Abstract
Objective:
Several immunotherapies have shown efficacy in slowing C-peptide decline in new-onset type 1 diabetes. Although most of these biologic drugs are expensive, they offer the opportunity to reduce downstream disease management costs and risk of complications. The objective of this study is to examine the cost-effectiveness of immunotherapies versus no treatment for patients with new-onset type 1 diabetes.
Methods:
Using Markov microsimulation modeling and efficacy data from immunotherapy trials, we examined the cost-effectiveness of six immunotherapies for new-onset type 1 diabetes, namely, low-dose (2.5 mg/kg) antithymocyte globulin (ATG), high-dose (6.5 mg/kg) ATG, abatacept, alefacept, rituximab, and teplizumab, versus no treatment. Effectiveness was measured by quality-adjusted life-years (QALYs). Costs were estimated from a health system perspective.
Results:
Low-dose ATG treatment saves US$10,270, on average, over a patient's lifetime and generates 0.09 additional QALYs compared with no treatment. These cost savings arise as low-dose ATG generates downstream savings in disease management costs that more than offset its cost. In contrast, treatment with other immunotherapies yields smaller QALY gains (0.02–0.05 additional QALYs) and increases lifetime costs by US$9500–US$168,380 relative to no treatment, with incremental cost-effectiveness ratios that exceed the willingness-to-pay threshold of US$100,000 per QALY.
Conclusions:
Low-dose ATG treatment is both less costly and more effective relative to other immunotherapies and no treatment for new-onset type 1 diabetes.
Introduction
Type 1 diabetes is estimated to affect ∼1.6 million individuals in the United States, 1 with >60,000 individuals diagnosed with the disease annually. 2 The annual health care costs attributed to type 1 diabetes are estimated at >US$14.4 billion. 3 Furthermore, with rising insulin costs, insulin treatment is increasingly becoming inaccessible to thousands of Americans. 4
Several immunotherapies have recently demonstrated the ability to transiently preserve C-peptide among patients with new-onset type 1 diabetes. 5 Among these, low-dose antithymocyte globulin (ATG) and teplizumab have been found to be the most effective with 103% and 63% increase in C-peptide preservation at the end of 2 years, respectively, relative to no treatment. 5 Consequently, these therapies offer the potential to slow disease progression and reduce risk of downstream complications.
From a cost perspective, greater C-peptide preservation from immunotherapy treatments implies savings in day-to-day costs of managing type 1 diabetes. However, immunotherapies themselves are expensive. Per capita costs of these drugs range from US$5000–US$7000 for low-dose ATG to anticipated cost of as much as US$100,000 for teplizumab. 6,7 Adoption of these drugs into clinical care, therefore, requires an assessment of not only their efficacy but also their cost-effectiveness.
The objective of this study is to examine the cost-effectiveness of six immunotherapies for new-onset type 1 diabetes, namely, teplizumab, low-dose ATG, high-dose ATG, abatacept, alefacept, and rituximab, relative to no treatment. Findings from this study provide important insights for both policymakers and clinicians, especially as the first immunotherapy (teplizumab) has already been designated as a “breakthrough therapy” by the U.S. Food and Drug Administration (FDA). 8 Teplizumab is nearing potential approval as an agent to delay progression from stage 2 to stage 3 type 1 diabetes, and is undergoing a phase-3 clinical trial in patients with new-onset type 1 diabetes.
Methods
Model structure
We used a Markov microsimulation model to estimate the cost and effectiveness of seven treatment strategies: (1) teplizumab, (2) low-dose ATG (2.5 mg/kg), (3) high-dose ATG (6.5 mg/kg), (4) abatacept, (5) alefacept, (6) rituximab, and (7) no treatment. Although alefacept and high-dose ATG did not meet their primary clinical trial endpoints, we included these immunotherapies as potential comparators to low-dose ATG in our analysis as alefacept still met its preplanned 2-year endpoint, whereas post hoc analyses showed that high-dose ATG was effective in young adults.
Treatment dosing and regimen for each immunotherapy are provided in Table 1. The microsimulation model, which was adapted from the Sheffield Type 1 Diabetes Policy Model, 9 simulates a hypothetical cohort of 1000 patients aged 12–43 years 10 and newly diagnosed with type 1 diabetes from diabetes to diabetes-related complications and death.
