Abstract
Background:
Severe hypoglycemic episodes are life-threatening events demanding rapid administration of glucagon by a caregiver or bystander. The glucagon analog dasiglucagon is stable in aqueous formulation and therefore suitable for delivery in a ready-to-use autoinjector, potentially increasing speed and ease of use compared with standard glucagon emergency kits (GEKs).
Methods:
In an open label, randomized, crossover, comparative device handling study, trained caregivers and untrained bystanders administered the dasiglucagon autoinjector or Eli Lilly GEK to manikins in a simulated emergency hypoglycemia situation.
Results:
In total, 54 participants were randomized (18 patient-caregiver pairs and 18 bystanders). Overall, 94% of trained caregivers were able to administer the dasiglucagon autoinjector successfully within 15 min, compared with 56% for the GEK (P < 0.05). A greater proportion of trained caregivers and untrained bystanders successfully prepared and administered the dasiglucagon autoinjector within 2 min compared with the GEK (P < 0.005 and P < 0.05, respectively). Time to successful completion was also significantly faster with the dasiglucagon autoinjector than with the GEK (P < 0.005 for both groups). Most study participants preferred the dasiglucagon autoinjector over the GEK (94%, P < 0.001) and rated it as easier (90%, P < 0.001) and less stressful to use (94%, P < 0.001) than the GEK.
Conclusion:
Dasiglucagon autoinjector was more rapidly and reliably administered, and users reported greater ease of use and usage satisfaction than with the GEK. Thus, dasiglucagon autoinjector has the potential to improve speed and ease of treatment in severe hypoglycemic events, providing a better usage experience for rescuing individuals and enabling faster recovery for patients.
Introduction
Hypoglycemia is an episode of abnormally low plasma glucose concentration that exposes an individual to potential harm. 1 The most common cause of hypoglycemia is treatment with diabetes medications such as sulfonylurea, glinide, or insulin, and it is most likely to occur in patients with profound endogenous insulin deficiency, such as type 1 diabetes (T1D) and type 2 diabetes (T2D) after prolonged therapy. 2 Other causes of hypoglycemia include congenital hyperinsulinism, hormonal deficiencies, pancreatic tumors, and kidney or hepatic failure. 3 Acute hypoglycemia can lead to confusion, loss of consciousness, seizures, and death. 1,4 Long-term cohort studies have reported 4%–9% of deaths in patients with T1D to be caused by hypoglycemia. 5 –7
Severe hypoglycemia (SH), also called level 3 hypoglycemia, is defined as a severe event characterized by altered mental and/or physical functioning that requires assistance from another person for recovery. 1,8 The American Diabetes Association (ADA) recommends prescribing glucagon for all individuals at an increased risk of level 2 (blood glucose <54 mg/dL) and level 3 hypoglycemia, so that it is available if needed. 8 The guidance specifically states that glucagon administration is not limited to health care professionals, but that family members, caregivers, and school personnel of the affected individual should know when and how to administer it. 8 An SH event is an emergency situation that can necessitate use of rescue medication administered to the patient by family members, caregivers, or even bystanders.
The majority of glucagon prescribed and used for SH events are glucagon emergency kits (GEKs), such as GlucaGen® HypoKit (Novo Nordisk) and Glucagon for Injection (Eli Lilly). 9 These GEKs require reconstitution of lyophilized glucagon powder immediately before use, requiring a caregiver to be trained to carry out multiple steps when administering the treatment. The complexity of the multistep process is a known barrier to timely and effective administration, and lack of training and confidence in the use of GEKs may delay or prevent glucagon delivery. 10 –13 Consequently, there are significant data demonstrating that glucagon is both underprescribed and underused. 10,12,14,15
Fear of hypoglycemia among individuals with diabetes is widespread, and may have a negative impact on diabetes management, glycemic control, and subsequent health outcomes. 16 Similarly, caregivers of people with diabetes may feel concerned about administering glucagon during an emergency for fear of harming the patient. 11,17 This may be related to the fact that timely and accurate administration is a crucial part of the rescue procedure. Rapid, intuitive, easy to use methods of administering glucagon rescue therapy may help to alleviate anxiety about hypoglycemia for both patients and caregivers and increase effective utilization in emergency situations. 11
Ready-to-use glucagon products for subcutaneous injection (Gvoke® liquid formulation in the organic solvent dimethyl sulfoxide; Xeris Pharmaceuticals) and intranasal dry powder administration (Baqsimi™; Eli Lilly) have recently become available. 9 The novel glucagon analog dasiglucagon (Zegalogue®; Zealand Pharmaceuticals) was recently approved for the treatment of SH in adults and children ≥6 years of age with diabetes and is the first glucagon product to be provided in a ready-to-use aqueous formulation. 18 The increased physical and chemical stability of dasiglucagon in aqueous solution compared with native human glucagon eliminates the need for reconstitution before injection, and, as such, dasiglucagon is suitable for delivery in a ready-to-use autoinjector as well as in a prefilled syringe. 18
Autoinjectors are generally easier to use and allow for accurate and safer handling than conventional syringe and vial solutions for delivering medications, thus increasing confidence in patients and caregivers to use the device. 19 Therefore, dasiglucagon is anticipated to facilitate rapid and easier administration in both of its delivery devices, and to increase patient and caregiver confidence during a hypoglycemic emergency. The aim of the current study was to assess the speed, accuracy, and participant experience in using the dasiglucagon autoinjector (Fig. 1A) compared with using a commercially available GEK (Eli Lilly (Fig. 1B)), during simulated hypoglycemic events.
