Abstract
Background:
Automated insulin delivery systems are associated with improved glycemic outcomes for patients with diabetes. Ultrarapid lispro (URLi), which has an accelerated pharmacokinetic profile and shows superior postprandial glucose control compared to lispro (Humalog®), is a potential candidate for use in these systems.
Methods:
In this double-blind, crossover trial over two 4-week treatment periods, we evaluated URLi in a hybrid closed-loop system using the Medtronic MiniMed™ 670G system (670G). After a 2-week lead-in on lispro, 42 adults with type 1 diabetes were randomized to 1 of 2 treatment sequences of URLi and lispro delivered via the 670G. Primary endpoint was the percentage of time with glucose values within target range 3.9–10.0 mmol/L (70–180 mg/dL; %TIR).
Results:
Both treatments achieved %TIR over the 24-h period that was above the 70% minimum recommended by the International Consensus Guidance: URLi, 77.0%; lispro, 77.8%; P = 0.339. %Time <3.0 mmol/L (54 mg/dL) was similar between treatments (URLi, 0.3%; lispro, 0.4%; P = 0.548) and %time <3.9 mmol/L (70 mg/dL) was lower with URLi (1.5%) versus lispro (2.2%); P = 0.009, while %time >10.0 mmol/L (180 mg/dL) was higher with URLi (21.5% [309.4 min] vs. 19.9% [287.2 min]; P = 0.088). Mean sensor glucose was significantly higher with URLi versus lispro with least squares mean difference of 0.17 mmol/L or 3.0 mg/dL (P = 0.011) between treatments. Insulin dose, %time in Auto Mode per week, and pump settings were similar between treatments. No serious adverse events (AEs) (including severe hypoglycemia) or discontinuations occurred, and the incidence of treatment-emergent AEs was similar between treatments. Although the overall incidence and rate of unplanned infusion set changes were similar between treatments, a significantly higher rate of unplanned infusion set changes due to infusion site reactions was seen during URLi treatment compared with lispro: 0.12 versus 0.00 events/30 days (P = 0.063).
Conclusions:
URLi demonstrated good glycemic control that was comparable to lispro and showed a similar safety profile to lispro with the 670G hybrid closed-loop system. Trial registration: ClinicalTrials.gov, NCT03760640.
Introduction
Integrated insulin pump and continuous glucose monitoring (CGM) systems, also known as closed-loop systems, have a greater potential of improving glycemic control and reducing disease burden. 1 Fully automated closed-loop systems mimic the human pancreas by using real-time CGM data to determine and automatically adjust the pump's basal and bolus insulin delivery. Hybrid closed-loop systems such as the Medtronic MiniMed™ 670G (Medtronic Diabetes, Northridge, CA; 670G) automatically adjust basal insulin delivery only, while the user manually programs meal and correction boluses.
Over the years, there has been increasing evidence to show that integrated systems can significantly improve hemoglobin A1c (HbA1c) outcomes without increasing the risk of hypoglycemia. 2 –5 In adolescents and adults >14 years of age using the 670G system, significant improvements in HbA1c and time in range, and less overall hypoglycemia and hyperglycemia were seen at the end of the study phase compared to baseline. 6 While closed-loop systems show these advantages over nonintegrated systems, their performance is potentially limited by the relatively slow absorption of current rapid-acting insulin analogs, which have a delayed onset and prolonged duration of action. This can be challenging during times of rapid glucose changes, such as the postprandial period.
Ultrarapid-acting insulins, which have an accelerated pharmacokinetic (PK) and pharmacodynamic (PD) profile, could help minimize these rapid glucose changes and potentially increase the time patients spend in the target glucose range. However, their use with hybrid closed-loop systems must be evaluated to ensure that the basal algorithm accommodates this accelerated PK/PD profile. Recently published data from an open-label, crossover trial, comparing efficacy and safety of fast-acting insulin aspart (Fiasp®; Novo Nordisk; Bagsvaerd, Denmark) versus insulin aspart (NovoLog®; Novo Nordisk Bagsvaerd, Denmark) when used in the 670G system in Auto Mode in patients with type 1 diabetes, showed greater reduction in the 1 h postprandial glucose rise following a standardized meal test and increased time in the range 3.9–10.0 mmol/L (70–180 mg/dL) with fast-acting insulin aspart compared with insulin aspart. 7
Ultrarapid lispro (URLi), another ultrarapid-acting insulin, is a new formulation of insulin lispro with two enabling excipients (treprostinil and citrate) that facilitate rapid absorption of insulin lispro into the blood stream. In previous studies URLi showed faster onset of appearance in serum, with an earlier glucose-lowering effect, and a shorter duration of action when compared with insulin lispro (lispro; Humalog®; Eli Lilly and Company; Indiana, ). 8 Furthermore, compared to other rapid-acting insulins (lispro, insulin aspart, and fast-acting insulin aspart), URLi demonstrated the fastest insulin absorption, greatest reduction in late insulin exposure and shortest exposure duration, while the early postprandial glucose profile following URLi administration more closely matched that in healthy subjects with normal endogenous insulin secretion. 9 In phase 3 trials, URLi demonstrated superior postprandial glucose control to lispro in patients on multiple daily injection regimens 10,11 or using continuous subcutaneous insulin infusion (CSII). 12
The aim of this study was to compare URLi to lispro with respect to the percentage of time with sensor glucose values within target range 3.9–10.0 mmol/L (70–180 mg/dL; %TIR) when both were delivered by CSII with the Medtronic MiniMed 670G System using the Auto Mode feature.