Immunotherapy Dosage, Costs, and Efficacy
Converted at 1 USD = 1.259 CAD as on October 6, 2004. 44
Percentage effects between treated and control groups based on the cross-trial comparison. 5
ATG, antithymocyte globulin; AUC, area under the curve; NA, not applicable.
All newly diagnosed patients entered the model with type 1 diabetes but without diabetes-related complications. Disease progression in our model was captured through reduction in C-peptide (area under the curve [AUC]) measured by a mixed meal tolerance test, and associated increases in HbA1c. Each patient entering the model was thus assigned a starting value for C-peptide AUC and an associated HbA1c value. These starting values were drawn from distributions mirroring that of participants across five TrialNet type 1 diabetes intervention studies. 11 Over time, rate of decline in C-peptide AUC and rate of increase in HbA1c in the absence of treatment followed the average rates observed in the TrialNet study. 11 Patients receiving immunotherapy treatment experienced smaller declines in C-peptide and thus, smaller increases in HbA1c (as detailed hereunder).
Driven primarily by their HbA1c, patients with type 1 diabetes faced risks of microvascular complications (neuropathy, nephropathy, and retinopathy), macrovascular complications (myocardial infarction, cerebrovascular accident, and angina pectoris), diabetic ketoacidosis, and/or hypoglycemia. 9 Death could occur due to cardiovascular events, end-stage renal disease, or causes unrelated to diabetes. 9 The model was run for a lifetime horizon and each cycle lasted 1 year. All analyses were performed using TreeAge 2020 v2.1. 12
Model inputs
C-peptide AUC and HbA1c dynamics in the absence of immunotherapy
Percentage reduction in C-peptide AUC over the first 4 years was assumed to equal that in the observational TrialNet study of C-peptide decline in new-onset type 1 diabetes patients. 11 Beyond year 4, we assumed that rate of decline equaled that between years 2 and 4. In the TrialNet observational study, HbA1c values increased during years 1–4 for patients aged 12–17 years. Meanwhile, for those aged 18 years, HbA1c declined in the first 2 years and stabilized in years 3–4. Using these rates of change in HbA1c, for ages 12–17 years in our model, we, therefore, assumed that HbA1c increased in years 1–4, declined in years 5–6 (at the rate for age ≥18 years), and remained at the level achieved at the end of year 6 thereafter. For ages 18 years and above, HbA1c declined in years 1–2 and stabilized at that level thereafter.
Efficacy of drugs and effects on HbA1c and insulin needs
Efficacy of each drug was captured by the percentage improvement in C-peptide AUC levels. 5 Greater improvement in C-peptide AUC implied lower HbA1c levels, which, in turn, meant lower risk of diabetes-related complications, better quality of life, and lower downstream complication costs. Specifically, we assumed that 0.1 pmol/mL increase in C-peptide AUC as a result of immunotherapy led to 0.1% decline in HbA1c. 13 Data on drug efficacy from clinical trials of immunotherapies were available for a maximum duration of 2 years. Therefore, in the base case analysis, we assumed that drug efficacy (in terms of percentage improvement in C-peptide AUC) in year 2 was maintained in each year beyond year 2. We varied this assumption in the sensitivity analyses as described hereunder.
Insulin requirements in our model increased in the first 4 years at the rate observed in the TrialNet study. 11 However, as HbA1c levels plateau, we assumed that insulin requirements beyond year 4 were the same as those in year 4. Although immunotherapy can reduce insulin needs, no significant reductions in insulin use were observed for low-dose ATG, high-dose ATG, rituximab, and abatacept. 10,14 –16 Furthermore, even as clinical trials for alefacept 17 and teplizumab 18 reported lower insulin needs for treated patients, it is unclear whether these reductions in exogenous insulin needs are sustained. Thus, in our base case analysis, we assumed that immunotherapies did not affect insulin requirement (we varied this assumption in a sensitivity analysis hereunder).
