Abstract
Objective:
Clinical guidelines now define the standard of diabetes care, but few health care jurisdictions systematically assess their practicality and impact. The Canadian LMC Diabetes Registry includes the electronic health records of >50 endocrinologists in three provinces and provides quarterly real-time outcome reports to each endocrinologist. This retrospective cohort study aimed to characterize the demographics, treatment regimens, and outcomes of the type 1 diabetes (T1D) patient population in the registry.
Research Design and Methods:
Adults were included if they had a clinical diagnosis of T1D, had seen an LMC endocrinologist between July 1, 2015 and June 30, 2018, and had follow-up >6 months. This study is registered on
Results:
The resulting cohort included 3600 individuals with mean age of 43.9 ± 15.3 years and duration of diabetes of 21.5 ± 13.9 years. Mean hemoglobin A1C (HbA1c) was 8.1% ± 1.5% and only 22.5% had achieved HbA1c ≤7.0%. In each measure, individuals in younger cohorts showed poorer glycemic control than older cohorts. Within each age cohort, insulin pump users showed a lower mean HbA1c than those using multiple daily injections, especially in cohorts who were also not using a continuous glucose monitor. Overall, 63.1% reported at least weekly hypoglycemia, whereas 3.6% reported severe hypoglycemia ≥1 per year.
Conclusions:
Despite receiving care in an advanced well-resourced environment, within a public health care system, from specialists armed with regular patient outcomes feedback, most individuals with T1D are unable to achieve the goals recommended by clinical practice guidelines.
Introduction
Diabetes represents a growing burden on global health care systems and is arguably unique as a chronic disease in that accessible current health outcome measures (e.g., glycemia, blood pressure, and lipids) directly predict the resulting health burden. As such, most health care authorities have developed or adopted comprehensive care guidelines to optimize these outcomes. However, few have taken the important step of maintaining a systematic awareness of those outcomes to reconcile the practicality and the impact of well-intended practice guidelines.
In type 1 diabetes (T1D), several large-scale population registries have emerged, including the Swedish National Diabetes Register (NDR), 1 the Diabetes-Patienen-Verlaufsdokumentation databases (DPV), 2 the German Diabetes Versorgungs-Evaluation (DIVE), 3 and others. 4,5 In the past decade, the Type 1 Diabetes Exchange Clinic Registry (T1DX), 6 an invited clinic-based cooperative, and the Optum Humedica database 7 have provided similar reports in the United States. In Canada, the Canadian Study of Longevity in T1D has particularly studied adults with T1D >50 years. 8,9 These registries have provided meaningful snapshots of pediatric and adult T1D populations by characterizing patient demographics, management regimens, and outcomes. 6,10 –13 For example, several registries have recently updated their reports to again confirm that even with recent advances in therapies and technologies, only a minority actually achieves recommended targets. 7,10,11
An accurate and constant awareness of the clinical status of people with T1D is necessary to inform the feasibility of current guidelines and the need for future strategy redevelopment. The Canadian LMC Diabetes Registry comprises the health records of one of the largest endocrine practice groups globally, with >50 endocrinologists across three Canadian provinces. Since 2015, the registry has uniquely provided quarterly real-time outcome reports, aligned to clinical guidelines, to each endocrinologist for their respective cohorts, with their centile rankings. The registry has also provided unique insights into the impact of new therapies and of novel education delivery systems and measures. 14,15 This report aims to characterize the Canadian T1D patient population within the registry based on their demographics, their treatment regimens and technology utilization, and their metabolic outcomes.
Methods
Study design and data source
This study was a retrospective cohort analysis of the LMC Diabetes Registry. The LMC Diabetes Registry represents the shared electronic health records of 42,000 patients with diabetes actively attending 1 of 13 LMC clinics in Canada (10 in Ontario, 2 in Quebec, and 1 in Alberta). 14,16 Patients are usually assessed quarterly by 1 of >60 endocrinologists and physician assistants, supported by >50 diabetes educators, all practicing in alignment to the Diabetes Canada Clinical Practice Guidelines, 17 within a publicly funded health care system. The study was conducted in compliance with the ethical principles of the Declaration of Helsinki. An independent ethics committee approved the protocol and patients provided consent for their medical data to be used for research purposes (NCT04162067).
