Abstract
Numerous studies have demonstrated the clinical benefits of continuous glucose monitoring (CGM) in individuals with diabetes. Within ongoing innovations in CGM technology, individuals now have an expanding array of options that allow them to select the device that meets their individual needs and preferences. Although demand for CGM in primary care continues to grow, many clinicians are reluctant to prescribe this technology due to their unfamiliarity with the various devices, uncertainty about which devices are best suited to each patient and the feasibility of using CGM. This article reviews the features and functionality of the most recent commercially available CGM devices and provides guidance for integrating CGM use into clinical practices.
Introduction
Continuing advances in glucose monitoring technologies have led to the development of innovative continuous glucose monitoring (CGM) systems that enable individuals with type 1 diabetes (T1D) and type 2 diabetes (T2D) to optimize glycemic control and improve the quality of their lives. Within the past decade, real-time CGM (rtCGM) and flash CGM have emerged as a standard of care for individuals with diabetes who are treated with intensive insulin regimens. 1 –6
The clinical benefits of these systems have been demonstrated in numerous studies of individuals with T1D regardless of their insulin delivery method. 7 –22 These benefits include reductions in hemoglobin HbA1c, 7,9,11,17,18,23 –27 fewer severe hypoglycemia events, 24,25,28 increased time within target glucose range (time in range [TIR]), 11,18,19,29 and reductions in time below range. 11,18 A significant association is found between CGM use and reductions in both HbA1c and diabetes-related hospitalizations in individuals with T1D 24,25,28 and T2D treated with intensive and nonintensive therpaies. 30 –33
Despite the demonstrated clinical efficacy and safety of CGM use, many clinicians may be reluctant to prescribe this technology due to their unfamiliarity with the various devices, uncertainty about which devices are best suited to each patient, and the feasibility of using CGM. This article reviews the features and functionality of the most common commercially available CGM devices and provides guidance for integrating CGM use into clinical practices.
Personal CGM
Current technologies
Personal CGM refers to patient use of the generated data to monitor glycemic control and guide therapy. Two types of personal use CGM technologies are commercially available: rtCGM and flash CGM. Both systems continuously monitor interstitial glucose levels and transmit these data in numerical and graphical formats to the user's handheld reader or a smartphone app. While rtCGM devices automatically transmit the data to the reader or smartphone, flash CGM users must manually scan the sensor with their reader or smartphone to transfer data.
Until recently, the major difference between the devices as that rtCGM was provided real-time alerts and alarms that warned users when their current or impending glucose exceeded their glycemic targets. This immediate information enables users to quickly identify and mitigate acute glycemic events, providing significant safety advantages and enhanced decision support. These alerts also provide immediate feedback to a user about the lifestyle or therapy decisions that were recently made. In a 2016 survey of 300 individuals with T1D or T2D who regularly use CGM, most respondents reported that they rely heavily on their CGM data, specifically, the rate of change arrows, to make adjustments in their insulin dosages, and the real-time alarms and alerts to prevent and manage hypoglycemia. 34
With the introduction of the FreeStyle Libre 2 system (Abbott Diabetes Care, Inc., Alameda, CA) in 2020, both types of systems offer this added safety net. Table 1 presents the key features of the current CGM systems.
Key Features of Current Personal Continuous Glucose Monitoring Systems
Requires surgical insertion procedure.
System is designed for data sharing and is currently under FDA review.
Does not connect with current insulin pump systems
Currently under FDA review.
Confirmatory testing is advised under certain circumstances as indicated in each system's safety information.
APAP, Paracetamol; DMS, Data Management System; FDA, Food and Drug Administration.
rtCGM Versus flash CGM
Because rtCGM and flash CGM differ in the way users obtain their data and interact with their reader or smartphone, there has been considerable debate about whether these differences change outcomes. Two randomized trial have reported greater glycemic improvements (e.g., increased TIR, lower glycemic variability) with rtCGM use compared with flash CGM. 35,36 However, two recent real-world, large database studies have shown similar outcomes with use of either rtCGM or flash CGM. 37,38 Both studies assessed clinical outcomes in head-to-head comparisons of the FreeStyle Libre 14-Day (FSL) system versus the Dexcom G5 or G6 (DEX) systems (Dexcom, Inc., San Diego, CA) in both T1D and T2D populations.
