Abstract
Background:
The aim of this prospective open-label single-arm single-center follow-up study was to analyze glycemic control in children and adolescents with type 1 diabetes treated with the advanced hybrid closed loop (AHCL) system in relation to a sensor-augmented pump with low-glucose suspend (SAP-LGS) or predictive low-glucose suspend (SAP-PLGS).
Materials and Methods:
The data for 50 children and adolescents (age 5.5–19.6 years) with type 1 diabetes, receiving insulin through an AHCL system after being switched from SAP-LGS/PLGS systems, were included in the analysis. The SAP-LGS/PLGS records from 2 weeks preceding the AHCL connection were compared with the records from the first 4 weeks of AHCL use, represented as two separate 2-week intervals.
Results:
Significant improvements in most of the parameters, namely time spent in the range of 70–140 mg/dL (from 53.80% ± 12.35% to 61.70% ± 8.42%, P < 0.001) and 70–180 mg/dL (from 76.17% ± 10.28% to 81.32% ± 7.71%, P < 0.001), average sensor glucose (from 138.61 ± 16.66 to 130.02 ± 10.91 mg/dL, P < 0.001), and glucose management indicator (from 6.54% ± 0.45% to 6.27% ± 0.29%, P = 0.001), were observed within 2 weeks of switching to the AHCL. More evident improvements were observed for the parameters monitored at night than during the day. The potential limitations of this study were the short observation time, lack of glycated hemoglobin measurements, and no control arm.
Conclusion:
The AHCL system can significantly improve glycemic control even in well-controlled children and adolescents with type 1 diabetes by increasing the proportion of time spent in the narrower range of 70–140 mg/dL and decreasing the mean glucose concentration, especially during the night.
Introduction
Type 1
In line with the American Diabetes Association (ADA), the International Society for Pediatric and Adolescent Diabetes (ISPAD), and Diabetes Poland (PTD) recommendations, glycemic goals in type 1 diabetes care should include maintaining glycated hemoglobin (HbA1c) levels below 7.0%, and 6.5%, respectively, minimizing hypoglycemia, and achieving at least 70% of the time in range (TIR) of 70–180 mg/dL and glycemic coefficient of variation (CV) <36%. 3 –7 Unfortunately, available data show that the ADA, ISPAD, and PTD glycemic goals are not met by a considerable proportion of children with type 1 diabetes. 8,9 The causes of this unfavorable situation include a plethora of physiological, developmental, and psychosocial factors, with one specific issue being the inconvenience of maintaining a status close to normoglycemia with currently available insulin delivery systems. 4
The issue mentioned above can be addressed adequately with state-of-the-art insulin pumps, such as the advanced hybrid closed loop (AHCL) system. 10 –12 AHCL (MiniMed® 780G Medtronic) can maintain glucose concentration at a prespecified level based on the information obtained through continuous glucose monitoring (CGM), using an advanced algorithm to automatically estimate the required insulin dose, along with manual initiation of postmeal boluses. The system received regulatory approval in the European Union (Conformité Europëenne Mark, CE) in June 2020. According to the product resume, the AHCL system is indicated for type 1 diabetes patients aged 7–80 years whose total daily insulin (TDI) dose is eight units per day or more. The MiniMed 780G system can work in both manual and automated modes (SmartGuard) with three glucose targets—100, 110, and 120 mg/dL, along with a temporary target of 150 mg/dL. 13
While the AHCL systems have been studied quite extensively in adult patients, comparably less is known about their application in children with type 1 diabetes, who should theoretically benefit the most from the treatment. To the best of our knowledge, only a few studies of children being switched from a sensor-augmented pump predictive low-glucose suspend (SAP-PLGS) pump to a MiniMed 780G system have been conducted thus far, with the studies often pooling the children with older patients. 14 –20
The aim of this prospective open-label, single-arm, single-center, follow-up study was to analyze glycemic control in children and adolescents with type 1 diabetes treated with the AHCL system in relation to a sensor-augmented pump with low-glucose suspend or predictive low-glucose suspend (SAP-LGS/PLGS).