Individuals ≥18 years of age were included in the analysis if they had a clinical diagnosis of T1D, had a visit between July 1, 2015 and June 30, 2018, and were followed by an endocrinologist for >6 months. All data used for analysis were the most recently available up to June 30, 2019.
Sociodemographic information included age, gender, duration at LMC, ethnicity, education level, household income, health benefits, marital, and employment status.
Outcomes
Outcomes included hemoglobin A1C (HbA1c), fasting plasma glucose, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein (HDL) cholesterol, non-HDL cholesterol, triglycerides, estimated glomerular filtration rate (eGFR), urine albumin-to-creatinine ratio (uACR), weight, body mass index (BMI), waist circumference, blood pressure, and hypoglycemia.
Hypoglycemia frequency is collected at each patient visit using a structured interview by a trained nonphysician interviewer. Patients report their frequency of “any hypoglycemia” (symptomatic and/or confirmed) in the prior week, as well as severe hypoglycemia, defined as requiring the assistance of another person, within the prior year.
At each visit, any insulin pump, continuous glucose monitoring (CGM) device, and self-measured blood glucose (SMBG) meter brands were recorded and the device-stored data were uploaded (Glooko®) or shared (Clarity®, CareLink™ or LibreView®).
A microvascular condition was defined as a clinical diagnosis of neuropathy, retinopathy, or nephropathy (also defined as uACR ≥2.0 mg/mmol or eGFR <60 mL/min/1.73 m2). A macrovascular condition was defined as the presence of either coronary, cerebral, or peripheral vascular disease or revascularization. A mental health condition was defined as a clinical diagnosis or the use of a prescription antidepressant, antianxiety, or antipsychotic therapy.
CGM or flash glucose monitoring (FGM) outcomes included mean glucose, standard deviation (SD), coefficient of variation, time in range (TIR, 3.9 to 10.0 mmol/L), time above range (>10.0 mmol/L), and time below range (<3.9 mmol/L). The most recently available CGM data were used, using a minimum of 5 days and a maximum of 90 days, where the active status of the device was >70%.
Individuals were considered as “carbohydrate counting” for bolus dosing if a carbohydrate count was recorded for >50% of their total bolus dose volume, over the prior 7 days. Daily SMBG frequency was assessed in a randomly selected one-third subgroup of non-CGM/FGM users, over their prior 14 days of SMBG data.
Statistical analyses
All demographic and clinical data were tabulated by age cohort (18–25 years, 26 to 49 years, ≥50 years). Continuous variables are presented as mean ± SD and categorical variables are presented as percentages. The main variables of interest for comparison between age cohorts, and between multiple daily injections (MDI) versus continuous subcutaneous insulin infusion (CSII) users, were HbA1c, weight, BMI, LDL-C, blood pressure, hypoglycemia, and CGM metrics. Multivariable linear regression was used to determine differences between age cohorts in HbA1c, weight, BMI, LDL-C, blood pressure, and continuous CGM metrics, adjusting for duration of diabetes and type of insulin therapy (MDI vs. CSII). The model for HbA1c was also adjusted for CGM/FGM use, the model for LDL-C was additionally adjusted for use of lipid lowering therapy, the models for blood pressure were additionally adjusted for use of antihypertensive therapy, and the models for CGM metrics were also adjusted for device type (CGM or FGM). Multivariable regression was used to test HbA1c differences in SMBG frequency <2 versus ≥2/day (adjusting for age, diabetes duration, and mode of insulin therapy); and in carbohydrate counting <20% and ≥50% of bolus dosing, (adjusting for age and duration of diabetes). Multivariable logistic regression was used to evaluate differences between age cohorts in the proportions of individuals with HbA1c ≤7.0% and >9.0%, those with LDL-C ≥2.0 mmol/L, and those reporting hypoglycemia and severe hypoglycemia. Similar multivariable linear regression models and multivariable logistic regression models were used to assess differences in clinical outcomes between MDI and CSII users, also adjusting for age. Differences between age cohorts in the proportion of individuals using CSII therapy, and a CGM/FGM device, were evaluated with a chi-square test. All regression models that compared the three age cohorts used a Tukey–Kramer adjustment to account for multiple comparisons.
