Abstract
Continuous glucose monitoring devices have significantly improved in many respects compared with earlier versions. As sensor accuracy improved, U.S. Food and Drug Administration approved a nonadjunctive indication for use that no longer required confirmatory blood glucose monitoring. This article discusses the performance characteristics and regulatory classifications for the current systems that are relevant to informed clinical decision-making.
Introduction
Although ongoing advances in reliability and convenience have led to the growing adoption of continuous glucose monitoring (CGM) among individuals with diabetes who are treated with intensive insulin therapy, sensor accuracy, which can impact both the clinical utility and patient acceptance of CGM, has long and appropriately been the dominant focus in assessing the efficacy and safety of these devices. However, it is difficult for clinicians to compare the accuracy and precision of CGM devices because of differences in assessment protocols and how results are reported. Sensors may also differ with regard to duration of wear and the impact of activity and interfering substances. In this article we discuss the performance characteristics and regulatory classifications for the current systems that are relevant to informed clinical decision-making.
Continuous Improvement in CGM Accuracy
Mean absolute relative difference
A common metric for assessing CGM accuracy is the aggregate mean absolute relative difference (MARD), which compares all temporally matched sensor data with reference measurements (e.g., Yellow Springs Instrument [YSI]) across all subjects of a given study. Reported as a percentage, MARD is the average of the absolute error between the CGM values and matched reference values. A small MARD number indicates that the CGM readings are close to the reference glucose value, whereas a larger MARD number indicates greater discrepancies between the CGM and reference glucose values.
As given in Figure 1, we have seen significant improvements in CGM sensor accuracy as measured by MARD over the past 20 years. Improvements in CGM accuracy have also permitted the integration of CGM into advanced automated insulin delivery (AID) systems, which have been shown to be safe and effective in reducing HbA1c and hypoglycemia and increasing users' time spent in their target glycemic range. 1 –7
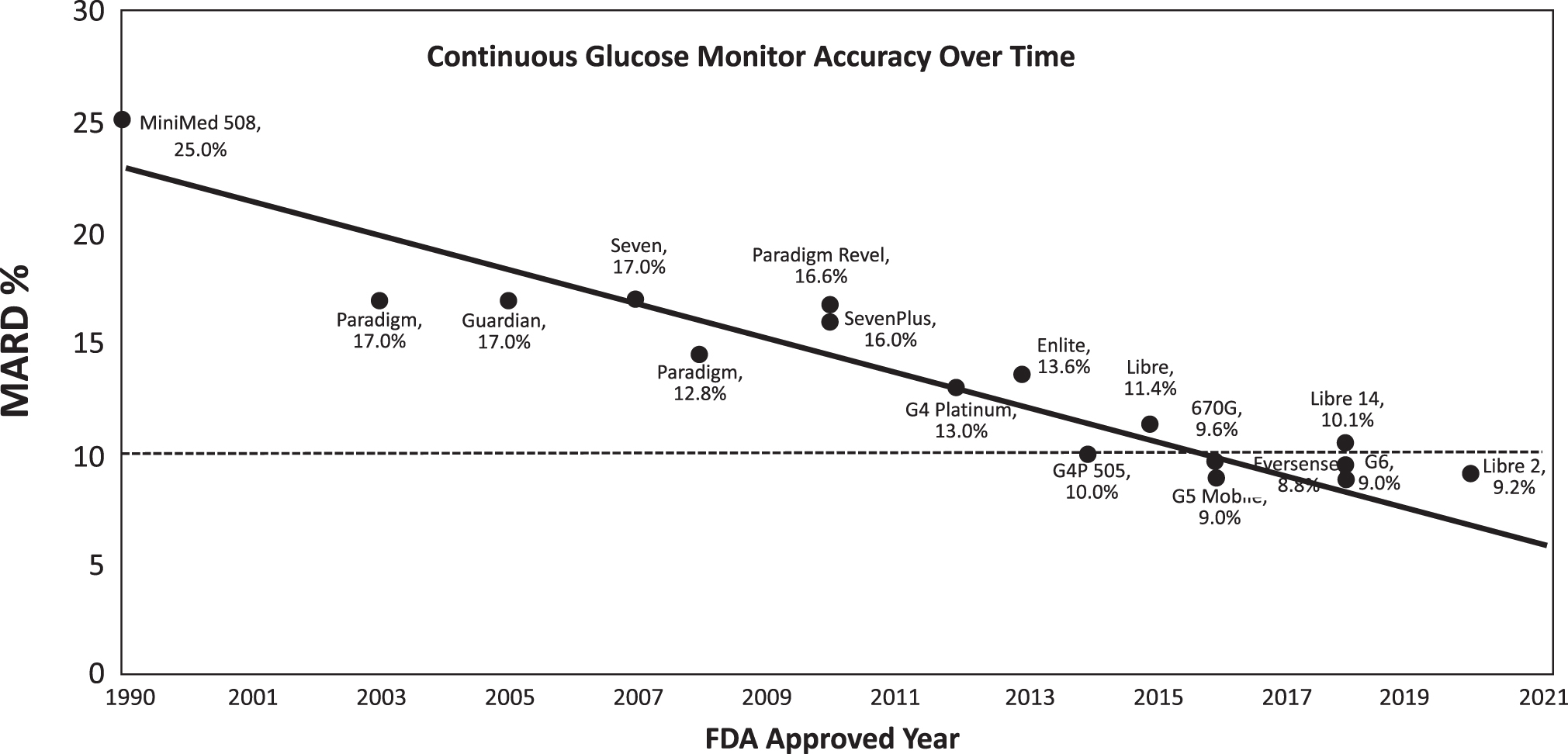
Incremental improvements in MARD accuracy over the past 20 years. MARD, mean absolute relative difference.
