Abstract
Background:
Diabetes-related distress is common among persons affected by diabetes and is associated with suboptimal glycemic control and complications, thus constituting a relevant patient-report outcome (PRO). Improving glycemic control may reduce diabetes distress and improve treatment satisfaction. This post hoc analysis evaluated PRO data for a pooled cohort of adults with type 1 diabetes (T1D) receiving sotagliflozin as adjunct to optimized insulin in the inTandem1 and inTandem2 studies.
Methods:
Clinically meaningful changes in the Diabetes Treatment Satisfaction Questionnaire status version (DTSQs) and the two-item Diabetes Distress Scale (DDS2) total and individual scores were examined in the pooled data from the first 24 weeks of the studies.
Results:
In the cohort of patients with a baseline DTSQs total score ≤32 (∼76% of entire cohort), nearly twice as many patients treated with sotagliflozin 200 (45.9%) or 400 mg (42.3%) experienced a >3-point improvement from baseline versus those treated with placebo (24%). Treatment with sotagliflozin led to statistically significant (P < 0.05) improvements across all DTSQs items. Approximately 42% of all patients were considered to have a high risk of diabetes distress (total DDS2 score ≥6) at baseline following insulin optimization. More patients shifted from high to low risk with sotagliflozin compared with placebo (∼40% vs. 23%; P ≤ 0.0002). The baseline-adjusted difference in DDS2 from placebo was significantly (P < 0.001) reduced by −0.5 and −0.6 for sotagliflozin 200 and 400 mg, respectively.
Conclusions:
Patients with T1D treated with sotagliflozin in addition to optimized insulin therapy reported meaningful improvements in treatment satisfaction and diabetes distress.
NCT02384941 and NCT02421510
Background
Diabetes-related distress is common, with elevated levels of distress present in as many as 40% of patients or more, and once present it tends to persist. 1 –3 Patients should, therefore, be monitored for diabetes distress, particularly when they fail to meet treatment targets or develop complications. 4
Patients with type 1 diabetes (T1D) cite a need for simple and predictable disease management, a greater time in range (TIR) of glycemic target, reduced mental effort to manage their disease, and avoidance of hypoglycemia and weight gain as important unmet needs. 5 Successful management of T1D should therefore go beyond glycated hemoglobin (A1C) control to encompass patient-centered care, including consideration of nonglycemic effects of treatment and patient-reported outcomes (PROs) such as quality of life and satisfaction with treatment. 4,6 It has been recognized that higher levels of satisfaction with treatment contribute to better adherence, which can improve glycemic control and may reduce diabetes distress. 7
Sotagliflozin, a novel dual sodium glucose cotransporter (SGLT) 1 and 2 inhibitor, has been shown to improve A1C, postprandial and fasting glucose control, and TIR of glycemic target, with less documented hypoglycemia and severe hypoglycemia when compared with placebo in adults with T1D. 8 –10 The adverse event profile of sotagliflozin is consistent with that observed for other SGLT inhibitors in T1D study populations, with an increased risk of diabetic ketoacidosis and genital mycotic infections. 8 –10 In two such studies, PROs related to treatment satisfaction and diabetes distress were improved with sotagliflozin (vs. placebo) in adults with T1D. 8,9
The present analysis evaluated pooled PRO data from the inTandem1 and inTandem2 studies to make additional post hoc assessments of the Diabetes Treatment Satisfaction Questionnaire status version (DTSQs) total and individual scores and responder subpopulation (i.e., patients with meaningful improvements in PROs), as well as two-item Diabetes Distress Scale (DDS2) total and individual scores.
Methods
Data for this post hoc analysis of DTSQs and DDS2 scores were pooled from the first 24 weeks (primary efficacy time point) of the two double-blind, 52-week, phase 3 inTandem1 and inTandem2 clinical trials. Trial methodology and clinical results have been reported in detail elsewhere. Since this was a secondary analysis of existing data, no approval from an institutional review board (IRB) was required. The appropriate IRB approval was obtained for the original studies upon which the current secondary pooled analysis is based. 8,9
Treatment satisfaction was evaluated using the eight-item DTSQs, a validated instrument available in multiple languages and approved by the World Health Organization and the International Diabetes Foundation. 6 For each item, patients provide their responses using a seven-point scale (0–6), with higher numbers representing increasingly greater satisfaction; by contrast, lower numbers for the two items of perceived frequency of hyperglycemia and hypoglycemia are associated with greater satisfaction. 11 The treatment satisfaction score was the sum of six of the DTSQs items (range of 0–36), excluding the perceived frequency of hyperglycemia and hypoglycemia scores; all eight individual items were also evaluated separately. The DTSQs responder analysis was conducted in the subset of patients whose scores were able to be improved by >3 points (i.e., those with a baseline score ≤32). A clinically meaningful change was defined as >3-point increase in DTSQs total score based on a 0.5 standard deviation of the baseline score in the pooled data set. 12
Diabetes distress was measured using the DDS2, a patient-completed instrument that has demonstrated acceptable levels of sensitivity and specificity to screen people for potential distress associated with managing and living with diabetes. 13 Each item has a six-point scale that ranges from 1 (“Not a Problem”) to 6 (“A Very Serious Problem”); the DDS2 total score is the sum of scores for each item (range 2–12). A DDS2 total score of 6 represents an experientially based cutoff for high (≥6) and low (<6) risk of diabetes distress; patients with high diabetes distress should be further evaluated with the original 17-item DDS (DDS17), from which DDS2 was derived, to inform subsequent interventions. 13 We used this cutoff to define high- and low-risk categories and investigated the shifts in risk category from baseline to week 24.