Missing data were not replaced. All analyses were conducted with SAS v. 9.4 (SAS Institute, Inc., Cary, NC). A P-value <0.05 was considered statistically significant.
Results
Of the 5113 patients with T1D in the LMC Diabetes Registry, 4107 had a visit with an endocrinologist between July 1, 2015 and June 30, 2018, and 3600 had been followed by an endocrinologist for ≥6 months and had given prior consent for their data to be used, forming the final cohort. Their mean age was 43.9 ± 15.3 years, with an age range of 18–92 years and mean duration of diabetes was 21.5 ± 13.9 years (Table 1). Males made up 55%, 80% identified as Caucasian, and 53% reported a family history of diabetes. Half were married and 85.4% were employed or studying. In the cohort, 39.4% were using CSII therapy (n = 1417) and were numerically younger, and had been in care at LMC longer, compared with the MDI cohort (n = 2183, Supplementary Table S1).
Participant Characteristics
n = 3014 for ethnicity; b n = 2526 for education; c n = 2086 for household income; d n = 3076 for marital status; e n = 3106 for occupation; f n = 3111 for smoking status.
T1D, type 1 diabetes.
HbA1c outcomes
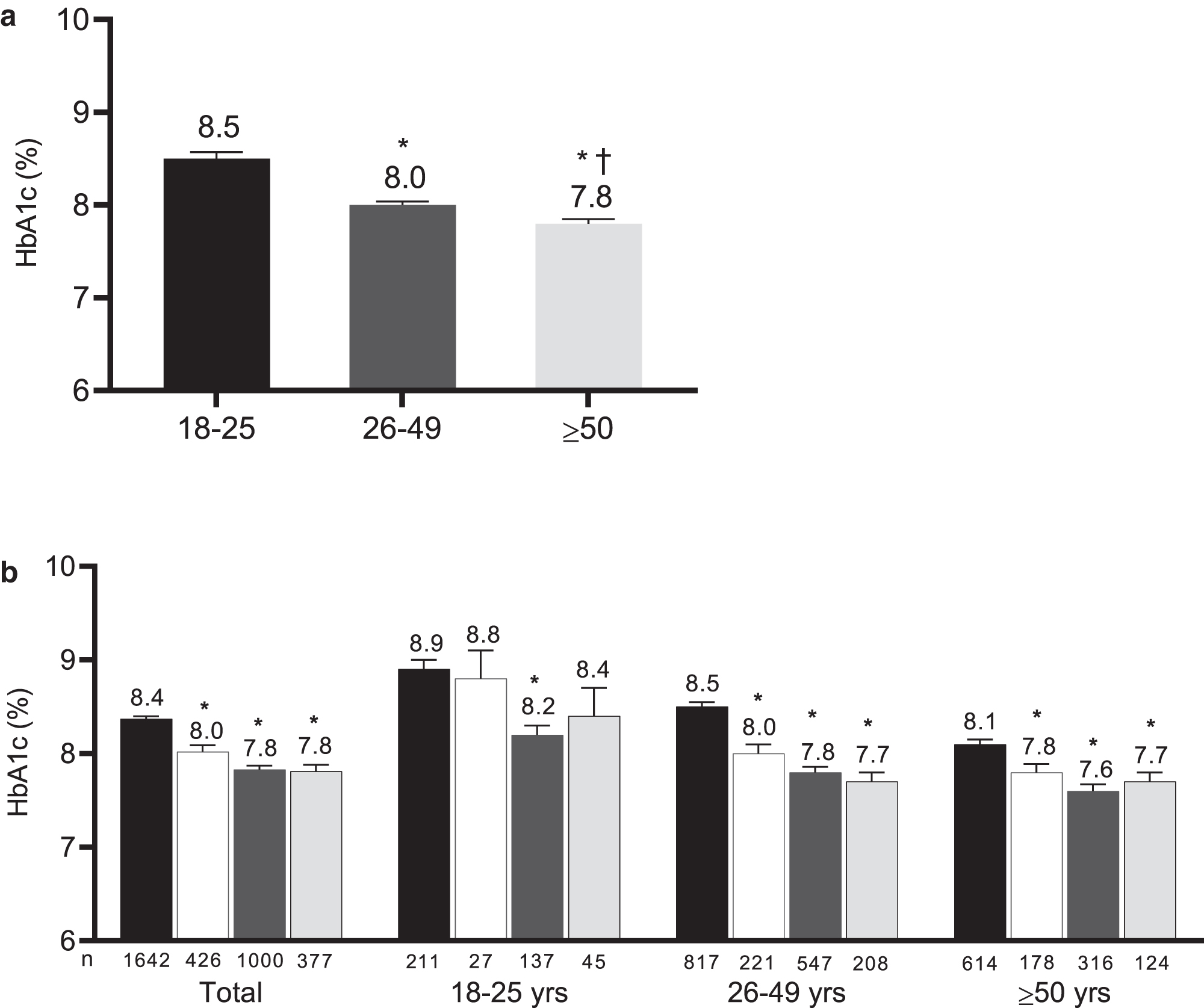
Metabolic outcomes of the total cohort and by age cohort are presented in Table 2. The most recent HbA1c mean was 8.1% ± 1.5% and 22.5% had achieved the target HbA1c ≤7.0%. Mean HbA1c was highest, and the proportion with HbA1c ≤7.0% was lowest, in the 18–25 age cohort (P < 0.01) (Fig. 1a). The proportion of individuals with HbA1c >9.0% was lowest in the ≥50 cohort (14.0%) compared with the 18–25 age cohort (35.1%) (P < 0.001) and the 26–49 age cohort (21.9%) (P < 0.001). When examined by ethnicity, the highest HbA1c levels were seen among the non-Hispanic black cohort with a mean of 9.0% ± 2.1% versus 8.3% ± 1.6% for the Asian and 8.0% ± 1.4% for non-Hispanic white populations.
