Abstract
Background and Aims:
Nocturnal hypoglycemia is mainly a consequence of inappropriate basal insulin therapy in type 1 diabetes (T1D) and may compromise optimal glycemic control. Insulin degludec is associated with a lower risk of nocturnal hypoglycemia in T1D. As nocturnal hypoglycemia is often asymptomatic, we applied continuous glucose monitoring (CGM) to detect a more precise occurrence of nocturnal hypoglycemia in the HypoDeg trial, comparing insulin degludec with insulin glargine U100 in people with T1D and previous nocturnal severe hypoglycemia.
Materials and Methods:
In the HypoDeg trial, 149 people with T1D were included in an open-label randomized cross-over trial. Sixty-seven participants accepted optional participation in the predefined substudy of 4 × 6 days of blinded CGM requiring completion of at least one CGM period in each treatment arm. CGM data were reviewed for hypoglycemic events.
Results:
Treatment with insulin degludec resulted in a relative rate reduction (RRR) of 36% (95% confidence interval [CI]: 10%–54%; P < 0.05) in nocturnal CGM-recorded hypoglycemia (≤3.9 mmol/L), corresponding to an absolute rate reduction (ARR) of 0.85 events per person-week. In nocturnal CGM-recorded hypoglycemia (≤3.0 mmol/L), we found an RRR of 53% (95% CI: 36%–65%; P < 0.001), corresponding to an ARR of 0.75 events per person-week. At the lower detection limit of the CGM (≤2.2 mmol/L), treatment with insulin degludec resulted in a significant RRR of 58% (95% CI: 23%–77%; P = 0.005). The reductions were primarily due to significant RRRs in asymptomatic hypoglycemia.
Conclusion:
In people with T1D, prone to nocturnal severe hypoglycemia, insulin degludec compared with insulin glargine U100 significantly reduces nocturnal CGM-recorded hypoglycemia.
Introduction
Nocturnal hypoglycemia is most often asymptomatic and thus passes unrecognized. 1 Frequent nocturnal asymptomatic hypoglycemia may promote impaired awareness of hypoglycemia, 2 –4 which increases the risk of severe hypoglycemia. 5 –7 The concern of severe hypoglycemia, occurring during the night, often results in avoidance behavior that may impair glycemic control. Thus, people with type 1 diabetes (T1D) who have experienced nocturnal severe hypoglycemia may aim at higher blood glucose at bedtime resulting in overnight hyperglycemia. 8 Therefore, reduction in nocturnal hypoglycemia is a key clinical challenge.
The long-acting insulin analogs insulin glargine U100 and insulin detemir reduce the risk of nocturnal hypoglycemia compared with neutral protamine hagedorn (NPH) insulin. 9 Insulin degludec provides a further reduction of nocturnal hypoglycemia than insulin glargine U100. 10 –13 A limitation of the trials performed with insulin degludec so far is the exclusion of subjects with recurrent severe hypoglycemia, and none specifically including people prone to nocturnal severe hypoglycemia. Furthermore, the studies have not applied continuous glucose monitoring (CGM) for recording of hypoglycemia, which is necessary to capture the extent of asymptomatic hypoglycemia, constituting the majority of nocturnal hypoglycemic events. 1
The objective of the HypoDeg trial was to investigate whether insulin degludec U100 in comparison with insulin glargine U100 reduces the risk of nocturnal hypoglycemia in people with T1D prone to nocturnal severe hypoglycemia. The primary objective of the HypoDeg trial was the occurrence of nocturnal symptomatic hypoglycemia as evaluated by blood glucose monitoring (BGM). There was a significant relative rate reduction (RRR) of 28% in hypoglycemic events at BGM ≤3.9 mmol/L and an RRR of 37% at BGM ≤3.0 mmol/L during treatment with insulin degludec. 14
In this study, we report the data on hypoglycemic endpoints recorded by blinded CGM from a predefined substudy of the HypoDeg trial.
Materials and Methods
Study design
HypoDeg is a 2-year investigator-initiated controlled multicenter cross-over study conducted in a prospective, randomized, open, blinded endpoint adjudication (PROBE) design. The study investigated the effect of insulin degludec on hypoglycemic events compared with insulin glargine U100 in people with T1D prone to nocturnal severe hypoglycemia. 15
We included adults (>18 years) diagnosed with T1D for >5 years and, most notably, one episode or more of nocturnal severe hypoglycemia within the last 2 years (as defined by the need of external assistance for recovery). Recruitment and screening are previously described. 15
Participants were randomized 1:1 to treatment with insulin degludec or insulin glargine U100 administered with the evening meal. Administration of insulin glargine U100 at this time provides a more consistent overnight glycemic control than administration in the morning. 16
Participants took their short-acting insulin (insulin aspart) three times daily before main meals, and they reduced both basal and prandial insulin doses by 20% at the beginning of the trial period. After 12 months of treatment (3 months run-in and 9 months maintenance), participants crossed over to the other treatment for 12 months (3 months run-in and 9 months maintenance). We aimed at maintaining baseline hemoglobin (HbA1c) at the discretion of the local investigator because this was the most pragmatic glycemic target in these hypoglycemia-prone participants.
