Abstract
Use of telemedicine and remote monitoring technologies can significantly improve glycemic control in patients with type 1 diabetes and type 2 diabetes. Patients' ability to interact remotely with their health care providers via smartphones and other communication devices can increase their access to clinical care and online coaching and support programs. The establishment of metrics for clinical use of continuous glucose monitoring data and standardization of data reporting has enabled clinicians to maintain high-quality diabetes care through remote monitoring and telemedicine visits during the COVID-19 pandemic. This article discusses our experiences using remote monitoring and telemedicine visits during this time.
Introduction
Advances in connected digital technologies have created opportunities to improve both the quality and efficiency of diabetes care delivery. Improvements in the accuracy and functionality of continuous glucose monitoring (CGM) devices have prompted more widespread adoption of this technology, whereas the establishment of glycemic targets has further increased the usefulness of this technology in clinical care.
During the past decade, studies have demonstrated that use of telemedicine and remote monitoring technologies can significantly improve glycemic management in patients with type 1 diabetes (T1D) and type 2 diabetes (T2D). 1 –6 Moreover, patients' ability to interact remotely with their health care providers via smartphones and other communication devices can increase their access to clinical care and online coaching and support programs. 7 –10
This is particularly true with today's CGM systems, which can automatically transmit patients' glucose data to be shared with their health care providers for analysis. Health care providers can review the data with their patients via telemedicine consultations and make therapy adjustments as needed. Numerous studies have shown an association between remote monitoring of CGM data via telemedicine visits and improved clinical outcomes. 6,10 –14 The value and utility of this approach to patient care have been clearly demonstrated throughout the COVID-19 pandemic. 15 –17
For clinicians, the pandemic has raised obstacles to delivering quality in-person care to our patients who are unable to attend in-person clinic visits. This article discusses our experiences using remote monitoring and telemedicine visits during this time.
CGM Metrics and Data Reporting
In 2019, an international panel was convened to develop a set of CGM metrics providing guidelines for health care providers, researchers, and individuals with diabetes in interpreting, reporting, and using CGM data in clinical practice. Among the 10 measures identified as key metrics for clinical use, the panel identified three metrics as most relevant for assessing daily glycemic management. These were percentages of time in target range (%TIR, 70–180 mg/dL); time below range (%TBR, <70 mg/dL, and <54 mg/dL); and time above range (%TAR, >180 mg/dL, >250 mg/dL). Importantly, the panel highlighted the utility of a composite metric of %TIR/%TBR, focusing on reducing %TBR while increasing %TIR. This would, by default, reduce hyperglycemia while minimizing hypoglycemia. Other key metrics include the average glucose and the glucose management indicator (GMI) calculation, a new indicator of overall glycemic status, which closely aligns with glycated hemoglobin (HbA1c) levels. 18
The panel also endorsed use of the Ambulatory Glucose Profile (AGP) as a template for presenting CGM data in a standardized report. 19 The AGP report streamlines data analysis and interpretation, enabling health care providers to rapidly assess patient status based on the established metrics, facilitating more efficient and informed decision-making, as well as enhanced patient understanding and engagement. 20 Although the AGP was initially developed as a standalone tool for glycemic assessment, most CGM manufacturers have incorporated the AGP template into their reporting software.
Case Studies
Case 1: Pregnancy (Anders Carlson, MD)
This patient was a 31-year-old female with 10-year duration of T1D and no known complications. The patient was managing her diabetes using the Medtronic 670G system (Medtronic, Inc., Northridge, CA). She reported that she runs the device in Auto Mode >90% of the time and prefers to keep her glucose “tight.” Her goal is to keep her time in range (TIR, 70–180 mg/dL) at >80%, which she has been successful in doing.
At the initial visit, we planned routine care and discussed her plans to become pregnant within the next year. She indicated that she had discontinued birth control 3 months before the consult. We remotely downloaded her insulin pump information and CGM data. As shown in Figure 1A, her TIR was 80% with 0% %TBR (A) and average glucose was 150 mg/dL with a GMI of 6.9% (B), which was the same as her laboratory HbA1c of 6.9%. She ran her pump in Auto Mode 88% of the time with 84% use of the sensor (C). Her glucose appeared well controlled overnight but with some hyperglycemia spikes after meals (D).
