Abstract
Advances in glucose monitoring and insulin delivery technologies have led to the development of innovative self-management tools, such as continuous glucose monitoring, automated insulin delivery systems, and smart connected insulin pens. Although the clinical advantages of today's emerging diabetes technologies are well documented, the cost of integrating these tools into clinical practice must be considered to sustain the financial viability of both public and private insurers. Most clinicians are unfamiliar with the process the commercial insurers follow when making these decisions. This article reviews the key factors the insurers consider when determining eligibility criteria.
Introduction
Advances in glucose monitoring and insulin delivery technologies have led to the development of innovative self-management tools, such as continuous glucose monitoring (CGM), automated insulin delivery (AID) systems, and smart connected insulin pens. These technologies enable individuals with type 1 diabetes (T1D) and type 2 diabetes (T2D) to optimize glycemic control and improve the quality of their lives. The clinical efficacy of these technologies has demonstrated in numerous studies of individuals with T1D and T2D. 1 –34
The benefits of CGM in this population include reductions in glycated hemoglobin (HbA1c), 1 –10 fewer severe hypoglycemia events, 7,8,24 increased time within target glucose time in range (TIR), 4,5,22,23 and reductions in time below range. 4,5 CGM use has also been shown to be strongly associated with the reduction of diabetes-related hospitalizations in individuals with diabetes treated with intensive and nonintensive therapies. 7,8,24 –27
Improvements in CGM accuracy and dosing algorithms have made possible a new generation of AID systems that can detect and or predict dangerous glycemic events and then automatically suspend or increase insulin infusion. These devices have been shown to improve overall glycemic control and reduce hypoglycemia in pediatric and adult T1D populations. 13,28 –34
Individuals who prefer multiple daily insulin injection therapy now have the opportunity to improve their diabetes control with smart connected insulin pens, which automatically track insulin injections and provide dose decision support via a bolus calculator. These devices store the date, time, and amount of the previous doses and are integrated with USB or Bluetooth features for more effective data management. Use of connected smart pens have been shown to increase time in glycemic target range, decrease time spent below range, and reduce the number of missed insulin bolus doses. 21 A recent cost-effectiveness analysis by Jendle et al. projected that the use of a connected smart pen would improve clinical outcomes and lower costs compared with standard of care in a Swedish T1D population. 20
In light of the growing demand for public and private insurers to provide coverage for these technologies, the clinical benefits must be weighed against the initial and ongoing costs. This is particularly important for commercial health plans. According to the U.S. Census Bureau, commercial health insurance coverage was more prevalent than public coverage, covering the vast majority of the U.S. population. 35
Although clinicians are well aware of the coverage policies as they relate to diabetes technologies, most are unfamiliar with the process commercial insurers follow when making these decisions. This article reviews the key factors that payers and/or employers/organizations consider when determining eligibility criteria.
Critical Factors Considered
When determining whether to provide coverage for a new medical technology and who will be eligible for coverage, the overarching consideration is how these decisions will impact the organization's approach to the Quadruple Aim (QA), which focuses on improved patient and provider experience with improved quality outcomes while reducing the per capita cost of health care. What is the expected return on investment (ROI) relating to QA? Will the ROI be high enough to offset additional cost of the new technology? Making these decisions requires a comprehensive assessment of several factors.
Clinical efficacy
The first factor payers and/or member employers must consider is whether the new technology confers any clinical advantages over the current technologies. For example, does use of an AID system lead to better clinical outcomes than traditional insulin pump therapy? To make this determination, insurers rely on published results from well-designed randomized controlled trials (RCTs) and, more recently, from observational and prospective real-world studies that assess patient data from large databases and registries. Several insurers and regulatory agencies now require this information in conjunction with RCT results when evaluating the safety and effectiveness of new drugs and medical devices. 19 –22
Although proof of efficacy from RCTs is foundational in the decision-making process, real-world studies provide critical data about correlations between improvements in clinical outcomes (e.g., reductions in HbA1c, severe hypoglycemia, diabetic ketoacidosis) and health care resource utilization. With this information, payers can calculate the potential savings specific to their payment structure and clinical status of their members. For example, if a payer covers a large population of individuals with poorly controlled diabetes (e.g., >9% HbA1c), recent real-world CGM studies that report reductions in inpatient hospitalizations for diabetes-related events within a similar population would allow payers to calculate the financial impact of these reductions relevant to their organization's or member employer's population.