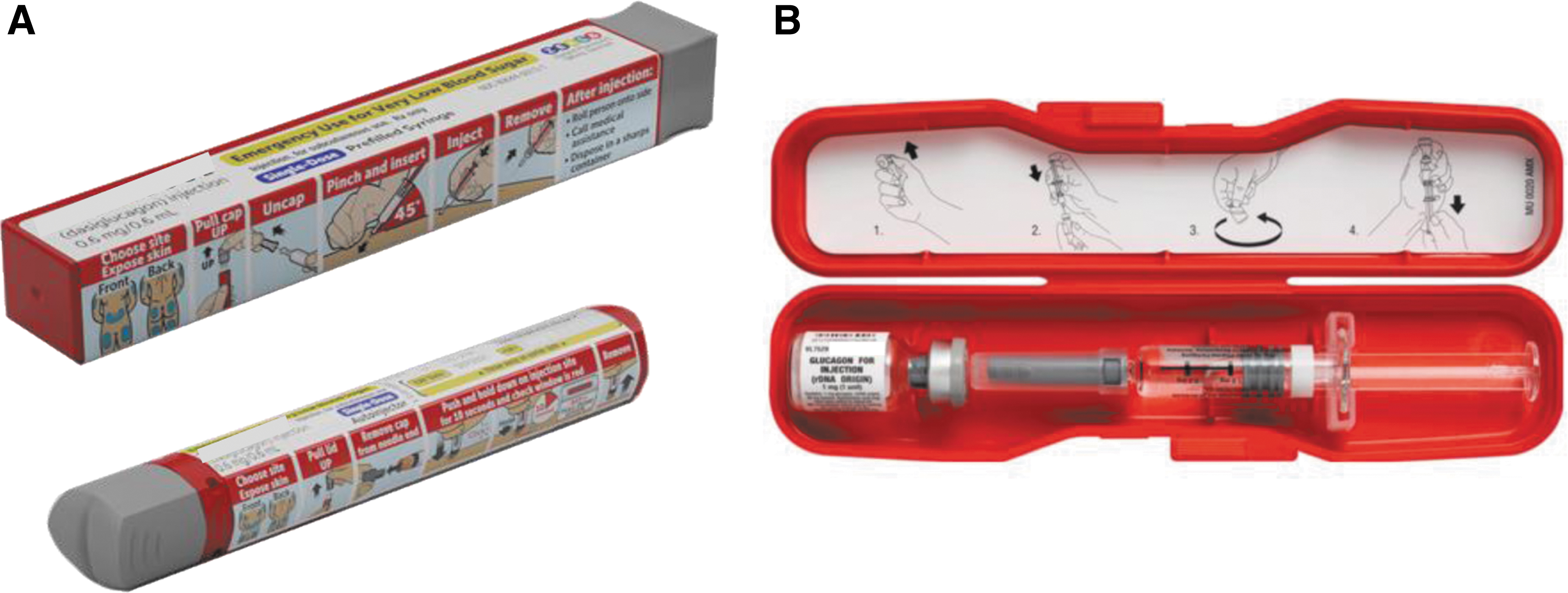
Methods
Participants, interventions, and study design
This was an open label, randomized, crossover, comparative device handling study conducted at the AMCR Institute, Inc., in Escondido, California, U.S., between November 23, 2020 and December 18, 2020. All study participants provided written informed consent approved by the Salus Institutional Review Board (IRB), approval number 20-122, and were recruited from a proprietary database of volunteers in accordance with IRB-approved processes. The study was conducted under simulated-use conditions on a manikin, thus avoiding the use of glucagon rescue treatment on human participants.
Study participants ≥18 years of age were recruited into three groups: (1) people with T1D or T2D, (2) caregivers (close friends or relatives of the people with diabetes) without any prior experience with glucagon or rescue medications; and (3) adults without any medical training or experience with diabetes, glucagon, or rescue medications, hereafter called “bystanders.”
Figure 2A and B illustrate the study design and flow of participants through the study. Within each group, individuals were randomized to first use dasiglucagon autoinjector or lyophilized powder GEK, and then the procedures were repeated with the other device. In group 1, patients were trained by study staff on the first device to which they were randomized, and ∼30 min later patients trained their corresponding caregivers on the device.