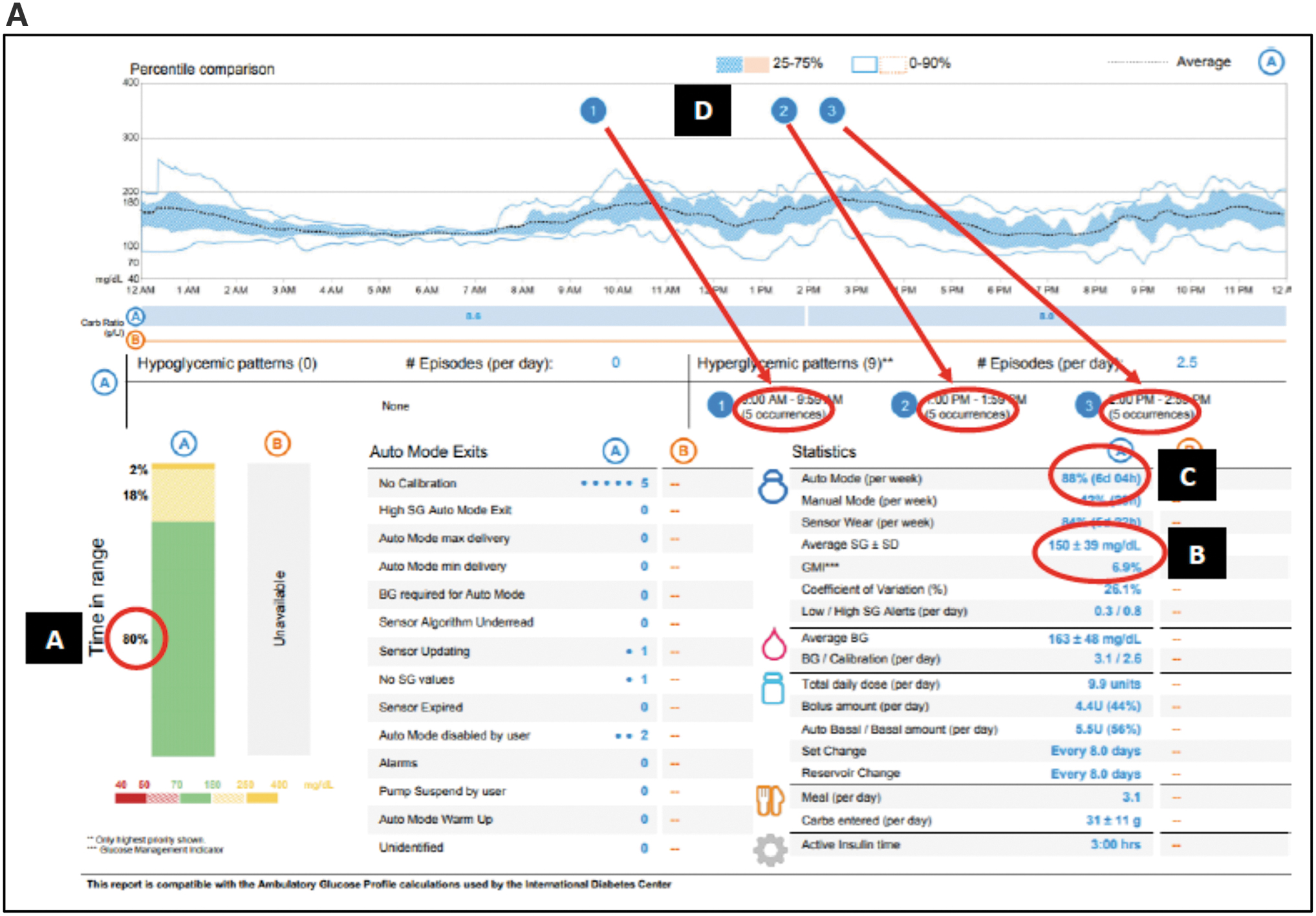
Glucose and insulin profiles at first and follow-up visits.
We scheduled her next follow-up telemedicine in 2 months. However, before the consult, she sent an electronic chart message explaining that she was pregnant. We were able to set up another telemedicine visit within the next few days.
As before, we remotely downloaded her CGM and insulin pump data. She continued to do excellent. However, we discussed that the set point for the 670G Auto Mode was 120 mg/dL, which is above the recommended targets for pregnancy, and the 670G Auto Mode has not been well-studied in pregnancy. We readjusted her settings to the recommended glucose targets for pregnancy, and she was asked to use her insulin pump in the manual mode for the remainder of her pregnancy. She was asked to download her CGM and pump data weekly and send a chart message that she had done so. I then reviewed her data and sent a note back with any recommended changes.
At 27 weeks into her pregnancy, the patient's downloaded data indicated good glycemic control within the parameters of her new targets (Fig. 1B). Her TIR continued to be 88% with only 2% between 50 and 70 mg/dL (A). Her average glucose was 124 mg/dL with a 6.3% GMI (B). Even with her pump running in manual mode 100% of the time, the number of postprandial hyperglycemic events dropped from 15 to 8 (D). Postpregnancy, we continued scheduling our telemedicine every 4 weeks to download her data and monitor her status.