Research Design and Methods
Study design
This was a double-blind, 2-period, crossover, randomized controlled trial evaluating the efficacy of URLi compared with lispro in adults with type 1 diabetes on CSII therapy using the 670G hybrid closed-loop system. The study included a 2-week lead-in period before randomization, followed by two 4-week treatment periods (with no washout between treatments) and a 2-week safety follow-up (Supplementary Fig. S1). It was conducted at two sites within the United States of America (USA) and in accordance with the Declaration of Helsinki, the International Conference on Harmonization guidelines for Good Clinical Practice, and applicable laws and regulations in the USA. All procedures were approved by an Independent Ethics Review Board and all patients provided written, informed consent before participating in the study.
Participants
Main inclusion criteria were age ≥18 years; clinically diagnosed with type 1 diabetes and continuously using insulin for at least 1 year; using CSII therapy for ≥6 months; using a 670G insulin pump in Auto Mode for ≥90 days before screening; using Auto Mode at least 70% of the time per week during the 4 weeks before screening; HbA1c ≥6.0% and ≤8.0%; and body mass index (BMI) ≤35 kg/m2.
Patients were excluded from the study if they had hypoglycemia unawareness; experienced >1 episode of severe hypoglycemia within the 6 months before screening; had any emergency department visit or hospitalization due to poor blood glucose control (hyperglycemia or diabetic ketoacidosis) within 6 months before screening; had significant lipohypertrophy, lipoatrophy, or scars within subcutaneous tissue in areas of infusion; had an abscess at an infusion site within 90 days before screening; or used a total daily insulin dose >100 U/day, on average, over the 3 days before screening. Further details on inclusion and exclusion criteria can be found in Supplementary Table S1.
Study procedures
At the start of lead-in, patients who were not using lispro were switched from their rapid-acting insulin analog to lispro unit-for-unit. Following the lead-in period, a 3-day pilot safety assessment was conducted at a single site for the first 10 patients randomized before expanding enrollment in the study. After evaluating patient safety (including episodes of severe hypoglycemia and severe hyperglycemia), it was determined to continue with the study.
Eligible patients were then randomized 1:1 to 4 weeks of URLi or lispro then crossed over to the other insulin for an additional 4 weeks.
Assignment to treatment groups was determined by a computer-generated random sequence using an interactive web-response system and stratified by HbA1c stratum at screening (≤7.0%, >7.0%) and %TIR over the 2 weeks before randomization (≤75%, >75%).
Throughout the study, patients used their personal 670G insulin pump and CareLink Personal Software, and the study provided MiniMed reservoirs, Mio infusion sets, Contour Next Link 2.4 Glucose Meter, Medtronic Guardian Sensor 3, and Guardian Link 3 Transmitter. Patients filled their pump reservoirs from blinded vials with each infusion set change, and boluses were delivered at mealtime (0–2 min before meals) using the same bolus delivery speed used at screening that is, Standard (1.5 U/min) or Quick bolus delivery speed (15 U/min). Patients were required to use the Auto Mode insulin delivery function as much as possible throughout the study, completing any actions indicated on the pump screen to return to Auto Mode, if exited.
The pump reservoir and infusion sets were to be changed every 3 days unless a change was required due to a failure of the infusion set. Unplanned infusion set changes were documented in the patient's study diary.
Optimization of insulin dose
The basal and bolus delivery of insulin in this study were individualized for each patient. All patients were carbohydrate-counting and used the Auto Mode bolus feature for calculation of meal and correction bolus doses utilizing carbohydrate ratio (carb ratio) and active insulin time. It was recommended that all therapy adjustments be based on measurements obtained using the study-provided blood glucose meter and not on values from the Guardian Sensor 3.
Assessments
The primary endpoint was the percentage of time with sensor glucose values between 3.9 and 10.0 mmol/L (both inclusive), during the last 2 weeks of each 4-week treatment period.
Key secondary endpoints included the mean sensor glucose value, percentage of time spent in Auto Mode per week, and the percentage of time with sensor glucose values <3.0 mmol/L (54 mg/dL) during the last 2 weeks of each 4-week treatment period.
Statistical analyses
It was estimated that 42 randomized patients would provide 90% statistical power to detect a 5% difference between URLi and lispro in %TIR during the last 2 weeks of each 4-week treatment period, with assumptions of a standard deviation of between-period differences of 10%, at two-sided alpha-level 0.10, and a 12% dropout rate.
All efficacy and safety analyses were conducted based on all randomized patients who received at least one dose of the randomly assigned study treatment.
Treatment comparisons for the primary objective were performed at the full significance level of 0.10. No multiplicity adjustment was made for secondary and exploratory objectives.