Risks of drug-related adverse events, diabetes complications, and mortality
Serum sickness, cytokine release syndrome, and lymphopenia are the primary adverse events associated with ATG treatment. 19 However, only individuals experiencing serum sickness typically require intervention (oral prednisone). 19 Teplizumab and ATG are both associated with similar risks for cytokine release syndrome, although this is limited to reactions that are self-limited and occur only during infusions. In addition, both drugs are associated with significant lymphopenia, and specifically, reductions in CD4 T cells. That said, rates of infection (a consequence of lymphopenia) were similar between patients receiving teplizumab or ATG and their placebo groups. 18,19
Furthermore, no adverse events involving significant clinical management costs were noted for abatacept, alefacept, or rituximab. 14,16,17 Consequently, our model accounted for the possibility of serum sickness after low-dose or high-dose ATG treatment, but no adverse events related to other immunotherapies. The risk of diabetes-related complications and progression through these complications were based on the Sheffield Type 1 Diabetes Policy Model 9 and other published studies. 20 –22 Data on death due to competing causes was age- and gender-specific and were obtained from the 2017 U.S. life tables. 23
Costs
Costs were estimated from a health care system perspective. The cost of each strategy included cost of treatment with immunotherapies (as applicable), cost of managing adverse events (i.e., serum sickness, if applicable), and annual health care costs depending on existence of diabetes complications. As described in Table 1, cost of treatment with immunotherapies included drug costs (including cost of premedication with methylprednisolone for patients treated with ATG) and injection or infusion cost (infusion costs included cost of personnel physician, nurse, etc., infusion supplies, and overhead costs). Infusion costs were obtained by applying the hourly infusion costs for biologic drugs estimated in the literature. 24 All drugs were delivered in an outpatient setting.
Cost of managing low-dose or high-dose ATG-related serum sickness included cost of oral prednisone for a subset of patients (72% of patients developed serum sickness of whom 81% required prednisone treatment 19 ). Costs of diabetes management included cost of insulin, cost of four physician visits per year, and costs of managing diabetes-related complications that were sourced from the published literature 25 –27 (Table 2). All costs were discounted at 3% per year. 28
Diabetes Management Costs and Utilities
Total costs of insulin treatment depend on body weight that depends on age and gender and is obtained from Fryar et al. 45
ESRD, end-stage renal disease.
Effectiveness
Effectiveness was measured by quality-adjusted life years (QALYs). QALYs captured both a patient's time spent in a particular health state and the quality of life (or utility) in that state. Utility in the diabetes state was specific to age group (age below or above 18 years). The model also captured disutility due to diabetes-related complications (Table 2). Utility values were obtained from the published literature. 9,29 –31 All utility values were discounted at 3% per year. 28
Cost-effectiveness analysis
We estimated the total costs and QALYs of the seven strategies. A treatment strategy was considered to be “dominant” if it cost less and yielded more QALYs than the other strategy. After removing any strategies that were dominated (i.e., strategies that cost more but yielded fewer QALYs), a strategy was considered cost-effective relative to another if the incremental cost-effectiveness ratio (ICER) (calculated as difference in total costs of two treatment strategies divided by difference in their total QALYs) was lower than the willingness-to-pay (WTP) threshold of US$100,000 per QALY. 32,33
We conducted several scenario and sensitivity analyses to examine the robustness of our results. First, to address parameter uncertainty, we performed one-way sensitivity analyses in which we varied key costs and utilities over ±25% of base case values. 34 We also conducted probabilistic sensitivity analyses (PSA) in which we assigned distributions to input parameters and performed 1000 second-order Monte Carlo simulations. Standard deviations for these distributions were derived from the literature where available; where unavailable, these were assumed to be 25% of base case values of parameters. 35 Two exceptions were the probability of transition from microalbuminuria to macroalbuminuria and from healthy to background retinopathy for which we assumed standard deviation of 5% to prevent the probabilities from exceeding 1.
Second, we considered two alternative scenarios for long-term efficacy of the drugs. In the first scenario, we considered a gradual annual reduction of 10% in efficacy such that the efficacy dropped to 10% (of year 2's efficacy) in year 11 and stayed at this level beyond that. In the second scenario, we assumed that efficacy dropped to 25% of that in year 2 from year 3 onward. Third, we examined robustness of our results to potentially lower insulin requirements with alefacept and teplizumab (28% and 43% lower, respectively). Fourth, in our base case analysis, we allowed HbA1c in any cycle to be distributed over a range of 5%–12% to prevent transition probabilities from exceeding bounds in the PSA. In this sensitivity analysis, we expanded this range to 5%–14%.