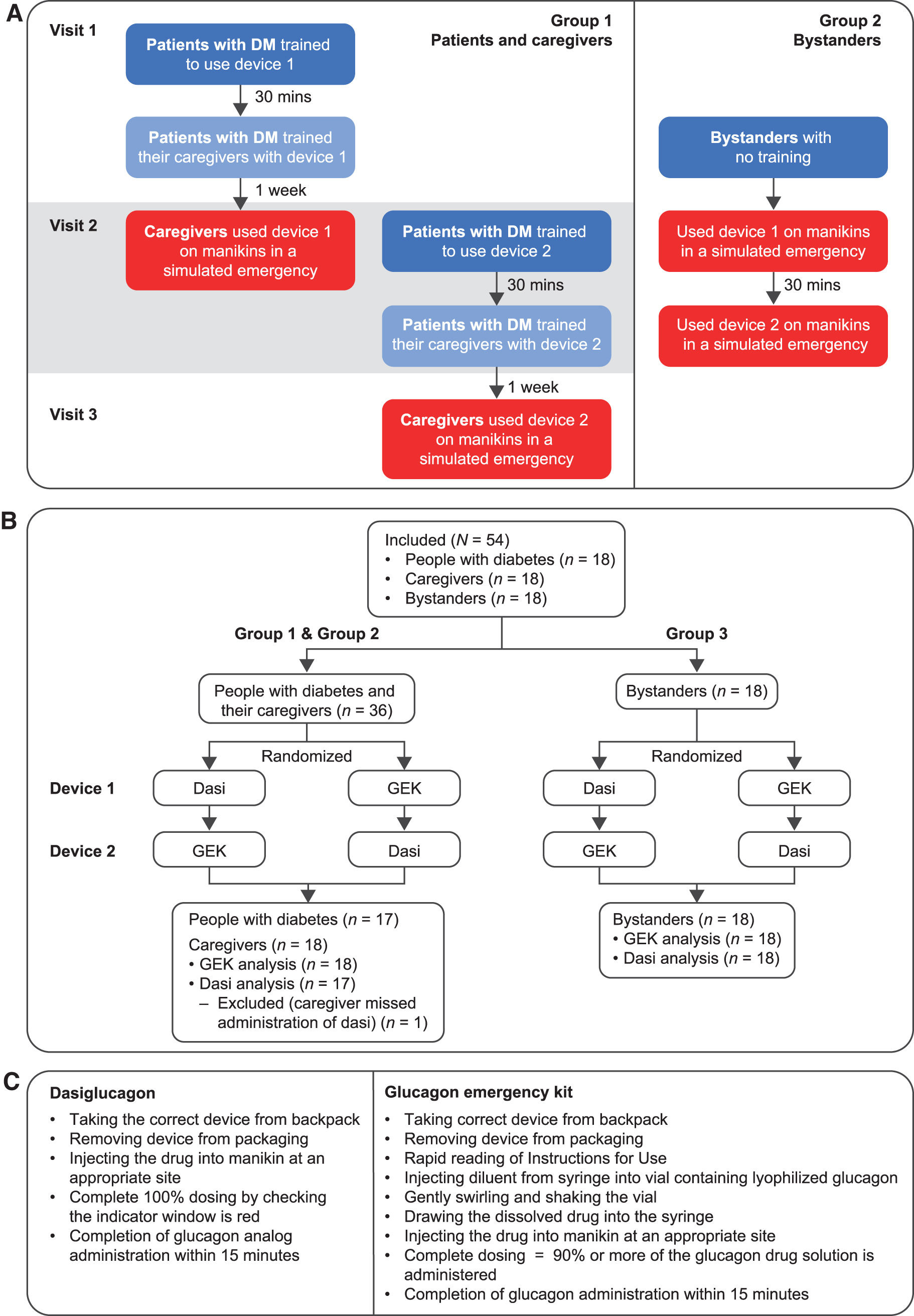
One week later, to allow for memory decay, caregivers used the device on a manikin during a simulated hypoglycemic emergency. At the same visit, patients were trained on the alternate device and subsequently trained their caregivers. One week later, caregivers used the second device on a manikin in a simulated hypoglycemic emergency. In group 2, bystanders used both devices on a manikin during a simulated hypoglycemic emergency without prior training on the same day, in the order in which they were randomized to them, with a 30-min break in between.
A constant loud sound of sirens was played during the simulations to mimic the stress of a real-life emergency situation. Three staff members administered and recorded the test data during the simulations. In the simulation room, a test administrator monitored activity using a video recorder and a data logger inputted scores and comments related to the simulations. In a remote room, a test observer viewed the testing sessions in real-time through a computer link to the video and identified problems, concerns, and procedural errors.
Evaluations
The parameters used to monitor task completion are listed in Figure 2C, and included taking the correct device from the backpack, correct removal of packaging, injecting the drug into an appropriate site on the manikin, administering a complete dose, and completing the task within 15 min for both devices. For the GEK, the additional tasks of injecting the diluent from the syringe into the vial, gently swirling the vial to mix, and drawing the glucagon mixture into the syringe were also assessed. Timing of task completion was recorded.
At the end of the study, following completion of emergency simulations with both devices, caregivers and bystanders completed two questionnaires. The first questionnaire asked them to compare their experiences with each of the devices and to answer eight questions about their preferences (indicating whether they favored dasiglucagon, GEK, both, neither, or that the question did not apply to them). The second questionnaire asked about features of the individual devices using Likert scale scores to assess responses (1 = very difficult; 2 = somewhat difficult; 3 = neither easy nor difficult; 4 = fairly easy; 5 = very easy).
Statistical analysis
All analyses and figures were derived using SAS software version 9.4. Caregiver and bystander groups were either compared directly or compared using a nonparametric paired test. Paired success rates and success within 1 and 2 min were analyzed using the McNemar's test. Paired time-to-success was assessed using the signed rank test, and log-transformed time-to-success was assessed using a two-way log-normal model with random participant effect. The binary participant-level preference proportions were treated using an exact binomial test of the P = 0.5 fifty-fifty null-hypothesis. The Likert scale questionnaire data were tested for difference within participant strata, using the Friedman rank test.
Results
Study participants
Overall, 18 participants per group (i.e., 18 patient-caregiver pairs and 18 bystanders) were recruited to the study, giving a total of 54 participants (Fig. 2B). One patient was withdrawn after the initial training owing to an unrelated adverse event. Their caregiver was trained on both devices but missed the dasiglucagon autoinjector administration and completed the GEK administration only and was therefore excluded from the dasiglucagon analysis (n = 17). Baseline characteristics of the study population are shown in Table 1.
Baseline Characteristics of the Study Population
T1D, type 1 diabetes; T2D, type 2 diabetes.
Administration success rate and completion time
Figures 3A–C show overall success rates (within 15 min), rates of completion within 1 and 2 min, and average completion times for either dasiglucagon or the GEK in each group of study participants. Overall, 94% (16/17) of the trained caregivers were able to administer the dasiglucagon autoinjector successfully within 15 min when treating a simulated episode of SH, compared with 56% (10/18) for the GEK (P < 0.05). Within 1 min, 63% (10/16) of the trained caregivers were able to successfully administer the dasiglucagon autoinjector, whereas none (0/10) of the caregivers achieved this with the GEK (P < 0.005). Within 2 min, 88% (14/16) of trained caregivers were able to successfully administer the dasiglucagon autoinjector; 40% (4/10) accomplished this for the GEK (P < 0.005) (Fig. 3B). The mean completion time for trained caregivers was 75 s (median 51 s) for the dasiglucagon autoinjector and 133 s (median 126 s) for the GEK (P < 0.005) (Fig. 3C).