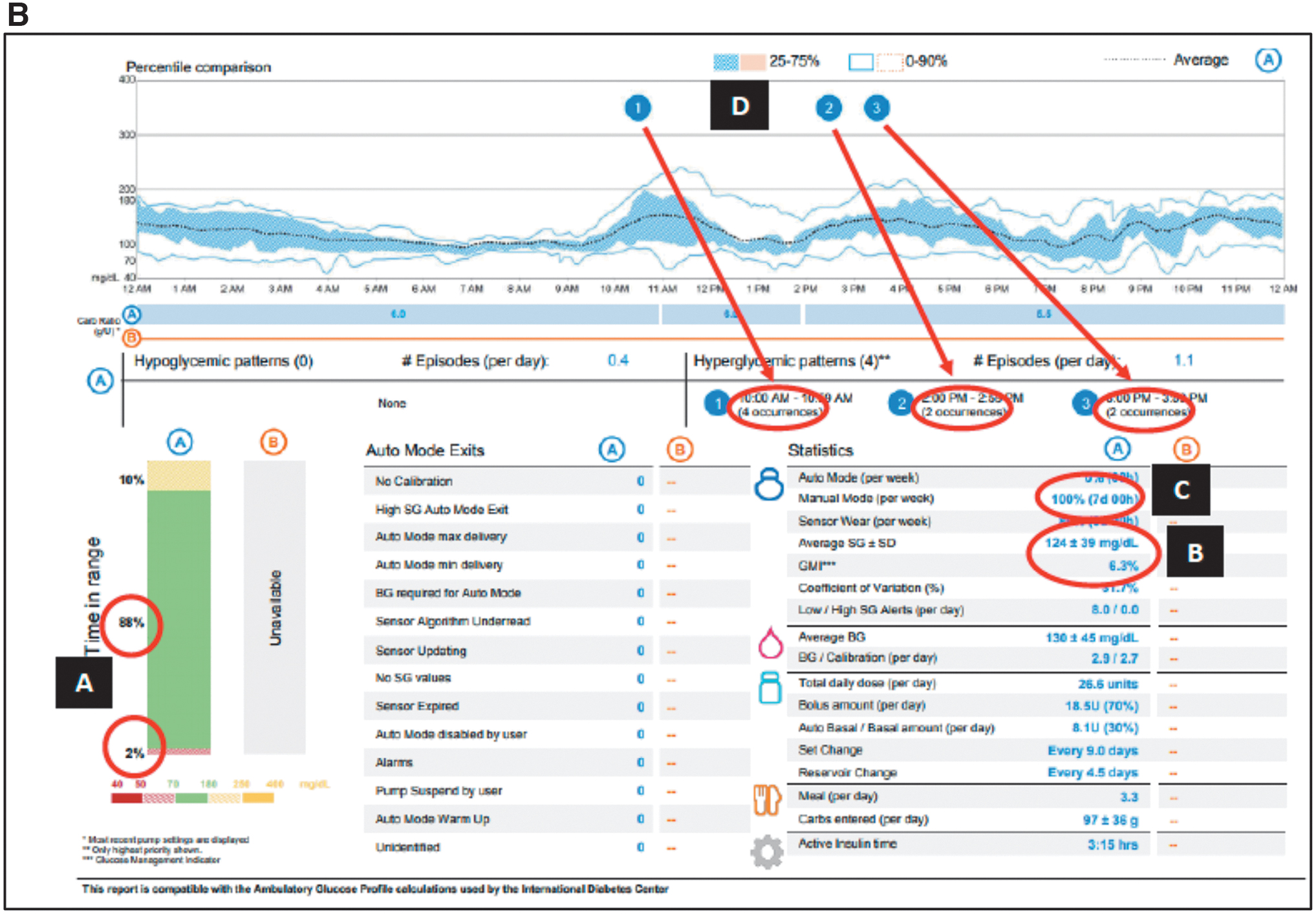
Glucose and insulin profiles at first and follow-up visits.
The ability to remotely download the patient's data allowed me to make small adjustments every week, as needed. This enabled her to achieve and maintain good glycemic management throughout her pregnancy. In addition to my recommendations, the patient often made appropriate adjustments on her own, which indicated a high level of engagement in her diabetes self-management. I found these telemedicine consults to be extremely easy and efficient.
Moreover, it significantly lessened the burden on the patient. Due to the COVID-19 pandemic, she has not wanted to leave the house to come into the clinic for visits. Generally, the telemedicine visits are rather quick in making fine tuning adjustments with her pump, which we can do very efficiently by telemedicine. It also saves her travel time to and from the clinic. Not having to take time off from work or be away from her family has been an additional benefit. Overall, use of telemedicine consults has been a seamless way to manage her diabetes during pregnancy with frequent touch points, which traditionally are done in clinic.
Case 2: Noninsulin therapy (Libby Johnson, RDN, LD, CDCES)
Background
This patient was a 58-year-old female. Her diabetes medications included metformin extended release (XR) 2000 mg daily, liraglutide 1.8 mg daily, and glipizide extended release (XL) 20 mg daily. She was using the Freestyle Libre® 14 day system.
When she met with her primary care provider in early February 2021, her updated HbA1c result was 8.6%, up from 8.3% 4-month prior. Options for next medications were discussed. The patient was opposed to initiating insulin but agreed to start dapagliflozin at 5 mg daily. The glipizide XL was reduced to 10 mg daily. She was referred by her physician to meet with a diabetes educator due to rising HbA1c and to review CGM use. The physician had noted that the patient was initially reluctant to meet with a diabetes educator but eventually agreed.
One month later, the patient met with the certified diabetes care and education specialist (CDCES) for her initial telemedicine visit. The patient worked from home, so the remote visit removed one more barrier as she did not need to leave home to attend the visit. At this time, the patient was using CGM device reader to scan her sensor, but she had interest in using her smartphone instead. She verbally reported her CGM data from the reader device.
Her average glucose for the last 14 days was 182 mg/dL. TIR was 50% and TAR was 50%. She was scanning on average three times a day, with 77% of the data captured. The patient reported she typically ate two structured meals a day, breakfast and dinner, with a snack or multiple snacks in between. During the visit, patient received further training on using her CGM device to evaluate her usual food choices. No changes were made to the medication plan. A follow-up telemedicine visit was scheduled in 1 month.
At her follow-up visit, she was using the smartphone app to scan her sensor and was sharing her CGM data with the clinic through the cloud. Her CGM data reports were now available on LibreView. Over the last 14 days, the AGP report (Fig. 2A) showed that her average glucose had improved to 147 mg/dL and her GMI was 6.8% (A) based on 73% of active CGM use (B). Her TIR increased to 79%, with significant reductions in TAR and negligible TBR (C). She reported that she had made changes to her diet, including being more mindful of food choices and portions, drinking more water, and including at least 1 cup of vegetables with at least one meal a day. She noted she was not exercising as much as she would like. However, the patient reported she was pleased that her glucose readings were improving. No changes were made to the medication plan. A follow-up visit was scheduled in 1 month.