Fifth, since most studies target children and adolescents, we considered a study sample that comprised only adolescents aged 12–17 years. Lastly, we accounted for the time costs to patients for receiving immunotherapy treatment. Specifically, we assumed that the patient (or family member of the child) receiving low-dose ATG required 2 full days off work, those receiving high-dose ATG required 4 full days off work, whereas those receiving abatacept, alefacept, rituximab, or teplizumab required 27, 24, 4, and 14 (per treatment course) half-days off work, respectively. Each day off work was valued at US$190 based on median weekly earnings of full-time workers in the United States. 36
As this was a modeling study, no ethics approval was required.
Results
Base case analysis
Panel A of Table 3 gives results of the base case analysis. There are several notable results. First, among all seven strategies, low-dose ATG treatment entails the lowest lifetime costs (US$448,480) and generates the highest QALYs (20.05 QALYs). Thus, low-dose ATG dominates all other treatment strategies.
Base Case Cost-Effectiveness Results
All costs are in 2020 U.S. dollars (US$). This base case analysis is based on a lifetime horizon.
ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life-years.
Second, compared with no treatment, low-dose ATG generates cost savings of US$10,270 and 0.09 additional QALYs. These cost savings and QALY gains arise as greater C-peptide preservation reduces risk of downstream diabetes complications. Specifically, savings in downstream cost of diabetes management ($18,090 on average) more than offset costs of low-dose ATG treatment (US$7820 on average).
Third, the cost savings with low-dose ATG are in contrast with other immunotherapies that increase lifetime costs by US$9500–US$168,380 compared with no treatment. QALY gains with other immunotherapies (relative to no treatment) are also smaller than that with low-dose ATG (0.02–0.05 QALYs vs. 0.09 QALYs). The ICERs for each of the other immunotherapies exceed the WTP threshold of US$100,000 per QALY (Panel B, Table 3), indicating that these therapies are not cost-effective versus no treatment. These findings are explained by the lower costs and higher efficacy of low-dose ATG compared with other immunotherapies.