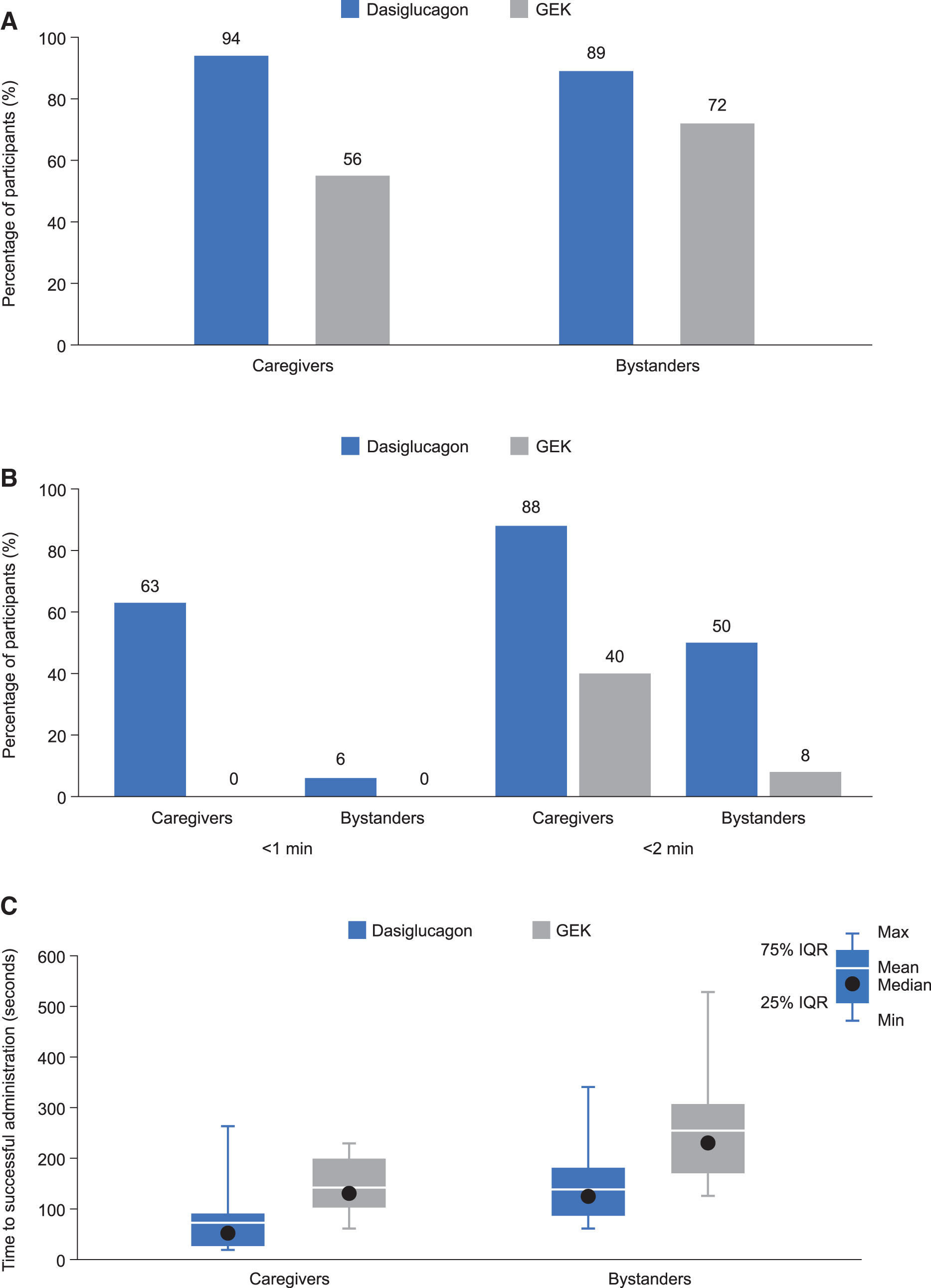
Among untrained bystanders, successful administration was achieved within 15 min by 89% (16/18) for the dasiglucagon autoinjector and 72% (13/18) for the GEK (P = not significant). Of those bystanders who successfully administered either device, 6% (1/16) successfully administered the dasiglucagon autoinjector within 1 min and none (0/13) administered the GEK at the same time (0%, P < 0.05). Within 2 min, 50% (8/16) of these bystanders administered the dasiglucagon autoinjector successfully compared with 8% (1/13) for the GEK (P < 0.05) (Fig. 3B). The mean completion time in this group was 137 s (median 116 s) for the dasiglucagon autoinjector and 251 s (median 227 s) for the GEK (P < 0.005) (Fig. 3B).