Clinical effectiveness
In addition to RCT reports and findings from real-world studies, payers also review budget impact and cost-effectiveness analyses that are relevant to the technology being considered. While a budget impact analysis provides an economic assessment of the estimated financial consequences of adopting a new technology, a cost-effectiveness analysis assesses both the costs and health outcomes of using the new technology in comparison to the current technology by estimating the cost needed to gain a defined unit of a health outcome (e.g., life year). Because theses analyses use cost data derived from various studies to determine an average cost of a given treatment or service (e.g., inpatient hospitalization for severe hypoglycemia), the most impactful data provided by these models is reduction in incidence of health care resource utilization for disease-related events admissions. Payers must perform additional calculations using their own reimbursement schedules to determine the actual cost impact on their organization. The projected cost savings can vary depending upon the line of business (e.g., Commercial, Medicare Advantage).
Direct and administrative costs
The next consideration is how coverage will impact a payer's and/or employer's overall costs. Costs include both the direct medical cost of the technology and any relevant administrative expenses. Is the out-of-pock cost of the new technology more or less than the current technology covered? Will the benefits support attainment of other priority goals such as improvement in quality of care, reduced avoidable utilization and member satisfaction that would justify the expense? Since we know that the costs of the new diabetes technologies are higher than older devices, the question is whether their use will lower the direct medical costs of care (e.g., fewer avoidable hospitalizations and emergency room visits, etc.) and if adapted internally to manage the membership, will it provide a more efficient workflow that will allow an organization to reallocate staff (e.g., case managers) to other priority areas of its disease management programs?
Ability to retain members
In today's competitive health insurance environment, payers must consider the impact of their coverage policies on their ability to retain members, which is an ongoing challenge for all commercial plans. A 2016 survey of 1000 consumers found that almost half of respondents reported changing their health plan in the last 3 years. 36 Coverage policies are also a key consideration for employers, who must provide a variety coverage policies (Health Maintenance Organization [HMO], Preferred Provider Organization [PPO]) as a benefit option to attract and retain employees.
Because potential or current members often consider the medications or technologies covered in the health plan's formulary, both payers and employers must assess whether and to what degree making new diabetes technologies available impacts member/employee satisfaction and promotes retention. The same is true for medications. For example, if an individual with insulin-treated diabetes feels that they are well-controlled on insulin lispro, but the plan they are considering only covers insulin aspart, they may be reluctant to choose (or stay with) that plan. This also applies to large employers who are familiar with the medical literature or survey their employees (or both) when reviewing a plan's formulary.
However, payers and member employers must determine whether coverage of diabetes medical devices will offset both the rising cost of health care and any additional direct/administrative costs. Over the past 20 years the annual average cost of health care has risen almost twice as much (5.0%) compared with nonhealth care costs (2.6%) as reflected in the Consumer Price Index for each category. 37,38 As such, payers and member employers must generate additional premium or product price increases of at least 5% per year to offset increasing costs and maintain its profit margins. Thus, the cost of technologies such as CGM will be weighed against the cost of the current technology in the market (e.g., traditional blood glucose monitoring), taking into consideration all of the variables discussed earlier.
Finally, payers must assess how providing diabetes medical device coverage impacts quality scoring. Higher scores in both quality of care and treatment satisfaction are critical to a payer's ability to market their plans to individuals, self-insured employers, and other organizations.