A restricted maximum likelihood-based, mixed-effects model with repeated measures analysis was used to analyze continuous variables. The model included the fixed class effects of treatment, period, sequence, strata (HbA1c [≤7.0%, >7.0%] and %TIR over the 2 weeks before randomization [≤75%, >75%]), and the continuous, fixed covariate of baseline value. Significance tests were based on least squares mean (LSM) and type III tests. SAS PROC MIXED was used to perform the analysis.
Analyses of adverse events (AEs) included all data collected during the entire 4-week treatment period for each treatment regardless of investigative product (IP) use. Analyses of hypoglycemia and CGM measures were conducted using the data from first to last dose of IP in each 4-week treatment period. Due to the crossover design of the study, treatment comparisons (except severe hypoglycemia) were based on the derived outcome variables for week 2–4 in each 4-week treatment period. Pump-related safety analyses and CGM analyses excluded data that were collected while patients were temporarily off pump or off IP. Wilcoxon Signed-Rank test and Prescott's exact test were used to analyze the rate and incidence of events, respectively.
To ensure that the CGM outcome variables were only calculated from CGM session days with sufficient data, a day was counted as valid only when it had at least 70% of the total CGM measures intended to be obtained. Similarly, for the by-meal outcome variables (e.g., incremental area under the curve [iAUC] from 0 to 2 h), only meals with at least 70% of the total measures intended to be obtained were included.
Meal-related CGM measures were derived for breakfast, lunch, dinner, and overall (average across all meals). To assess the glucose control over the course of ∼3 days of continuous insulin infusion, the duration and percentage of time in ranges (target, hypoglycemia, or hyperglycemia) and iAUCs after meals were derived based upon the CGM raw data collected on each of the infusion set wear days, including day 1 (>0 and ≤24 h), day 2 (>24 and ≤48 h), and day 3 (>48 and ≤72 h). For analyses of CGM data by infusion set wear day, only data from the infusion sets with ≥60 h (2.5 days) of wear were included so that adequate data could be used for by-day (day 1, 2, and 3) comparison.
To evaluate consistency of treatment effects on the primary endpoint, analyses were conducted on the two bolus delivery speed subgroups (Standard and Quick). Only data from patients who maintained the same bolus delivery speed during lead-in and the two 4-week treatment periods were included in the analyses.
Results
Table 1 provides a summary of the baseline characteristics for the total population. Mean age was 47.8 ± 13.8 years, with BMI 27.2 ± 4.2 kg/m2 and HbA1c 7.07% ± 0.47%. Duration of CSII and 670G use was 16.9 ± 9.5 and 1.4 ± 0.7 years, respectively, with an average baseline %TIR of 77.9% ± 7.8%. Overall, 42 patients were randomized to treatment, and all completed the study with no discontinuation of treatment.