Sensitivity analyses
Results of the one-way sensitivity analyses are presented in the Tornado diagram in Figure 1. The diagram shows that low-dose ATG remained dominant relative to no treatment for all values of key costs and utilities in the range of ±25% of base case values. Cost-effectiveness acceptability curves from the PSA indicate that at the WTP threshold of US$100,000 per QALY, low-dose ATG was cost-effective or dominant in 99% of iterations (Fig. 2).
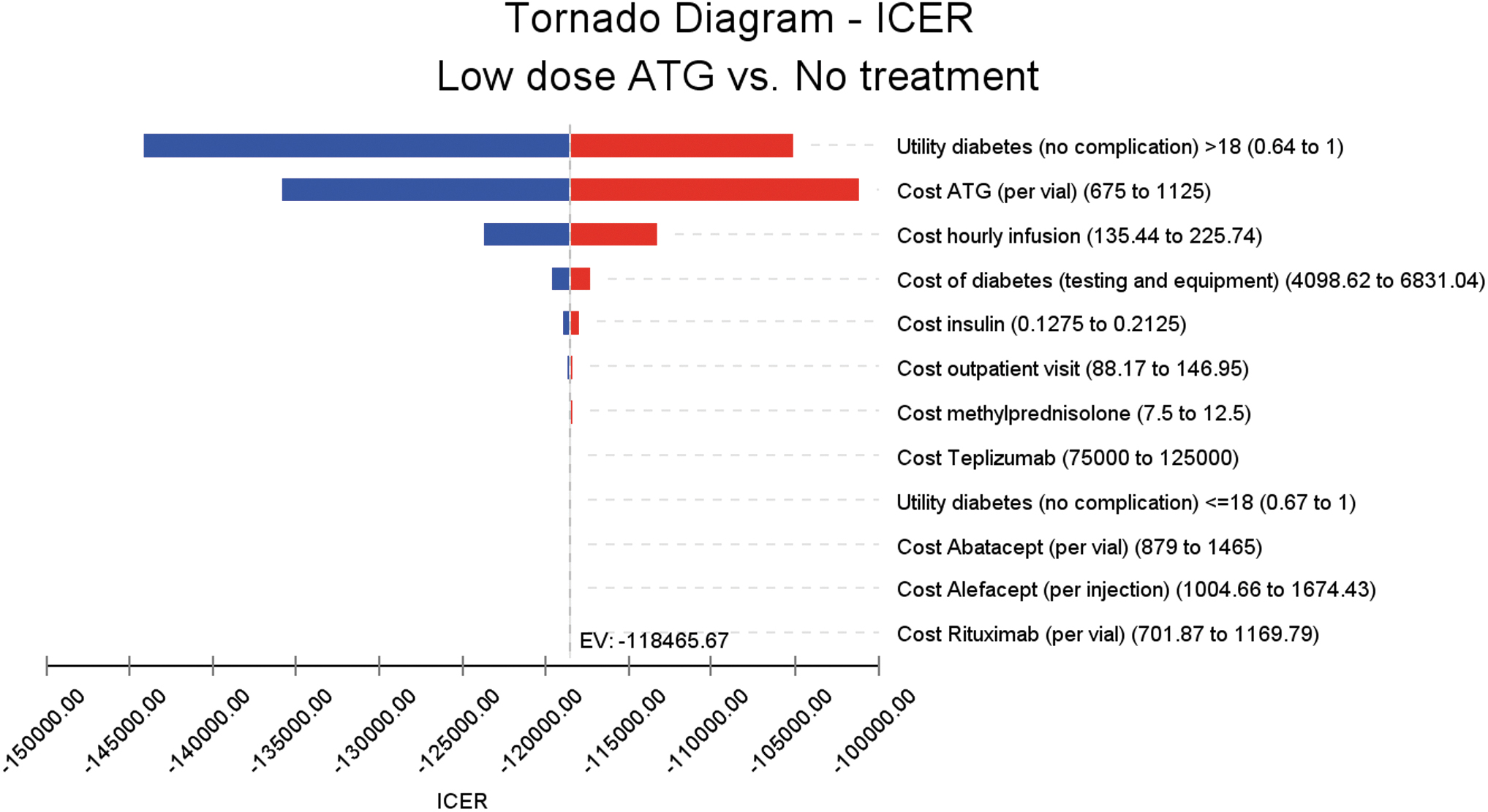
Tornado diagram. Color graphics are available online.
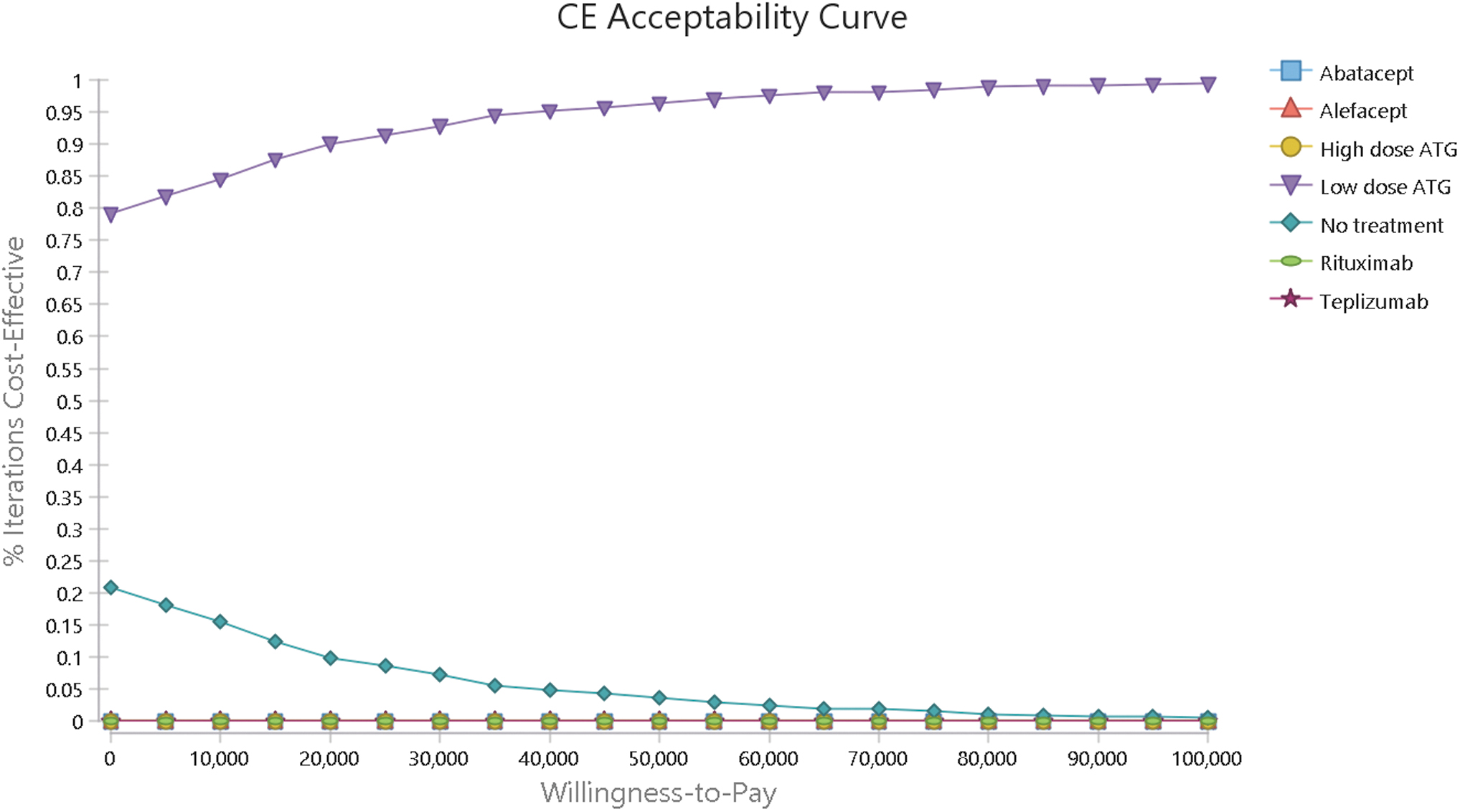
Cost-effectiveness acceptability curve. Color graphics are available online.
Table 4 gives the results of the additional scenario and sensitivity analyses. We obtained similar results to the base case analysis even when we assumed a gradual 10% annual decline in efficacy beyond year 2 to 10% of efficacy in year 2 beyond year 10 (Panel A). When we assumed that only 25% of the drugs' efficacy is retained after year 2, lifetime costs with low-dose ATG became higher than with no treatment (US$461,570 vs. US$458,750). QALY gains (over no treatment) were also smaller than in the base case analysis (0.05 instead of 0.09 additional QALYs). However, low-dose ATG was still cost-effective versus no treatment at the WTP threshold of US$100,000 per QALY, with an ICER of US$55,270 per QALY gained (Panel B, Table 4).
Sensitivity and Scenario Analyses
All costs are in 2020 U.S. dollars (US$).
Our base case findings also continued to hold even when we allowed for lower insulin requirements after treatment with alefacept and teplizumab (Panel C), considered only a sample of adolescents aged 12–17 years (Panel D) and allowed HbA1c levels to vary in a wider range of 5%–14% (Panel E). Although total costs for all immunotherapies were higher and cost savings from low-dose ATG (relative to no treatment) were smaller when we accounted for time costs to seek immunotherapy treatment, low-dose ATG continued to be the dominant treatment strategy (Panel F).