Study task success rate
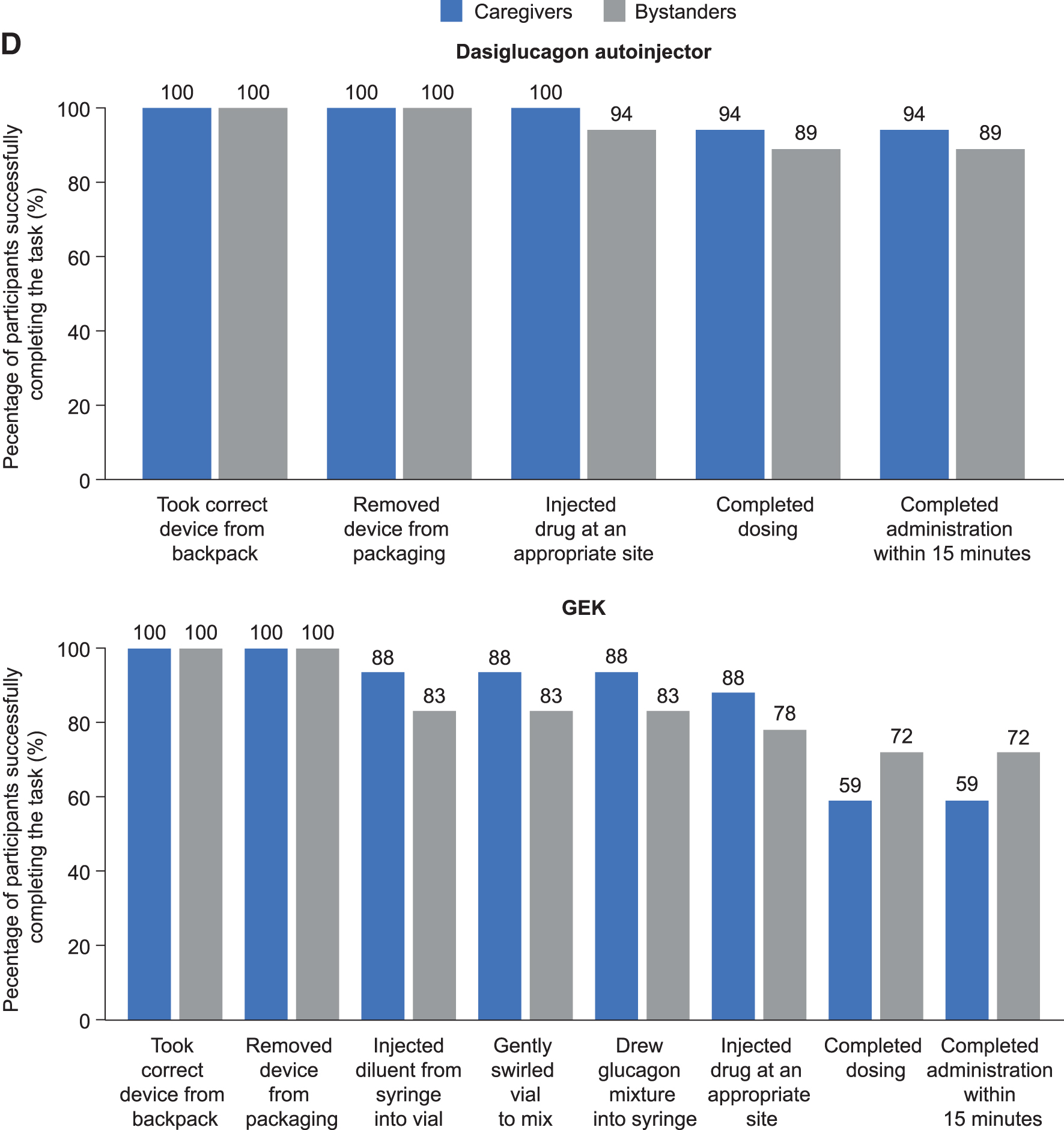
A total of 35 participants (17 caregivers and 18 bystanders) were rated by the study team on how successfully they were able to complete each of the study tasks involved in administering the two devices (outlined in Fig. 2C). The results of this evaluation are shown in Figure 3D. Overall, at least 94% of the trained caregivers and at least 89% of the untrained bystanders were able to successfully complete all tasks involved in administering the dasiglucagon autoinjector. All participants (100% [17/17] of caregivers, 100% [18/18] of bystanders) took the correct device from the backpack and removed it from packaging, 100% (17/17) of caregivers and 94% (17/18) of bystanders were able to inject the dose at the appropriate site on the manikin, and 94% (16/17) of caregivers and 89% (16/18) of bystanders were able to complete the dosing and completed administration within 15 min (Fig. 3D).
For the GEK, all participants (100% in both groups) were able to remove the device from the backpack and remove the packaging, and many of the participants (88% [15/17] of caregivers and 83% [15/18] of bystanders) successfully completed the tasks involved in reconstituting and drawing the glucagon into the syringe. However, although a considerable proportion (88% [15/17] of caregivers and 78% [14/18] of bystanders) were able to inject the glucagon at an appropriate site on the manikin, only 59% (10/17) of caregivers and 72% (13/18) of bystanders were able to complete the dosing and complete the administration within 15 min.
User preference questionnaire
A total of 52 participants (17 patients, 17 caregivers, and 18 bystanders) completed the user preference questionnaire. Results are shown in Figure 4. For all categories, the dasiglucagon autoinjector was preferred to the GEK by most participants. Overall, 94% (49/52) preferred the dasiglucagon autoinjector and 6% (3/52) preferred the GEK (P < 0.001). Most participants (90%, 47/52) considered the dasiglucagon autoinjector “easy to use from beginning to end,” 2% (1/52) found the GEK easier, 4% (2/52) rated them as equally easy, and 4% (2/52) did not answer this question (P < 0.001 for overall differences in values).