Metabolic Status of Type 1 Diabetes Registry
Data are presented as mean ± SD, or as a percentage.
BMI, body mass index; eGFR, estimated glomerular filtration rate; HDL, high-density lipoprotein; LDL-C, low-density lipoprotein cholesterol; SD, standard deviation; uACR, urine albumin-to-creatinine ratio.
There was a significant interaction between mode of insulin delivery and CGM use (P < 0.01). MDI only was associated with a significantly higher HbA1c compared with pump only, MDI+CGM, and pump+CGM (P < 0.001) (Fig. 1b). In individuals not using a CGM/FGM device, pump users showed a lower mean HbA1c than MDI users (CSII 7.8% ± 0.1% vs. MDI 8.4% ± 0.1%, P < 0.001). Among CGM/FGM users, pump users continued to show a smaller, but statistically significant, HbA1c benefit (CSII 7.8% ± 0.1% vs. MDI 8.0% ± 0.1%, P = 0.03). This pattern of results was consistent in the 26–49 and ≥50 age cohorts (P < 0.01).
Among MDI users, use of CGM/FGM was associated with a lower HbA1c (8.4% ± 0.1% vs. 8.0% ± 0.1%, P < 0.001). Among pump users, the mean HbA1c was 7.8% regardless of CGM/FGM use (P = 0.79).
Among 524 individuals with pump data available, there was a mean of 5.0 ± 2.4 bolus insulin doses administered/day, and a carbohydrate count was entered 3.2 ± 1.6 times/day. Carbohydrate counting (defined as a carbohydrate entry associated with >50% of bolus dose volume) was associated with a lower HbA1c (7.7% ± 0.9% vs. 8.2% ± 1.2%, P < 0.01). Individuals who “rarely” carb counted (<20% of their bolus insulin dosing) were few (n = 35) but showed no difference in HbA1c versus “regularly” carb counters (8.0% ± 1.6% vs. 7.7% ± 1.0%, respectively, P = 0.29). Highest mean HbA1c was seen in the group who could be seen as using carbohydrate counting intermittently (associated with 20%–50% of their bolus doing) at 8.6% ± 1.3% (Supplementary Table S2).