Materials and Methods
All 56 consecutive children and adolescents with type 1 diabetes, receiving insulin through an AHCL system at the Regional Pediatric Diabetology Center, University Clinical Hospital, the Medical University of Silesia in Katowice (Upper Silesia, Poland) between December 2020 and June 2021 after being switched from the SAP-LGS/PLGS, were screened for the eligibility for this follow-up study.
Pediatric diabetology care in Poland is highly centralized, with all children with type 1 diabetes treated at regional, mainly academic, centers. The center in Katowice is the largest Polish pediatric diabetology center experienced in pump therapy, a member of the SWEET-certified Centers of Reference. 21
The inclusion criteria of the study were age younger than 21 years, more than 70% of sensor wear. Eventually, the data for 50 children and adolescents were included in the analysis. Before being switched to the AHCL system, all patients used SAP-LGS/PLGS, MiniMed 754 (12%) or MiniMed 640 (88%), for at least 6 months. The switching to the Medtronic MiniMed 780G system was preceded by a 2-h pump training. The automatic mode was started after 7 days of manual mode. The target glucose level was set at 100 mg/dL for all patients and autocorrect was activated with active insulin time set at 2 h. Carbohydrate-to-insulin ratios remained unchanged. Patients and their caregivers were instructed to use a temporary target of 150 mg/dL during physical activity. The high alarm was set at 180 mg/dL and the low alarm at 60 or 65 mg/dL.
The data from SAP-LGS/PLGS were obtained using CareLink® Professional software before connecting the MiniMed 780G system. The data from the system were automatically sent to the CareLink server and retrieved with CareLink Professional software. The SAP-LGS/PLGS records from 2 weeks preceding the AHCL connection were compared with the records from the first 4 weeks (after the initial training) in the automatic insulin dose adjustment system to current blood glucose levels (SmartGuard, AHCL), separated into two 2-week intervals.
Very detailed data curation was performed before the statistical analysis. The three 2-week time series (one for SAP-LGS/PLGS and two consecutive 2-week intervals for the MiniMed 780G system) were summarized by the following parameters: average sensor glucose (Avg SG), standard deviation (SD) of blood glucose level, CV, and robust coefficient of variation (CVR) for blood glucose level. Unlike classical CV being the ratio between SD and mean value, CVR is the ratio between the interquartile range and the median value. In addition, the ratio and difference between the analyzed parameters for two consecutive time points were considered in the analysis. Also, the percentages of blood glucose data points below 54 mg/dL and within the intervals 54–70, 70–140, 140–180, 180–250 mg/dL, and above 250 mg/dL were calculated. In addition, the range from 70 to 180 mg/dL was considered to enable comparison with previous studies.
These parameters, describing the dynamics of patient blood glucose level, were supported by the information from the SAP-LGS/PLGS systems, such as total daily dose, basal, bolus, sensor wear, autocorrection amount, and SmartGuard (the last two parameters for AHCL only). Routine clinical information (patient age, disease duration, body weight, body height, BMI, sex) was also used to characterize the study group.
Descriptive statistics (mean, SD, median, interquartile range, minimum and maximum values, CV, and CVR) and their 95% confidence intervals were calculated for each parameter. The statistics describing each of the 2-week time series and the results split into daytime (from 6 am to 10 pm) and nighttime were estimated. The Lilliefors test was applied to verify the hypothesis of the normality of parameter distribution. The Friedman analysis of variance (ANOVA) for repeated measures, accompanied by Kendall's concordance W-coefficient as the effect size and intragroup homogeneity measure, was applied for each parameter. The Wilcoxon test was used in the post hoc analysis with the Bonferroni method adjusting for multiple comparisons.