Baseline Characteristics
Data are mean (standard deviation) unless otherwise stated.
BMI, body mass index; CSII, continuous subcutaneous insulin infusion; HbA1c, hemoglobin A1c; T1D, type 1 diabetes.
Time in ranges
The 24 h ambulatory glucose profiles for the two treatments were generally similar (Fig. 1A).
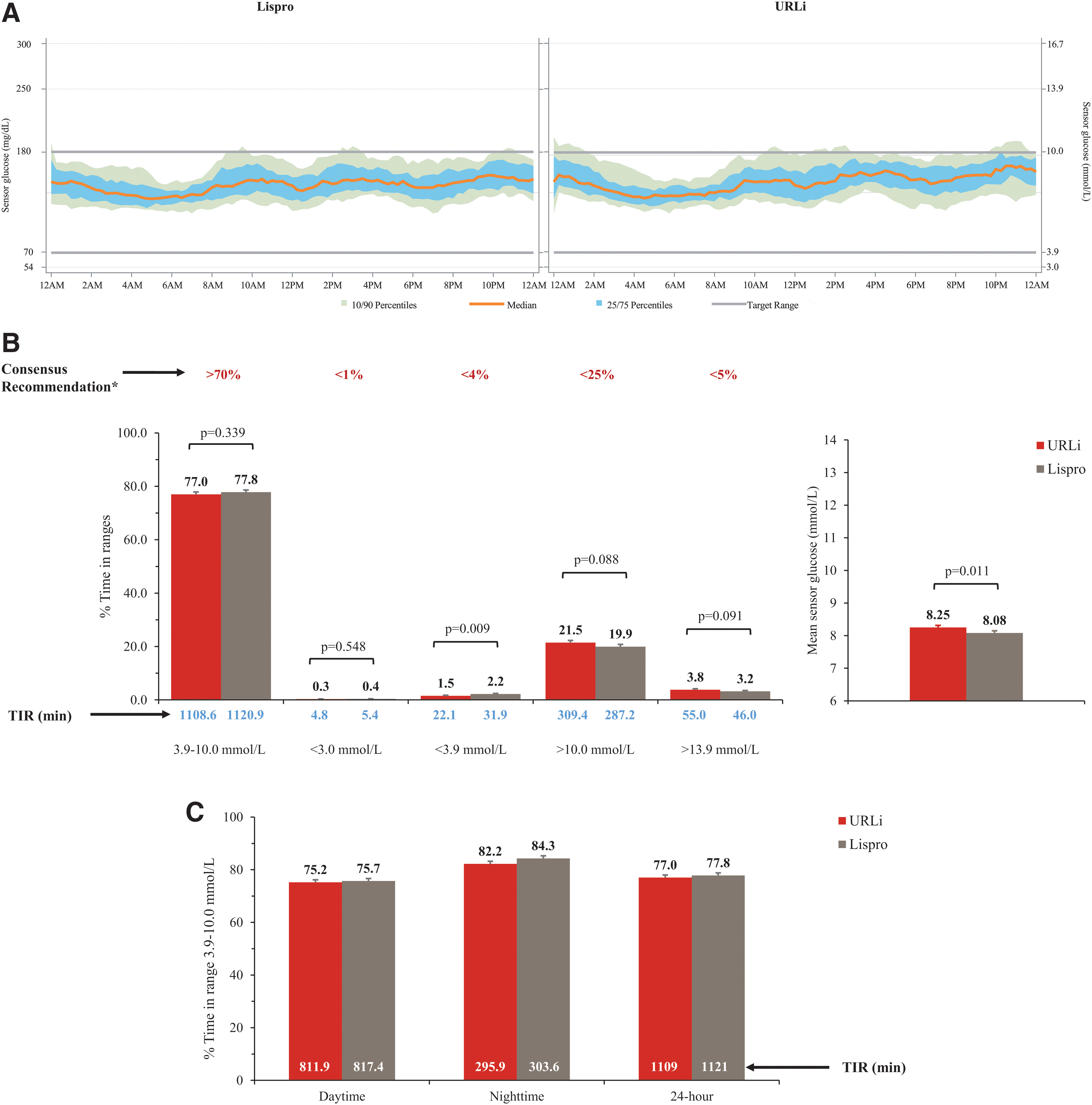
Treatment level ambulatory glucose profiles
There were no statistically significant differences between treatments in the mean time in target range for the daytime, nighttime, and 24 h periods (all P > 0.1). Both treatments achieved an average %TIR >75% in the three time periods and resulted in %TIR, as well as above and below range, that was within the targets recommended by the International Consensus Guidance on Time in Range 13 (Fig. 1B, C). Mean sensor glucose was slightly higher with URLi treatment (Fig. 1B) with a LSM difference of 0.17 mmol/L or 3.0 mg/dL (P = 0.011) between treatments.
Time in hypoglycemia with sensor glucose <3.0 mmol/l (54 mg/dL) was similar between treatments, but significantly lower with URLi at sensor glucose <3.9 mmol/L (70 mg/dL) during the daytime (URLi, 17.5 min [1.6%]; lispro, 26.4 min [2.4%]; P = 0.002) and 24 h period (URLi, 22.1 min [1.5%]; lispro, 31.9 min [2.2%]; P = 0.009). While time in hyperglycemia was generally similar between treatments, URLi resulted in significantly more time in hyperglycemia >10.0 mmol/L (180 mg/dL) during the nighttime (URLi, 59.2 min [16.4%]; lispro, 50.3 min [14.0%]; P = 0.083) and 24 h period (URLi, 309.4 min [21.5%]; lispro, 287.2 min [19.9%]; P = 0.002) (Supplementary Table S2). Overall, glucose control was generally maintained across all 3 days of infusion set wear as reflected by similar time above, below, and in target range, although small but statistically significant between-treatment differences (i.e., P-value <0.1) were observed at times (Table S2).
Postmeal glycemic control
The mean glucose excursion following meals was significantly lower with URLi compared to lispro treatment between 0 and 1 h postmeal (0.5 vs. 0.7 mmol/L [8.7 vs. 12.7 mg/dL]; P = 0.012) and 0–2 h postmeal (0.5 vs. 0.8 mmol/L [8.7 vs. 13.8 mg/dL]; P = 0.015) (Fig. 2A). Lower glucose excursions were also observed with URLi between 0–3 h and 0–4 h postmeal, but the differences did not reach statistical significance. Similarly, the iAUC across all meals combined was significantly lower with URLi compared to lispro between 0–1 h postmeal and 0–2 h postmeal, corresponding to a 29% and 38% reduction with URLi in iAUC 0–1 h and 0–2 h, respectively (Fig. 2B). Numerically lower iAUCs were also seen with URLi between 0–3 h and 0–4 h postmeal.