Influence of Quality Measures
Value-based care
Value-based care (VBC) is a relatively new approach to reimbursement that links payments for health care delivery to the quality of care provided. Unlike traditional fee-for-service models, in which health care providers charge was based on individual services rendered, VBC models reward health care providers for both effectiveness and efficiency. Because provider reimbursements are calculated using various quality measures, VBC is data driven in that health care providers must report to payers on specific metrics and demonstrate improvements in treatment, prevention, patient satisfaction and cost of care. Due to the prevalence of diabetes and the impact this chronic condition has on decreasing quality of care and increasing costs, improving care in this population is typically a focus in VBC.
Role of the National Committee for Quality Assurance
The National Committee for Quality Assurance (NCQA) utilizes a comprehensive framework for evaluating and improving key areas of health care. For a health care organization to earn an NCQA Health Plan Accreditation, it must meet specific standards encompassing more than 100 facets of health care delivery. Although NCQA accreditation is not required for commercial health plans, many large companies and organizations will only consider an accredited health plan. As such, more than 90% of health plans use and report the required metrics.
For commercial plans that offer Medicare Advantage plans, the NCQA publishes its annual Health Insurance Plan Ratings. The report provides a rating of each health plan on its quality of care and treatment satisfaction on a scale of 0 to 5 (5 = highest, 0 = lowest) based on its combined Health Care Effectiveness Data and Information Set (HEDIS), Consumer Assessment of Health Care Providers and Systems (CAHPS), and NCQA accreditation scores. These ratings allow current and potential consumers to compare the various insurers when selecting a plan based on customer satisfaction, disease prevention, and treatment 39 (Fig. 1).
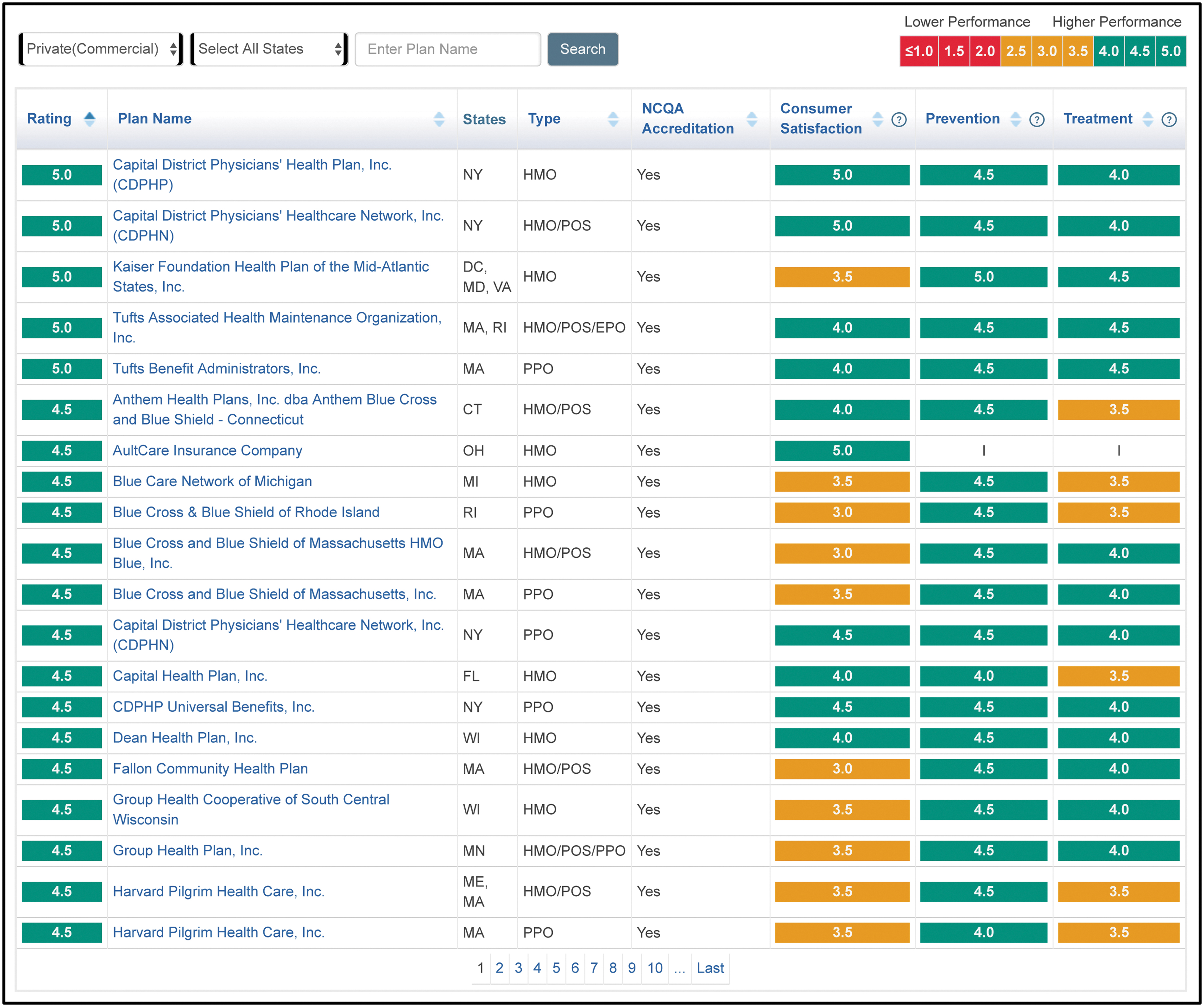
NCQA Health Insurance Plan Ratings (2019–2020). NCQA, National Committee for Quality Assurance.
Quality metrics