The adjusted P values were provided together with rank biserial correlation coefficient (rbc) as the effect size measure, which enables the quantification and comparison of results across different studies independent of the sample size. 22 The P-values below 0.05 were treated as statistically significant. The concordance coefficient (W) was used to qualify the agreement in time response, with W < 0.2 interpreted as slight agreement, 0.2–0.4 as fair agreement, 0.4–0.6 as moderate agreement, 0.6–0.8 as substantial agreement, and 0.8 and more as an almost perfect agreement in time response among patients. In the case of the rbc, values below 0.1 were interpreted as a negligible effect, 0.1–0.3 as a small effect, 0.3–0.5 as a moderate effect, and more than 0.5 as a large effect.
The protocol of the study was approved by the Local Bioethics Committee at the Medical University of Silesia in Katowice (Decision No. PCN/0022/KBI/83/2 of March 30, 2021).
Results
The study included the data for 50 children with a mean age of 11.0 ± 3.4 years (median: 10.4, range: 5.5–19.6), among them 22 (44%) boys. The mean age at the onset of type 1 diabetes was 6.3 ± 3.3 years (median: 5.8, range: 0.8–15.5), with a mean duration of the disease of 4.7 ± 2.8 years (median: 4.1, range: 0.7–11.0). The anthropometric characteristics of the study participants are summarized in Table 1.
Characteristics of the Participants (50 Children and Adolescents)
BMI, body mass index.
Avg SG, whether during the day or at night, decreased significantly after 2 weeks of AHCL use (P < 0.0005 and a large effect size with rbc >0.6 in all cases) and then did not undergo statistically significant changes until the end of the study period (Table 2 and Supplementary Table S1).
Glycemic Outcomes, Insulin Therapy, and Using the Sensors: After 2 Weeks of Sensor-Augmented Pump with Low-Glucose Suspend or Predictive Low-Glucose Suspend, the First 2 Weeks After Advanced Hybrid Closed Loop System Training and the Second 2 Weeks After Advanced Hybrid Closed Loop System Training
AHCL, advanced hybrid closed loop; ANOVA, analysis of variance; Avg SG, average sensor glucose; CV, coefficient of variation; GMI, glucose management indicator; SAP-LGS/PLGS, sensor-augmented pump with low-glucose suspend or predictive low-glucose suspend; TDI, total daily insulin.
A slight decrease in CVR over time was observed only for the measurements taken at night (Friedman ANOVA, P = 0.0156, W = 0.08). Both the basal insulin and glucose management indicator (GMI) decreased significantly within the initial 2 weeks after switching (large effect size, rbc = 0.58 and rbc = 0.67, respectively) and did not undergo further significant changes during the following 2 weeks. No significant changes in TDI were noticed during the initial 2 weeks of the study or later (Table 2 and Supplementary Table S1).
A slight increase in bolus insulin was noted (Friedman ANOVA, P = 0.0407, W = 0.06), with the most evident, moderate-degree change (Wilcoxon's adjusted P = 0.0734, rbc = 0.40) observed between weeks 2 and 4. During AHCL use, between weeks 2 and 4, a significant moderate-degree decrease was observed in the autocorrect value (Wilcoxon's adjusted P = 0.0078, rbc = 0.50) and a significant large-degree increase in the frequency of SmartGuard use (Wilcoxon's adjusted P = 0.0001, rbc = 0.74). Meanwhile, the frequency of using the sensor increased significantly over time (Friedman ANOVA P < 0.0001, W = 0.42 corresponding to a moderate agreement) within the initial 2 weeks and then remained unchanged between weeks 2 and 4 (Table 2 and Supplementary Table S1).
Significant changes in glycemic control were observed solely for glucose concentrations of ≥70 mg/dL. The time spent in the 70–180 mg/dL range significantly increased from 76.2% to 81.3% and 81.5% by average for the first and second 2-week observation periods, respectively (Friedman ANOVA P < 0.000001), with fair agreement among patients (concordance index W = 0.31). If daytime and nighttime were considered separately, the described pattern was confirmed during the nighttime but not as unequivocally during the daytime (concordance index W = 0.04). Detailed analysis revealed a significant increase for the range of 70–140 mg/dL (P < 0.000001, W = 0.31) and a slightly less coherent decrease in the proportion of time spent in the ranges of 140–180 mg/dL (P = 0.0007, W = 0.14). These observations were similar for daytime and nighttime.