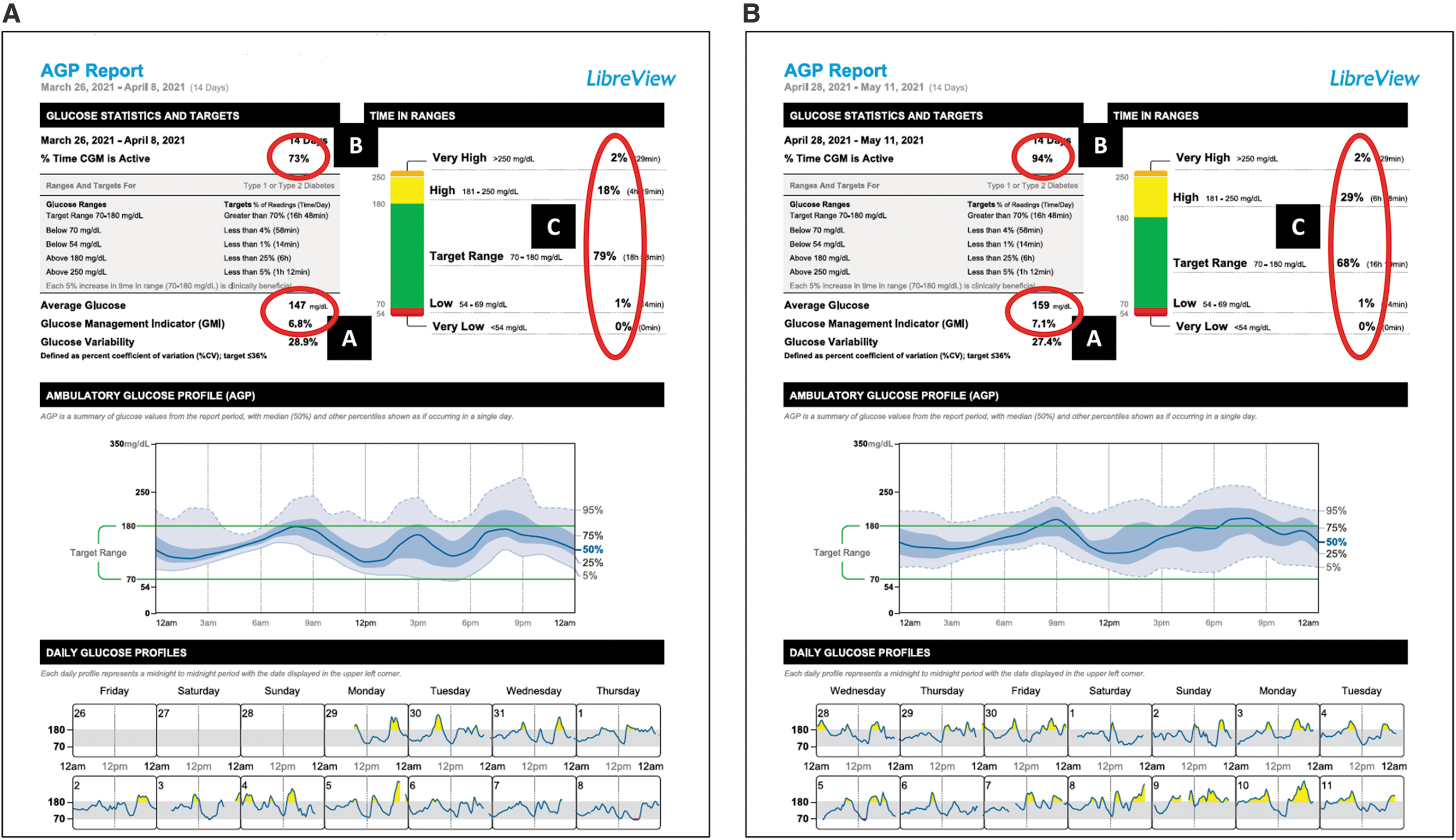
Initial and follow-up AGP reports. AGP, Ambulatory Glucose Profile.
At her third visit, patient was keeping detailed meal and snack notes in her FreeStyle LibreLink smartphone app. Review of her AGP report (Fig. 2B) showed that her average glucose had risen slightly to 159 mg/dL and her GMI was 7.1% (A) over the last 14 days based on 94% of active CGM use (B). TIR decreased to 68%, time in high range 29%, time in very high range 2%, time in low range remained at 1%, and time in very low range remained at 0% (C).
When reviewing the CGM data reports with the patient, she was able to see that her glucose rose from 86 mg/dL in the afternoon to 319 mg/dL 5-h later. She shared that she had much higher glucose readings over the weekend related to her food choices. The notes tracked in the app indicated that she ate three carb-containing snacks throughout the afternoon and then a sub sandwich with chips for dinner. She was able to describe the changes she could make to her choices to prevent her glucose from rising that high in the future.
She was also able to see that skipping a meal midday can lead to low glucose due to her diabetes medication plan. The patient had an updated HbA1c laboratory drawn and the result was 7.2%. The patient had a follow-up appointment scheduled with her physician and was looking forward to the visit now that she was able to understand her CGM data and felt confident in the changes she had made.
Case 3: COVID-19 (Thomas W. Martens, MD)
This patient is a 62-year-old female with T2D diagnosed in 1992. She is morbidly obese with a body mass index of ∼40 kg/m2, hypertension, hyperlipidemia, obstructive sleep apnea, depression, and chronic low back pain. She works the night shift in a medical setting and does not smoke.
