively analyzed data from hybrid closed-loop studies involving young children (2–7 years), children and adolescents (8–18 years), adults (19–59 years), and older adults (≥60 years) with type 1 diabetes. Main outcome was time spent in hypoglycemia <3.9 mmol/L (<70 mg/dL). Eight weeks of data for 88 participants were analyzed.
Results:
Median time spent in hypoglycemia over the 24-h period was highest in children and adolescents (4.4% [interquartile range 2.4–5.0]) and very young children (4.0% [3.4–5.2]), followed by adults (2.7% [1.7–4.0]), and older adults (1.8% [1.2–2.2]); P < 0.001 for difference between age groups. Time spent in hypoglycemia during nighttime (midnight–05:59) was lower than during daytime (06:00–23:59) across all age groups.
Conclusion:
Time in hypoglycemia was highest in the pediatric age group during closed-loop insulin delivery. Hypoglycemia burden was lowest overnight across all age groups.
Introduction
Hypoglycemia remains an important limiting factor in achieving optimal glycemic control in persons with type 1 diabetes. 1 Closed-loop insulin delivery systems, which consist of an insulin pump, a continuous glucose monitor (CGM), and an algorithm that directs insulin delivery in response to real-time glucose levels, have been shown to improve glycemic control while not leading to an increased time spent in hypoglycemia. 2
However, despite use of closed-loop systems, hypoglycemia continues to pose a challenge in the management of type 1 diabetes. 2 To the best of our knowledge, no study has directly compared the percentage of time spent in hypoglycemia across different age groups and by time of day in closed-loop insulin therapy. In the present analysis, we assess whether percentage of time spent in hypoglycemia during closed-loop insulin delivery differs by age group and time of day.
Research Design and Methods
We retrospectively analyzed 8 weeks of data from four multicenter multinational studies (Austria, Switzerland, Germany, Luxembourg, the United Kingdom, and the United States) using hybrid closed-loop insulin delivery in persons with type 1 diabetes (trial registrations NCT04055480, NCT04025762, NCT02925299, and NCT03784027). 3 –6 Participants were divided into four age groups: young children (2–7 years), children and adolescents (8–18 years), adults (19–59 years), and older adults (≥60 years).
Inclusion criteria included type 1 diabetes diagnosis (World Health Organization criteria) for >6 months (young children and adults) 4,5 or 12 months (adolescents and older adults) 3,6 and insulin pump therapy for minimum 3 months. Inclusion criteria for glycated hemoglobin (HbA1c) at screening varied between studies: upper limit was 10% (86 mmol/mmol) in adolescents, adults, and older adults 3,4,6 and 11% (97 mmol/mol) in young children. 5 There was no lower threshold for HbA1c in all but one study (lower limit set at 7.0% [53 mmol/L] for children and adolescents). 6
Participants and/or parents/caregivers gave written informed consent. All studies received national regulatory and independent ethical approval.
Closed-loop insulin delivery
The hybrid closed-loop system comprised the CamAPS FX app (CamDiab, Cambridge, United Kingdom) residing on an unlocked Android smartphone, a CGM device (Dexcom G6; Dexcom, San Diego, CA), and an insulin pump (Dana Diabecare RS; Sooil, Seoul, South Korea). Every 8 to 12 min, the adaptive model predictive control algorithm residing on the CamAPS FX app automatically calculated the insulin infusion rate that was then communicated wirelessly to the study pump. 4 The closed-loop algorithm has a default target glucose level of 5.8 mmol/L (104 mg/dL), which is adjustable between 4.4 and 11.0 mmol/L (79 and 198 mg/dL) across different times of day and night. 3
Data analysis and statistical methods
The main outcome was time spent in hypoglycemia (sensor glucose <3.9 mmol/L per 70 mg/dL) by time of day. Daytime was defined as 06:00 to 23:59 and nighttime was defined as midnight to 05:59. To ensure equal representation across all age groups, 22 participants were included from each study. For three out of the four studies, the 22 participants were randomly selected using random sampling. For one study (among children and adolescents), only data from 22 participants using the CamAPS FX configuration were available as the other participants in the study used the FlorenceM configuration.