Subgroup analyses
Specific subgroups related to efficacy response of treatment at week 24 were evaluated for change from baseline in both DTSQs and DDS2 scores to determine if there were any differences across the categories defined for the subgroups. The following subgroups were analyzed for between-treatment differences: (1) patients with a change in A1C from baseline of <0.5% or ≥0.5%; (2) patients with A1C <7.0% or ≥7.0%; (3) patients with a change from baseline in body weight of <3% or ≥3%; (4) patients with a change in TIR of glycemic target (70–180 mg/dL) of <10% or ≥10%; and (5) patients meeting the net composite of A1C <7.0% (8.6 mmol/L) with no severe hypoglycemia and no diabetic ketoacidosis versus those not meeting the net composite.
Statistical analyses
In the pooled cohort, treatment-group comparisons in the change from baseline to week 24 mean total and individual DTSQs score values were evaluated using the modified intention-to-treat population and mixed-model repeated-measures methods. The proportion of patients achieving a clinically meaningful change in DTSQs score from baseline to week 24 was calculated for each treatment group. Group-level changes in diabetes distress from baseline to week 24 were assessed in a modified intention-to-treat population, which included all randomized patients who had taken at least one dose of the study drug (consistent with previously described primary efficacy analyses). 8,9 To estimate the proportion of patients at high risk of diabetes distress, randomized patients who completed the DDS2 questionnaire at baseline (i.e., after insulin optimization) were included and deemed as being at either high or low risk. Comparisons were made between frequency distribution, based on chi-squared tests, if frequencies of all response/nonresponse ≥5; otherwise, a Fisher's exact test (two-sided) was used. To investigate the proportion of patients who changed risk categories during the studies, analyses were conducted on patients who completed the DDS2 questionnaire at baseline and at week 24; records with missing values for factors or responses were excluded from the statistical analyses. A threshold analysis on the proportion of patients improving (from high to low risk), remaining the same, or worsening (from low to high risk) was conducted using chi-squared statistic tests. A Cochran–Mantel–Haenszel test (stratified by randomization strata of insulin delivery and A1C at week −2 ≤ 8.5% or >8.5%) was used to calculate the proportion of patients whose scores could be improved by >3 points.
Results
Baseline and demographic characteristics for the pooled cohort are summarized by groups in Table 1. The results were generally well balanced across treatment groups. Overall, the mean age was ∼44 years, mean baseline A1C was 7.7%, and mean duration of disease was ∼21 years. Almost half of the patient population were women, and the majority (>90%) were Caucasian. At baseline, the overall mean DTSQs score was 28.5, and mean DDS2 score was 5.2.
Pooled Baseline and Demographic Characteristics for Participants in the InTandem1 and InTandem2 Studies
No patients started continuous/flash glucose monitoring while receiving treatment in the study.
A1C, glycated hemoglobin; BMI, body mass index; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DDS2, two-item Diabetes Distress Scale; DTSQs, Diabetes Treatment Satisfaction Questionnaire status version; IU, international units; SD, standard deviation.
Least squares mean (LSM) changes in DTSQs total scores were significantly (P < 0.001) greater with sotagliflozin than with placebo at week 24 (Fig. 1). The baseline-adjusted difference in LSM change in DTSQs total score from placebo inTandem1 and inTandem2 was 2.3 for sotagliflozin 200 mg and 2.1 for sotagliflozin 400 mg. In the cohort of patients with a baseline DTSQs total score ≤32 (i.e., patients whose scores could be improved by >3 points: ∼76% of entire cohort), nearly twice as many patients treated with sotagliflozin 200 or 400 mg (45.9% and 42.3%, respectively) experienced a >3-point improvement from baseline vs. those treated with placebo (24%; P < 0.0001 for both doses vs. placebo).