In the first study, Miller et al. conducted a retrospective analysis of the IBM Explorys U.S. EHR Database to assess changes in HbA1c during the 6 months following CGM initiation in adult T1D and T2D populations treated with short- or rapid-acting insulin. 35 In both T1D and T2D cohorts, FSL and DEX users were propensity score matched (2:1) on demographic and baseline clinical characteristics. At 6 months post-CGM initiation, no differences were observed in HbA1c reductions using either DEX or FSL among T1D (−0.35% and −0.37%, respectively, both <0.001) and T2D adults treated with short- or rapid-acting insulin (−0.73 and −0.79, respectively, both P < 0.001). Reductions were notably greater in T1D patients with ≥8.0% HbA1c at baseline among both FSL and DEX users (−0.62% and −0.66%, respectively, both P < 0.001) with no significant between-group differences. However, in the T2D population with baseline HbA1c ≥8.0%, reductions were only slightly greater. An analysis of the baseline demographics of the unmatched T1D and T2D cohorts revealed that the FSL population was more racially and socioeconomically diverse and included a higher percentage of patients with comorbidities.
In a similar analysis of the IBM MarketScan Commercial Claims and Medicare Supplemental dataset, Hirsch et al. looked at all-cause hospitalizations (ACH) and acute diabetes events (ADE) in T1D and intensively treated T2D populations. 36 At ≥6 months post-CGM acquisition, FSL and DEX users within the T1D cohort experienced similar event-free survival rates of ACH (94.9% and 95.4%, respectively) and ADE (94.9% and 94.8%, respectively) for ADE with no significant between-group differences. Within the T2D cohort, the 6-month post index event-free survival rates for FSL and DEX patients were 90.6% versus 90.4%, respectively, and 96.7% versus 96.4%, respectively for ADE. Again, there were no significant between-group differences. However, in this study, assessment of the unmatched T1D and T2D cohorts showed that FSL users were older, and more likely to live outside a metropolitan area and have more comorbidities, whereas DEX users had more ADEs and hypoglycemia events at baseline and were more likely to wear an insulin pump and visit an endocrinologist and/or dietician.
In summary, these studies demonstrate that use of rtCGM and flash CGM in both T1D and T2D populations can be expected to achieve similar clinical outcomes in patients using multiple injections.
Professional CGM
Professional CGM devices are used intermittently by patients and are owned by the clinic. The glucose data gathered through intermittent use allow clinicians to make informed treatment decisions by retrospectively assessing the patient's glycemic control throughout the day. When used in the “unblinded” mode, these devices can help patients better understand how their medications, dietary habits, and other lifestyle behaviors impact their glucose levels. These data can also be used for documentation purposes when submitting for insurance coverage for long-term CGM use. When used in the “blinded” mode, clinicians can capture the data without influencing patient behavior.
Two professional CGM systems are currently available: The FreeStyle Libre Pro system (Abbott Diabetes Care, Inc.) and Dexcom G6 Pro system (Dexcom, Inc.). The FreeStyle Libre Pro system can only be used in the blinded mode, whereas the Dexcom G6 Pro system can be used in both blinded and unblinded modes. Table 2 presents the key features of the current Professional CGM systems.
Key Features of Current Professional Continuous Glucose Monitoring Systems
Personalized Self-Management
Matching the CGM device to patient needs
Although effective diabetes care relies predominately on patients' willingness and ability to integrate the numerous tasks inherent to self-management into the reality of their daily lives, there is a tendency to take a “one size fits all” approach when prescribing CGM, which can negatively impact adherence and subsequent outcomes. Therefore, it is critical that health care providers take time to assess both the clinical requirements and individual needs and preferences of each patient.
The criteria for helping patients determine the most appropriate CGM system can be broken down into three base questions: Is it indicated for the patient? Does it address the clinical requirements of the patient? Does it meet the patient's individual needs and preferences? Consideration of the key features listed in Table 1 provides a reasonable starting point for making these determinations; however, there are varying “shades of gray” that must be considered when discussing CGM options with each patient.