The most common measures used for quality assessment are the HEDIS, Prevention Quality Indicators (PQI) and CAHPS.
Health Care Effectiveness Data and Information Set
The NCQA publishes annual Health Insurance Plan Ratings, based on measures of quality of clinical care, member satisfaction, and NCQA Accreditation Survey results. Included in the NCQA survey is the HEDIS, which assesses effectiveness of care, access and availability of care, patient satisfaction, and health care resource utilization. This information is collected through a combination of surveys, claims data, and medical record audits. HEDIS reporting is a requirement of health plans and Centers for Medicare and Medicaid Services for use in health plan accreditation, Star Ratings, and regulatory compliance.
Prevention Quality Indicators
The NCQA also uses the PQI, which are measures of potentially avoidable hospitalizations for Ambulatory Care Sensitive Conditions. Utilizing hospital discharge data, this assessment reflects issues of access to, and quality of, ambulatory care in a given geographic area. The purpose of the PQI composites is to improve the statistical precision of the individual PQI, facilitating greater discrimination in performance among areas and improved ability to identify potentially determining factors in performance. While an overall composite connects potentially avoidable hospitalization to individual PQI measures, separate composite measures are used for acute and chronic conditions to explore various factors that may be influencing hospitalization rates for each condition.
Consumer Assessment of Health Care Providers and Systems
This survey was developed by the Agency for Health Care Research and Quality (AHRQ) and was initially used as research tool for assessing patient experiences with health care. It has since become the national standard for measuring and reporting on the experiences of consumers with their health plans, focusing on the quality of communication with health care providers and ease of access to health care services.
As shown in Figure 2, the HEDIS, PQI, and CAHPS measures and other metrics, such as cost of care and large commercial group customer satisfaction, align with common clinical outcomes.
