Abstract
We evaluated the use of hybrid closed-loop (HCL) insulin delivery with faster insulin aspart (Fiasp) in very young children with type 1 diabetes (T1D). In a double-blind, multicenter, randomized, crossover study, children aged 2–6 years with T1D underwent two 8-week periods of HCL using CamAPS FX with Fiasp and standard insulin aspart (IAsp), in random order. Primary endpoint was between-treatment difference in time in target range 3.9–10.0 mmol/L. We randomized 25 participants: mean (±standard deviation) age 5.1 ± 1.3 years, baseline HbA1c 55 ± 9 mmol/mol. Time in range was not significantly different between interventions (64% ± 9% vs. 65% ± 9% for HCL with Fiasp vs. IAsp; mean difference −0.33% [95% confidence interval: −2.13 to 1.47; P = 0.71]). There was no significant difference in time with glucose <3.9 mmol/L. No post-randomization severe hypoglycemia or diabetic ketoacidosis events occurred. Use of Fiasp with CamAPS FX HCL demonstrated no significant difference in glycemic outcomes compared with IAsp in very young children with T1D. Clinical trials registration: NCT04759144.
Introduction
Hybrid closed-loop therapy has been shown to improve glycemic control and quality of life in very young children with type 1 diabetes (T1D) compared to standard therapies, 1 –3 but some challenges remain due to the high glycemic variability caused by variable insulin needs and unpredictable eating and activity patterns in this age-group. 4,5 Ultrarapid-acting insulin “Fiasp” has faster onset and offset than currently used rapid-acting insulins, and has been shown to improve HbA1c compared to mealtime standard insulin aspart (IAsp) given by injection, 6 but performance of ultrarapid insulins with hybrid closed-loop (HCL) therapy has not been assessed in very young children. The adaptive CamAPS FX HCL algorithm automatically and continuously modifies active insulin time, enabling it to accommodate a variety of insulin action profiles.
We aimed to evaluate whether using faster insulin aspart (Fiasp) with the CamAPS FX HCL system could improve glucose control compared to CamAPS FX HCL system with standard rapid-acting insulin in this vulnerable and challenging population.
Research Design and Methods
Study participants
Key inclusion criteria were age 2–6 years, T1D for ≥6 months, insulin pump therapy for ≥3 months, and screening HbA1c ≤11% (97 mmol/mol). Key exclusion criteria included use of diluted insulin, and concomitant disease affecting metabolic control (Supplementary Table S1, Supplementary Appendix).
Eligible children were recruited from diabetes clinics at Addenbrooke's Hospital (Cambridge, UK), including three local Patient Identification Centers, and Alder Hey Children's Hospital (Liverpool, UK).
Study oversight
Before study commencement, approval was received from an independent Research Ethics Committee in the UK. All parents/guardians gave written informed consent. Safety aspects were overseen by an independent data safety monitoring board. Trial registration NCT04759144.
Study design and procedures
The study adopted a double-blind, multicenter, randomized, crossover design comparing 8-week use of HCL insulin delivery using Fiasp (Novo Nordisk, Bagsvaerd, Denmark) followed by 8-week use of HCL using standard IAsp (Novo Nordisk), in random order. A 2–4 week run-in period preceded randomization, during which participants used the study HCL system with their prestudy insulin.
At enrolment, blood samples were taken for local analysis of glycated hemoglobin, using an International Federation of Clinical Chemistry and Laboratory Medicine (IFCC)-aligned method and following NGSP standards. At the start of run-in, participants received training on the study insulin pump, glucose sensor, and HCL system.
Participants were randomly assigned to receive either 8 weeks of HCL with standard IAsp followed by HCL with Fiasp or vice versa. Permuted block randomization was applied. Assignment was blinded to study participants and study personnel.
Participants continued the allocated intervention without remote monitoring by study personnel. Participants were advised to bolus 15 min before eating throughout the study as per standard clinical practice, but were free to adjust this as required. A 24 h telephone helpline to contact the local study team was provided.