Outcomes were calculated using GStat software, version 2.3 (University of Cambridge, Cambridge, United Kingdom), and statistical analyses were performed using R (version 4.4.1; R Foundation for Statistical Computing, Vienna, Austria). Data are presented as mean ± standard deviation for normally distributed data or median (interquartile range [IQR]) for non-normally distributed data. To compare the difference between age groups for time spent in hypoglycemia, data were winsorized at the 10th and 90th percentile, and one-way analysis of variance with post hoc analysis using the Tukey test was performed. The reported P-values are not adjusted for multiple testing. P-values <0.05 were considered statistically significant.
Results
Eight weeks of data for 88 participants were analyzed. Table 1 gives the demographic characteristics of the study population. Mean age was 5.5 ± 1.5, 12.5 ± 2.1, 38.3 ± 9.2, and 66.8 ± 5.5 years for very young children, children and adolescents, adults, and older adults, respectively. Around 41% of very young children were females, and 54% of children and adolescents, adults, and older adults were females. Duration of diabetes ranged from 1.1 ± 1.0 among very young children to 38.3 ± 12.0 among older adults. Mean HbA1c at baseline was 7.3% ± 0.7% (56.1 ± 8.1 mmol/mol), 8.0% ± 0.9% (64.0 ± 9.9 mmol/mol), 7.3% ± 0.8% (56.7 ± 8.5 mmol/mol), and 7.7% ± 0.8% (60.3 ± 8.6 mmol/mol) for very young children, children and adolescents, adults, and older adults, respectively.
Characteristics of the Participants at Baseline by Age Group
Data are n (%) or mean (SD).
BMI percentile.
BMI, body mass index.