For several years, she managed her diabetes with metformin and lifestyle interventions to some extent. Her metformin dose was metformin XR 1000 mg in the evening; she was intolerant of higher doses because of diarrhea. Over the years, she had also been found to be intolerant of glipizide, thiazolidinediones, and statins. She was reluctant to start glucagon-like peptide-1 (GLP-1) therapy because of concern about side effects.
In November of 2019, her HbA1c was 8.6%; a recheck in December showed that it had risen to 9.8%. Although therapy intensification was advised, the patient did not pursue it. Subsequent follow-up was disrupted because of the onset of the COVID-19 pandemic.
In March of 2020, our health care organization moved to a care model, in which all primary care visits were conducted remotely unless they were for evaluation of acute symptoms, because of a shortage of personal protective equipment (PPE) in the face of the expanding pandemic. Our CDCES diabetes educators moved to a 100% remote model for provision of care at this same time. Laboratory measured HbA1c data became unavailable as primary care moved to remote management because of the PPE shortage.
The patient reconnected with her primary care physician via a telephone visit in mid-April of 2020, after a period of disrupted care because of the move to remote management. At that time, fingerstick glucose readings were ranging in the upper 100 s to low 300 s mg/dL, and an insulin start was strongly recommended. Although the patient declined to start insulin therapy, she did agree to a diabetes education follow-up visit, which she subsequently missed. She did have a remote visit with the CDCES by telephone at the end of April 2020.
At that time, she expressed discouragement with her diabetes management (“everything I've tried hasn't worked”) and again reinforced that she is very sensitive to medication side effects. Her blood glucose readings continued to range between 100 and 300 mg/dL. She was asked to reconsider insulin and research GLP-1 therapy, but she was not ready to start insulin therapy at that time.
In mid-May of 2020, she did agree to start insulin-based therapy in addition to metformin XR. Basal insulin was initiated at 10 U/day, which was increased to 20 U/day a week later, with subsequent increases up to 40 U/day by June 1. At that time, we discussed the possibility of monitoring glucose with CGM after the patient expressed concerns about hypoglycemic symptoms with a measured glucose of 143 mg/dL. Her perception of hypoglycemia with glucose readings in the mid 100 s continued to be a concern, resulting in very slow titration of insulin. The patient also complained of weight gain and “feeling sick for hours” when her glucose dropped to 130 mg/dL. However, we continued to slowly increase her basal insulin dose.
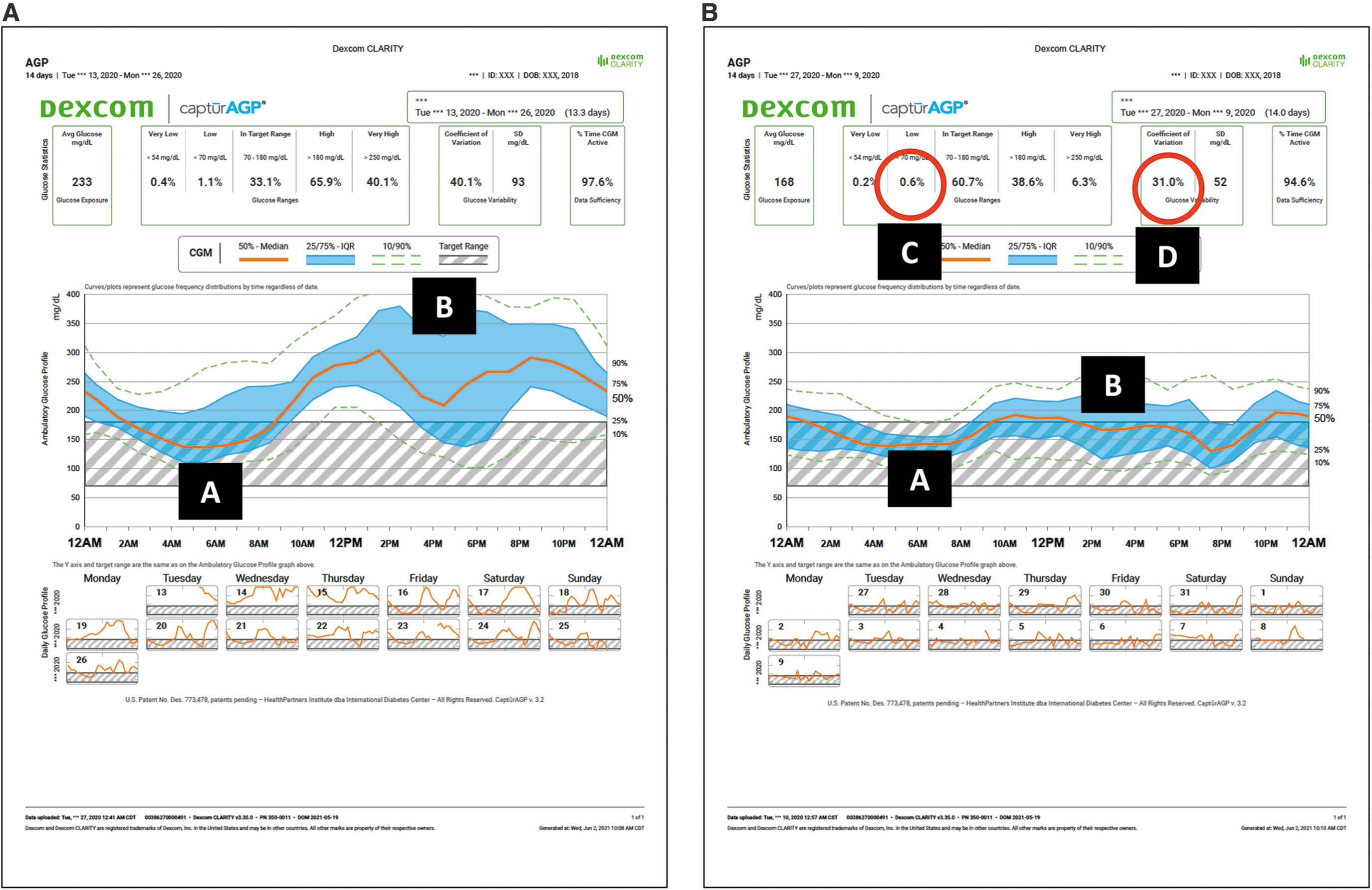
In late June 2020, the patient was able to obtain the Freestyle Libre 14 day system to facilitate remote insulin titration. Up to this point, all diabetes management had been done remotely via telephone. Her initial CGM data indicated extremely poor glycemic control with only 17% TIR and 62% TAR (Fig. 3A). Over the summer, we continued to slowly titrate her insulin dose upwards, which, again, was limited by the patient's perception of hypoglycemia even with glucose readings in the low to mid 100 mg/dL range. By the end of August 2020, her basal insulin dose was 55 U.