A significant decrease in time spent was also observed for the range of 180–250 mg/dL both during the day and at night, with slightly more evident changes seen during the night (as shown by the values of concordance coefficients and rank biserial correlation coefficients; see Table 2 and Supplementary Table S1). The changes in the proportion of time with glucose concentrations >250 mg/dL during the day and night were slight although significant (Friedman ANOVA P = 0.0017 and P = 0.0079, respectively; W = 0.13 and W = 0.10, respectively). All significant changes occurred during the initial 2 weeks of the new system implementation, and the results remained subsequently unchanged until the end of the observation period (Table 2 and Fig. 1; Supplementary Table S1).
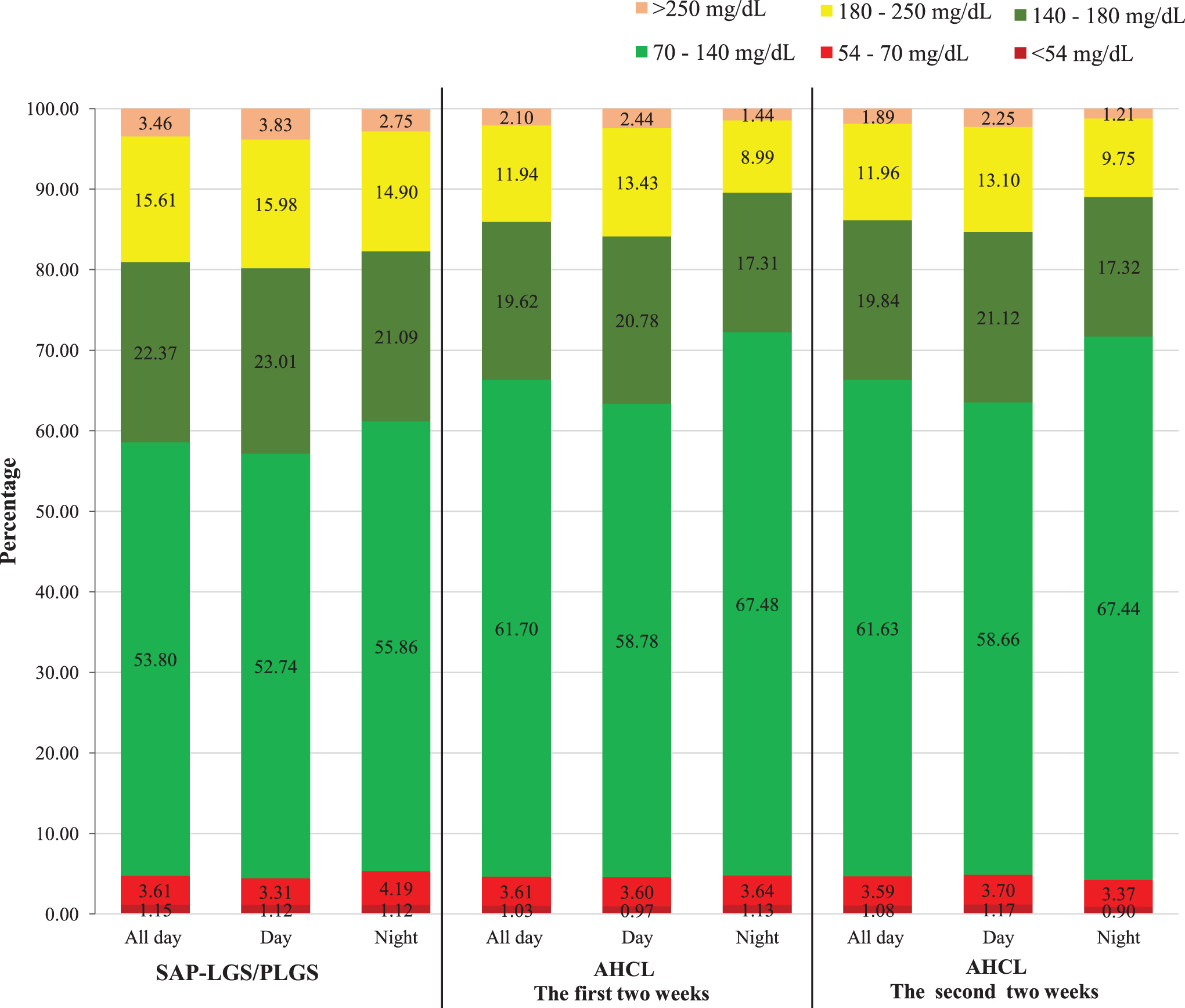
Percentages of sensor glucose values in range after SAP-LGS/PLGS treatment, 2 weeks after AHCL system training, and after 4 weeks of the AHCL system use. AHCL, advanced hybrid closed loop; SAP-LGS/PLGS, sensor-augmented pump with low-glucose suspend or predictive low-glucose suspend.