The primary endpoint of the trial was the incidence of nocturnal symptomatic hypoglycemia as recorded by BGM. The secondary endpoints were severe hypoglycemia incidence, glycemic control as evaluated by HbA1c, and mean insulin doses. 14 All endpoints were recorded during the 9 months of maintenance treatment. We have described the protocol previously in detail. 15
The participants were offered optional participation in a predefined CGM substudy consisting of 6 days of blinded CGM after 6, 12, 18, and 24 months of treatment, securing two CGM periods during each treatment arm. The blinded CGM monitoring (iPro®2 with the Enlite® sensor; Medtronic Minimed, Northridge, CA) was initiated at the participant's site, or at the Clinical Research Unit at Copenhagen University Hospital - North Zealand, Hillerød.
The sensor was inserted into the abdominal region, and participants were instructed to record BGM four times daily, the first 2 h after insertion and for later calibration as recommended by the manufacturer. After 6 days, the CGM recordings were uploaded to Medtronic Carelink iPro Therapy Management Software for Diabetes. The data remained blinded to the investigators until the end of the trial and were not used to adjust insulin dosage.
Participants kept a diary of activities, mealtimes, and insulin doses. We asked them to keep notes of symptoms of hypoglycemia in the diary and confirm hypoglycemic events with BGM.
A valid CGM period was from the first CGM measurement with a corresponding BGM calibration to the last CGM measurement within 12 h since the previous calibration. We excluded missing signals for >30 min from the total observation time, as we considered it enough to miss a hypoglycemic event.
The study was approved by the regional committee on biomedical research ethics (#H-3-2014-101), the Danish Medicines Agency (#201407615), and the Danish Data Protection Agency (I-suite no: 02945; #NOH-2014-018). The study is registered at (
All participants provided written informed consent.
Data are available from the corresponding author upon request.
Definitions of CGM-recorded hypoglycemic endpoints
CGM data were reviewed for hypoglycemic events with CGM values of ≤3.9 mmol/L (≤70 mg/dL), CGM ≤3.0 mmol/L (≤54 mg/dL), and CGM ≤2.2 mmol/L (≤40 mg/dL), corresponding to the lower detection limit of the CGM system. We assessed CGM data in accordance with the protocol of the main study, which was written before the current recommendations concerning hypoglycemia reporting established by The International Hypoglycaemia Study Group and the International Consensus on use of CGM. 17,18 Thus, hypoglycemic events with CGM ≤3.9 mmol/L include all events equal to and <3.9 mmol/L; furthermore, hypoglycemic events are reported as ≤3.0 mmol/L, not as <3.0 mmol/L, consistent with data from the main trial on BGM. 14,15
We included events lasting at least 15 min (four consecutive measurements in the CGM data), and we defined the termination of a hypoglycemic event as interstitial glucose concentration >3.9 mmol/L for 15 min. 18
Notes in the diary on symptoms of hypoglycemia and a CGM-verified low glucose value confirmed an event of symptomatic hypoglycemia. Asymptomatic events were CGM-verified low glucose values without corresponding notes in the diaries. A few events were classified as “unknown” when notes on single events were unclear.
The definition of nighttime was periods from 23:00 to 06:59 or 00:00 to 05:59. Hypoglycemic events starting 15 min or less before 23:00 or 00:00 were also considered as nocturnal, based on a period lasting ≥15 min (four consecutive measurements). The definition of daytime was periods from 07:00 to 22:59 or from 06:00 to 23:59. We also considered hypoglycemic events starting 15 min or less before 07:00 or 06:00 as daytime. There is currently no consensus on defining the night period. To compare the results according to time periods with previous studies and make sure that there were no differences in the occurrence of hypoglycemia based on the period, we used two different periods.
Statistical analyses
According to the data distribution, we performed baseline comparisons between groups for continuous variables using independent samples t-test or Mann–Whitney U test. For categorical variables, we compared differences in proportions between groups using the chi-square test.
As the distribution of hypoglycemia rates is skewed, we present medians and ranges in addition to means and SD. Through a Poisson log-linear model with fixed effects of treatment, treatment sequence, and period (first or last CGM period in each treatment period), we modeled the number of events to compare rates of hypoglycemia between groups. Owing to the cross-over design, the participants act as their own control. We used the generalized estimation equation (GEE) to compare the occurrence of hypoglycemia. The GEE considers the cluster of repeated measurements within each subject with fixed effects of treatment, treatment sequence, and period.
We quantified treatment comparisons as incidence rate ratios, which can be interpreted as a RRR and is presented as such.
To translate hypoglycemia rates into a clinically relevant number, we calculated the time needed to treat one person (TNT1) with insulin degludec to prevent one event of CGM-recorded hypoglycemia. We calculated the TNT1 using the following formula
19
:
Since the frequencies of CGM-recorded hypoglycemic events are unknown in patients prone to nocturnal severe hypoglycemia and that hypoglycemic rates are subject to unpredictable study effects, we did not consider a formal sample size calculation feasible. 20
We set a level of statistical significance at 5% (two sided) and used the statistical software SPSS (IBM SPSS statistics 25) for all analyses.
Results
A total of 149 participants were randomized in the main HypoDeg trial. A subgroup of 89 subjects agreed to participate in the CGM substudy. Seventy-four participants fulfilled the predefined criterion of at least one completed CGM period in each treatment arm. Seven participants were excluded from the analysis because the notes in their diaries were unclear or insufficient. Hence, we ended up with 67 participants in total.