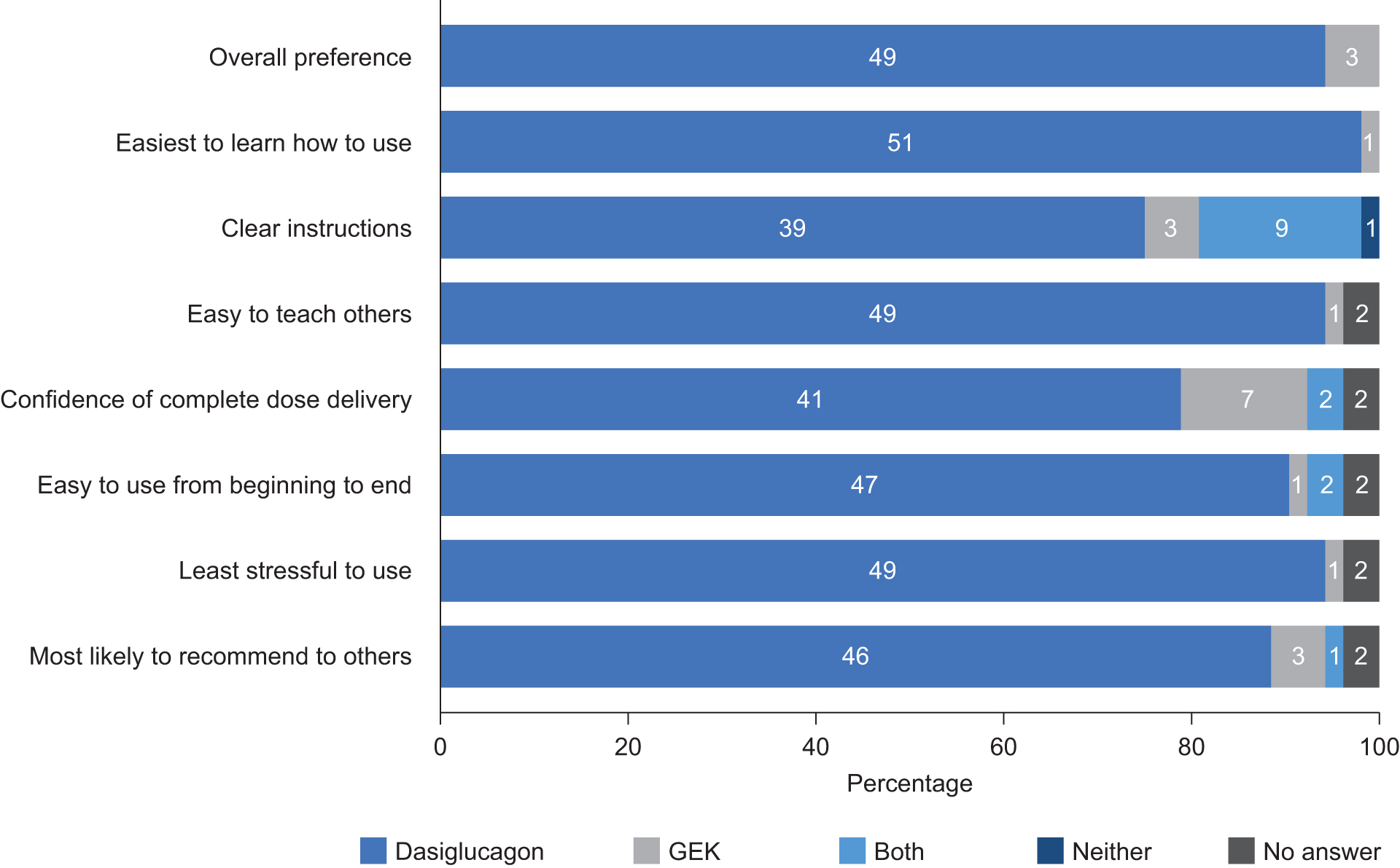
Device preferences among participants (n = 52) who completed the study.
Individual device questionnaire
A total of 35 participants (17 caregivers and 18 bystanders) completed the individual device questionnaire. The participants' average responses to questions on each of the tasks involved in administering the devices are shown in Figure 5. For five out of the six tasks, the participants rated the dasiglucagon autoinjector as easier to use than the GEK. The dasiglucagon autoinjector received an average Likert scale score of ≥4.5 (4 = fairly easy, 5 = very easy) for the tasks involving the preparation of the device, holding the device stable while injecting, administering the injection, recalling all steps, and following the instructions.
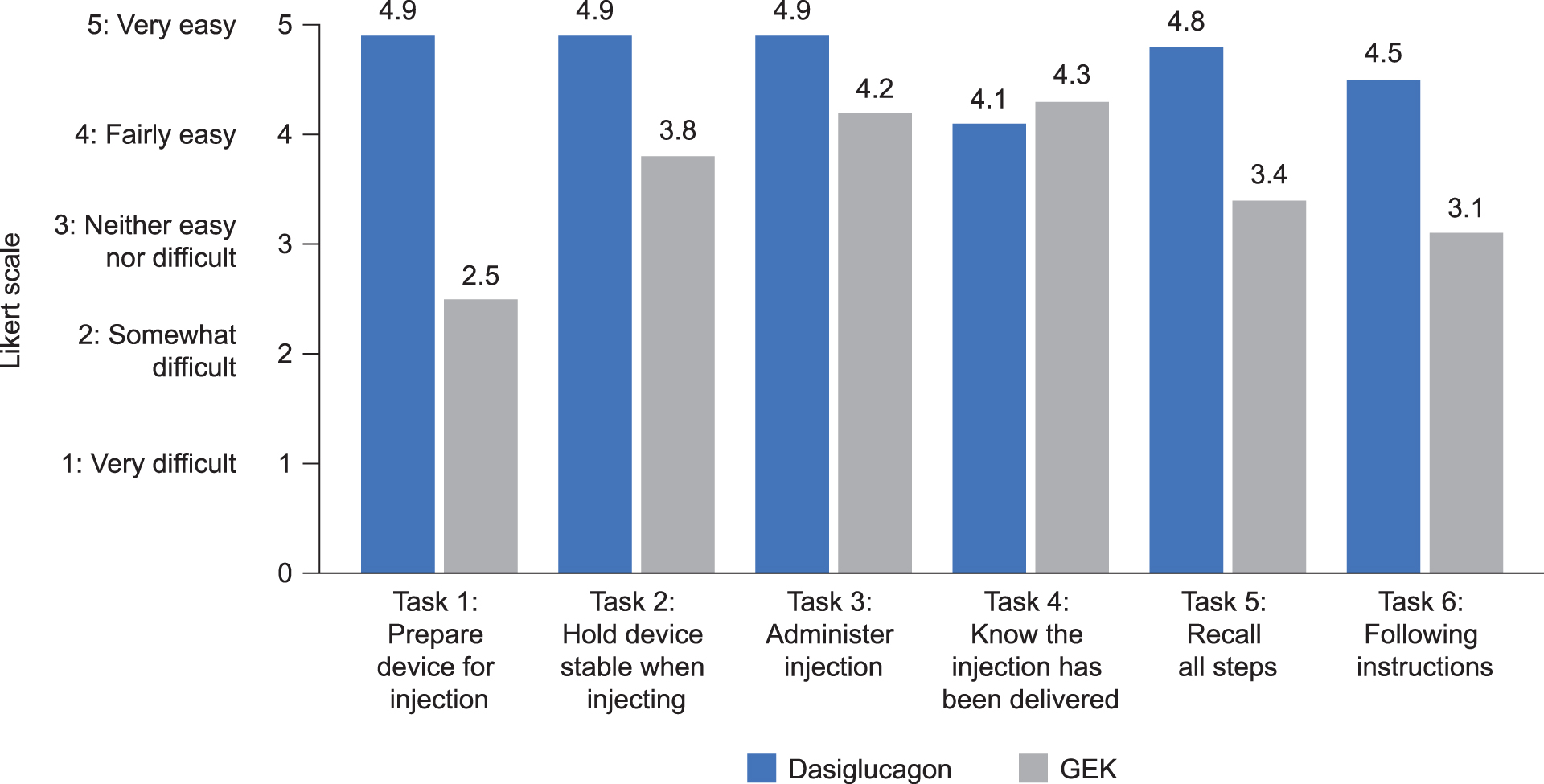
Mean caregiver and bystander ratings (n = 35) in the individual device questionnaire across six device handling tasks (1 = preparing the device for injection, 2 = holding the device stable when injecting, 3 = administering the injection, 4 = knowing the injection has been delivered, 5 = recalling all steps, and 6 = following the instructions) using Likert scale scores to assess responses (1 = very difficult; 2 = somewhat difficult; 3 = neither easy nor difficult; 4 = fairly easy; and 5 = very easy).
In comparison, the GEK received average Likert scale scores of 2.5, 3.1, and 3.4 (2 = somewhat difficult, 3 = neither easy nor difficult, 4 = fairly easy) for preparing the device, following the instructions and recalling all steps, respectively, whereas the tasks involving holding the device stable and administering the injection received average scores of 3.8 and 4.2, respectively. The task of knowing that the injection was delivered was rated with an average score of 4.1 for the dasiglucagon autoinjector and 4.3 for the GEK.
Discussion
The aim of this study was to compare the successful administration of the dasiglucagon autoinjector with a commercially available GEK among trained caregivers and untrained bystanders when treating a simulated episode of SH. The stability of the glucagon analog dasiglucagon in aqueous solution eliminates the need for reconstitution before injection, making it suitable for delivery in a ready-to-use autoinjector and prefilled syringe. 18 Dasiglucagon has been shown to provide rapid and consistent reversal of hypoglycemia in adults with T1D with a significantly faster time to plasma glucose recovery (median of 10 min) when compared with a placebo (median of 40 min). 18