Discussion
This study investigated the cost-effectiveness of six immunotherapies that have demonstrated the capacity to provide significant beta cell preservation in subjects with new-onset type 1 diabetes. Our results suggest that low-dose (2.5 mg/kg) ATG treatment is both least costly and most effective compared with other immunotherapies and versus no treatment. These findings are explained by ATG's lower drug costs and higher efficacy compared with other immunotherapies. Meanwhile, none of the other immunotherapies were cost-effective versus no treatment.
Given the current unsustainable rise in health care costs, the cost savings associated with low-dose ATG treatment versus other potential immunotherapies could have considerable implications for payers and policymakers. With >60,000 patients diagnosed with type 1 diabetes each year 2 , our estimated lifetime cost savings of US$10,300 per patient imply annual cost savings of nearly US$618 million for the United States over patients' lifetimes if all patients with new-onset type 1 diabetes receive low-dose ATG therapy. Thus, it will be critical that payers factor cost-effectiveness of these novel immunotherapies into their decisions to prevent excessive burden on health care resources.
Even as we found low-dose ATG to offer greater economic value than other immunotherapies, it is not yet known whether low-dose ATG treatment will be equally effective for all patients. If individuals with specific genetic and/or clinical characteristics do not sufficiently respond to ATG treatment, clinicians will need to consider “test-and-treat” strategies and/or second-line treatment with other immunotherapies. The development of predictive biomarkers allowing for “precision treatment” is critically needed to aid in these decisions and to lower health care costs.