Improvements in MARD have also led to changes in how CGM data are used in daily self-management, specifically in individuals treated with intensive insulin management. Before 2016, all CGM systems were indicated for use as an adjunct to traditional blood glucose monitoring (BGM). Users were advised to confirm their CGM values with a fingerstick BGM result before making any adjustments in their insulin dosing. This was referred to as adjunctive use with BGM. As sensor accuracy improved, U.S. Food and Drug Administration (FDA) approved a nonadjunctive indication for use that no longer required confirmatory BGM.
Pitfalls of MARD and other accuracy measurements relative to clinical decision-making
Physiological differences in changes in blood versus interstitial glucose
MARD may be significantly impacted by “lag time” between blood glucose and interstitial glucose, particularly with rapid rates of glucose change. As demonstrated in early studies by Basu et al., 8 the mean time to appearance of tracer glucose in the abdominal subcutaneous tissue after intravenous bolus is between 5 and 6 min in the resting, overnight fasted state. However, the glucose transport lag time may vary when the patient is not in the fasting state (e.g., rapidly rising or falling glucose levels). 8 As a result, the CGM and blood glucose values that are paired according to their measurement time stamp may be erroneously paired from the physiological point of view. 9 However, significant improvements have been made in glucose algorithms to address this. In addition, if a manually calibrated CGM system is used, other influencing factors should be considered, such as possible bias between the reference method and the BGM device used for calibration. It is also important to consider the blood samples used for comparison. If the device is calibrated using capillary blood glucose, use of other samples (e.g., venous blood or arterialized venous blood) for the reference method can introduce additional bias. 10,11
Lack of standardization in assessing accuracy
Although the FDA has clear expectations as to required accuracy for approval of CGM devices, there is no standardization with regard to testing methodology among manufacturers. Therefore, comparing results between studies performed differently is difficult; a true comparison requires a head-to-head comparison in the same study. Specific differences among studies include the populations studied (e.g., pediatric or adult), parameters assessed (e.g., glucose ranges, day of sensor wear, various rates of changing glucose), and other variables (number of paired data points).