Among those not using CGM/FGM, a randomly selected subgroup of 495 patients showed a mean daily SMBG frequency of 3.4 ± 2.2 (median 3.3). CSII users utilized SMBG more frequently (4.0 ± 3.7) compared with MDI users (2.9 ± 2.1). Overall, a higher mean SMBG frequency was associated with a lower mean HbA1c (r = −0.30). A mean testing frequency of <2 times/day was associated with a higher HbA1c (8.4% ± 0.1%) compared with a mean testing frequency of at least two or more times/day (7.8% ± 0.1%) (P < 0.001), adjusted for age, duration of T1D, and mode of insulin delivery (Supplementary Table S3).
Other metabolic outcomes
In the total cohort, mean weight was 79.6 ± 18.2 kg, and 63.3% had a BMI >25 kg/m2 (Table 2). Weight and BMI were both significantly higher in the two older age cohorts compared with the 18–25 age cohort (P < 0.05). There were no differences in weight (P = 0.37) or BMI (P = 0.35) between MDI and CSII users (Supplementary Table S4).
Mean LDL-C was 2.3 ± 0.8 mmol/L. Statin use in the overall cohort was 44.5% but among those >30 years of age and with diabetes duration >15 years, 62.7% were using statin therapy.
Mean systolic blood pressure was 121.6 ± 15.3 mmHg (Table 2) and 60.9% of the cohort had achieved a blood pressure of <130/80 mmHg. Systolic blood pressure increased with age (P < 0.001) and antihypertensive therapy was more prevalent with increasing age (18–25 age cohort 4.6%; 26–49 age cohort 26.0%; ≥50 age cohort 68.7% (Table 3).
Therapies, Devices, and Comorbidities in the Type 1 Diabetes Registry Cohort
All data are presented as a percentage, unless otherwise specified.
Nephropathy was defined as a clinical diagnosis of nephropathy or microalbuminuria, or a most recent uACR ≥2.0 mg/mmol or eGFR <60 mL/min/1.73m2. Neuropathy and retinopathy were based on clinical diagnoses. Coronary artery disease was defined as presence of angina, cardiovascular disease, congestive heart failure, coronary vascular disease, myocardial infarction, coronary artery bypass grafting or angioplasty; cerebral vascular disease was defined as history of stroke or cerebral vascular accident; peripheral vascular disease was defined as history of atherosclerotic disease, intermittent claudication, aorto-femoral bypass or femoral popliteal bypass. A mental health condition was defined as a history of anxiety, depression, bipolar affective disorder, schizophrenia or an eating disorder, or usage of an antidepressant, antianxiety, or antipsychotic therapy. Erectile dysfunction was defined as history of erectile dysfunction or usage of erectile dysfunction therapy.
CSII, continuous subcutaneous insulin infusion; CGM, continuous glucose monitor; DPP-4i, dipeptidyl peptidase-4 inhibitors; FGM, flash glucose monitor; GLP-1 RA, glucagon-like peptide-1 receptor agonists; MDI, multiple daily injections; RAAS, renin angiotensin aldosterone system; SGLT2i, sodium glucose co-transporter 2 inhibitors.
Hypoglycemia
For any hypoglycemia, 63.1%, reported at least one event weekly and the proportion trended lower from the youngest to oldest (18–25 age cohort 67.0%; 26–49 age cohort 63.5%; ≥50 age cohort 61.2%) reaching significance in the ≥50 age cohort (P < 0.05). The mean incidence of self-reported weekly hypoglycemia was 1.2 ± 2.2 and similarly trended downward with advancing age group (1.5 ± 0.1; 1.3 ± 0.1; and 1.1 ± 0.1, respectively; P < 0.05). For severe hypoglycemia, 3.6% of patients reported at least one event in the prior year and this proportion did not differ across age groups (P = 0.13). Self-reported hypoglycemia frequency did not differ between MDI and CSII cohorts (MDI 1.2 ± 0.1; CSII 1.3 ± 0.1, P > 0.05) but severe hypoglycemia was significantly lower in the CSII cohort (2.6%) than the MDI cohort (4.3%) (P = 0.03).