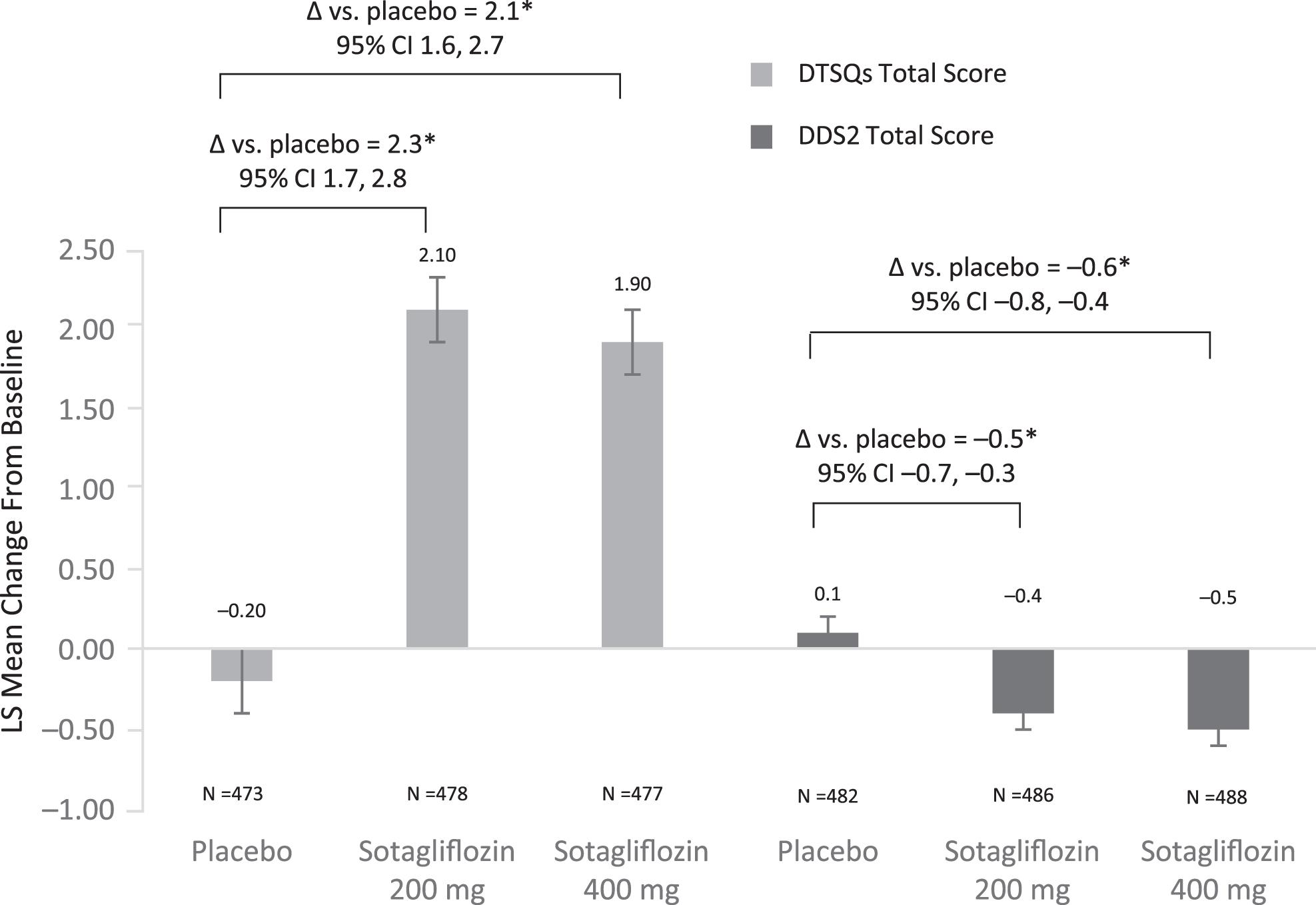
Change from baseline to week 24 in DTSQs total score and DDS2 total score in the pooled cohort.a aAn improvement in PROs is indicated by an increase in DTSQs scores and a decrease in DDS2 total scores. *P < 0.001 for treatment with sotagliflozin 200 or 400 mg versus placebo. CI, confidence interval; DDS2, two-item Diabetes Distress Scale; DTSQs, Diabetes Treatment Satisfaction Questionnaire status version; LS, least squares; PRO, patient-reported outcome.
Separately, a cumulative distribution function (CDF) curve for the placebo and sotagliflozin 200 and 400 mg groups (Fig. 2) was plotted to compare the proportion of patients experiencing a change in total DTSQs score from baseline between the groups. The separation of the CDF curves supports improved treatment satisfaction between the placebo and active treatment arms.