Discussion
This follow-up study demonstrated that switching to the AHCL resulted in a significant improvement of glycemic control in a group of 50 children with type 1 diabetes previously using SAP-LGS/PLGS; to the best of our knowledge, this is the first study involving a generally satisfactorily controlled, young homogenous cohort experienced in the use of SAP-LGS/PLGS. Importantly, statistically significant improvements in most parameters (time spent in the range of 70–140 and of 70–180 mg/dL, Avg SG, and GMI) were observed already, 2 weeks after switching to the AHCL. More evident improvements were observed for parameter values captured at night. Management of children with type 1 diabetes constitutes multiple challenges, with one of them being the difficulty of achieving the recommended TIR.
Switching to the AHCL system enabled us to overcome some of those obstacles, specifically to improve the time spent in the range of 70–140 and of 70–180 mg/dL; these beneficial outcomes were achieved without jeopardizing the treatment's safety, as shown by the lack of significant changes in time in hypoglycemia and TDI.
The effectiveness of the AHCL MiniMed 780G system in children and adolescents with type 1 diabetes has been recently analyzed by a few authors. However, most of those studies included the heterogeneous age groups of patients, with children and adolescents representing only a fraction of the participants. Among the 113 patients included in the FLAIR study, 73 participants were aged 14–20 years. 14 The mean age of patients examined by Beato-Víbora et al. was 43 ± 12 years, with only 5 out of the 52 participants aged <21 years. 15 A multicenter study conducted by Carlson et al. included 39 adolescents aged 14–21 years, along with 118 adults.
Collyns et al. examined 59 patients with type 1 diabetes, with a mean age of 23.3 ± 14.4 years, among them 33 younger than 21 years. 16,17 The mean age of 37 type 1 diabetes patients included in the study conducted by Piccini et al. was 15 ± 7.4 years. To the best of our knowledge, except for the present study, the effectiveness of the MiniMed 780G system in the purely pediatric population has been analyzed only by Petrovski et al. in a group of 34 type 1 diabetes patients with a mean age of 12.5 ± 3.7 years, previously treated with multiple daily injections (MDI). 19,20 The differences in the age structures and models of insulin therapy of the analyzed patients are worth emphasizing, given that according to literature, achieving the ISPAD/ADA glycemic targets in the pediatric population can be much more challenging than in adults. 23,24
Notably, the participants of our study presented a markedly better baseline degree of glycemic control than type 1 diabetes patients examined by other authors, with Avg SG of 138.61 mg/dL (median: 134.97, range: 103.09–173.89), GMI of 6.54% (median: 6.52, range: 5.65–7.81), 76.17% of time spent in the target range of 70–180 mg/dL, and 53.8% of time spent in the range of 70–140 mg/dL. This was undoubtedly associated with the fact that all children used the SAP-LGS/PLGS system before switching to AHCL, which represented another unique feature of our cohort. 25,26 Furthermore, our observations are consistent with the results of the PolPeDiab HbA1c study, a nationwide cross-sectional evaluation of glycemic control, in which the ISPAD target for HbA1c was achieved by up to 45.2% of 1255 Polish children with type 1 diabetes. 27
Meanwhile, in the FLAIR study, the mean baseline concentration of glucose was 173 mg/dL, with 57% of time spent in the target range and 34% of time spent in the range of 70–140 mg/dL. 24 Aside from our study, a relatively good degree of glycemic control at the baseline (patients switched from PLGS/HCL) was also reported by Ruiz Arroyo et al., with a mean glucose concentration of 154 mg/dL, GMI of 6.75%, and 70.73% of time spent in the target range of 70–180 mg/dL. 28 In the study conducted by Petrovski et al., including patients with a similar age structure as our participants who switched from MDI with real-time CGM, the degree of glycemic control at the baseline was markedly lower, with target HbA1c achieved by only 8.6% of the patients. 20