Although use of low-dose ATG to prevent or delay type 1 diabetes onset is still being studied, the extensive economic appeal of low-dose ATG in the new-onset setting suggests that the drug may also be valuable in the prevention space. Our preliminary cost-effectiveness analysis of low-dose ATG versus teplizumab (the only immunotherapy having demonstrated an ability to delay type 1 diabetes to date 37 ) indicated that if low-dose ATG can be at least as effective as teplizumab in delaying type 1 diabetes onset and is priced similar to that for new-onset treatment, low-dose ATG treatment will be cost-effective versus teplizumab. Similar analyses around price and efficacy can inform pricing for the future market basket of therapies for prevention and delay of type 1 diabetes.
This study has several limitations. First, no head-to-head trials have directly compared these immunotherapies. Thus, we had to rely on cross-trial comparisons to source efficacy data. Furthermore, data on efficacy of immunotherapies were only available for a maximum duration of 2 years. However, to address these data limitations to the extent possible, we considered alternative long-run scenarios in our sensitivity analyses and our conclusions continued to hold.
Second, our model assumed an average, fixed relationship between C-peptide preservation and HbA1c, that is, 0.1 pmol/mL increase in C-peptide AUC led to a 0.1% decline in HbA1c. 13 However, this relationship varies across studies and patient populations given that reported insulin doses are highly affected by patient characteristics such as eating habits and physical activity levels. Third, our study did not capture heterogeneity in treatment effects for different patient groups as such data were not available for all immunotherapies and additional mechanistic work remains in progress.
Fourth, patients can potentially be treated again with the same or alternative drug to slow diabetes progression. However, except for teplizumab, such retreatment scenarios have not been studied in clinical trials, and thus, could not be captured in our model. Fifth, although the assumed price of US$100,000 for teplizumab was based on current prices of other monoclonal antibodies, 6,38 actual price of teplizumab is yet to be determined. Similar uncertainty exists for prices of other immunotherapies as some of these drugs are either not yet available on the market (e.g., alefacept), or have biosimilars in development or approved (e.g., rituximab and abatacept). However, we varied the costs of immunotherapies in the sensitivity analyses, and our results were robust.
Finally, although outside the scope of this analysis, we would be remiss not to acknowledge that diabetes technologies such as continuous glucose monitoring and automated insulin delivery are providing marked improvements in the day-to-day lives of people living with type 1 diabetes. Although many, if not most, providers and patients would argue against the notion, some might reasonably suggest that diabetes technologies seeking to reduce the mental and emotional burdens of living with the type 1 diabetes may ultimately prove more cost-effective than immunotherapies.
Despite these limitations, our study can serve as a useful starting point to stimulate and guide future discussions and decisions on optimal strategies for type 1 diabetes treatment and management. For example, our finding that low-dose ATG can lead to both cost savings and better health outcomes compared with other immunotherapies warrants further research and policy consideration of this drug as a type 1 diabetes immunotherapy.
A large study of low-dose ATG in stage 2 diabetes is planned to start in early 2022. This research will add to our understanding of the potential for low-dose ATG to interdict the disease. If significant delay of the disease is observed, the trial could support an application to the FDA for approval in the prevention space. That said, Sanofi has not indicated any plans, at this point, to seek a label for ATG as a type 1 diabetes immunotherapy. As such, even if low-dose ATG's efficacy is observed, it may continue to be utilized as an “off-label” type 1 diabetes immunotherapeutic.
Conclusion
Immunotherapeutics capable of preserving beta cell function create opportunities to treat new-onset type 1 diabetes but simultaneously raise questions about cost-effectiveness. Our analysis indicates that, of all immunotherapeutics having demonstrated efficacy in preserving C-peptide, low-dose ATG is superior and, in particular, could lead to both cost savings and better health outcomes than with no treatment. Future research is needed to establish the clinical efficacy and cost-effectiveness of low-dose ATG for type 1 diabetes prevention.
Footnotes
Authors' Contributions
M.J.H. and D.A.S. conceptualized the idea. H.V.N. led data analysis, article writing, and revisions. S.M. contributed to data analysis and article writing. L.M.J. contributed to article writing and revisions. All authors have read and approved the final article.
Author Disclosure Statement
H.V.N., D.A.S., S.M., and L.M.J. have no conflicts of interest to disclose. M.J.H. is a member of the Scientific Advisory Board for SAbBiotherapeutics, a company developing a humanized polyclonal ATG.
Funding Information
No funding was received for this article.