Glycemic outcomes per study are presented in Table 2. The median percentage of time when sensor glucose was below target glucose range (<3.9 mmol/L/70 mg/dL) over the 24-h period was highest for children and adolescents at 4.4% (IQR 2.4–5.0), followed by very young children at 4.0% (IQR 3.4–5.2), then adults (2.7%; IQR 1.7–4.0), and lowest for older adults (1.8%; IQR 1.2–2.2). The mean percentage of time when sensor glucose was in target glucose range (3.9–10.0 mmol/L per 70–180 mg/dL) was highest among older adults (78.4% ± 8.2%) and lowest among children and adolescents (67.8% ± 6.2%).
Glycemic and Insulin Outcomes per Age Group
Data are mean ± SD or median (IQR).
The distribution of the median time in hypoglycemia over the 24-h period across the four age groups is shown in Figure 1. Percentage time spent in hypoglycemia appeared to be greatest between 12:00–14:00 and 18:00–20:00 among very young children and between 12:00–14:00 and 18:00–21:00 among children and adolescents. Among adults and older adults, time spent in hypoglycemia was more evenly distributed throughout the 24-h period but tended to be higher between 12:00–14:00 and 18:00–20:00.
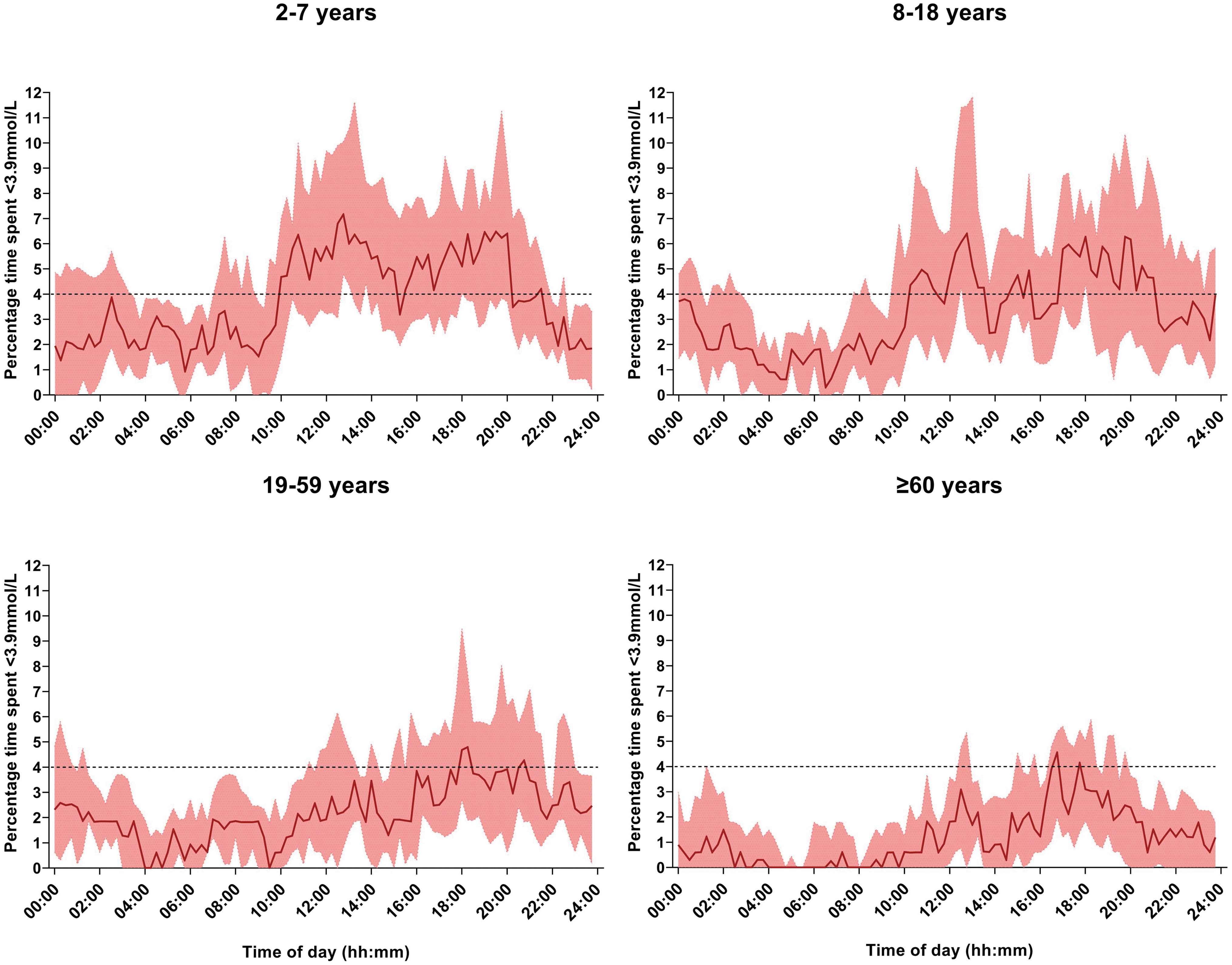
Median percentage time with sensor glucose level <3.9 mmol/L (70 mg/dL) across the four age groups for 8 weeks (dark red line). The red shaded area indicates the interquartile range. The dashed black line indicates the target percentage time spent below range <4%.
Median percentage time spent in hypoglycemia by age group and time of day (daytime versus nighttime) is shown in Figure 2. During daytime, median time spent in hypoglycemia was highest in children and adolescents (5.1%; IQR 2.8–5.8), followed by very young children (4.6%: IQR: 3.8–5.9), adults (3.1%; IQR: 1.7–4.4), and finally older adults (2.1%; IQR: 1.4–2.5); P < 0.001 for the difference between age groups. Similarly, during nighttime, median time spent in hypoglycemia was highest in very young children at 2.6% (IQR: 1.3–3.7), whereas older adults spent the least amount of time in hypoglycemia at 1.0% (IQR: 0.6–1.3); P < 0.001 for the difference between age groups.