The participants in the CGM substudy, who were included in our analysis, were characterized by a long duration of diabetes (mean 28 years), acceptable HbA1c (7.8%), C-peptide negativity (88%), and a high prevalence of impaired awareness or unawareness (83%). The participants had experienced a mean of 2.5 nocturnal severe hypoglycemic episodes (range 1–12) in the preceding 2 years (Table 1).
Baseline Characteristics of Participants
Data are mean ± SD or number (%), unless indicated otherwise.
Significant difference P = 0.038 Mann–Whitney U test for non-normal distribution.
Significant difference P = 0.048 Mann–Whitney U test for non-normal distribution.
Significant difference P = 0.003 Pearson chi-square.
Macrovascular complications: hypertension, myocardial infarction, ischemic heart disease, heart failure, stroke, and/or peripheral vascular surgery.
C-peptide negative = below detection limit (<20 pmol/L or <0.02 nmol/L).
1 unit = 15 g of alcohol.
Significant difference P = 0.041 Pearson chi-square.
CGM, continuous glucose monitoring.
The total valid observation time was 1441 days (715 days in the insulin degludec arm and 726 in the insulin glargine U100 arm) with a mean total valid observation time per participant of 21.5 (SD 4.5) days. During treatment with insulin degludec and insulin glargine U100, the mean total valid observation time per participant was 10.7 (SD 2.7) days and 10.8 (SD 2.5) days, respectively.
Fifty participants (75%) completed 24 days of CGM, 12 participants (18%) completed 18 days of CGM, and 5 participants (7%) completed 12 days of CGM. The mean number of any CGM-recorded hypoglycemic event per participant was 31 (95% confidence interval: 26–36).
A total of 1301 CGM-recorded hypoglycemic events (≤3.9 mmol/L) were recorded, corresponding to a mean of 6.4 (SD 4.7) events per person-week (“A” in Table 2). Of CGM-recorded hypoglycemic events (≤3.0 mmol/L), we observed a total of 595 events corresponding to a mean of 2.9 (SD 3.5) events per person-week (“A” in Table 2). We recorded 185 events at the lower detection limit of the CGM (≤2.2 mmol/L), corresponding to a mean of 0.9 (SD 1.9) events per person-week (“A” in Table 2). The mean number of events per participant according to treatment and time interval is depicted in Figure 1.
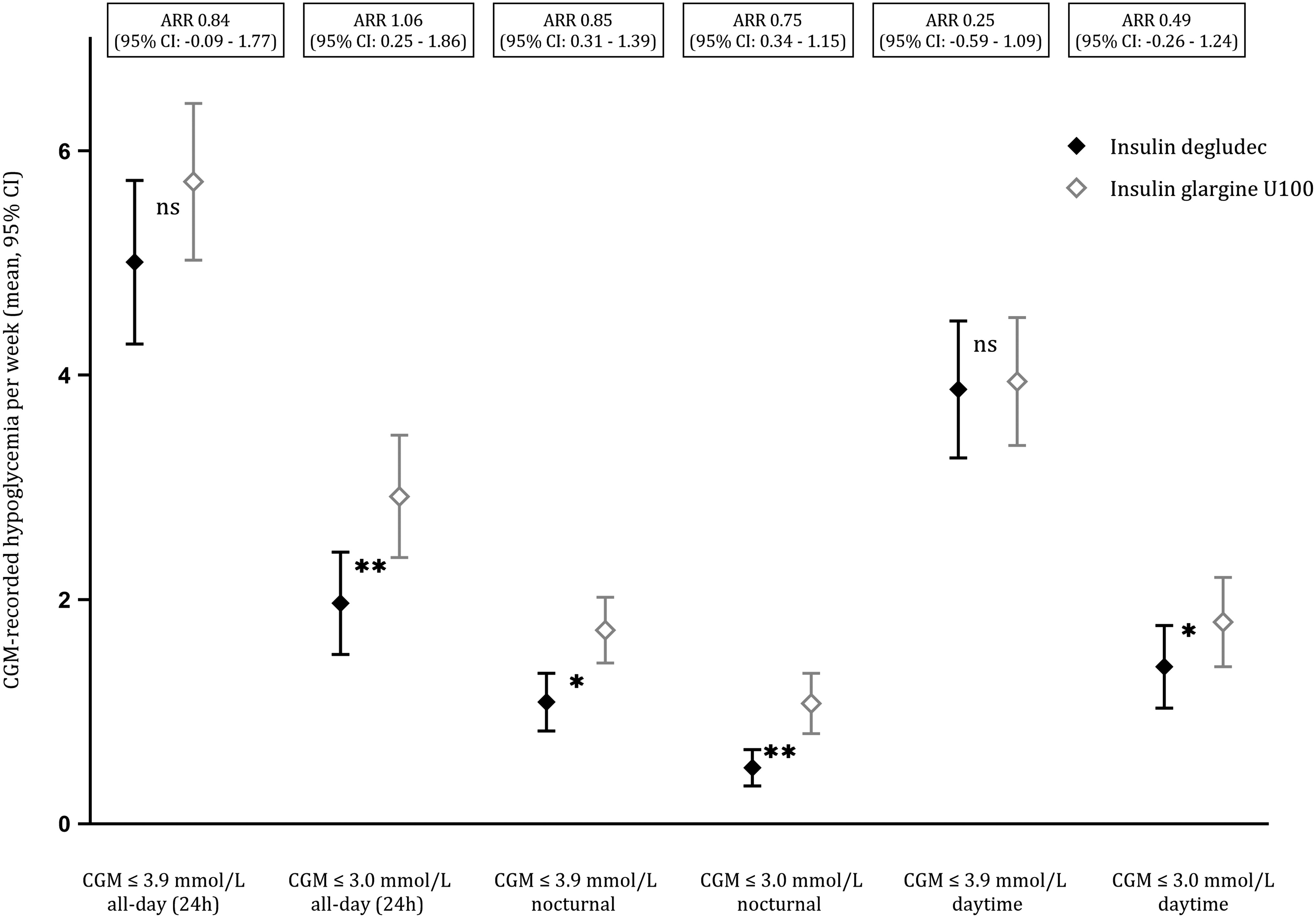
CGM-recorded hypoglycemic event rates with 95% CI according to glucose threshold and time period. Significant differences (*p < 0.05, **p < 0.001). All-day 24 h, nocturnal 23:00–06:59, daytime 07:00–22:59. CGM, continuous glucose monitoring; CI, confidence interval.