Clinical considerations
In terms of matching the various devices to the clinical requirements of each patient, the ability to share data with family/friends and caregivers and active alarms/alerts are clearly beneficial for patients treated with intensive insulin management or at elevated risk for severe hypoglycemia. All current CGM devices except the FreeStyle Libre 15-day system offer alarms/alerts that warn users when glucose levels have crossed upper and lower glucose thresholds. The Dexcom G6, Guardian Sensor 3 and Eversense XL systems also offer predictive alarms that provide advanced warning of impending hypoglycemia and hyperglycemia. The clinical benefits of this additional safety feature have been demonstrated in recent studies. 39 –42 However, the benefits of alarms and alerts must be weighed against each patients' personal preferences. For example, patients who experience frequent alarms may begin to ignore them or discontinue CGM use altogether. 43 –45 In these situations, clinicians may want to consider various strategies for minimizing these occurrences, such as adjusting the glucose threshold levels. However, the G6 and Guardian Sensor 3 have a default “urgent” alarm that cannot be silenced. Clinicians may want to consider the FreeStyle Libre 2 with optional alarms for patients who are not at high risk for hypoglycemia and prefer to control all of their alarm settings. However, all of the CGM systems that feature alarms/alerts have an automatic low alarm at 55 mg/dL that cannot be turned off as required by regulatory agencies.
Patients who are considering or currently using insulin pump therapy can use any of the current integrated CGM devices, such as the Dexcom G6 or FreeStyle Libre 2, for glucose monitoring or choose from one of the new CGM-integrated AID insulin pump systems. These include the Tandem Basal-IQ and Control-IQ (Tandem Diabetes Care, Inc., San Diego, CA) and OmniPod 5 (Insulet Corporation, San Diego, CA), which use the Dexcom G6 sensor, and the MiniMed 770G, which is paired with the Guardian Sensor 3. The MiniMed 780G with the Guardian Sensor 3 is also under Food and Drug Administration (FDA) review.
With the recent introduction of connected smart insulin pen technologies, patients who prefer using multiple daily insulin injection (MDI) therapy can accurately track dosages and injection times without sacrificing data and decision support. Data are transmitted to a smartphone device for review and shared with the health care provider. The Medtronic InPen, which connects with the Dexcom G6 and Medtronic Guardian Sensor 3, is the only device that is currently available; however, other similar smart pens with these capabilities are likely to receive FDA approval soon. We believe that use of insulin smart pens will be a standard of care within the next few years. Taking a somewhat different approach, the Bigfoot Unity (Bigfoot Biomedical, Milpitas, CA) utilizes a connected “smart cap” that fits onto the user's disposable insulin pen. Paired with the FreeStyle Libre 2 sensor, the cap provides insulin dosing recommendations based on the CGM data and health care provider instructions.
Convenience features
Advances in CGM technology have also led to significant improvements in usability and convenience. Perhaps the most significant advance in CGM technology is the ability to make insulin dosage adjustments without confirmatory blood glucose monitoring (BGM), thereby eliminating the need for fingerstick testing. In 2016, the Dexcom G5 Mobile device (Dexcom, Inc.) became the first CGM system to receive a nonadjunctive indication. Nonadjunctive indications for other systems, including the FreeStyle Libre system (Abbott Diabetes Care, Inc.) and Eversense implantable CGM sensor (Senseonics Holdings, Inc., Germantown, MD) soon followed. Today, there are four CGM systems indicated for nonadjunctive use: FreeStyle Libre 14-Day, FreeStyle Libre 2, Dexcom G6, and Eversense XL. The only CGM system without this indication is MiniMed Guardian Sensor 3 (Medtronic, Inc., Northridge, CA). FreeStyle Libre 2 and G6 devices have further reduced the need for fingerstick testing by providing devices that are factory calibrated and do not require BGM calibration, although G6 devices have the option of BGM calibration by users.
Another convenience factor to consider is the sensor wear time. Over the past years, sensor wear time has increase from 3 days up to 90 days (which may increase to 180 days pending FDA clearance) with the Eversense XL system. The Eversense XL system must be implanted and explanted by a provider. Patients who are not interested in an implanted sensor can choose from devices that require a sensor change from every 10 days (G6) to every 14 days (FreeStyle Libre 14-Day, FreeStyle Libre 2).