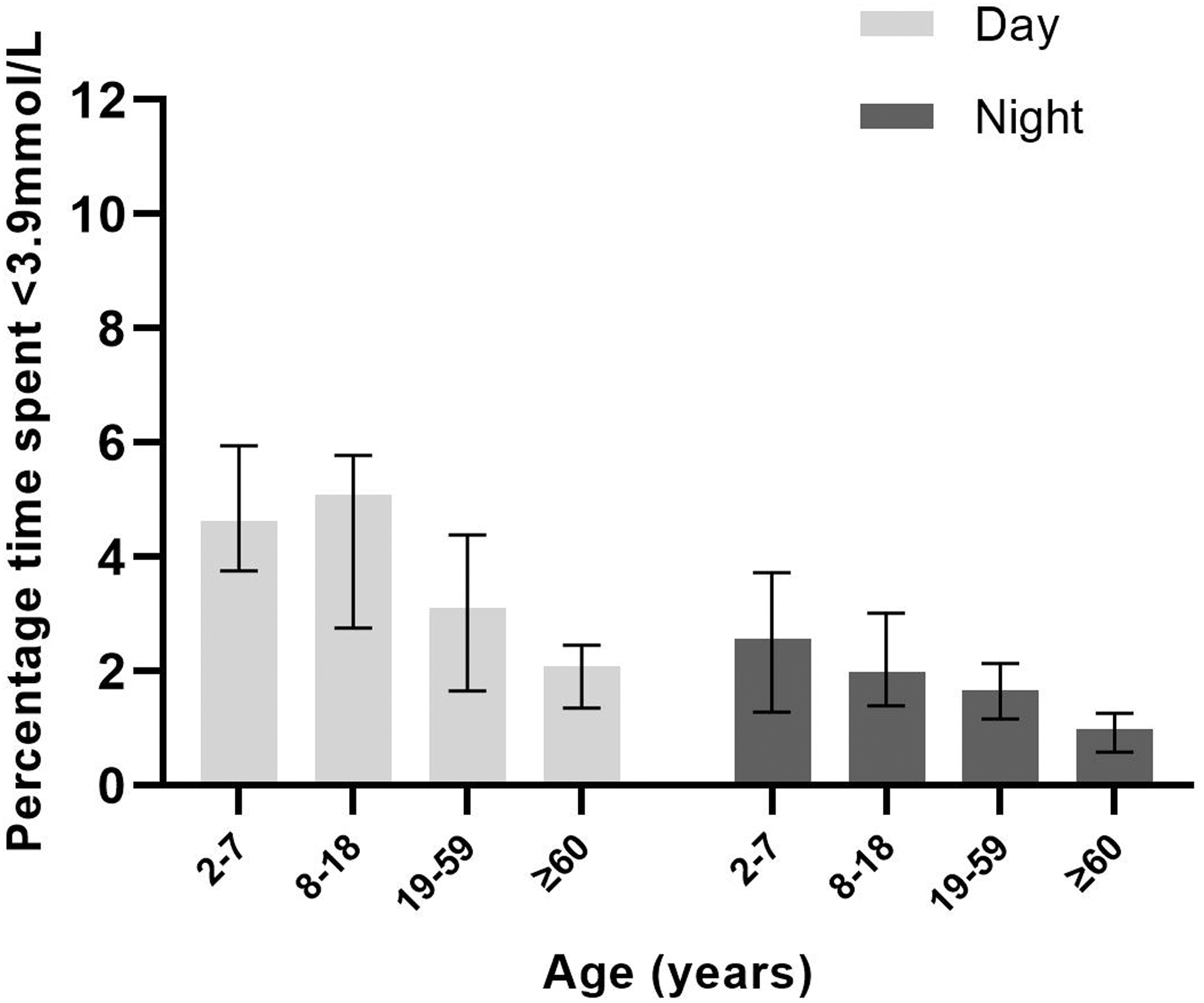
Median percentage time spent with sensor glucose level <3.9 mmol/L (<70 mg/dL) by age group and time of day. Each bar represents an age group (2–7, 8–18, 19–59, ≥60 years). Data are median (IQR). For daytime (06:00–23:59), P-values <0.01 except for 8–18 versus ≥60 years and 19–59 versus ≥60 years. For nighttime (midnight–05:59), P-values <0.01 for ≥60 years versus the other three age groups.
Discussion
In this retrospective analysis of data from four hybrid closed-loop studies across all age groups from very young children to older adults, we found that hypoglycemia burden is highest in the pediatric age groups and lowest in older adults during both daytime and nighttime.
International guidelines recommend that persons with type 1 diabetes should spend >70% of time within target glucose range 3.9–10.0 mmol/L (70–180 mg/dL) and <4% of time (or <1% of time for older adults) below target glucose range <3.9 mmol/L (70 mg/dL). 7,8 Studies have shown that the use of hybrid closed loop can improve glycemic outcomes as compared with usual care across all age groups. 9 –14 We found that adults were able to achieve these glycemic targets during both daytime and nighttime.