Persistent perceptions based on early generation CGM systems
It is also important to consider how the accuracy of earlier CGM systems can influence clinician perceptions of more recent generation devices. For example, the concurrence assessments of the original FreeStyle Libre® sensor with reference values within the 40–60 mg/dL glucose range was only 25.2% 12 ; the percentage of concurrent values within this range has doubled to 52.9% in the FreeStyle Libre 2 system. 13 Despite significant improvement in accuracy within the lower glucose range, perceptions of inaccuracy persist.
These are just a few of the confounding factors that challenge clinicians when assessing the accuracy of the CGM systems they are considering for their patients. However, from our perspective, these issues are not those most relevant to clinical decision-making. What really matters is how these devices perform in patients' hands and used in real-world conditions.
Considerations for Clinical Decision-Making
Evidence of efficacy and safety
Large randomized controlled trials (RCTs) have shown that all of today's CGM technologies improve glycemic control and reduce hypoglycemia risk. 14 –21
Although RCTs are still considered the “gold standard” of scientific research, 22 there is an increasing appreciation for the value of real-world evidence (RWE) in demonstrating how medical devices function in the hand of patients in real-life settings. 23 –26
Numerous real-world studies have also shown that use of CGM improves overall glycemic control, 27 –34 reduces hypoglycemia risk, 27,28,33 –35 and has been shown to reduced the incidence of diabetes-related events and hospitalizations. 27,28,32,36 In addition, current AID systems with integrated CGM (iCGM) can automatically suspend or increase insulin infusion in response to current and/or predicted hypoglycemic and hyperglycemic events in children and adults with type 1 diabetes (T1D). 21,37,38
Interferents
FreeStyle Libre 2
Taking ascorbic acid (vitamin C) while wearing the sensor may falsely raise glucose readings. 39 The level of inaccuracy depends on the amount of the interfering substance active in the body. The sensors have been tested for common interfering compounds such as acetaminophen, ibuprofen, levodopa, methyldopa, dopamine, salicylic acid, uric acid, and hydroxyurea. At the highest concentrations under therapeutic treatment, these compounds do not show clinically significant interference. However, glucose readings may be falsely elevated at higher doses of ascorbic acid (500 mg/day) supplements. As such, the sensor may potentially miss hypoglycemia, specifically severe hypoglycemia.
In a clinical evaluation of the effect of ascorbic acid on sensor accuracy, data from 57 adults with diabetes were collected over a 13-h period 40 (Table 1). Each subject had a 1-h baseline phase where venous blood was collected every 10 min. After this first hour, a dose of 1000 mg of ascorbic acid was given with a meal and venous samples were collected every 20 min for the next 4 h. Participants received a second dose of 1000 mg of ascorbic acid with a meal and the same process was continued for another 4 h. A third dose of 1000 mg of ascorbic acid was then given, and study subjects were followed for four more hours.
The Maximum Average Bias from the Reference and Baseline at Each Ingestion of Ascorbic Acid on the FreeStyle Libre 2
Abbott cautions patients to remove the sensor before magnetic resonance imaging (MRI), computed tomography (CT) scan, or high-frequency electrical heat (diathermy) treatment. 38 Although the evidence is limited, the sensor glucose data do not appear to be affected by in vitro exposure to X-ray and radiographic testing (RT), computed tomography (CT) or magnetic resonance imaging (MRI) as recorded by the FreeStyle Libre Pro sensors. 41
Dexcom G6
The accuracy of Dexcom G6 system is affected by hydroxyurea medications; individuals treated with these medications should be advised to use an alternative glucose monitoring method. 42 Although acetaminophen is contraindicated with previous versions of the Dexcom sensors, G6 systems can be used when taken up to a maximum dose of 1000 mg every 6 h.
The effect of repeated doses of acetaminophen on G6 sensor performance was evaluated in a clinical study. 43 The study involved 15 adults with diabetes who wore one sensor in the abdomen; data from 14 adult subjects were collected over one 13-h period during 3–7 days of the sensor wear. Each subject had a 1-h baseline phase in which venous blood was collected every 10 min. A dose of 1000 mg acetaminophen was given with a meal after the first hour and venous samples were collected every 20 min over the next 4 h. Acetaminophen ingestion and blood sample collection was repeated two more times in 4-h intervals. Table 2 presents the maximum average bias from YSI and baseline after each administration of acetaminophen. When taken at higher doses, it may falsely elevate sensor glucose readings. Use of repeat doses of acetaminophen is shown to increase the interference from acetaminophen.