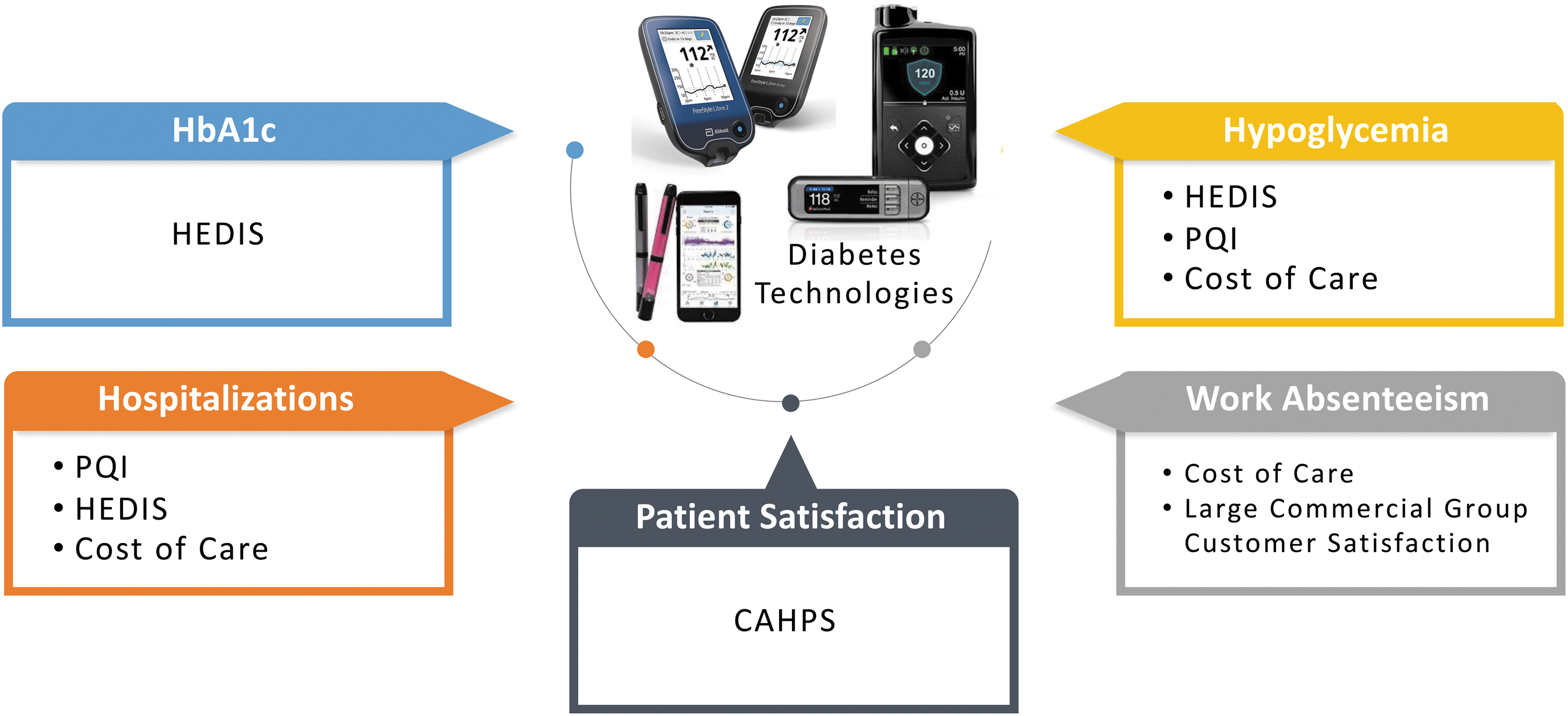
Alignment of clinical outcomes and quality measures.