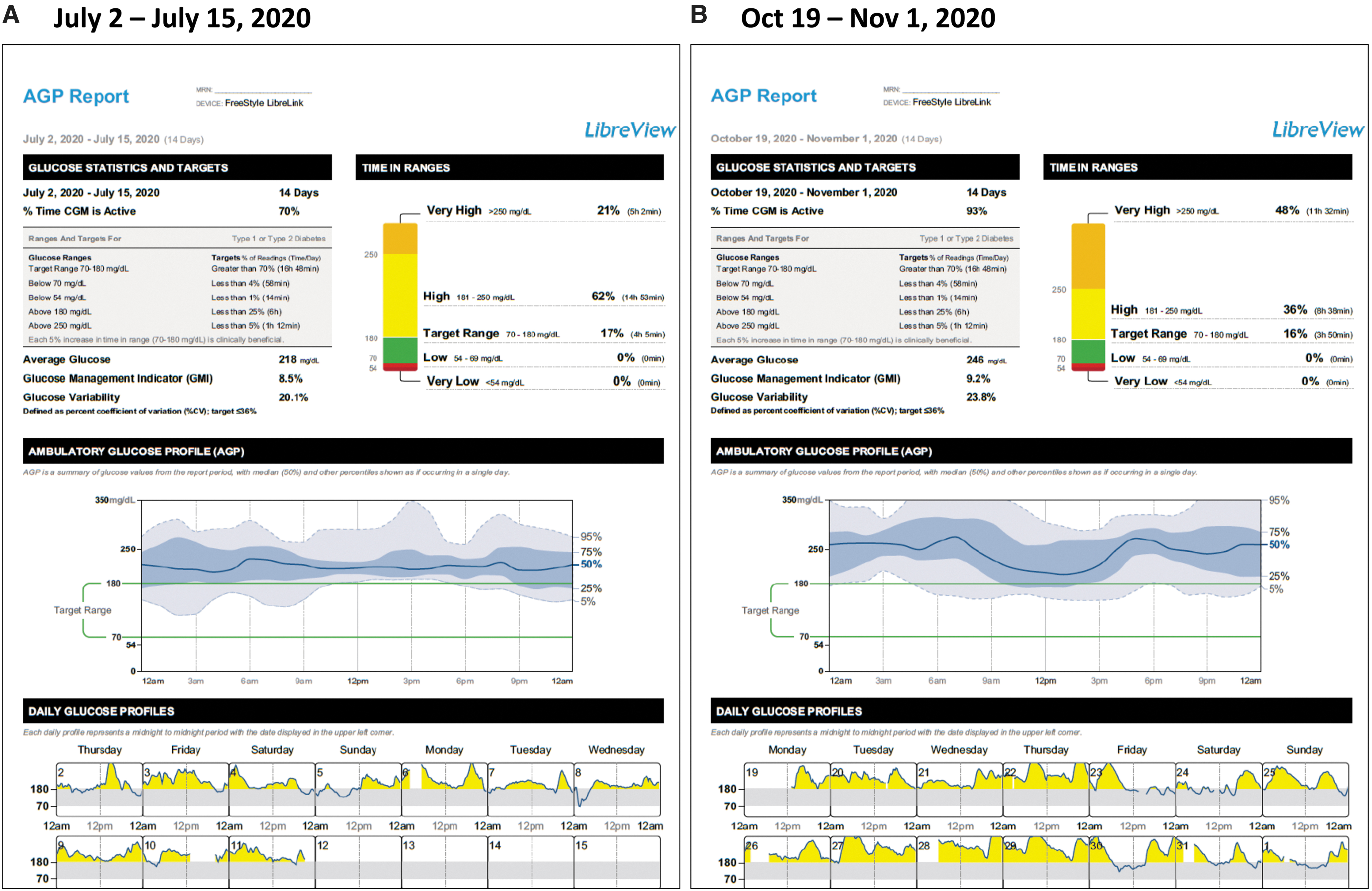
Glycemic status during COVID-19 recovery.