The Maximum Average Bias from the Reference and Baseline at Each Ingestion of Acetaminophen on Dexcom G6
Dexcom cautions against wearing the sensors for MRI, CT scan, or diathermy treatment. However, no effects from X-ray, CT, or angiography have been observed, 44 but some interference from MRI has been reported. 45
Eversense CGM system
Medications such as mannitol intravenous and irrigation solutions may increase blood mannitol concentrations and cause falsely elevated readings of sensor glucose results. 46 On the other hand, due to this sensor's different chemistry, interference with both ascorbic acid and acetaminophen is nil. Additionally, the Eversense system is contraindicated in individuals for whom dexamethasone or dexamethasone acetate may be contraindicated. Senseonics cautions against Lithotripsy, diathermy or electrocautery procedures or massage therapy near the inserted sensor. Senseonics cautions patients to remove their transmitter before MRI procedures, medical X-rays or CT scans. Close contact with direct electromagnetic interference may interfere with the smart transmitter's ability to send data to the mobile device. 47 However, the sensor can be safely scanned in a closed boar MR scanner that meets the conditions specified in the Eversense safety information. 47
MiniMed Guardian Sensor 3
Medications with acetaminophen may falsely raise sensor glucose readings. 48 The level of inaccuracy depends on the amount of acetaminophen active in your body and may be different for each person. Individuals treated with hydroxyurea medications are advised to use an alternative glucose monitoring method. 49 The sensor should not be exposed to MRI equipment, diathermy devices, or other devices that generate strong magnetic fields as the performance of the sensor has not been evaluated under those conditions and may be unsafe.
FDA classifications: class II versus class III
The FDA assigns each approved medical device to one of three medical device classifications: class I, class II, and class III. These classifications are based on the device risks and the regulatory controls necessary to provide a reasonable assurance of safety and effectiveness. Class I devices are not intended for use in supporting or sustaining life or of substantial importance in preventing impairment to human health, and they may not present a potential unreasonable risk of illness or injury. 50 Class II medical devices are considered to be moderate-to-high risk because they are more likely to involve sustained use by the patient. 50 Class II devices require device-specific special controls in addition to the general controls, such as performance standards, postmarket surveillance, and special labeling. 51 Class III devices are considered the highest risk in that they are life-supporting or life-sustaining, for use in preventing impairment of human health or if the device presents a potential unreasonable risk of illness or injury. 50
The relevance of these classifications to CGM relates to the speed of device development and FDA clearance. For example, in addition to general controls for safety and performance, all the current class III CGM devices required filing an investigational device exemption, a more extensive time-consuming evaluation, and premarket approval. 51 Because of the filing of the Dexcom G6 device as de novo 510(k), there is a new pathway to approval as a class II device. The special controls that enable this are new and include more stringent performance standards. Devices meeting these performance standards are identified as iCGM devices. Only the FreeStyle Libre 2 and G6 are classified as iCGM (e.g., class II devices) to date.
The special controls specified for the products meeting the FDA requirements are listed hereunder. 52
Design verification and validation must include the following:
○ Robust clinical data demonstrating the accuracy of the device in the intended use population.
○ The clinical data must include a comparison between iCGM values, and blood glucose values in specimens collected in parallel that are measured on an FDA-accepted laboratory-based glucose measurement method that is precise and accurate, and that is traceable to a higher order (e.g., an internationally recognized reference material and/or method).
○ The clinical data must be obtained from a clinical study designed to fully represent the performance of the device throughout the intended use population and throughout the measuring range of the device.
○ Clinical study results must demonstrate consistent analytical and clinical performance throughout the sensor wear period.
○ Clinical study results in the adult population must meet the performance requirements listed in Table 3.