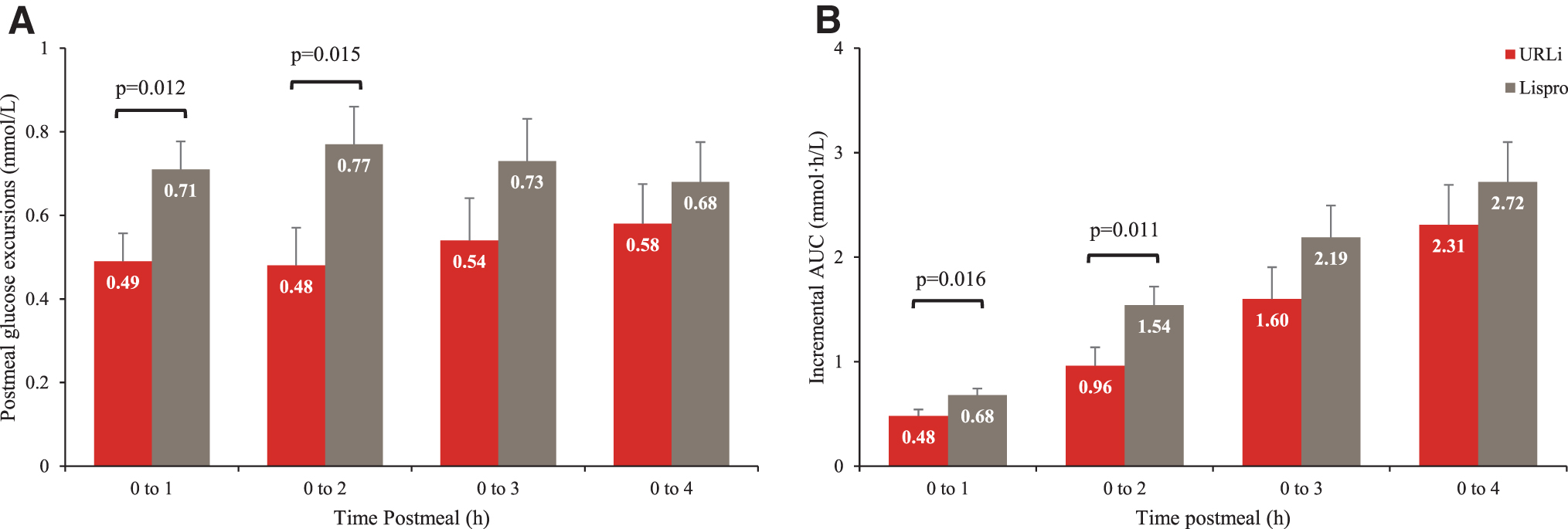
Average postmeal glucose excursions
Ambulatory glucose profiles for the postmeal period following breakfast, lunch, and dinner were comparable between URLi and lispro. Both treatments maintained median glucose values within the target range 3.9–10.0 mmol/L (70–180 mg/dL) and showed no significant differences in the mean TIR following the three meals and overall.
Overall, there were no significant between-treatment differences in the mean time spent in hypoglycemia <3.0 mmol/L (54 mg/dL) during the postmeal period. However, significantly less time in hypoglycemia <3.9 mmol/L (70 mg/dL) was observed with URLi in the period between 0–3 h (URLi, 1.4%; lispro, 2.3%; P = 0.063), 0–4 h postmeal following lunch (URLi, 1.3%; lispro 2.5%; P = 0.006), and across all meals combined (URLi, 2.0%; lispro, 2.6%; P = 0.095).
Meal bolus doses and postprandial glucose control were similar for both treatments regardless of bolus delivery speed. However, numerically reduced time below target range was observed with both treatments up to 2 h postmeal for Quick compared to Standard bolus delivery speed. In addition, visually flatter postmeal glucose curves were observed with both treatments for Quick compared to Standard bolus delivery speed (Figure 3).
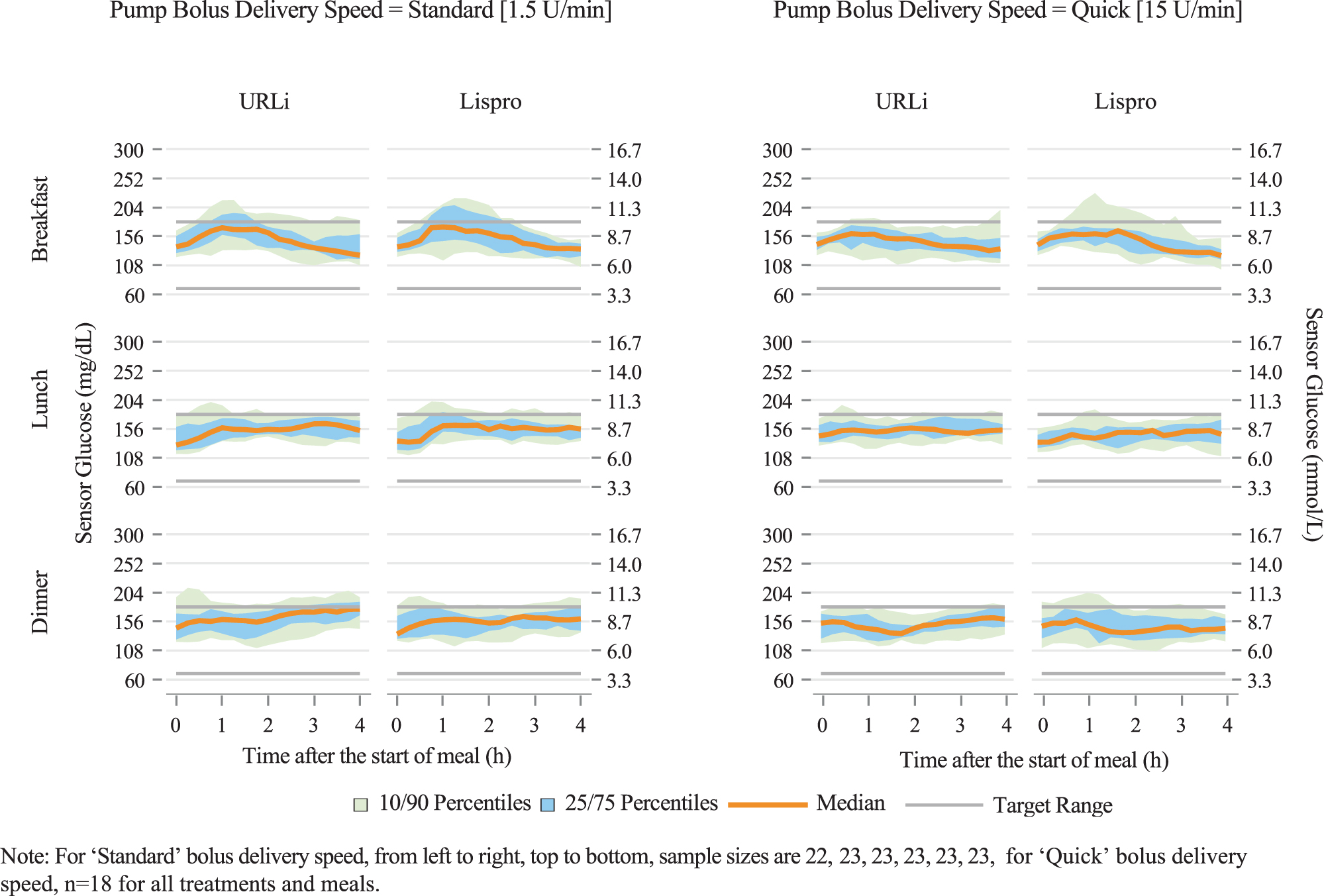
Ambulatory glucose profiles by bolus delivery speed.
Insulin dose and pump settings
For both treatments, the mean time spent in Auto Mode per week was above 90% (URLi, 91.98%; lispro, 91.42%; P = 0.557).
The mean active insulin time was 3.4 h for both URLi and lispro (Supplementary Table S3). Overall, carb ratio was similar between treatments, although a statistically significant difference was observed for the lunchtime carb ratio: URLi, 8.8 g/U; lispro, 8.4 g/U; P = 0.036 (Supplementary Table S3). The mean insulin dose (basal, bolus, and total daily insulin dose; units/day) was similar between URLi and lispro and remained