Metabolic therapies and devices
Nearly all (98.1%) were using analogue insulins. Individuals using CSII therapy (39.3%) were numerically younger, and had been in care at LMC longer, compared with the MDI cohort (60.7%, Table 3, Supplementary Table S5). Insulin pump use was lower with increasing age (18–25 age cohort 43.5%; 26–49 age cohort 42%; ≥50 age cohort 36.1%; P < 0.001). Insulin pump use was also lowest among the black population at 24.2% versus 36.5% among Asians and 43.2% among non-Hispanic whites. Of the total cohort, 22.7% were using or had used a wearable glucose sensor device (CGM 6.1%; FGM 16.6%) and was more prevalent with increasing age (16.4%, 23.4%, and 23.8%, respectively; P < 0.01). CSII users were more likely to also be using a CGM/FGM device (27.1%) compared with the MDI cohort (19.8%) (P < 0.01) (Supplementary Table S5).
Adjunct antihyperglycemic therapy was used in 11.9% of the patient cohort, with metformin (6.2%) and SGLT2 inhibitors (5.5%) used commonly (Table 3). Adjusted mean HbA1c was slightly lower among individuals using adjunct antihyperglycemic therapy (8.0% ± 0.1% vs. 8.2% ± 0.1%).
Comorbidities
A microvascular condition was present in 34.9% of the cohort (Table 3). Nephropathy was present in 26.8%, retinopathy in 14.1%, and clinically significant neuropathy in 6.5%. Erectile dysfunction was reported in 4.9%. Each disorder was increasingly prevalent with advancing cohort age. Macrovascular disease was less common, affecting 4.8% of the overall cohort but almost entirely affecting the age ≥50 cohort where 8.8% had a history of coronary artery disease, 2.2% had cerebral vascular disease, and 1.6% had peripheral vascular disease. There was a slightly higher prevalence of microvascular conditions in MDI users (36.4%) than CSII users (32.6%) (P = 0.04) and no difference in prevalence of macrovascular conditions (Supplementary Table S6). In the total cohort, 15.9% had a diagnosed and/or treated mental health condition, with a prevalence of nearly 20% in the ≥50 age cohort (Table 3).
CGM/FGM data
Of the total cohort, 22.7% (n = 816) were using a CGM/FGM device. Data from 623 individuals were available (68.6% FreeStyle Libre, 31.4% Dexcom), with a mean of 64.7 ± 32.1 days of data (within the prior 5 to 90 days) and with 91.4% ± 8.6% active CGM time (Supplementary Table S7). In the total cohort, mean TIR was 53.4% ± 17.0%, mean CGM/FGM glucose was 9.6 ± 2.0 mmol/L and only 36.3% showed CV <36%. Time above range was 40.3% ± 18.7% and time below range was 6.3% ± 5.9%. The 18–25 age cohort showed the highest time below range and highest variability compared with both older cohorts (P < 0.05) (Supplementary Table S7). There were no significant differences in CGM-measured outcomes between CSII and MDI users (Supplementary Table S8). Finally, across all CGM/FGM users, CGM users showed a higher TIR (58.8% ± 18.5% vs. 51.0% ± 15.8%, P < 0.001), lower variability (SD: 3.4 ± 0.8 vs. 3.9 ± 1.0, P < 0.001), (Supplementary Table S9).
Discussion
This study reports on the current state and management of a large Canadian cohort of 3600 adults with T1D and provides comparisons across different age cohorts and different insulin delivery modes. Majority of these adults with T1D did not meet metabolic targets, particularly if they were younger. The use of advanced technologies has also remained relatively low.