Making the Decision for Coverage
Impact on the QA
After reviewing the clinical efficacy and conducting a cost analysis (e.g., modeling, direct/administrative costs, ability to generate additional revenues through retention, or quality incentives) relevant to the specific diabetes technology being considered, the next step is to conduct a risk stratification of the member population, using quality metrics to identify the population at the highest risk for poor clinical outcomes. Risk stratification is critical because improving quality while reducing costs in these populations will yield the greatest cost savings. According to a recent AHRQ report, only 5% of the U.S. population accounted for over 50% of total health care spending in 2017. 40 Among these individuals, the upper 1% ranked represented ∼22% of total health care spending that year, whereas the bottom 50% represented only 2.9% of health care costs. Within the diabetes population, the highest risk patients are generally those who are in the poorest glycemic control based on the HEDIS metric of >9.0% HbA1c and who have the highest short-term complication admission rates, all-cause admission rates, and pharmaceutical costs. These components are significant drivers of the total cost of care for the payer. Based on their risk stratification findings, many commercial payers will then implement a proof-of-concept project to demonstrate whether and to what degree utilization of the technology will improve care and reduce costs within the identified high-risk population.
Although most commercial payers currently provide CGM coverage for individuals with T1D and T2D treated with intensive insulin regimens, evidence from recent studies and growing consumer demand are prompting payers and employers to investigate the potential benefits of providing CGM coverage to individuals within their broader T2D populations. Figure 3 presents an example of a proof-of-concept demonstration project to assess the impact of expanding CGM coverage to include individuals with T2D who are treated with less intensive therapies.

Proof-of-concept demonstration project workflow.
Impact of telemedicine/remote monitoring
Another consideration is how use of telemedicine and remote monitoring will impact quality, treatment satisfaction, and cost. Utilizing the connectivity of many current diabetes technologies, the efficacy and efficiency of telemedicine interventions and remote monitoring in diabetes management have been clearly demonstrated. 41 –50 Moreover, the ability of patients with diabetes to interact remotely with their health care providers via smartphones and other communication devices can significantly increase patient access to clinical care and support programs (e.g., diabetes coaching, online support groups). 51 –54 For example, two recent studies demonstrated that intermittent use of CGM in individuals with noninsulin-treated T2D who were enrolled in a comprehensive telemedicine program was associated with significant reductions in HbA1c 54 and diabetes-related distress, a known risk factor for poor glycemic control 55 –59 and a higher prevalence of complications. 59,60
Although once considered to be more futuristic than practical, the COVID-19 pandemic has, by necessity, accelerated the adoption of telehealth in clinical practice. A global survey of 7477 individuals with T1D from 89 countries found that use of telemedicine visits and remote monitoring of CGM and insulin data was perceived as positive, with the majority (75%) reporting that they would consider telemedicine visits beyond the pandemic. 61 Given the positive impact of telemedicine consultations on clinical outcomes and treatment satisfaction, use of these interventions will likely continue as a component of usual care.
Impact on stakeholders
The plan also considers how the adoption of medical device technology will impact key partnerships such as their Pharmacy Benefit Manager (PBM) (if applicable) and customer contracts such as large employer and provider/health system contracts.
PBMs play an integrate role in pharmacy benefit decision-making. PBM's are contracted by health plans, labor unions, and corporate employers to interface with drug and device manufacturers and process prescription-related claims with the goal of improving quality of care and reducing costs. Customer contracts must also be considered as many contracts involve some form of risk sharing. Input and alignment from stakeholders must be obtained and potential direct relationship impact evaluated.
Summary
The clinical advantages of today's emerging diabetes technologies over traditional tools are well documented. However, the cost of integrating these technologies into clinical practice must be considered to sustain the financial viability of both public and private insurers. Making these determinations requires insurers to evaluate both the effectiveness and efficiency of these technologies relative to their internal costs and the needs of their members. Consideration around the cost of the technology, impact on quality outcomes, member and customer satisfaction, as well as impact on plan's stakeholders are key to consider when making a final formulary determination.
Footnotes
Acknowledgments
The authors thank Christopher G. Parkin, MS, CGParkin Communications, Inc., for editorial support.
Author Disclosure Statement
T.N. reports no disclosures. M.F. is an employee of Abbott Diabetes Care.
Funding Information
Funding for the development of this article was provided by Abbott Diabetes Care.