All current CGM systems require a warm-up period before the data can be used for therapy adjustments. The warm-up time required before using the CGM data are comparable between the G6, Guardian Sensor 3, FreeStyle Libre 14-Day, and FreeStyle Libre 2 systems, the Eversense XL requires 24 h of warm-up. However, as a safeguard, the data are not made available to the patient during the warm-up period.
When discussing CGM options with patients, a final consideration is cost. Based on current wholesale pricing, the costs for the Medtronic and Dexcom sensors are notably higher than the FreeStyle Libre systems. However, this may not be an issue for patients who have insurance coverage that requires little or no copay for CGM.
Considerations when integrating CGM into clinical practice
When making the decision to integrate CGM into a clinical setting, it is important for health care providers to consider a number of factors, including their familiarity with CGM, practice demographics, staffing resources, and office workflow. What is the provider's level of expertise with CGM? Is a diabetes educator or staff readily available to provide training and patient follow-up? Is the practice adequately staffed to handle the additional documentation and billing requirements? Consideration of these factors is helpful when determining the type of CGM they will utilize (e.g., personal or professional) as well as the specific CGM device(s) that best align with their patients and practice infrastructure.
For example, a practice that sees mostly older T2D patients treated with nonintensive insulin or noninsulin therapy may want to start with professional CGM, especially if the health care provider and staff are relatively unfamiliar with using CGM or staff resources are limited. Conversely, practices in which the providers have received some training in CGM may want to consider starting with the personal CGM devices. Based on our clinical experience, we have identified four levels in CGM complexity as a starting point for the best approach to integrating CGM into the practice (Fig. 1).
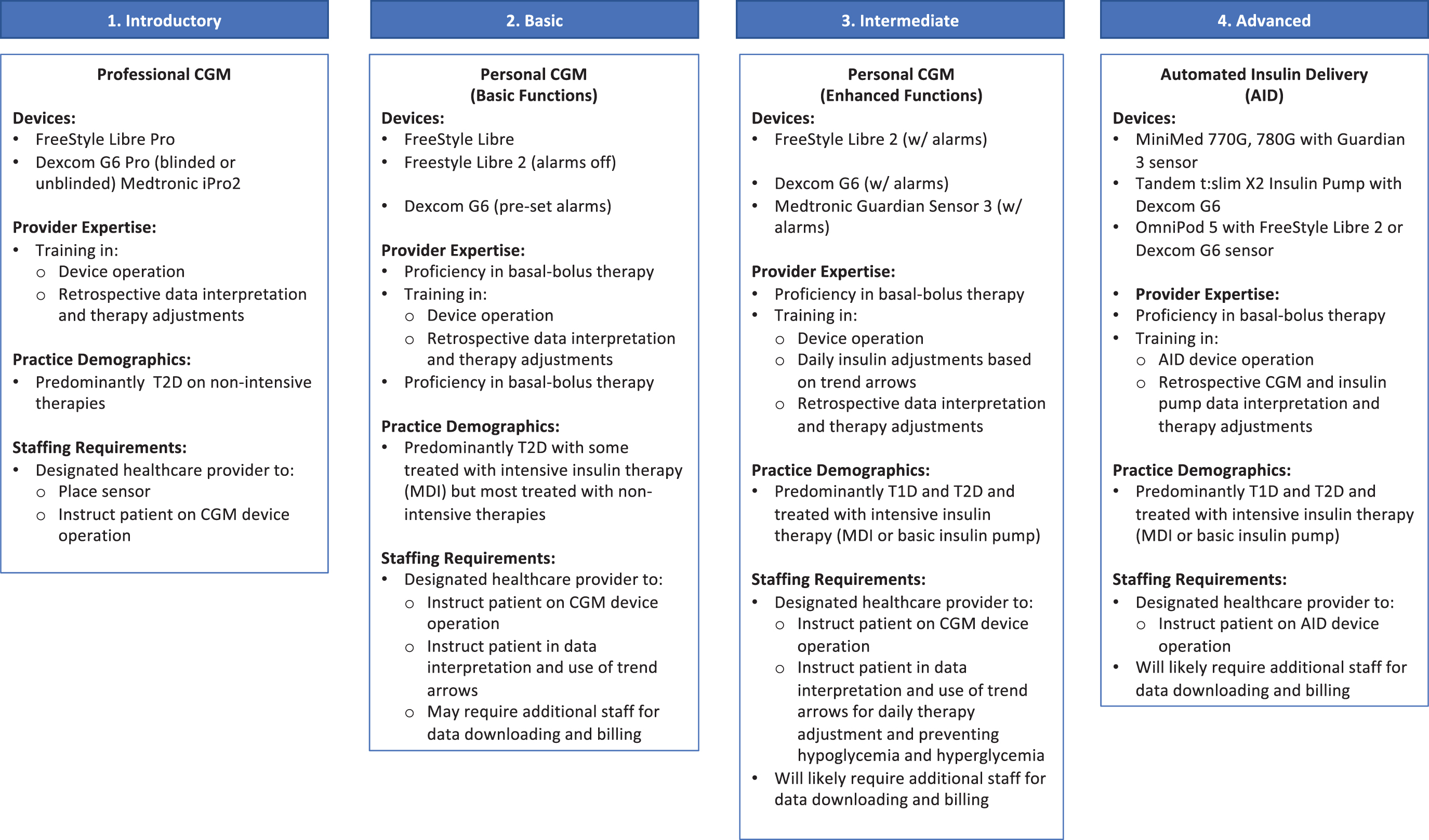
Levels of CGM integration into clinical practice. CGM, continuous glucose monitoring; T1D, type 1 diabetes; T2D, type 2 diabetes.
As shown in Figure 1, only basic expertise in CGM data interpretation is needed to initiate professional CGM in the clinic. However, the more advanced expertise is required if the clinic decides to expand into personal CGM use. Although basic CGM use (Level 2) recommends starting with no (or preset) alarms, health care providers may want to transition their patients to the use of more advanced features (e.g., threshold alarms) and begin instructing patients on use of trend arrows for insulin dosing.