relatively unchanged from day 1 to 3 of infusion set wear (Supplementary Table S3). Similar ratios of bolus-to-total insulin dose were shown with the two treatments: 54.0% with URLi and 54.2% with lispro (P = 0.851). The insulin sensitivity factor did not change or show treatment differences in this study.
Adverse events
No serious AEs (including severe hypoglycemia) or discontinuations from study or treatment due to AEs were reported in the study. The incidence of treatment-emergent AEs (TEAEs) was similar between treatments (URLi, 11 [26.2%]; lispro, 9 [21.4%]), with the most frequently reported being infusion site reactions (Table 2).
Adverse Events
MeDRA, medical dictionary for regulatory activities; TEAEs, treatment emergent adverse events; URLi, ultrarapid lispro.
The reported potential treatment-emergent infusion site reactions using MedDRA search terms were infusion site pain, infusion site reaction, and infusion site erythema (Table 2).
Infusion set changes
Overall, the mean time interval to infusion set changes was similar between groups (URLi, 63.6 h; lispro, 64.5 h). No between-treatment differences were noted for the overall rate of planned and unplanned infusion set changes (Table 3). However, a significantly higher rate of unplanned infusion set changes due to infusion site pain, redness, or swelling was seen during URLi treatment compared with lispro: 0.12 versus 0.00 events/30 days (P = 0.063). Unplanned infusion set changes due to an occlusion alarm and due to unexplained hyperglycemia were similar between groups. Similar trends in rates of unplanned infusion set change by reason were observed across the 3 days of infusion set wear (Supplementary Table S4).
Summary of Unplanned Infusion Set Changes
Prespecified significance level was set to P < 0.1.
Infusion site reaction includes pain, redness, or swelling at infusion site.
infusion set problem includes empty reservoir, infusion set kinked, was pulled out, or was leaking.