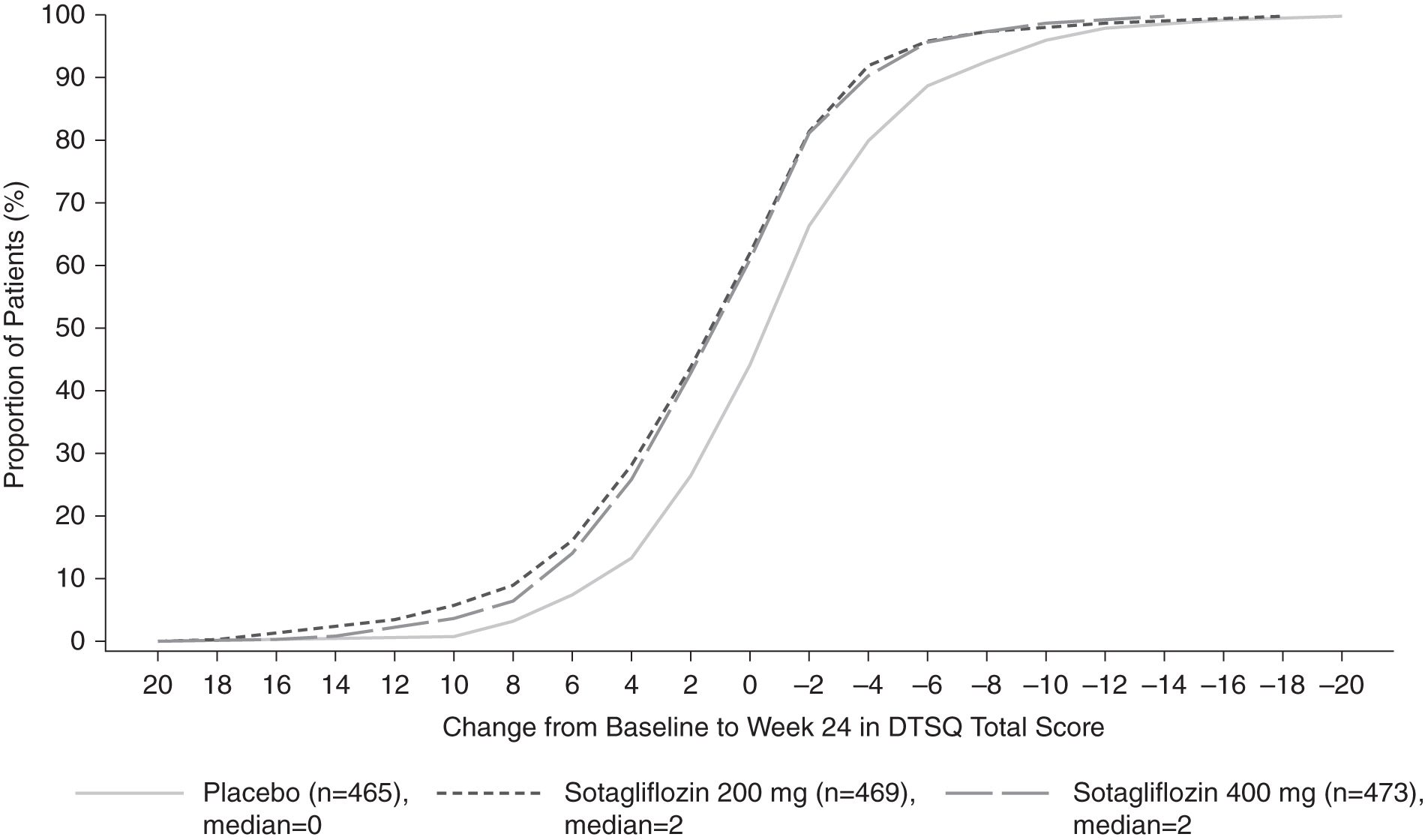
Cumulative distribution of change from baseline in DTSQs total score over time.a aAn improvement in PROs is indicated by an increase in DTSQs total scores.
Figure 3 shows the between-group comparisons for the individual items of the scores. Treatment with both doses of sotagliflozin led to statistically significant (P < 0.05) improvements in all items of the DTSQs. When evaluated by specific subgroups of change from baseline in efficacy parameters, the change in the DTSQs total score was greater with sotagliflozin (vs. placebo) and generally similar to the results observed in the overall cohort (Supplementary Fig. S1).