Among all individuals, only 22% had achieved the American Diabetes Association 18 and Diabetes Canada 17 recommended target HbA1c of ≤7.0%. These results are virtually identical to the latest data from the T1DX, which reported that 21% of U.S. adults had an HbA1c ≤7.0%. 10 Interestingly, T1DX also showed little change in HbA1c levels between 2010 and 2018, despite increases in insulin pump and CGM use. Our finding of higher HbA1c levels among younger age groups have similarly been reported in earlier registry reports. 11,19,20
One of the unique elements in this registry is that each endocrinologist receives a quarterly report outlining their patient populations' achievements in metabolic outcomes, expressed both as means and as centile ranking within the group, along with the proportion using evidence-based cardiovascular disease prevention therapies. In other jurisdictions, 21 quality improvement initiatives have similarly involved outcomes reporting for pediatric T1D populations. In a comparison of pediatric outcomes among eight high income countries, optimal glycemic outcomes appeared to be achievable where specific center performance data were publicly accessible, and where professional development initiatives were additionally targeted to results. In adult health care, although tempting to progress beyond individual physician feedback reporting, the transparent publication of performance data has not been consistently effective, at least in hospital-based care. 22 Novel approaches are still needed.
Lower HbA1c in CSII users has also been reported in clinical trials 23,24 as well in the T1DX, DIVE, and DPV registries. 10,11 Insulin pump use in T1D varies significantly by country, with pump use in adults ≥25 years as low as 1% in Latvia and as high as 83% in Austria. 13 In this Canadian cohort, only 40% were using CSII therapy, compared with the 63% reported in the United States, 10 indicating an underutilization of advanced technologies in Canada. Although our data have also shown a glycemic benefit in CSII versus MDI users, the advantage was much smaller in the subgroup using CGM, demonstrating the potential glycemic benefit of CGM in reducing the impact of the chosen mode of insulin delivery itself. However, CGM use in this cohort was also relatively low (22% vs. the T1DX experience of 30% 10 ) and was especially low among the 18–25 age cohort.
Many individuals were also not achieving other metabolic targets. Achievement of evidence-based targets for LDL-C and blood pressure is important to reduce the risk of cardiovascular disease and microvascular complications. 17 Canadian clinical practice guidelines call for statin therapy for people >30 years of age with a diabetes duration >15 years 17 and in this group, only 62.7% were using statin therapy. Only 61% of the total cohort had achieved target blood pressure, whereas antihypertensive therapy was only in place for 26.0% of the 26–49 cohort and 68.7% of the ≥50 age group.
Among CGM users, mean TIR was 53.4% and the recommended target of >70% 25,26 was only achieved by 15.7% of CGM users. Mean time in hyperglycemia and hypoglycemia were also well above the recommended targets of <25% and <4%, respectively. 27 As seen in the other real-world reports, CGM/FGM users in this cohort showed no difference in glycemia between individuals using MDI versus CSII therapy. 28 In this cohort, CGM users showed improved TIR and lower variability than FGM users. This difference appears to have been driven by approximately equal increases in time below range and time above range. Given the more recent introduction of FGM versus CGM, the glycemic outcomes differences may be due to considerably different respective durations of device use.
Certain self-care behaviors have become tenets in the effort to achieve optimal glycemic control. The relationship between SMBG frequency and glycemia has been well established 29,30 and testing 6–10 times daily is broadly endorsed. 31,32 Similarly, carbohydrate counting has undeniable value in helping an individual achieve an optimal glycemic outcome. In this “real-world” analysis, both behaviors were similarly associated with glycemic control. However, individuals who had self-selected to “rarely” carb count did not appear to have a resulting glycemic handicap. Similarly, lower SMBG frequencies (twice daily or more) were still associated with significant HbA1c benefit. Prior observational reports had not explored the benefits of SMBG frequencies below four times daily. Both of these self-care behaviors are sometimes prerequisites for funding, 33 often requiring arbitrary thresholds such as SMBG >4 times daily or advanced carbohydrate counting skill that are not fully evidence based. The resulting restrictions to access funding or programs may create unintended barriers that do not align with our preferred approach of individualizing care.