Continuous Glucose Monitoring-Recorded Hypoglycemia in Maintenance Periods According to Treatment
ARR, absolute rate reduction with IDeg; CI, confidence interval; E/week, events per person-week; IDeg, insulin degludec; IGlar, insulin glargine U100; RRR, relative rate reduction with IDeg.
During the main study, we included 136 episodes of severe hypoglycemia in the analysis. 14 Among these episodes, we registered one during the CGM substudy. The episode had a BGM of 1.8 mmol/L; in the CGM data, the interstitial glucose value was 2.7 mmol/L.
Event rates according to treatment
CGM ≤3.9 mmol/L
At this threshold of hypoglycemia, we registered 591 events (45%) during treatment with insulin degludec and 710 events (55%) during treatment with insulin glargine U100. There were no significant differences in all-day (24 h) event rates between treatments (“A” in Table 2).
Of these events, 342 (26%) were nocturnal (23:00–06:59). A 36% RRR (P = 0.009) during treatment with insulin degludec was observed for nocturnal CGM-recorded hypoglycemia as compared with insulin glargine U100 corresponding to an absolute rate reduction (ARR) of 0.85 events per person-week when treated with insulin degludec (“A” in Table 3). The reduction was due to a 32% RRR (P = 0.004) in asymptomatic hypoglycemia with insulin degludec compared with insulin glargine U100, corresponding to an ARR of 0.55 events per person-week (“C” in Table 3) (Fig. 2 and Supplementary Fig. S1). In contrast, we did not detect any significant differences between treatments in the occurrence of nocturnal symptomatic hypoglycemia (“B” in Table 3).
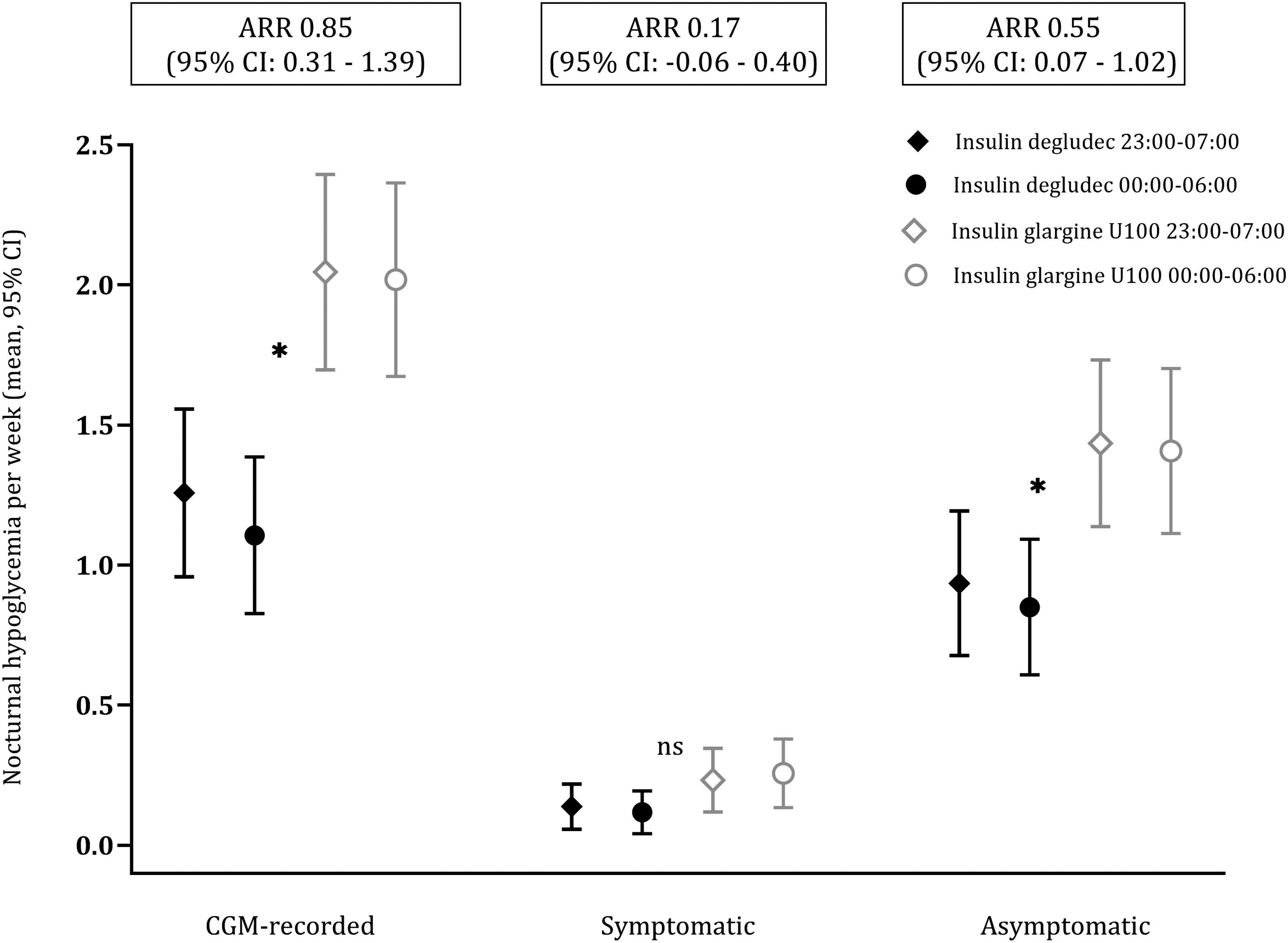
Nocturnal CGM-recorded hypoglycemia (≤3.9 mmol/L). Event rates with 95% CI according to treatment 23:00–06:59. Significant differences (*p < 0.05).
Continuous Glucose Monitoring-Recorded Hypoglycemia in Maintenance Periods According to Treatment