The differences in the baseline degree of glycemic control should be considered an important determinant of the outcome, as both literature data and the results of our present study suggest that children with satisfactory glycemic control can benefit more from the treatment. 17
In the present study, the use of the AHCL system contributed to a significant increase in the proportion of time spent in the strict range of 70–140 and of 70–180 mg/dL, as well as to a decrease in the mean concentration of glucose and GMI, with the CV maintained during the day and no additional risk of hypoglycemia despite a good degree of glycemic control at the baseline. Our findings are consistent with the results of previous studies using the MiniMed 780G system. In the FLAIR study, treatment with the new system resulted in a decrease in mean glucose concentration and HbA1c level, along with an increase in the proportion of time spent in the range of 70–140 and of 70–180 mg/dL.
Also, according to other authors, the use of the Medtronic system was associated with a decrease in mean glucose concentration and GMI without concomitant changes in the CV and the proportion of time spent in hypoglycemia. 15,19,28 In summary, our findings and the results of previous studies clearly demonstrate that the use of the AHCL MiniMed 780G system could improve metabolic control of type 1 diabetes in all children, increasing the proportion of time spent in the target range without an increase in the time in hypoglycemia. In addition, our study showed that the use of the new system was associated with a slight decrease in CVR during the night, which should be considered important in preventing future vascular complications.
In this study, an increase in the proportion of time spent in the target range was more evident at night than during the day. This observation is consistent with the results reported by Da Silva et al., who analyzed the data from 4120 users of the MiniMed 780G system uploaded to CareLink Personal software. According to those authors, the proportion of time spent in the target range was 76.2% overall, 73.9% during the day (6:01–11:59), and 83% during the night (12:00–6:00). 29 The difficulty of achieving glycemic control during the day is not surprising given that the number of factors that interfere with glycemia in the daytime is higher than at night; this might still constitute a problem for the already advanced algorithm of the device.
Also, Weinzimer et al. demonstrated that the high glycemic control results from improved control at night and a later postprandial period (after 3 h). The glycemic control does not appear to stem from the early (first 3 h) postprandial glycemia improvement. 30 However, glycemic control at night also constitutes a challenge in the pediatric population, among others, because of the pubertal growth spurt. In this context, the ability to control glycemia during the night represents, without a doubt, an important feature of the AHCL MiniMed 780G system. 31
The improvement in the glycemic control of our patients was not associated with an increase in TDI. In the study conducted by Da Silva et al., information about TDI before implementing the Medtronic system was available for 812 patients. The use of the new system in those patients was associated with a significant increase in TDI, which was probably associated with a lower degree of glycemic control at the baseline (mean glucose concentration 162.2 mg/dL, GMI 7.2%, 63.4% of time spent in the target range). 29 Our patients had better glycemic control at the baseline, and thus, the further glycemic improvement did not require an increase in TDI. This observation is worth emphasizing, given that the maintenance of TDI at the lowest possible level is important in preventing cardiovascular complications. 32
Similar to the studies conducted by Beato-Víbora et al. and Weinzimer et al., using the AHCL MiniMed 780G system in our patients was associated with a decrease in basal insulin with a concomitant increase in bolus insulin, which was probably caused by autocorrection. 15,30 Active insulin time in our patients was set at 2 h, with a target glucose level of 100 mg/dL and the autocorrect function activated. Beato-Víbora et al. used similar settings, which, similar to in our study, were not reflected by a significant increase in the proportion of time spent in hypoglycemia.