Closed-loop system
The HCL system comprised an unlocked smartphone (Galaxy S8, Samsung, South Korea) hosting the CamAPS FX app (CamDiab, Cambridge, UK) running the Cambridge model predictive control algorithm (version 0.3.71), which communicated wirelessly with both the Dana Diabecare RS or Dana i insulin pump (Sooil, Seoul, South Korea), and Dexcom G6 transmitter (Dexcom, San Diego, CA) (further details in Supplementary Fig. S1, Supplementary Appendix).
Study end points
The primary endpoint was the between-treatment difference in time in target glucose range 3.9–10.0 mmol/L during the study periods. Secondary endpoints included mean sensor glucose; standard deviation (SD) and coefficient of variation of glucose; time in hypo- and hyperglycemia; and insulin metrics. All glycemic endpoints were based on sensor glucose data. Secondary endpoints were calculated over the whole 8-week study periods, fortnightly and during daytime and nighttime periods.
Three validated questionnaires were administered at baseline and at the end of each study period to evaluate hypoglycemia fear, 7 diabetes distress, 8 and closed-loop treatment satisfaction. 9
Safety evaluation comprised the frequency of severe hypoglycemia and diabetic ketoacidosis events and other adverse events.
Statistical analysis
This was an exploratory analysis aiming for 24 completed participants. All analyses were carried out on an intention-to-treat basis. We analyzed endpoints from participants with a minimum of 48 h of sensor data in at least one study period. The treatment interventions were compared using a repeated measures linear mixed model adjusting for period as a fixed effect and site as a random effect and accounting for the baseline value as a separate period. A 95% confidence interval (CI) was reported for the difference between interventions and P-values <0.05 were considered significant. Non-normally distributed data were winsorized. Missing data were not imputed for the primary analysis. Outcomes were calculated using GStat software, version 2.3 (University of Cambridge, Cambridge, UK), and statistical analyses carried out using SPSS Statistics software, version 28 (IBM Software, Hampshire, UK).
Results
Between March 2021 and March 2022, 27 participants were enrolled. During run-in, one participant chose to withdraw. Another participant was withdrawn on safety grounds. Twenty-five participants were randomized and all completed the trial. Study flowchart and Consort flow diagram are shown in Supplementary Figures S2 and S3, Supplementary Appendix. Participants had a mean age of 5.1 ± 1.3 years (range 2.1–6.8 years), 68% (n = 17) were male, and the majority were of white ethnicity (n = 20, 80%) (Supplementary Table S2, Supplementary Appendix). Baseline HbA1c was 7.2% ± 0.8% (55.5 ± 8.6 mmol/mol) with time in target range 3.9–10.0 mmol/L of 63.9% ± 8.5%, total daily insulin requirements of 0.74 ± 0.14 units/kg/day and a mean duration of diabetes of 2.4 ± 1.2 years. Nineteen participants (76%) were using HCL therapy at enrolment.
Primary and secondary endpoints for all randomized participants are shown in Table 1. Time in target range 3.9–10.0 mmol/L was not significantly different between interventions (mean ± SD 64.2% ± 8.8% vs. 64.6% ± 8.8% for HCL with Fiasp vs. HCL with standard IAsp, respectively), with mean adjusted difference of −0.33% points (95% CI −2.13, 1.47; P = 0.71). Figure 1 shows 24 h sensor glucose profiles.