Discussion
Evolving technologies, such as the hybrid-closed-loop system, offer patients with diabetes increased potential to achieve glycemic goals with improved diabetes management. In this randomized, crossover trial, we evaluated the use of URLi in the 670G hybrid closed-loop system to determine if glycemic outcomes were comparable to lispro. URLi achieved good glycemic control that was similar to lispro, averaging a percent time in range of 77.0% compared to 77.8% with lispro over the 24 h period. Time in range during the daytime and nighttime was also similar between treatments. This was achieved with no differences in pump settings or insulin dose between the two treatments. On average, treatment with URLi resulted in significantly reduced time in hypoglycemia (<3.9 mmol/L) following meals and during the daytime and 24 h period. However, URLi treatment also resulted in increased time in hyperglycemia during the nighttime and over the 24 h period, with a corresponding increase in the mean sensor glucose compared with lispro. Despite these findings, both treatments achieved good glycemic control overall, with percent time in range, below range (<3.0 and <3.9 mmol/L [54 and 70 mg/dL, respectively]), and above range (>10.0 and >13.9 mmol/L [180 and 250 mg/dL, respectively]) falling within the ranges recommended by the International Consensus Guidance on Time in Range. 13 Glycemic control was maintained over the 3 days of infusion set wear.
As mentioned, a small but significant benefit was seen with URLi in the time spent in hypoglycemia (sensor glucose <3.9 mmol/L [70 mg/dL]) during the daytime and 24 h period, with an average between-treatment difference of about -0.7% (equivalent to 10 min per day). A similar reduction in the time spent in hypoglycemia was also seen with URLi in the postmeal period. While the differences in hypoglycemia in this study were small, they were consistent with previous findings of reduced time in hypoglycemia with URLi in the PRONTO-Pump-2 study 12 and overall reduced rates of hypoglycemia with URLi, particularly in the postmeal period, with both MDI 10 and CSII use. 12 Reduced postmeal hypoglycemia is reflective of URLi's faster onset and shorter duration of action compared to lispro as described in clinical pharmacology studies. 8
Further improvements were seen with URLi regarding postprandial glucose control in this study. While both treatments maintained a median glucose level between 3.9 and 10.0 mmol/L (70 and 180 mg/dL) following breakfast, lunch, and dinner, and the mean time in range in the postmeal period was similar between treatments, the magnitude of glucose excursions and iAUCs was reduced with URLi compared to lispro treatment. Lower glucose excursions and iAUCs with URLi treatment were apparent in all the timeframes assessed, with differences reaching statistical significance between 0–1 and 0–2 h postmeal. On average iAUCs were reduced by 29% and 38% during these two timeframes, similar to previous findings 12,14 and confirming the improved postprandial glucose control that URLi exhibits.
Despite these improvements in hypoglycemia and postprandial glucose excursions, there was no remarkable improvement in time in range with URLi and no obvious benefit of using this ultrarapid insulin in the 670G system. Similarly, when evaluating the use of fast-acting insulin aspart versus insulin aspart in the 670G system in the 2-week, randomized, double-blind, pilot study among established 670G users, researchers found no significant advantage of fast-acting insulin aspart use with the system. 15
It is likely that since the 670G algorithm modulates only basal insulin delivery, it may not be optimized for potential advantages of an insulin with an accelerated PK profile with which differential effects are seen primarily during bolus delivery. As the results show, much of the significant benefits of URLi were seen following meals, consistent with the timing and action of bolus insulin delivery. Outside of the mealtime bolus insulin action, significantly increased time in hyperglycemia was observed with URLi: time in hyperglycemia during the nighttime was higher, contributing to an overall increased time in hyperglycemia during the 24 h period and higher mean sensor glucose compared with lispro. Similar increases in nighttime hyperglycemia were seen with URLi in the PRONTO-Pump-212 and PRONTO-T1D CGM Substudy. 14
Utilizing URLi in the 670G system may require higher insulin doses to prevent late postmeal hyperglycemia between meals and going into the nighttime; this will reduce time in hyperglycemia and potentially increase %TIR. Higher total daily dose was evident with fast-acting insulin aspart in the 670G system, with a corresponding numerical increase in TIR and reduced time in hyperglycemia versus insulin aspart in the pilot study. 15 There is also potential for better outcomes with URLi in an advanced hybrid closed-loop system that automates correction boluses or a fully automated closed-loop system that automates meal and correction boluses. The advanced version of the 670G, the MiniMed 780G, and the Tandem X2™ Control IQ (Tandem Diabetes Care, San Diego, CA) are examples of advanced hybrid closed-loop systems that automatically correct high glucose levels and may enable improved outcomes with URLi and other ultrarapid insulin analogs.
In this study, data from patients who maintained the same prestudy bolus delivery speed (Standard or Quick bolus delivery speed) were analyzed to provide insight into glycemic control with different bolus delivery speeds. Numerically reduced time below target range and visually flatter postmeal glucose curves were observed with the Quick bolus delivery speed compared with Standard speed for both treatments. While the sample size was too small to conclusively determine the implications of this finding, it is reflective of earlier insulin absorption and action following the quicker bolus dose delivery. In a previous study assessing subcutaneous absorption kinetics of rapid-acting insulin using different bolus delivery speeds, investigators found that insulin bolus delivery with fast delivery speed resulted in more rapid insulin absorption and could potentially lead to better control of postprandial glucose excursions. 16 Further studies will be needed to confirm the glycemic control findings with increased bolus delivery speed seen in the current study, and identify what impact, if any, this has on clinical outcomes.