ARR, absolute rate reduction with IDeg; CI, confidence interval; E/week, events per person-week; IDeg, insulin degludec; IGlar, insulin glargine U100; RRR, relative rate reduction with IDeg.
During daytime, we recorded 946 events at this threshold of hypoglycemia without finding any significant differences between treatments (“A” in Table 4).
Continuous Glucose Monitoring-Recorded Hypoglycemia in Maintenance Periods According to Treatment
ARR, absolute rate reduction with IDeg; CI, confidence interval; E/week, events per person-week; IDeg, insulin degludec; IGlar, insulin glargine U100; RRR, relative rate reduction with IDeg.
The TNT1 for nocturnal CGM-recorded hypoglycemia (≤3.9 mmol/L) was 1.4 weeks. The TNT1 for nocturnal asymptomatic hypoglycemia at this threshold was 2 weeks.
CGM ≤3.0 mmol/L
At this threshold of hypoglycemia, we registered 236 events (40%) during treatment with insulin degludec and 359 events (60%) during treatment with insulin glargine U100. For all-day (24 h) events, there was a 34% RRR (P < 0.001) of CGM-recorded hypoglycemia in favor of insulin degludec. The difference was explained by a 25% RRR (P = 0.012) in asymptomatic hypoglycemia with insulin degludec, corresponding to an ARR of 0.49 events per person-week, and a 48% RRR (P = 0.022) in symptomatic hypoglycemia with insulin degludec compared with insulin glargine U100 (“A–C” in Table 2).
One hundred ninety-three (32%) events were nocturnal (23:00–06:59). When treated with insulin degludec, we observed a 53% RRR (P < 0.001) in nocturnal CGM-recorded hypoglycemia compared with insulin glargine U100, corresponding to an ARR of 0.75 events per person-week (“A” in Table 3). A 52% RRR (P < 0.001) in asymptomatic hypoglycemia with insulin degludec was the primary reason for the reduction in nocturnal CGM-recorded hypoglycemia. This reduction corresponded to an ARR of 0.43 events per person-week (“C” in Table 3). There were no significant differences between treatments in nocturnal symptomatic hypoglycemia (“B” in Table 3) (Fig. 3 and Supplementary Fig. S1).