In early September of 2020, she presented to the urgent care with several days of cough, chills, and myalgia. She had a measured temp of 101.3. A chest X-ray showed an early right lower lobe infiltrate, concerning for pneumonia. COVID-19 testing was done, and she was started on cefdinir and azithromycin for bacterial pneumonia.
Two days later, her COVID-19 test returned positive. Five days later she was transported to the emergency room with increasing shortness of breath. A chest X-ray showed moderate diffuse airspace opacities consistent with an infectious process. Glucose was 69 mg/dL, oxygen saturation 89%. She was hospitalized for 5 days and treated with dexamethasone and remdesivir. During her hospitalization her metformin was withheld, and she was treated with a combination of basal-bolus insulin, with additional NPH insulin to help with dexamethasone-related hyperglycemia.
She was discharged on a reduced dose of glargine (30 U/day) and resumed her metformin but did not have the availability of her Freestyle Libre 14 day system. She was hospitalized two additional times during September with a severe herpes virus outbreak, followed by rehospitalization with worsened dyspnea at the end of September. She was again treated with dexamethasone as well as rivaroxaban before discharge.
She next met remotely with a diabetes educator in mid-October. She continued to struggle with prolonged COVID symptoms. Her basal insulin had been titrated to 62 U in the interim; arrangements were made to resume the CGM. On subsequent follow-up in early November (Fig. 3B) she was seen remotely with concerns regarding uncontrolled hyperglycemia on her glargine dose of 62 U (0.56 U/kg). At that time, a decision was made to switch to basal-bolus insulin, and she was begun on glargine 36 U with rapid acting insulin 18 U before her two largest meals.
By December, her glargine had been increased to 54 U/day with 16 U of NovoLog at meals. Unfortunately, she continued to struggle with post-COVID symptoms and was felt to have “long haul COVID-19,” with symptoms of fatigability, dyspnea, and prolonged cough. Her activity level was poor.
Early 2021 was characterized by very slow improvement in her post-COVID symptoms and slowly increasing activity level. She had ongoing insulin titration through via remote visits with the diabetes educator, with improving AGP data. She was also becoming comfortable in using her insulin and recognized the dramatic improvement in her glucose management.
By April 2021, she had significantly improved her glycemic control, showing 93% TIR and 0% TBR. (Fig. 3C) At that time, she continued on metformin XR and her insulin regimen of 44 U/day of glargine with 8–14 U of rapid-acting insulin with meals. An interesting side note is that her laboratory HbA1c was 8.1% on April 1, 2021, while her CGM data indicated a GMI of 6.4%, which correlates with her improved %TIR. 18 This discrepancy highlights the limitations of reliance on HbA1c measurements, which can be influenced by a number of conditions such as anemia, 21 iron deficiency, 22 and other abnormalities. 23 –25
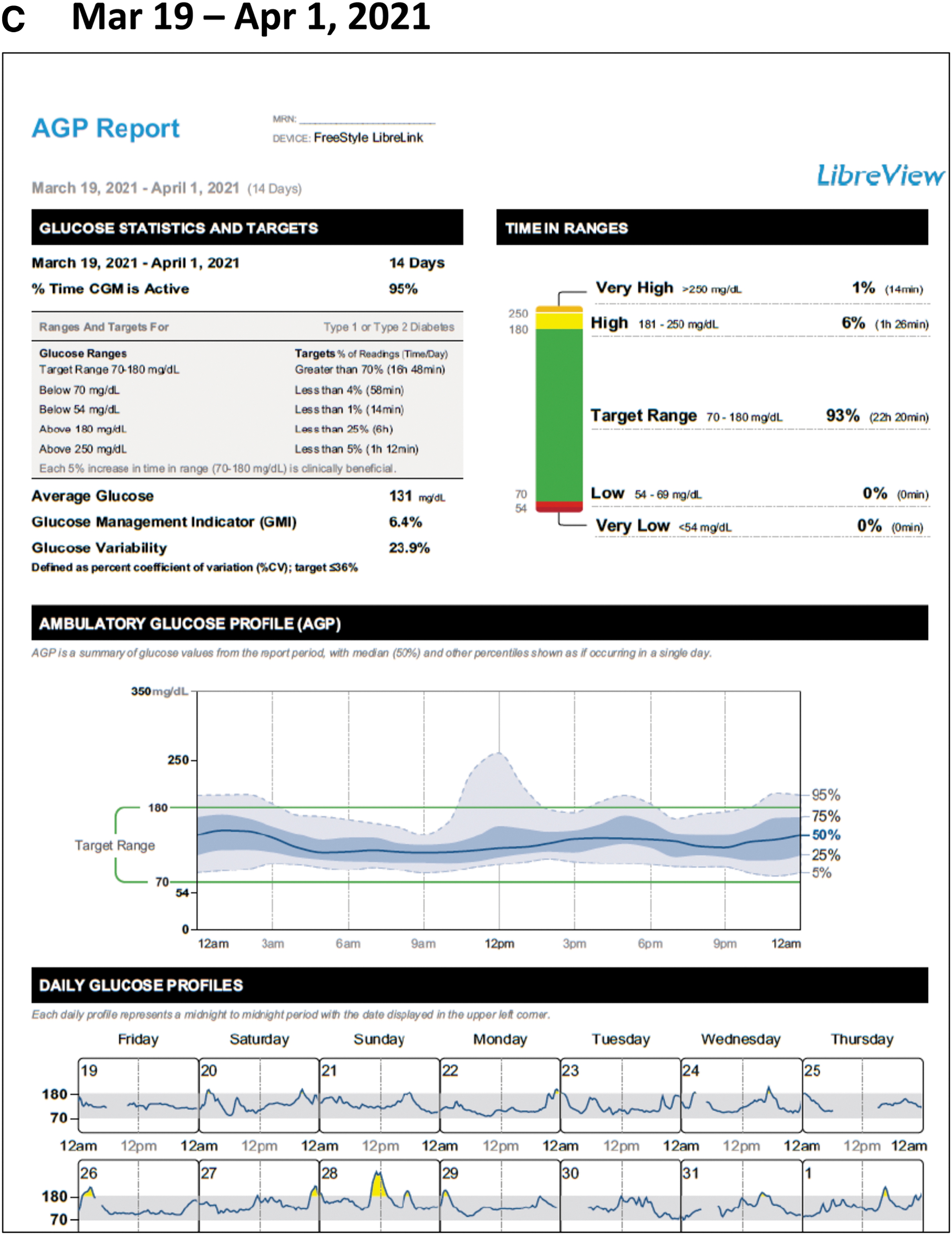
Glycemic status during COVID-19 recovery.