Generally, URLi was well tolerated in this study showing a safety profile similar to that of lispro. The incidence of TEAEs was similar between treatments, with infusion site reactions being the most frequently reported. Although mostly mild in severity, these events led to a higher rate of infusion set changes with URLi, translating to ∼1 additional infusion set change every 8–9 months based on routine changes every 3 days. The incidence of infusion set changes due to pump occlusion alarm and unexplained hyperglycemia were similar between treatments, indicating that URLi was compatible with the 670G system.
A potential limitation of this study is that the study population had generally well-controlled diabetes before enrollment, averaging %TIR of 77.9% and HbA1c 7.07%. To add to this, they were well-established users of the 670G system, averaging 1.4 years of use before enrollment. Altogether, these factors may have left little room for improvement in the primary and key secondary endpoint measures. For future direction, it would be of interest to investigate differential treatment effects on %TIR between URLi and lispro in patients naive to hybrid closed-loop systems and with higher baseline HbA1c. In addition, more comprehensive analysis of data that complements the ambulatory glucose profile as suggested in literature, 17,18 such as treatment-effects on lifestyle changes, including variations between weekdays versus weekend, physical activity, or different meals, could help elucidate key differences between treatments and inform diabetes management. Alternative methods of deriving the mean sensor glucose such as that described by Henry et al. 19 can also provide additional data on treatment effects.
This study was conducted in an outpatient setting providing insight into outcomes that would be achieved in the real-world where daily meals and activities are not standardized. While this was a significant strength of the study, it also served as a potential limitation as external factors of diabetes management could add noise to the data.
Other strengths are that the study was designed as a double-blind, randomized, clinical trial, providing an opportunity to reduce bias in the study; with a crossover design to reduce variability and increase study power, as each patient served as his or her own control. In addition, treatment comparisons only used data collected during the last 2 weeks of each treatment period, which helped to minimize the carryover effect on the primary analysis of efficacy, safety, and exploratory outcomes. In this study, each period of the crossover consisted of 4 weeks of treatment, which could be perceived as a short duration. However, this was considered appropriate for the assessment of the primary endpoint based on a recent retrospective analysis of two hybrid closed-loop studies, which concluded that a study of 4 weeks duration provides representative data for mean glucose and %TIR that is highly correlated with 12-week outcomes. 20
Conclusion
Over 4 weeks of treatment using the 670G hybrid closed-loop system, URLi provided good glycemic control that was comparable to lispro, showing that the system's basal algorithm accommodated URLi's accelerated PK/PD profile. Both URLi and lispro achieved International Consensus recommended targets for the percent time in range, percent time below range, and percent time above range. However, given that only modest improvements in glycemic control were seen with URLi, it is possible that the 670G system is not fully optimized for ultrarapid-acting insulin. With continued advancements in insulin delivery systems, it is likely that even greater benefit will be seen with the use of URLi in advanced hybrid closed-loop and fully automated closed-loop systems.
Footnotes
Authors' Contributions
B.B. and A.C. participated as trial investigators and reviewed and edited the article. R.L. contributed to the study design, the statistical analysis, the interpretation of the research, and writing of the article. T.H. contributed to the study design, medical oversight, interpretation of the research, and writing of the article. J.B., R.B., and S.M. contributed to the interpretation of the research and writing of the article. D.I. was responsible for medical oversight during the trial and contributed to the study design, the data analysis, interpretation of the research, and writing of the article. All authors approved the final article to be published. D.I. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Acknowledgments
The authors thank the study participants and the investigators and study coordinators who cared for them. The authors also thank Dr. John Shin (Medtronic, Northridge, CA) for providing consulting services for the interpretation of Medtronic pump data; Leilei Qian, Fei Yang, Brandon George, and Ming Hua (Eli Lilly and Company, Indianapolis, IN) for conducting the statistical analysis; Farai Chigutsa (Eli Lilly and Company, Indianapolis, IN) for medical writing and editorial assistance; and Dr. Janet Tobian (Eli Lilly and Company, Indianapolis, IN) for critically reviewing the article.
Author Disclosure Statement
B.B. is a shareholder of Aseko, Inc., and has received consultant fees from Eli Lilly and Company, Lexicon, Medtronic, Novo Nordisk, and Pfizer; speaker honoraria from Astra Zeneca, Boehringer Ingelheim, Eli Lilly and Company, Janssen, Mannkind, Medtronic, Novo Nordisk, Sanofi, Senseonics, and Xeris; he is an employee of Atlanta Diabetes Associates, which has received research grants and support from Boehringer Ingelheim, Dexcom, Diasome, Eli Lilly and Company, Insulet, Janssen, Lexicon, Mannkind, Medtronic, National Institutes of Health (NIH), Nova Biomedical, Novo Nordisk, Provention Bio, Sanofi, Senseonics, REMD Biotherapeutics, vTv Therapeutics LLC, and Xeris. R.L., S.M., and D.I. are employees and shareholders of Eli Lilly and Company. T.H. is a former employee and shareholder of Eli Lilly and Company. J.B. reports no conflicts of interest. No other potential conflicts of interest relevant to this article were reported.
Funding Information
This study was funded by Eli Lilly and Company.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