Older adults were within target for time below range during nighttime, and slightly above target during daytime. The pediatric age groups, however, spent a slightly longer time in hypoglycemia over the 24-h period than recommended in the guidelines. This finding is in line with previous studies that have shown that children experience hypoglycemia more frequently than older persons with type 1 diabetes 15 Factors that can increase the risk of hypoglycemic events in the pediatric age groups include increased physical activity as compared with their older counterparts and skipped meals. 15,16
We found that the pediatric age group spent more time in hypoglycemia between 12:00–14:00 and 18:00–20:00. Although information on participants' mealtimes is not available in the current analysis, the timing of these events suggests postprandial hypoglycemia. We found a similar, yet less pronounced, pattern for time spent in hypoglycemia for adults and older adults.
This finding reinforces the importance of accurate carbohydrate counting, the timely administration of bolus insulin before meals, use of extended boluses for meals high in protein/fat, as well as the use of ultrafast-acting insulin as measures to reduce the burden of hypoglycemia in persons with type 1 diabetes. 17,18 However, some causes of hypoglycemia are difficult to prevent and/or manage (e.g., skipped meals), which highlights the challenges involved in managing type 1 diabetes in these age groups despite use of closed-loop insulin systems.
Time spent in hypoglycemia was higher during daytime than during nighttime across all age groups, in line with previous studies. 10,11,19 Differences in time spent in hypoglycemia across age groups were more pronounced during daytime than during nighttime. This is unsurprising as most of the insulin delivered overnight is closed-loop-driven insulin, rather than user-driven bolus insulin 11 with safety measures to reduce the risk of hypoglycemia. Moreover, other factors that can influence glucose levels during the daytime (e.g., meals and exercise) are generally absent at night. 11
To our knowledge, this is the first study that aimed to compare the effect of closed-loop therapy on time spent in hypoglycemia by age group and time of day. Strengths of this study are, therefore, the inclusion of several age groups across the lifespan in the analyses, and the multinational unrestricted-living study design. The limitations of this study include the retrospective analysis, the relatively short follow-up period, and the minor differences in study design. Although the participants included in this analysis were randomly selected for three out of the four studies, we cannot rule out selection bias among the 8–18 years age group as only data for 22 participants who used the CamAPS FX configuration were available.
Moreover, participants in the ≥60 years age group might not be completely representative of the general population of older adults with type 1 diabetes due to the requirement for insulin pump therapy (although this was a requirement for participants in all included studies). Although older adults received the same training as participants in the other age groups, data on whether higher personal glucose targets were more widely applied in older adults were not recorded. A final limitation is that C-peptide was not measured in all studies and, therefore, not included in the present analysis.
Conclusions
In conclusion, in persons with type 1 diabetes using closed-loop therapy, hypoglycemia burden was low overall across all age groups but was higher in the pediatric age groups as compared with adults. Time in hypoglycemia was lowest during nighttime. Our results highlight the challenges involved in managing type 1 diabetes in the pediatric age groups, despite use of closed-loop insulin therapy. Strategies designed to reduce potentially avoidable causes of hypoglycemia such as accurate carbohydrate counting, and timely administration of prandial boluses, can be reinforced.
Footnotes
Acknowledgments
The authors thank study volunteers for their participation in the studies and acknowledge support by the staff at the Addenbrooke's Wellcome Trust Clinical Research Facility.
Disclaimer
The views expressed are those of the authors and not necessarily those of the funders. No sponsor had any role in the study design, data collection, data analysis, data interpretation, or writing of the article.
Authors' Contributions
H.A., C.K.B., M.N., M.E.W., J.W., and R.H. codesigned the analysis. H.A., R.L., M.E.W., J.W., and R.H. carried out or supported data analysis, including statistical analysis. HA wrote the article. M.E.W., J.M.A., C.K.B., L.B., C.d.B., R.E.J.B., F.M.C., E.F.-R., A.G., S.E.H., T.M.K., L.L., B.R.-M., H.T., A.T., P.N., and R.H. codesigned the clinical studies. R.H. designed the control algorithm.