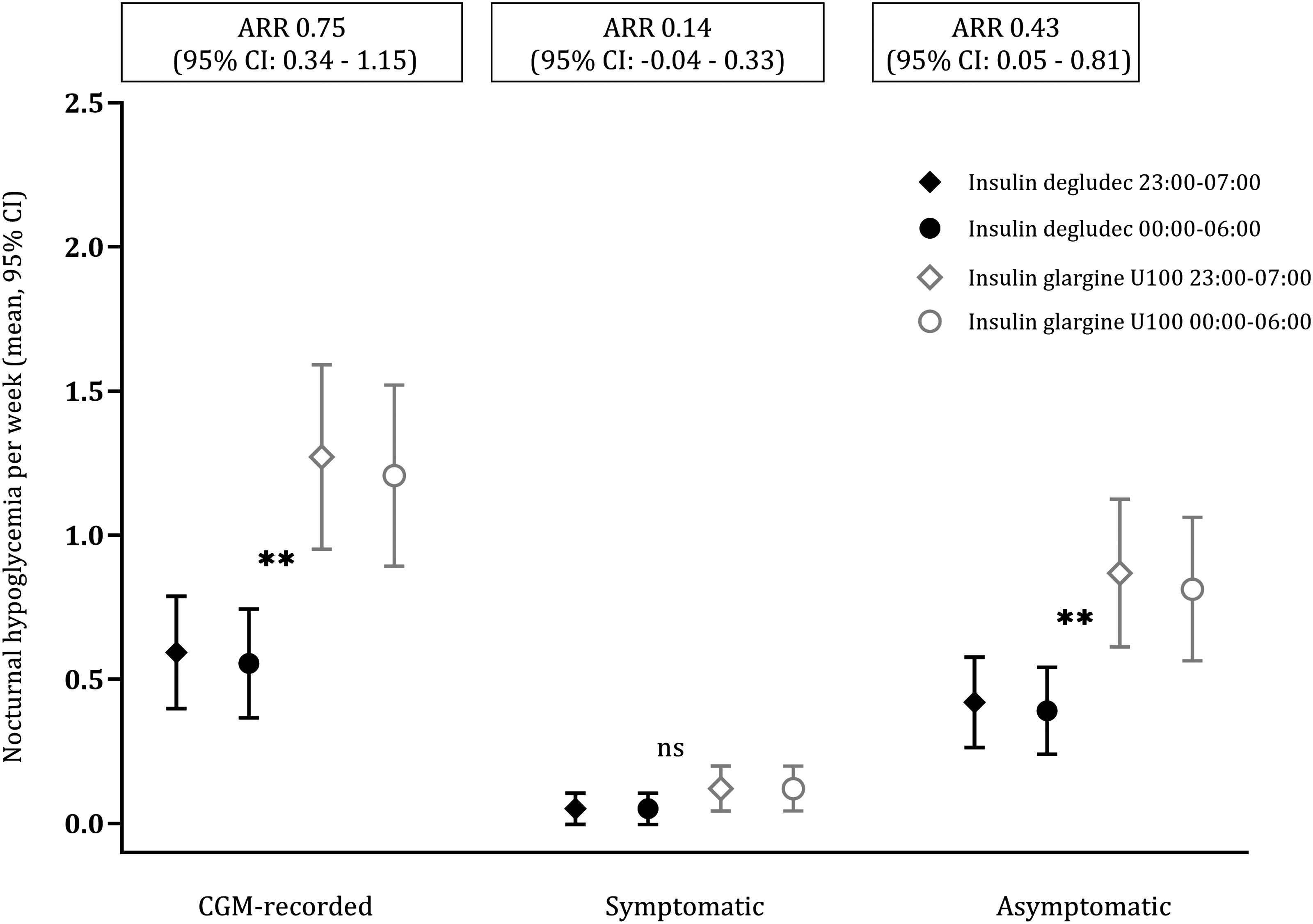
Nocturnal CGM-recorded hypoglycemia (≤3.0 mmol/L). Event rates with 95% CI according to treatment 23:00–06:59. Significant differences (**p < 0.001).
During daytime (07:00–22:59), we recorded 391 (66%) events. We observed a 24% RRR (P = 0.021) of CGM-recorded hypoglycemic events during treatment with insulin degludec compared with insulin glargine U100, corresponding to an ARR of 0.49 events per person-week (“A” in Table 4). This reduction was due to a 49% RRR (P = 0.027) in daytime symptomatic hypoglycemia with insulin degludec compared with insulin glargine U100, corresponding to an ARR of 0.14 events per person-week (“B” in Table 4).
The TNT1 at this threshold of nocturnal CGM-recorded hypoglycemia was 1.4 weeks. The TNT1 for nocturnal asymptomatic hypoglycemia was 2 weeks.
CGM ≤2.2 mmol/L
At this hypoglycemia threshold, we registered 55 events (30%) during treatment with insulin degludec and 130 events (70%) during treatment with insulin glargine U100. There was a 55% RRR (P = 0.003) of all-day (24 h) CGM-recorded hypoglycemia in favor of insulin degludec (“A” in Table 2). The reduction was partly due to a 79% RRR (P = 0.022) in all-day (24 h) symptomatic hypoglycemia with insulin degludec compared with insulin glargine and partly due to a 46% RRR (P = 0.017) in asymptomatic hypoglycemia with insulin degludec compared with treatment with insulin glargine (“B” and “C” in Table 2).
We recorded a total of 72 (39%) nocturnal (23:00–06:59) events of CGM-recorded hypoglycemia at the lower detection limit of the CGM. We found a 58% RRR (P = 0.005) in CGM-recorded hypoglycemia with insulin degludec compared with treatment with insulin glargine U100, corresponding to an ARR of 0.39 events per person-week (“A” in Table 3). There was a trend toward less asymptomatic hypoglycemia with insulin degludec than insulin glargine U100 (“C” in Table 3). We detected too few events of symptomatic hypoglycemia to allow comparison of treatments.
During daytime, at this threshold, we recorded 109 (59%) events (07:00–22:59). We observed a 53% RRR (P = 0.008) in CGM-recorded hypoglycemia during treatment with insulin degludec compared with insulin glargine U100 (“A” in Table 4), corresponding to an ARR of 0.50 events per person-week. The reduction was due to a 44% RRR (P = 0.017) in asymptomatic hypoglycemia with insulin degludec, corresponding to an ARR of 0.35 events per person-week (“C” in Table 4). There were too few symptomatic events during this period to test for differences between treatments.