A listing of recent consensus reports on use of trend arrows and general guidance for CGM use in clinical practice is presented in Table 3. 46 –51 For health care providers who are interested in the AID systems, Heile et al. recently published a comprehensive review of the components, differences, limitations, and considerations for integrating these devices into primary care practices. 52
Resources for Utilizing Continuous Glucose Monitoring in Primary Care
CGM, continuous glucose monitoring; ROC, rate of change; T1D, type 1 diabetes.
Summary
Innovations in CGM technologies have resulted in the development of an expanding array of products, ranging from devices with basic functionalities to advanced AID systems that adjust insulin infusion in response to current and impending glucose levels. Given these options, patients can now choose the CGM device that best meets their individual needs and preferences.
As the number of practicing endocrinologists continues to decline, 53 an increasing number of primary care providers are compelled to begin utilizing CGM with their diabetes patients. However, for many clinicians, the diversity in products creates uncertainty in how to integrate this technology into their practices.
Becoming familiar with all of the features and functionalities of the current devices and how they address the clinical needs of the various diabetes populations is clearly a critical first step. However, health care providers must also determine the level at which they will begin the integration process. This will require an objective assessment of their practice resources. What is the level of expertise with CGM within the practice? Do they have access to other clinical support (e.g., diabetes educator)? Is there adequate staffing to handle billing and the additional documentation associated with obtaining insurance coverage for their patients?
Although this article has discussed the key considerations for CGM integration, it is only intended as a starting point. We strongly recommend that interested health care providers access the resources listed in Table 3. Given the demonstrated benefits of CGM, and the growing demand for using this technology, we urge health care providers to become knowledgeable about these devices, begin to integrate CGM into their practices at the appropriate level, and then transition to more advanced levels of use.
Footnotes
Acknowledgments
The authors thank Christopher G. Parkin, MS, CGParkin Communications, Inc., for editorial support.
Author Disclosure Statement
I.B.H. receives research funding from Medtronic Diabetes, Insulet, and Beta Bionics, has received consulting fees from Abbott Diabetes Care Bigfoot, Gwave, and Roche. E.M. has received consulting fees from Abbott, Astra Zeneca, Novo Nordisk Boehringer Ingelheim, Eli Lilly, Merck, and Sanofi U.S., and has acted as a speaker for Abbott, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Funding Information
Funding for the development of this article was provided by Abbott Diabetes Care.