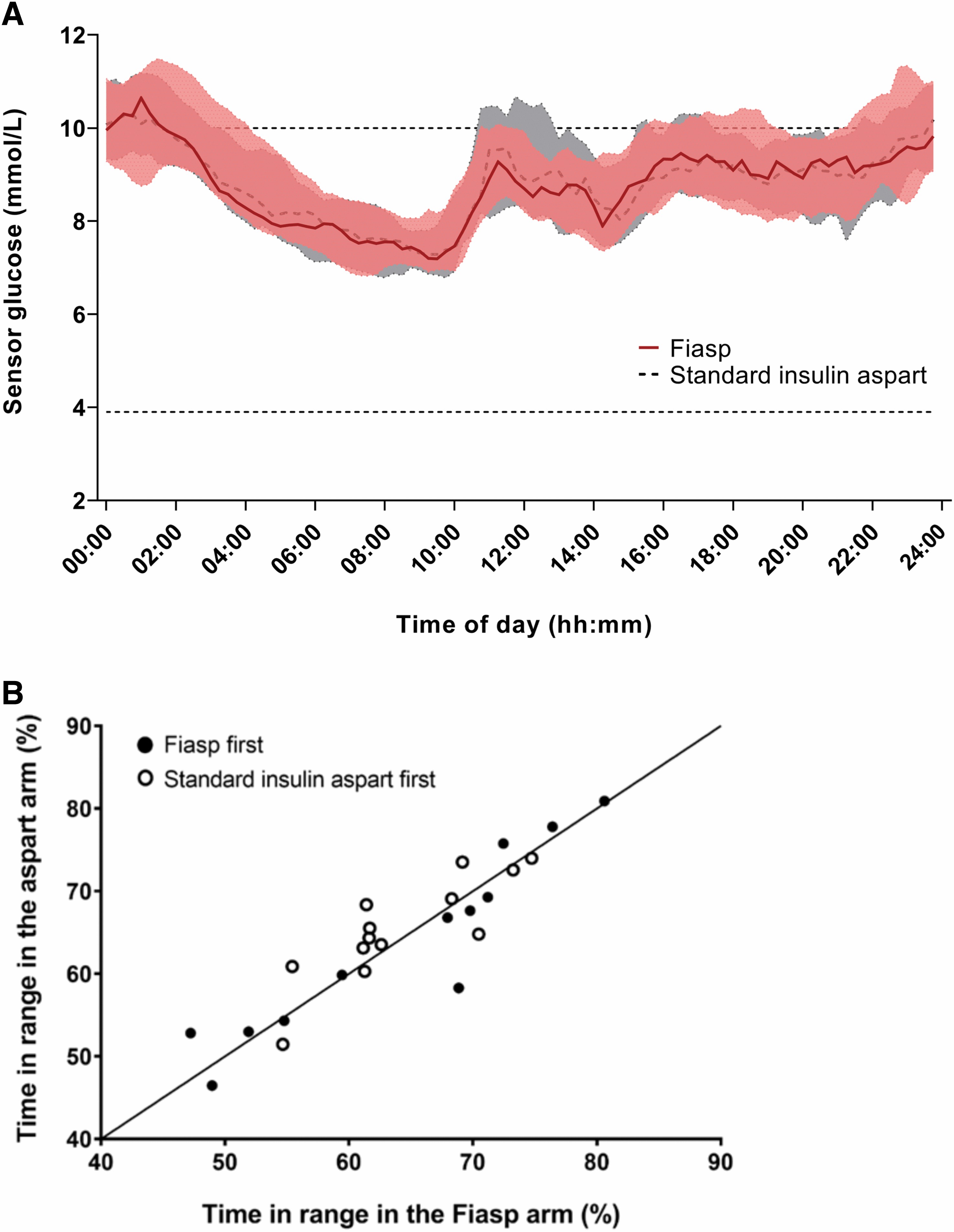
Glucose Control and Insulin Delivery Over 8 Weeks of Closed-Loop with Faster-Acting Insulin Aspart and Closed-Loop with Standard Insulin Aspart
Data presented are mean ± SD or median (Q1, Q3) throughout the 8-week study periods.
Glucose data are based on sensor glucose measurements.
Treatment difference is calculated as Fiasp minus standard insulin aspart.
Primary end point.
Based on linear mixed model adjusting for period as a fixed effect and site as a random effect and accounting for the baseline value as a separate period.
CGM, continuous glucose monitoring; CI, confidence interval; CV, coefficient of variation; SD, standard deviation.
There was no significant difference in time spent in hypoglycemia <3.9 mmol/L (median [interquartile range (IQR)] 3.5% [2.6, 6.3] Fiasp vs. 3.7% [2.6, 6.2] IAsp) or time spent in significant hyperglycemia >16.7 mmol/L between interventions (median [IQR] 4.5% [2.2, 6.6] Fiasp vs. 4.1% [1.7, 7.7] IAsp). Measures of glucose variability were not significantly different between interventions.