Both in our present study and in the study conducted by Beato-Víbora et al., switching to the MiniMed 780G system was associated with an increase in the frequency of sensor use, possibly motivated by the AHCL system's ability to respond to hyperglycemia when the CGM signal is available. Importantly, in both studies, SAP-LGS/PLGS was used before switching to the MiniMed 780G system. 15
As mentioned earlier, achieving near normoglycemia in pediatric patients is always a challenge. Both prior published evidence and our findings suggest that some of the hurdles on the way to normoglycemia could be addressed by the new automated Medtronic system. The normalization of glycemic parameters during the night, a problem that raises the concerns of all parents and caregivers, is a particularly promising development. 31
We are well aware of the potential limitations of this study—a relatively short observation time, no HbA1c measurements, and the lack of a control arm. The study participants were followed up for only 4 weeks, but the most beneficial effects of changing to the new system were observed within the initial 14 days from the switching, with subsequent stabilization during the following 2-week period. Additional factors might have contributed to the improvement of glycemia, such as frequent contact with educators and commitment resulting from new device use with new technology. Our patients did not require additional education in the field of pump therapy in manual mode (previously, they used SAP). Hence, the education was simplified and concerned only the technical part of the automatic mode.
Patients in our diabetic center are regularly educated on diet and self-control, as evidenced by good alignment before including patients in the study. The first personal contact with the medical staff took place 1 month after the AHCL was connected. Due to the short period of the study, GMI instead of HbA1c was used. 5 It was a prospective, open-label, single-arm, single-center study, and it was carried out at the largest Polish center of pediatric diabetology, a member of SWEET. 21 The study included a large group of mostly satisfactorily controlled young children and adolescents with type 1 diabetes, homogenous in terms of age unlike in prior studies. The time of observation of the use of AHCL was similar to other studies and publications, and the favorable results of the use of AHCL in children and adolescents with type 1 diabetes prompted us to publish the results.
Nevertheless, these promising observations need to be confirmed during a longer follow-up period.
Conclusions
The AHCL system can significantly improve glycemic control even in well-controlled children and adolescents with type 1 diabetes by increasing the proportion of time spent in the narrower range of 70–140 mg/dL and decreasing the mean glucose concentration, especially during the night, without a concomitant increase in the proportion of time in hypoglycemia, variability, and the total and basal dose of insulin.
Footnotes
Authors' Contributions
S.S. and E.R. researched the data, S.S., E.R., and P.J.-C. wrote the article, S.S., J.P., and P.J.-C. contributed to the discussion, J.P. developed the methodology and performed data analyses, and J.P. and P.J.-C. reviewed/edited the article.
Acknowledgments
The authors would like to thank Foundation Maxima for Children, Poland, for financial support for the publication and all patients with type 1 diabetes and their caregivers from the Regional Pediatric Diabetology Center, University Clinical Hospital, the Medical University of Silesia in Katowice, Poland. In addition, the authors acknowledge the editorial assistance of Dr Simon Kim, SciencePro.
Author Disclosure Statement
Jarosz-Chobot has received speaker honoraria from Medtronic, DexCom, Abbott, Ypsomed, and Roche, was a member of the advisory boards for Medtronic and Abbott and received research support from Medtronic. Sebastian Seget has received speaker honoraria from Medtronic and Ypsomed. Other authors declare no conflict of interest.
Funding Information
The work was partially supported by a grant for the support and development of research potential from the Silesian University of Technology (J.P.). Grant no SUT/02/070/BK_22/0033.
Supplementary Material
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