○ Data demonstrating similar accuracy and rate of change performance of the iCGM in the pediatric population compared with that in the adult population, or alternatively a clinical and/or technical justification for why pediatric data are not needed, must be provided and determined by FDA to be acceptable and appropriate.
○ Data must demonstrate that throughout the claimed sensor life, the device does not allow clinically significant gaps in sensor data availability that would prevent any digitally connected devices from achieving their intended use.
Design verification and validation must include a detailed strategy to ensure secure and reliable means of iCGM data transmission to provide real-time glucose readings at clinically meaningful time intervals to devices intended to receive the iCGM glucose data.
Design verification and validation must include adequate controls established during manufacturing and at product release to ensure the released product meets the performance specifications discussed previously.
The device must demonstrate clinically acceptable performance in the presence of clinically relevant levels of potential interfering substances that are reasonably present in the intended use population, including but not limited to endogenous substances and metabolites, foods, dietary supplements, and medications.
The device must include appropriate measures to ensure that disposable sensors cannot be used beyond its claimed sensor wear period.
Performance Requirements for the Adult Population
iCGM, integrated continuous glucose monitoring.
The performance of Decom G6 and FreeStyle Libre 2 against the iCGM performance requirements are given in Table 3. 52
CGM versus iCGM
In addition to their Class II designations, both the FreeStyle Libre 2 and G6 are also the first two systems to receive FDA clearance as iCGM devices owing to their additional feature of interoperability with other devices. As defined by the FDA, iCGM systems “are designed to reliably and securely transmit glucose measurement data to digitally connected devices, including automated insulin dosing systems.” 53 These devices can be used alone or in conjunction with digitally connected medical devices for the purpose of glycemic management. Of interest, the Guardian Sensor 3, a Class III device that still requires adjunctive use with BGM, is integral to the MiniMed 780G.
Summary
Over the past 10 years, there has been dramatic transition from traditional BGM to CGM. Initially limited to individuals with T1D, adoption of CGM is now rapidly increasing within the type 2 diabetes (T2D) population. Although the majority of T2D clinical trials have focused on patients treated with intensive insulin regimens, 18,34,54 a growing body of RWE is now demonstrating both the clinical and economic values of CGM use in T2D patients treated with nonintensive therapies. 29,32,55
Key drivers of CGM uptake have been the improved accuracy of current devices, expanded functionality of alarms and alerts, and the added convenience of fewer (or no) fingerstick requirements and extended sensor life. All these factors have significantly improved the utility and reliability of CGM use and reduced the burden of diabetes self-management. 56
Although the lack of standardization in assessing and reporting the accuracy of today's CGM devices has led to some confusion among clinicians and patients, the main take-away from this article is that all current CGM devices are accurate enough for safe and effective diabetes management. Moreover, the glycemic benefits of all the devices have been conclusively demonstrated in numerous clinical trials 14,16 –21,27,28,33,37,38,57 –60 and real-world studies. 27 –32,36,58,61 –63 Therefore, from the standpoint of clinical decision-making, the individual needs and preferences of each patient should be considered when helping them select a CGM system. This goes to the core of person-centered medicine.
Footnotes
Authors' Contributions
T.S.B. and S.A. wrote the article. All authors reviewed the article before submission and take full responsibility for the accuracy of its content.
Acknowledgments
The authors thank Christopher G. Parkin, MS, CGParkin Communications, Inc., for editorial support.
Author Disclosure Statement
T.S.B. has received research support from Abbott, Capillary Biomedical, Dexcom, Diasome, Eli Lilly, Kowa, Lexicon, Medtronic, Medtrum, Novo Nordisk, REMD, Sanofi, Senseonics, Viacyte, vTv Therapeutics and Zealand Pharma; consulting honoraria from Abbott, Lifescan, Novo Nordisk and Sanofi; and speaking honoraria from Medtronic and Sanofi. S.A. is an employee of Abbott Diabetes Care.
Funding Information
Funding for the development of this article was provided by Abbott Diabetes Care.