Total daily insulin delivery was slightly higher in the Fiasp period (mean ± SD 0.74 ± 0.12 vs. 0.72 ± 0.12 units/kg/day with IAsp; P = 0.04). The higher total daily insulin delivery in the Fiasp period was due to higher basal (i.e., automated) insulin delivery. Total daily bolus insulin was not significantly different between interventions (Table 1).
Closed-loop usage was high with median of 96.4% (IQR 91.7, 97.9) of the time with Fiasp and 96.7% (95.0, 98.0) with standard IAsp.
Sensor glucose metrics were similar during daytime and nighttime periods using Fiasp compared to standard IAsp (Supplementary Table S3, Supplementary Appendix). Time in range at fortnightly intervals over the 8-week periods remained stable (Supplementary Fig. S4, Supplementary Appendix).
Levels of diabetes distress and hypoglycemia fear were not significantly different between interventions, although there was a trend toward less hypoglycemia worry in the Fiasp period (Supplementary Table S4, Supplementary Appendix). Treatment satisfaction as measured by INSPIRE was similar between interventions.
Adverse events
One severe hypoglycemia event occurred in the run-in period. No severe hypoglycemia or diabetic ketoacidosis events occurred after randomization, one nonintervention-related serious adverse event (hospital admission for gastroenteritis) occurred in the IAsp period. Thirty other adverse events were reported (17 Fiasp period, 8 IAsp period, 5 run-in). Of these, 14 were hyperglycemia with ketosis (ketones ≥0.6 mmol/L) events, 9 in Fiasp period, 2 in IAsp period, and 3 in run-in. The nine hyperglycemia with ketosis events in the Fiasp period occurred in eight participants, and the two events in the IAsp period occurred in two participants. Four (all Fiasp period) were associated with intercurrent illness, the others were most likely secondary to cannula failure or occlusion. All events resolved at home with pump cannula change and/or pen corrections. Safety-related events are summarized in Supplementary Table S5, Supplementary Appendix.
Discussion
In the present study, we demonstrated that using Fiasp with the CamAPS FX HCL system over 8-weeks did not lead to any significant difference in glycemic control compared with using standard IAsp in very young children with T1D. In this age group, closed-loop algorithms improve glycemic control primarily by reducing time in hyperglycemia, and are able to achieve this improvement with standard IAsp when compared to sensor-augmented pump therapy. 1,2 Fiasp is only marginally faster-acting in children, 10 and this difference may not be sufficient to provide additional benefit over and above the inherent benefit of closed-loop glucose control itself, but may still confer benefit in those on standard therapies. 6
This is further reflected in our questionnaire outcomes, which were similar between interventions, and showed no significant change in diabetes distress or hypoglycemia fear with use of Fiasp. Treatment satisfaction was high at 86 (out of 100) at baseline and remained consistently high during both study periods, suggesting that Fiasp does not appear to offer additional clinical benefit over and above the benefit of HCL therapy as a whole in this age group.
Our efficacy outcomes are consistent with observations in several adult studies comparing HCL therapy using Fiasp to standard rapid-acting insulin, 11 –14 where measures of glycemic control were either clinically similar with both insulins or improvements were statistically significant, but clinically modest. 14 Two adult studies demonstrated a reduction in hypoglycemia, 11,14 an effect not observed in the present study. This may be due to a higher proportion of bolus insulin delivery (57% compared to 47% in adults) in the daytime in our cohort, limiting the closed-loop algorithm's ability to mitigate hypoglycemia during this time. In addition, inherent differences in eating behavior and insulin variability in very young children may be contributing to the observed differences in time in hypoglycemia.