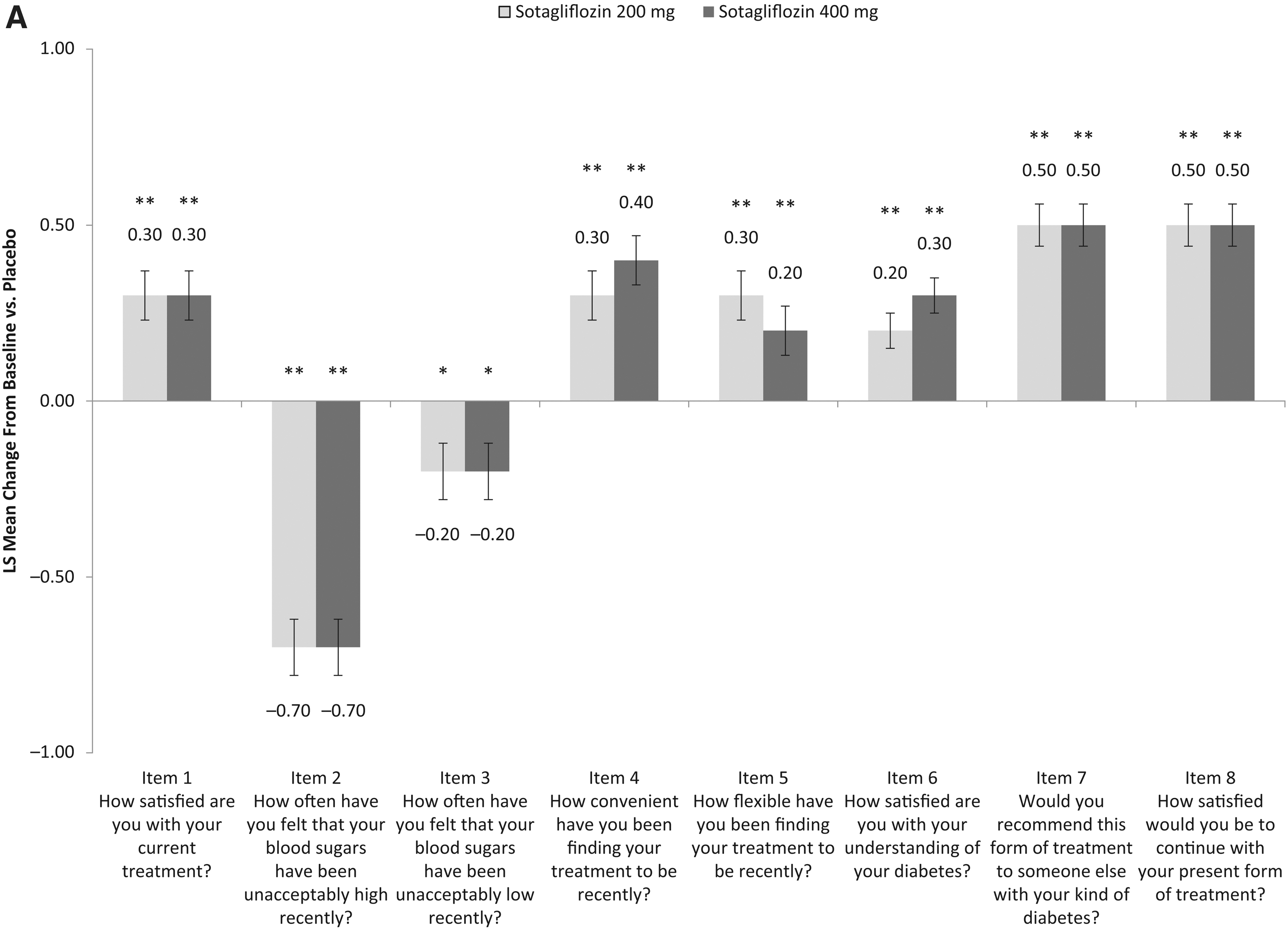
Difference from placebo in LS adjusted mean change from baseline to week 24 in the individual
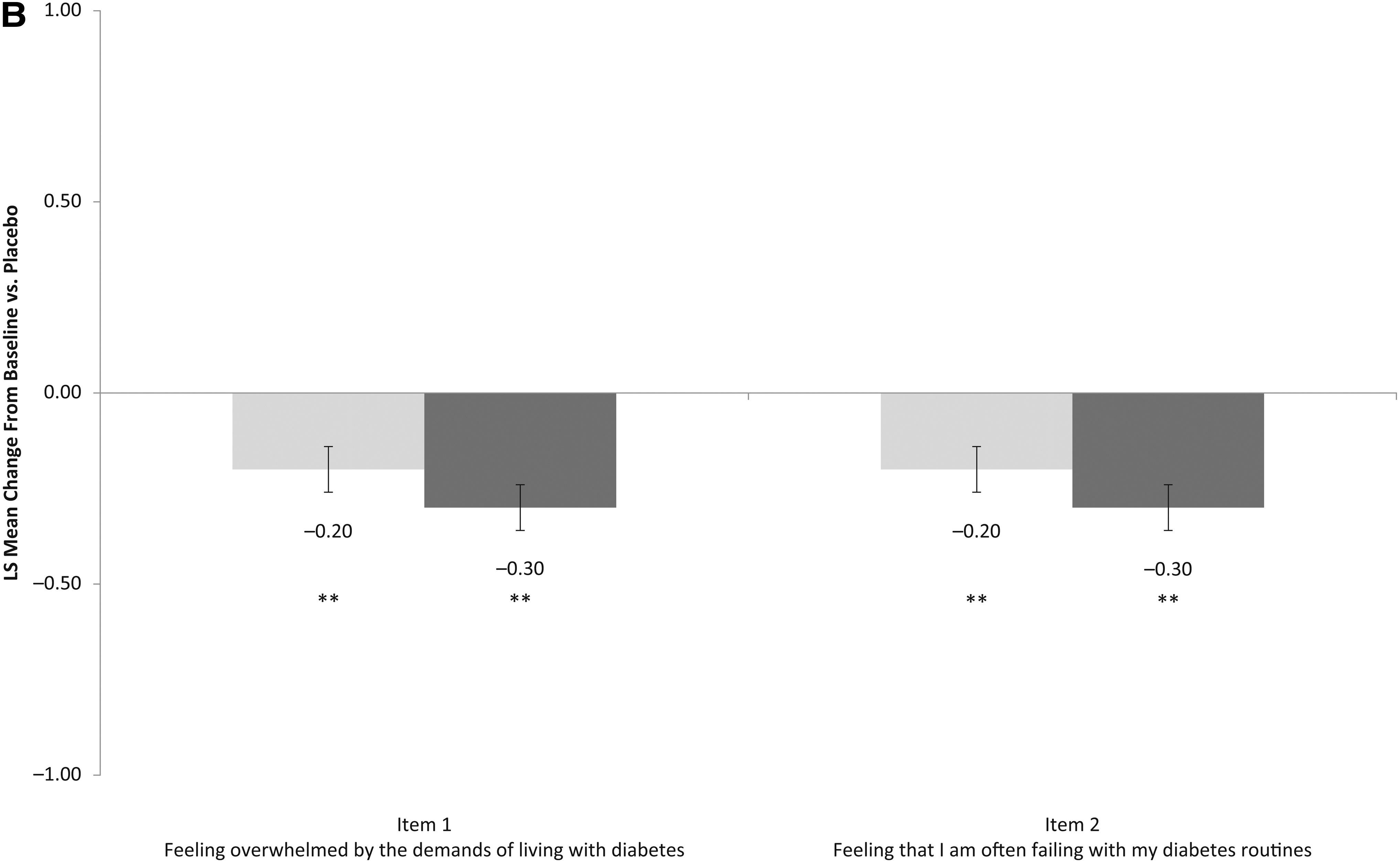
Two-item Diabetes Distress Scale
Approximately 42% of all patients were considered to have a high risk of diabetes distress at baseline following insulin optimization; total DDS2 score ≥6 (Table 2). Patients at high risk were significantly younger (42.2 ± 13.0 years vs. 44.8 ± 13.8 years; P ≤ 0.01), had shorter duration of T1D (19.6 ± 11.5 years vs. 22.7 ± 12.6 years; P ≤ 0.01), higher A1C levels (8.0% [10.2 mmol/L] ±0.8 vs. 7.7% [9.7 mmol/L] ±0.8; P ≤ 0.01), and a lower percentage of TIR of glycemic target (70–180 mg/dL) (48.6% ± 14.2 vs. 53.7% ± 14.7; P ≤ 0.01), all compared with patients at low risk of diabetes distress (total DDS2 score <6) (Supplementary Table S1).
Baseline Diabetes Distress Category and Shift in Diabetes Distress Categorization at Week 24
Proportion based on the number of patients in treatment group.
Proportion based on number of patients who screened for high diabetes distress at baseline (DDS2 score ≥6).
Proportion based on number of patients who screened for low diabetes distress at baseline (DDS2 score <6).
N represents the number of patients with a baseline assessment of DDS2.
Missing DDS2 total score values at week 24 were imputed using worst observation carried forward methodology.