Over the course of 1 year, the patient was able to work through her concerns regarding hypoglycemia, as well as intensification of therapy; initiating first basal insulin then basal-bolus insulin, using CGM for insulin titration. This was achieved 100% remotely by the diabetes educators in conjunction with primary care.
Case 4: Newly diagnosed T1D (Amy B. Criego, MD)
This was a 3-year-old child who was diagnosed in fall 2020. Hyperglycemia without evidence of instability or diabetic ketoacidosis (DKA) was noted by primary care provider late in the day and the child was seen in the Pediatric Endocrine Clinic the next morning. Insulin therapy with multiple daily injections was initiated with diabetes education and CGM was started that day. At the initial visit, the point of care HbA1c was just over 13%. The family had daily telephone visits with the physician and two additional telemedicine video visits for education during the initial 2 weeks after diagnosis with continued remote follow-up until 6 weeks after diagnosis. CGM data were reviewed remotely at each contact point.
Although administering long-acting insulin in the morning is generally recommended at diagnosis in younger children, the family felt that this would be a difficult time of day and opted to administer in the evening. However, as shown in Figure 4A, this caused a significant decrease in overnight glucose, which was evident in the CGM data (A). Uncertainty and caution with premeal insulin dosing initially was also evident with large postprandial excursions during the day (B). When reviewing and discussing the data with the family via a telephone visit, the family agreed to move long-acting insulin administration to the morning after the 1st week. Further discussion helped the family become more comfortable with premeal dosing and transition to more consistent premeal carbohydrate-based dosing after the 2nd week.
Initial and follow-up AGP reports.
In our review of the CGM data at our follow-up telemedicine visit (Fig. 4B), we observed significant improvements in both overnight glucose (A) and reductions in postprandial glucose (B). Although this is typical with as there is recovery of endogenous insulin, the use of CGM allowed us to make informed decisions about insulin dose adjustments and timing of insulin administration, which improved the time in range (C) and reduced glycemic variability (D) more safely and efficiently. The use of CGM during the remote visits allowed the family to be seen conveniently and helped the parents gain confidence in their ability to safely manage their child's diabetes.
Summary
We found that use of telemedicine patient consults and remote monitoring of CGM and insulin data enabled us to assess glycemic control and make therapy adjustments without the potential hazards and patient burden of in-person clinic visits. Moreover, our ability to review and discuss the data with our patients helped them better understand how their therapy impacted daily glucose management, which, in turn, enhanced their engagement in their daily self-management. Although once considered to be more experimental than practical, recent studies have clearly demonstrated the usefulness of telemedicine technologies throughout the COVID-19 pandemic. 15 –17 Given the growing body of evidence supporting use of remote monitoring and telemedicine visits, we believe that these technologies have the potential to reduce the need for in-person clinic visits.
Footnotes
Acknowledgments
The authors thank Christopher G. Parkin, MS, CGParkin Communications, Inc., for editorial support.
Author Disclosure Statement
A.L.C. has received research support, consulted or has been on the scientific advisory board for Abbott Diabetes Care, Dexcom, Insulet, Medtronic, Novo Nordisk, Roche, and Sanofi. His employer, nonprofit International Diabetes Center/HealthPartners Institute/Park Nicollet, contracts for his services and he receives no personal income from these activities. T.W.M. reports research and speaking support from Abbott Diabetes Care, Dexcom, Medtronic, Insulet, Lilly, and Novo Nordisk. He has received speaking support from Medscape, consulted for Bigfoot Biomedical, and received a grant to write an article from Eli Lilly. His employer, nonprofit International Diabetes Center/HealthPartners Institute/Park Nicollet, contracts for his services and he receives no personal income from any of these activities. His employer, nonprofit International Diabetes Center/HealthPartners Institute/Park Nicollet, contracts for his services and he receives no personal income from these activities. L.J. reports no disclosures. A.B.C. has received research support and/or consulted for Abbott Diabetes Care, DexCom, Insulet, Medtronic, Novo Nordisk Roche Diabetes Care and Sanofi. Her employer, nonprofit International Diabetes Center/HealthPartners Institute/Park Nicollet, contracts for her services and she receives no personal income from these activities.
Funding Information
Funding for the development of this article was provided by Abbott Diabetes Care.