We observed a higher rate of hyperglycemia with ketosis events (ketones ≥0.6 mmol/L) during treatment with Fiasp. An adult study investigating the safety of Fiasp in insulin pumps reported a higher number of unplanned infusion set changes in the Fiasp group, although unexplained hyperglycemia events were similar to standard IAsp. 15 Younger children produce ketones more readily when fasting due to inherent physiological mechanisms where there is decreased availability of gluconeogenic substrates and precursors in this age group, compared with older children and adults. 16,17 In a study comparing the effect of overnight suspension of insulin delivery using predictive low glucose suspend in young children (4–9 years) and older children (10–14 years), young children had ketones ≥0.6 mmol/L on 23% of mornings compared with 2% in older children following pump suspensions of >120 min. 18 A similar picture would also be expected to occur in other clinical scenarios such as insulin infusion set failure or intercurrent illness. Thus, any factor increasing the likelihood of infusion set failures may increase the risk of hyperglycemia with ketosis events in very young children.
Strengths of our study include the multicenter, double-blind, crossover design with each participant being their own control. There was 100% retention of randomized participants, suggesting high acceptability of closed-loop in this age group. Limitations include a relatively small sample size, and a study population with good glycemic control at baseline. All participants used hybrid closed-loop therapy during the baseline period, however, it has already been shown that closed-loop therapy improves glycemic control compared to sensor-augmented pump therapy in this age group. 1
Conclusion
The use of Fiasp with the CamAPS FX HCL system demonstrated no significant difference and clinically similar outcomes in glycemic control compared to standard IAsp in very young children with T1D, suggesting that use of Fiasp with closed-loop therapy does not offer any additional clinical benefit over using standard IAsp. In contrast to adult studies, we observed a higher rate of hyperglycemia with ketosis events in the Fiasp period. Future research should aim to trial newer ultrarapid insulins with faster onset and offset than Fiasp, as improved preparations in conjunction with HCL therapy may well be able to confer additional clinical benefit and address some of the remaining treatment challenges in this vulnerable age group.
Footnotes
Acknowledgments
We are grateful to study volunteers for their participation. We acknowledge administrative support from Alina Cezar, Matthew Haydock, and Josephine Hayes at the University of Cambridge, support by the staff at the Addenbrooke's NIHR/Wellcome Trust Clinical Research Facility Cambridge, support from Emma Patterson and the pharmacy team at Alder Hey Children's NHS Foundation Trust, Liverpool, and support from Yue Ruan and Rama Lakshman.
Authors' Contributions
R.H., J.W., J.M.A., C.K.B., M.E.W. and A.T. co-designed the study. J.W., J.M.A., S.H., M.D., H.L., K.P., and K.T. were responsible for screening and enrolment of participants, arranged informed consent, and provided patient care. A.C. supported study set up, coordination and randomization. J.W. and R.H. wrote the report. J.W., M.E.W., and R.H. carried out or supported data analysis, including the statistical analyses. R.H. designed and implemented the glucose controller. R.H., J.W., J.M.A., C.K.B., S.H., M.E.W., and A.T. contributed to the interpretation of the results. All authors critically reviewed the report. J.W., M.E.W. and R.H. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Disclaimer
The views expressed are those of the author(s) and not necessarily those of the funders.
Author Disclosure Statement
R.H. reports having received speaker honoraria from Eli Lilly, Dexcom and Novo Nordisk, receiving license fees from BBraun; patents related to closed-loop, and being director at CamDiab. J.W. reports receiving speaker honoraria from Ypsomed. J.M.A. reports training fees from CamDiab. C.K.B. reports receiving consultancy fees from CamDiab and speaker honoraria from Ypsomed. A.C. reports receiving consultancy fees from CamDiab. S.H. reports speaker and advisory board fees from Dexcom, Medtronic, Sanofi & Ypsomed; being director at ASK Diabetes Ltd and receiving consulting/training fees from CamDiab. M.E.W. is a consultant at CamDiab and reports patents related to closed-loop. A.T., M.D., H.L., K.P., and K.T. have no disclosures.
Funding Information
Supported by National Institute for Health Research Cambridge Biomedical Research Centre. Dexcom supplied discounted continuous glucose monitoring transmitters and sensors for the study.
Supplementary Material
Supplementary Appendix
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