J.W., J.M.A., C.K.B., S.H., L.B., C.d.B., R.E.J.B., F.M.C., M.L.E., E.F.-R., A.G., S.E.H., T.M.K., L.L., J.K.M., P.N., B.R.-M., H.T., A.T., L.D., and N.D. screened and enrolled participants, provided patient care, and/or took study samples. All authors critically reviewed the article. H.A. and R.H. are the guarantors of this study and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Author Disclosure Statement
R.H. reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk; receiving license fees from BBraun; patents related to closed loop; and being director at CamDiab. J.W. reports receiving speaker honoraria from Ypsomed. H.A. is a consultant at CamDiab. M.E.W. is a consultant at CamDiab and reports patents related to closed loop. C.K.B. reports receiving consultancy fees from CamDiab and speaker honoraria from Ypsomed. S.H. reports speaker and advisory board fees from Dexcom, Medtronic, Sanofi, and Ypsomed; being director at ASK Diabetes Ltd and receiving consulting/training fees from CamDiab.
L.B. reports receiving research support from Dexcom and CamDiab. R.E.J.B. reports receiving speaking honoraria from Eli Lilly and Springer Healthcare, and sitting as a voluntary unpaid member of the Novo Nordisk U.K. Foundation Research Selection committee. F.M.C. reports receiving speaker honoraria from Eli Lilly, Dexcom, Novo Nordisk and Insulet, and consultancy fees from Abbott Diabetes Care. E.F.-R. reports having received speaker honoraria from Eli Lilly and Novo Nordisk, serving on advisory boards for Eli Lilly and Sanofi. M.L.E. is a clinical triallist, has served on advisory boards, or has received speaker/writer honoraria from Medtronic, Dexcom, Abbott Diabetes Care, Roche, AstraZeneca, Novo Nordisk, Eli Lilly, Zucara, Pila Pharma, and Imcyse Pharma. The University of Cambridge has received salary support for M.L.E. through the National Health Service in the East of England through the Clinical Academic Reserve.
S.E.H. has received speaker honoraria by Eli Lilly, Vertex, Minimed Medtronic, Insulet, Ypsomed, Dexcom, and Sanofi. T.M.K. reports having received speaker honoraria from Eli Lilly and Novo. L.L. has received personal fees from Abbott Diabetes Care, Dexcom, Insulet, Medtronic, Novo Nordisk, Sanofi, and Diabetes Care. J.K.M. is a member on the advisory board of Becton-Dickinson, Eli Lilly, embecta, Medtronic, NovoNordisk, Pharmasens AG, Roche Diabetes Care, and Sanofi-Aventis, and received speaker honoraria from Abbott Diabetes Care, A. Menarini Diagnostics, Becton-Dickinson, Dexcom, Eli Lilly, Medtrust AG, Novo Nordisk, Roche Diabetes Care, Sanofi-Aventis, Servier, and Ypsomed.
H.T. reports receiving research support from Dexcom and speaker honoraria from Eli Lilly. B.R.-M. has received speaker honoraria from Abbott Diabetes Care, Eli Lilly, Medtronic, Novo Nordisk, Roche Diabetes Care, Sanofi, and A. Menarini Diagnostics, and has been on the advisory boards of Eli Lilly, Roche Diabetes Care, and Abbott Diabetes Care. CdB has received speaker honoraria from MiniMed and Medtronic and has been member of their European Psychology and e-learning Advisory Board. J.M.A. reports training fees from CamDiab. M.T. reports having received speaker honoraria from Eli Lilly, Novo Nordisk, and Medtronic and advisory board fees from Abbott Diabetes Care.
M.N. has received travel grant support from Sanofi, Janssen, and Eli Lilly and was previously chair of the Young Diabetologists' and Endocrinologists' Forum in the United Kingdom, which uses unrestricted sponsorship from industry partners to deliver educational programs for health care professionals. L.D. has received advisory board payment from Dexcom and a fee for chairing an educational session for Novo Nordisk. P.N., R.L., A.G., N.D., and A.T. have no disclosures.
Funding Information
This study was supported by the Swiss National Science Foundation (P1BEP3_200041), National Institute for Health Research EME Grant (14/23/09), JDRF, Helmsley Trust (#2016PG-T1D046), National Institute for Health Research Cambridge Biomedical Research Centre, and Horizon 2020 (grant agreement no. 731560). Dexcom supplied discounted continuous glucose monitoring systems for the studies.