We also analyzed other conventional time definitions: daytime defined from 06:00 to 23:59 and nighttime from 00:00 to 05:59. All significant differences at all three hypoglycemia thresholds were robust to these definitions (Supplementary Tables S1 and S2).
Discussion
In this study of people with T1D prone to nocturnal severe hypoglycemia, treatment with insulin degludec resulted in a significant reduction of nocturnal CGM-recorded hypoglycemic events compared with treatment with insulin glargine U100. The difference between treatments was consistent between definitions of nighttime. The difference in nocturnal CGM-recorded hypoglycemia increased with increasing depth of hypoglycemia with RRR of 36%, 53%, and 58% at CGM ≤3.9 mmol/L, CGM ≤3.0 mmol/L, and at the lower detection limit of the CGM ≤ 2.2 mmol/L.
At CGM ≤3.0 mmol/L and at the lower detection limit, we also found significant reductions in daytime CGM-recorded hypoglycemia in favor of insulin degludec. The CGM ≤3.0 mmol/L threshold corresponds to level 2 hypoglycemia as defined by the International Hypoglycaemia Study Group as indicating serious clinically important hypoglycemia. 17
These analyses are complementary to our previously reported primary endpoint of the trial, based upon nocturnal symptomatic BGM-confirmed hypoglycemia. We found an RRR of 28% at BGM ≤3.9 mmol/L and 37% at BGM ≤3.0 mmol/L in nocturnal symptomatic hypoglycemia during treatment with insulin degludec and an RRR of 35% in all-day (24 h) severe hypoglycemia. 14 Our present findings in CGM-recorded hypoglycemia are consistent with these relative differences.
However, the complete capture of hypoglycemic exposure by CGM translates the consistent relative differences into much more clinically relevant absolute differences. At CGM ≤3.9 mmol/L and CGM ≤3.0 mmol/L, the TNT1 with insulin degludec to avoid an episode of nocturnal CGM-recorded hypoglycemia was 1.4 weeks at both thresholds of hypoglycemia.
Our data on CGM-recorded hypoglycemia, in addition to BGM data, provide evidence of a reduction of all types of nocturnal hypoglycemia with insulin degludec, as significant reductions in asymptomatic hypoglycemia drove the ARRs in nocturnal CGM-recorded hypoglycemia. As for nocturnal symptomatic hypoglycemia in the CGM data, we did not find significant reductions, as observed in the BGM data, 14 probably because of the overall observation period with CGM being much shorter than the 2 years of BGM measurements. Furthermore, the symptomatic events relied on notes in the participants’ diaries and, for some participants, they may have been insufficient due to participant response bias.
Insulin degludec and insulin glargine U100 have been compared based on BGM in three published studies in T1D. 10,11,13 Birkeland et al. 10 published a phase 2, 16-week randomized controlled open-label three-arm parallel-group multicenter study in 178 participants with T1D. The Basal-Bolus Type 1 trial (BEGIN) was a phase 3 open-label parallel-group treat-to-target multicenter study of 629 participants with T1D. 11,12 The SWITCH 1 Randomized Clinical Trial was a double-blind randomized cross-over noninferiority multicenter trial involving 501 participants with T1D. 13 The studies demonstrated relative reductions in nocturnal hypoglycemia with insulin degludec amounting to 10%–36%.
Comparisons of hypoglycemia rates are difficult due to the pooling of hypoglycemic events (symptomatic and severe hypoglycemia). 21,22 The incidence of hypoglycemic events is influenced by the titration regimens used, as aiming for low target fasting glucose values causes more hypoglycemic episodes in patients already at hypoglycemia risk. 21 –23 Furthermore, hypoglycemia was only a secondary endpoint, and CGM was not applied in the studies. The Birkeland and BEGIN studies excluded patients with previous major/severe hypoglycemia and hypoglycemia unawareness. 10 –12
The SWITCH 1 study included patients with at least one risk factor for hypoglycemia. In the study, 25% of the participants had a history of one episode or more of severe hypoglycemia in the preceding year. Twenty-one percent of the participants in the SWITCH 1 study had hypoglycemia unawareness. 13 In comparison, in our cohort 46% of the participants had more than one episode of severe hypoglycemia in the past 2 years, and the majority had impaired hypoglycemia awareness assessed by validated methods (Table 1).
Our present data expand the findings in the previous three studies 10 –13 and show even greater absolute reductions in rates of hypoglycemia with the use of insulin degludec when CGM is applied. The use of CGM is critical when it comes to evaluating actual hypoglycemic rates, 22 especially asymptomatic and nocturnal events.
We found that the reduction in nocturnal hypoglycemia during treatment with insulin degludec compared with insulin glargine U100 was primarily due to a reduction in asymptomatic events. This finding, which can be detected only by CGM, is important because a high frequency of asymptomatic hypoglycemia among all hypoglycemic events is a significant predictor of future severe hypoglycemia. 5
Miura et al. did a head-to-head comparison of insulin degludec versus insulin glargine U300. The trial was a randomized controlled cross-over study in 46 people with T1D, excluding participants with frequently recurring severe hypoglycemia within the last year.