The primary strength of this study is the comprehensive nature of the LMC Diabetes Registry, including a variety of sociodemographic and clinical variables, as well as CGM/FGM and glucose meter data. In contrast, population registries reflect a more heterogeneous group; may lack insight into education, employment, and earning status; and may not have access to individual comorbidity or hypoglycemia experience. Volunteer-based registries may also reflect volunteer bias at either patient or clinic levels. Finally, the LMC cohort receives care within the Canadian public health system, minimizing obstacles to access evidence-based therapies and devices.
A limitation of this study is that LMC clinics primarily care for adults, and usually by referral from primary care. Individuals who had not provided prior consent and patients seen before the specified inclusion period could not be included. The results may, therefore, not be generalizable to the broader T1D population, especially children and adolescents, and individuals in primary care alone. The period of review ended in 2018 and findings such as device usage may not generalize to current populations with T1D. The LMC Diabetes Registry is based on an electronic medical record system so that information not routinely collected could not be included. Finally, only cross-sectional data were presented. Future studies should report on the clinical state and management of individuals with T1D in Canada over time.
Despite an advanced and well-resourced medical environment, including regular feedback on each specialist's individual cohort achievements, many individuals with T1D are not able to meet guidelines-based targets using guideline-derived systems and care programs. Younger adults appear to be the most deficient, in both control and in adoption of new technology. Use of adjunct risk-lowering therapies was incomplete, while use of advanced technologies was generally low. This pattern of limited response to our traditional medical systems appears to be consistent across multiple health jurisdictions and consistent over time. Although evidence-based development of care guidelines must continue, health care providers need real-time feedback about their respective patient outcomes to assess the impact and the feasibility of guideline-directed care. Even with this critical feedback in hand, there further appears to be a need of novel interventions and innovative communications to more effectively achieve outcomes in a larger proportion of individuals under our care.
Footnotes
Acknowledgment
The authors thank Richa Parihar for her contributions to the literature review and helping to draft the introduction.
Authors' Contributions
R.A. and R.E.B. designed the study and conducted the literature review. R.E.B. performed the data analysis. All authors interpreted the study data. R.A. and R.E.B. wrote the first draft of the article. A.A., R.G., B.A., Z.Y., and J-F.Y. provided critical revisions to the article. All authors approved the final version of the article.
Guarantor Statement
Ronnie Aronson is the guarantor of this article and takes responsibility for the contents of this article
Author Disclosure Statement
R.A. reports research grants from Sanofi, Novo Nordisk, Janssen, AstraZeneca, Becton Dickinson Technologies, Boehringer Ingelheim, Eli Lilly, Zealand, Xeris, Medpace, Kowa, Insulet, Dexcom, Bausch Health, Tandem Diabetes, and Bayer; and consultancies/personal fees from Sanofi, Novo Nordisk, Janssen, AstraZeneca, Becton Dickinson Technologies, Boehringer Ingelheim, Eli Lilly, HTL Strefa, Gilead, Merck. R.E.B. has no conflicts of interest to declare. A.A. reports grants and personal fees outside the submitted study from Sanofi, Novo Nordisk, Janssen, AstraZeneca, Eli Lilly, Merck, and Senseonics; personal fees from Boehringer Ingelheim and Dexcom, and grants from Amgen, Gilead, Glaxo-Smith-Kline, JDRF, Lexicon, Pfizer, Xeris, and Zealand. R.G. reports research support from Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Janssen, Medtronic, Merck, Novartis, Novo Nordisk, Roche, Sanofi, and Takeda; serving on advisory panels for AstraZeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Merck, Novo Nordisk, Roche, Sanofi, and Takeda; speaker bureaus for Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Merck, Novo Nordisk, Sanofi, and Servier; consulting for AstraZeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Novo Nordisk, and Takeda.
Funding Information
This study was supported by grants from Sanofi and Insulet. Neither Sanofi nor Insulet was involved in the writing of the article.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
Supplementary Table S8
Supplementary Table S9
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