This study expands on the potential for dasiglucagon to provide significant benefit for people with diabetes and their caregivers, demonstrating superior ease and speed of use of the dasiglucagon autoinjector over traditional GEK during a simulated emergency hypoglycemic situation. Trained caregivers showed a significantly greater success rate with the dasiglucagon autoinjector than with the GEK (P < 0.05), and a greater proportion of both trained caregivers and untrained bystanders successfully prepared and administered the dasiglucagon autoinjector within 2 min compared with the GEK (P < 0.005 and P < 0.05, respectively). Within 1 min, 63% of the trained caregivers were able to successfully administer the dasiglucagon autoinjector, whereas none of the caregivers achieved this with the GEK (P < 0.005). Time to successful completion was also significantly faster with the dasiglucagon autoinjector than with the GEK (P < 0.005 for both groups).
The advantages of the dasiglucagon autoinjector over the GEK observed in this study are similar to the observations reported in a similarly designed study of nasal glucagon versus GEK, 20 adding to the evidence base supporting use of next-generation ready-to-use glucagon rescue therapies. In addition, the successful administration rate of dasiglucagon by caregivers (94%) is similar to the rates reported for other ready-to-use glucagon formulations, with 94% of caregivers successfully administering a nasal glucagon and 100% of caregivers successfully administering a subcutaneous injection of glucagon. 20,21 Future studies are, however, needed to directly compare the administration rates of these ready-to-use glucagon rescue therapies.
The advantages demonstrated for the dasiglucagon autoinjector are likely to have clinical implications on time to administration, caregiver training needs, and reduction of anxiety associated with the use and correct administration of rescue glucagon. The superior ease of use and speed of administration support a much broader use of emergency rescue therapy for SH, which still constitutes a common emergency in people with diabetes.