As assessed by CGM, treatment with insulin degludec resulted in a significantly shorter duration of time below range <3.9 mmol/L. There were no differences between treatments on the duration of time below range <3.0 mmol/L. The frequency of hypoglycemic events confirmed by self-monitoring blood glucose did not differ between treatments. 24 The exclusion of high-risk participants, treat-to-target design, and relatively short duration of maintenance treatment makes the results difficult to compare with our study.
This study has several strengths. All participants had experienced at least one episode of nocturnal severe hypoglycemia within the past 2 years, and the majority had experienced more than two episodes. The long duration of run-in, cross-over, and maintenance periods approaches steady state and reduces the fluctuations in endpoints. By limiting the analysis of endpoints to the maintenance periods, we minimize any carry-over effects. The cross-over design makes it possible to compare the two treatments in the same person, thereby eliminating the interperson variability between treatment arms, particularly when analyzing hypoglycemic events with a skewed distribution.
This design also reduces the effect of confounding covariates. Another strength is that none of the participants used CGM, which makes the rates of hypoglycemia unmanipulated. Moreover, the CGMs were blinded and without alarms, eliminating bias in the data. The CGM periods each consisted of 6 days. Although not consecutive, we obtained a mean observation period of 11 days in each treatment arm per participant, which is close to the recommended 14 days of CGM data to estimate glycemic control. 25
The main limitation of our study is its open-label design. However, the limitation is minor in collecting blinded CGM data, especially asymptomatic hypoglycemia, as reporting bias is reduced. The precision of the interstitial glucose values recorded by CGM systems differs. The Enlite CGM system used in this substudy has a mean absolute difference in the hypoglycemic range of 0.4 mmol/L (glucose values 3.9–3.0 mmol/L) and 0.7 mmol/L (glucose values <3.0 mmol/L) in an unselected cohort.
In the same cohort, the CGM system showed 0.5 mmol/L higher values in the hypoglycemic range compared with BGM, 26 which could indicate that the incidence of asymptomatic hypoglycemia in our study is a conservative estimate. Future studies on CGM-recorded data should use the most accurate and available devices. We conducted the study from 2015 to 2017, and we used the best available CGM at the time.
The statistically significant reduction of nocturnal CGM-recorded hypoglycemia in our participants at all three hypoglycemia thresholds when treated with insulin degludec should lead to the consideration of changing the basal insulin therapy to insulin degludec in patients who experience difficulties with nocturnal hypoglycemia. Moreover, the results of our study call for future use of CGM in insulin trials to properly detect the actual occurrence of, especially asymptomatic, hypoglycemia.
Conclusion
In people with T1D prone to nocturnal severe hypoglycemia, treatment with insulin degludec, compared with insulin glargine U100, provides a clinically relevant relative and absolute reduction of nocturnal CGM-recorded hypoglycemia. CGM provides important supplementary glycemic information in clinical trials when added to conventional glycemic measures such as BGM and HbA1c.
Footnotes
Acknowledgments
We are grateful to the participants for their willingness and cooperation to participate in this trial. We thank the teams at each clinical site and at the Endocrine Research Unit at the Department of Endocrinology and Nephrology, Copenhagen University Hospital - North Zealand, Hillerød for their dedicated work. We also thank the team at the Clinical Research Unit at Copenhagen University Hospital - North Zealand, Hillerød for coordination and assistance during the trial.
Authors’ Contributions
U.P.-B., R.M.A., L.T., and B.T. initiated and designed the trial. R.M.A., A.C.A., H.U.A., H.B.-N., P.G., T.K.H., C.G.R.H., T.J.J., C.R.S., C.B.J., S.S.L., K.N., H.-H.P., L.T., B.T., and U.P.-B. participated in the coordination of the study and data collection. J.M.B.B. and R.M.A. planned and executed the statistical analyses. J.M.B.B. was responsible for the development of the article. All authors contributed to, read, and approved the final article.
Author Disclosure Statement
J.M.B.B., H.B.-N., T.K.H., C.G.R.H, T.J.J., C.R.S., S.S.L., H.-H.P., L.T., and B.T. have no competing financial interests. U.P.-B. has served on advisory boards for Novo Nordisk and Sanofi, and has received lecture fees from Abbott, Sanofi, and Novo Nordisk. A.C.A. and R.M.A. have been employed by Novo Nordisk A/S since September 2019 (after the finalization of the study). H.U.A. is on advisory boards for Abbott Laboratories, Astra Zeneca, and Novo Nordisk, has received lecture fees from Nordic Infucare and owns stock in Novo Nordisk.
P.G. has served on advisory boards for Abbott Laboratories, Astra Zeneca, Boehringer Ingelheim, Novo Nordisk, and Sanofi. C.B.J. serves on advisory boards for Novo Nordisk. K.N. serves as an advisor to Abbott Laboratories, Medtronic, and Novo Nordisk and has received fees for speaking from Bayer, Medtronic, Novo Nordisk, Roche Diabetes Care, Rubin Medical, Sanofi, and Zealand Pharma, and owns stock in Novo Nordisk.
Funding Information
Novo Nordisk A/S funded this study by an unrestricted grant and supply of study medications. The study funder was not involved in the study's design, data collection, data analysis, or interpretation of the data. Nor was the funder involved in writing the report and did not impose any restrictions regarding publication of the report.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