Use of sotagliflozin resulted in a significant reduction in DDS2 total score from baseline to week 24 (P < 0.001), whereas an increase was observed with placebo (Fig. 1). The baseline-adjusted difference from placebo in the pooled inTandem1 and inTandem2 cohort was −0.5 for sotagliflozin 200 mg and −0.6 for sotagliflozin 400 mg. When evaluated by specific subgroups, the reductions in DDS2 scores were greater with sotagliflozin than with placebo and were generally similar to results observed in the overall cohort (Supplementary Fig. S2).
A comparison of shifts in diabetes distress risk is shown in Table 2. Among patients who experienced a change in risk group, a greater proportion of patients on sotagliflozin shifted from high to low risk (40.4% for sotagliflozin 200 mg and 40.2% for sotagliflozin 400 mg vs. 23.2% for placebo; P ≤ 0.0002 for both active treatment groups vs. placebo), and more patients on placebo shifted from low to high risk (13.0% for sotagliflozin 200 mg and 9.4% for sotagliflozin 400 mg vs. 21.3% for placebo; P < 0.01 for both active treatment groups vs. placebo).
Discussion
Treatment satisfaction is relevant to medication adherence and constitutes an important factor in the successful management of T1D, 6,7 as patients with lower satisfaction scores are more likely to discontinue therapy. 6 This post hoc analysis of DTSQs scores shows that treatment satisfaction and concern about hypo- and hyperglycemia improved for patients with T1D treated with sotagliflozin. Based on a specified threshold and the CDF curves, a greater proportion of patients receiving sotagliflozin had a clinically meaningful improvement in treatment satisfaction at the end of week 24 versus placebo.
At baseline (i.e., post-insulin optimization), a high proportion of patients were deemed to be at risk for diabetes distress; this is as reported in other published studies, where the prevalence of diabetes distress in adults with T1D is estimated at ∼42%. 1,2 In the present analysis, younger age, shorter duration of T1D, higher A1C, and a lower percentage of TIR of glycemic target were recognized as characteristics of patients at high risk for diabetes distress, and introduction of sotagliflozin resulted in an improvement in diabetes distress at week 24 at both doses versus placebo.
Although no specific subgroup response appeared to contribute to the outcomes, the benefits observed with sotagliflozin, including improved glycemic control, TIR of glycemic target, and body weight, may have collectively contributed to the observed PRO benefits. We conducted exit interviews to understand why DTSQs and DDS2 scores improved in patients using sotagliflozin versus placebo: responses indicated that what patients valued most was the improvements they experienced in glycemic stability and control. 14 This predominant theme of glycemic stability is relevant to several dimensions covered by the PROs. The top reported benefits in the exit interviews were (in order of frequency of spontaneous reporting) as follows: fewer hyperglycemia events, greater blood glucose stability, lower A1C, insulin works better/reduced insulin requirements, fewer hypoglycemia events, and weight loss. Each of these benefits was much more common in the sotagliflozin groups than in the placebo group. 14 The known benefit of less documented hypoglycemia and severe hypoglycemia with treatment with sotagliflozin could have contributed to the improved PROs seen in our analysis.
Multiple studies have demonstrated the value of reduced diabetes distress and higher treatment satisfaction on a patient's emotional burden and outcomes. 7,15 –18 Prospective data from newly diagnosed patients show that A1C trajectories over time are not predicted from baseline distress, but, rather, they appear to be related to the experience during the course of the disease. 19 The results of the present study are in agreement with previous studies showing a relationship between poorer glycemic control and higher risk of diabetes distress. 15,16 Furthermore, in a study where the population was stratified by age, high diabetes distress was associated with greater A1C values regardless of age. 17 This is notable given that younger age was associated with higher risk of diabetes distress in both our study and in previous studies. 20,21 In addition, a noninterventional study in patients with T1D showed that baseline diabetes distress was associated with higher A1C and a greater percentage of missed bolus insulin doses. Over time, a decrease in diabetes distress was associated with a decrease in A1C. 18
This post hoc analysis was not specifically powered to detect differences between patients with high and low risk of diabetes distress or subtle changes in treatment satisfaction, but it is still interesting to see robust differences between sotagliflozin and placebo being reported in this analysis. TIR of glycemic target data were only available for a small subgroup of patients, further reducing statistical power. Although the DDS2 can be used to classify patients with low and high risk of diabetes distress, it remains a screening tool, and the actual prevalence of diabetes distress cannot be established without additional follow-up using the long form of the assessment. 13 However, in this study, patients with high diabetes distress were not further evaluated with the original DDS17, as previously recommended. 13 Strengths of the present analysis include the use of patient-level data combining patients from two large multinational, controlled trials.