A recent survey of 264 people with T1D and 58 caregivers demonstrated that 65% of patients reported experiencing a severe hypoglycemic event, while only 51% received glucagon despite a GEK being in proximity. 12 Only 18% of the 90 patients who received rescue glucagon reported that it was administered without difficulties. When the GEK was not administered, the reasons for this omission included use of oral treatment, and the rescuing individual being untrained, being unable to use the kit correctly, or being too fearful to use the kit. 12 Nearly a third of patients reported not receiving training on using the GEK. 12
Similarly, in a study of 136 caregivers of pediatric patients with T1D, all caregivers reported having only received verbal instructions on how to administer a GEK, with no hands-on experience reported. 10 When they were called upon to administer the GEK in a simulated hypoglycemic event in the study, 69% of caregivers experienced handling difficulties, which included opening the pack, sheath removal, mixing, and bending the needle. 10 This points toward a potential shortfall in the effectiveness of the training that caregivers typically receive. The superiority of dasiglucagon autoinjector administration over GEK administration by caregivers in the current study, in which caregivers were trained by patients rather than directly by health care professionals, suggests that the reduced complexity of the dasiglucagon autoinjector may reduce training needs. Furthermore, administration of the dasiglucagon autoinjector was faster and more accurate than administration of the GEK, even when given by bystanders with no training at all.
The differences in the speed of administration between the two devices are notable considering the importance of rapid administration of rescue therapy in an emergency situation to limit the risk of neurologic sequelae. 21 We found the mean time for preparation and administration of injectable glucagon to be longer than what was previously reported, 20 adding to the overall recovery time for patients. Furthermore, SH events are known to be very stressful for the rescuing individual, and the complexity of GEK preparation and administration has been reported to be challenging and intimidating for many caregivers; itself leading to the potential for delays in treatment. 12
The current study also found that the dasiglucagon autoinjector was preferred over the GEK by a majority of study participants (94%, P < 0.001), and that most study participants rated it as easier to use (90%, P < 0.001) and less stressful to use (94%, P < 0.001) than the GEK. Indeed, the faster administration observed with the dasiglucagon autoinjector than with the GEK may be related to reduced stress on the caregiver or bystander administering the treatment in a ready-to-use, autoinjector preparation.
Use of autoinjectors instead of GEKs may also have considerable health economic impacts. Severe hypoglycemic events in patients with diabetes are associated with frequent emergency services utilization and considerable health care cost for payers in the United States. 22 Recent economic models of the annual value of novel glucagons, including nasal and read-to-use injectable glucagon, showed reduced utilization of emergency medical services, emergency departments, and inpatient and outpatient costs for the treatment of severe hypoglycemic events compared with lyophilized powder GEK in the United States, suggesting that these novel glucagons can lead to significant annual cost savings for payers. 22,23
This was a randomized crossover study of robust design, similar to a recent comparative study of nasal glucagon and injectable glucagon. 20 The incorporation of three different stakeholder groups, two of whom administered treatment, allowed for a comprehensive investigation of how the dasiglucagon autoinjector might be used in both trained and untrained individuals, who might be called upon to provide glucagon rescue in real life. Care was taken to attempt to simulate the stress of a real-life emergency. However, because this was a simulation, it was not possible to fully replicate the fear and emotional distress a caregiver or bystander would likely feel in this situation. It is also likely that participants experienced less stress when using the second device because they had already been through one simulation, although the crossover design aimed to mitigate this effect.
Other limitations of the study include the use of a manikin instead of people. While the fact that caregivers were trained by patients and not health care professionals might seem to be a disadvantage, this more closely approximates what occurs in clinical practice. In addition, the participant groups were not completely equal in terms of demographics, because the bystander group had a much lower median age than the patient and caregiver groups. In addition, they were observed to have spent more time reading the instructions than the participants in the other groups, presumably because they had not received any training. Finally, it should be noted that the emotional impact of witnessing a severe hypoglycemic event may be more severe on caregivers than bystanders, due to their previous concern about a severe hypoglycemic event as well as their closeness to the patient.
Conclusions
This study showed that the dasiglucagon autoinjector is more easily, rapidly, and reliably administered than the current GEK, and that the autoinjector provides a better user experience. Compared to the current GEK, the dasiglucagon autoinjector offers the potential to enhance speed of treatment by reducing the complexity of preparation and administration. The ready-to-use device, delivering a stable aqueous formulation of a novel glucagon analog, may further enhance the ability of the rescuing individual to deliver essential treatment in emergency situations quickly and efficiently, potentially improving the patient's time to recovery from a severe hypoglycemic event.
Footnotes
Authors' Contributions
N.C.B. and T.S.B. developed the study protocol, collected the data, and analyzed the data. J.D., M.H. and D.M.K. contributed to the study design and data analysis. All authors critically revised the article drafts for intellectual content and gave final approval of the version to be submitted.
Acknowledgments
Medical writing and editorial assistance for this article were provided by Claudia Brockmeyer, PhD, Sarah Griffiths, PhD, and Jane Francis of Oxford PharmaGenesis (Oxford, United Kingdom).
Author Disclosure Statement
J.D., M.H., and D.M.K. are employees and shareholders of Zealand Pharma A/S.
T.S.B. received research support (independent of the submitted work) from Abbott Diabetes, Abbott Rapid Diagnostics, Biolinq, Capillary Biomedical, Dexcom, Eli Lilly, Kowa, Lexicon, Livongo, Medtronic, Novo Nordisk, REMD, Sanofi, Sanvita, Senseonics, Viacyte, vTv Therapeutics, and Zealand Pharma; consulting honoraria (independent of the submitted work) from Abbott, LifeScan, Medtronic, Novo Nordisk, and Sanofi; and speaking honoraria (independent of the submitted work) from BD, Medtronic, and Sanofi.
Funding Information
This study was funded by Zealand Pharma A/S. Medical writing assistance and publication of this research were supported by Zealand Pharma A/S.