More specific tools that are more sensitive to change (DTSQs and DDS17) are available, and a more robust change may be observed with these. However, the large treatment effects and significant P values observed with the tools used in this analysis suggest a robust effect of sotagliflozin on PROs. Patients diagnosed with T1D face a lifetime of challenges in managing their disease in the context of family, work, and social life, and these challenges can lead to poorer quality of life and other burdens. These PROs are clinically important to improving outcomes in patients with T1D, as poor satisfaction and/or diabetes distress have been implicated in poor adherence to treatment and poor glycemic control that increase patients' risk for complications.
Conclusion
Patients with T1D treated with sotagliflozin in addition to optimized insulin therapy reported significant improvements in treatment satisfaction and diabetes distress. When selecting the most appropriate treatment for patients with T1D, clinicians should consider how treatment affects the whole patient; they should look beyond A1C alone. Factors such as weight gain/loss, hypoglycemia risk, cardiovascular effects of treatment, and humanistic concerns about the impact of diabetes distress, satisfaction, and well-being should all be considered.
Footnotes
Acknowledgment
The authors would like to thank Leila Djebarri for assistance with the statistical analysis.
Prior Presentation
Parts of data from this study were presented at the 2019 European Association for the Study of Diabetes annual meeting in Barcelona, Spain, September 16–20, 2019, and the 2018 Academy of Managed Care Pharmacy—Nexus conference in Orlando, Florida, October 22–25, 2018.
Author Disclosure Statement
V.N.J., P.L., M.J.D., P.B., and S.S. are employees of Lexicon Pharmaceuticals, Inc; authors who are employees of Lexicon Pharmaceuticals, Inc., may own common stock or may have been granted stock options or other equity incentive awards. D.L. was a postdoctoral fellow at Sanofi US, Inc., during the study and now is employed at Janssen Scientific Affairs, LLC. M.A., M.R., and S.G. are employees at Sanofi; authors who are employees of Sanofi may own common stock or may have been granted stock options or other equity incentive awards. T.S.B. has received research support from Abbott, Capillary Biomedical, Dexcom, Diasome, Eli Lilly, Kowa, Lexicon, Medtronic, Medtrum, Novo Nordisk, REMD, Sanofi, Senseonics, ViaCyte, vTv Therapeutics, and Zealand Pharma; has received consulting honoraria from Abbott, LifeScan, Novo Nordisk, and Sanofi; and received speaking honoraria from Medtronic and Sanofi. J.B.B received contracted consulting fees paid to the University of North Carolina by Adocia, AstraZeneca, Dance Biopharm, Dexcom, Eli Lilly, Fractyl, GI Dynamics, Intarcia Therapeutics, Lexicon, MannKind, Metavention, NovaTarg, Novo Nordisk, Orexigen, PhaseBio, Sanofi, Senseonics, vTv Therapeutics, and Zafgen for 3 years, and by Adocia, AstraZeneca, Dance Biopharm, Eli Lilly, MannKind, NovaTarg, Novo Nordisk, Senseonics, vTv Therapeutics, and Zafgen for 1 year. He received grant support from AstraZeneca, Eli Lilly, Intarcia Therapeutics, Johnson & Johnson, Lexicon, Medtronic, Novo Nordisk, Sanofi, Theracos, Tolerion, and vTv Therapeutics for 3 years and from Novo Nordisk, Sanofi, Tolerion, and vTv Therapeutics for 1 year. He is a consultant to Cirius Therapeutics Inc., CSL Behring, Mellitus Health, Neurimmune AG, Pendulum Therapeutics, and Stability Health; holds stock/stock options in Mellitus Health, Pendulum Therapeutics, PhaseBio, and Stability Health; and has received grant support from the National Institutes of Health (UL1TR002489, U01DK098246, UC4DK108612, U54DK118612), PCORI, and ADA. T.D. is a consultant, advisory board member, steering committee member/speaker for and has received research support from Abbott, AstraZeneca, Bayer, Boehringer Ingelheim, Dexcom, Eli Lilly, Insulet, Medtronic, Novo Nordisk, and Roche; and is a shareholder of DreaMed Diabetes Ltd. F.J.S is a consultant and advisory board member for Abbott, Eli Lilly, Novo Nordisk, and Roche Diabetes Care; has received research support from Sanofi; and his contracted consulting fees and grants are paid to the Amsterdam University Medical Centers. W.H.P. is a consultant and advisory board member for Abbott, Ascensia, AstraZeneca, Dexcom, Eli Lilly, Insulet, MannKind, Novo Nordisk, Onduo, Sanofi, Roche, and Xeris.
Funding Information
This study was funded by Sanofi US, Inc., and Lexicon Pharmaceuticals, Inc., The authors received writing/editorial support provided by Grace Richmond, PhD, and Patricia Fonseca, PhD, of Excerpta Medica, funded by Sanofi US, Inc.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
