Abstract
Current intermittently scanned continuous glucose monitoring (isCGM) devices do not allow calibration and require confirmatory self-monitored blood glucose (SMBG) under specific defined conditions. The overall time spent in an SMBG-indicated state while using isCGM has not been reported in the existing literature. We present the time spent in an SMBG-indicated state and the daily number of transitions to an SMBG-indicated state during isCGM use in a high-risk cohort of adults with type 1 diabetes. This is a post hoc analysis of participants randomized to isCGM as part of a clinical study. The percentage of values meeting one or more of the criteria for SMBG testing (hours per day) and the number of transitions to an SMBG-indicated state per day were calculated according to the manufacturer's label in the United Kingdom. Complete data sets for 20 participants on isCGM for 8 weeks were included in the analysis. Participants spent a mean (SD) of 5.61 (1.81) h/day in an SMBG-indicated state and there were 5.43 (1.42) transitions to an SMBG-indicated state per day while using isCGM. These findings have important implications for cost-effectiveness and for decision-making around insulin dose calculations and driving. Our findings emphasize the need for continued SMBG testing among people at high risk of hypoglycemia who use isCGM. Cost-effectiveness analyses for isCGM may require revision.
Introduction
Monitoring of glucose is an essential component of self-management of type 1 diabetes mellitus (T1DM) and, for most people with T1DM globally, self-monitored blood glucose (SMBG) remains standard care. SMBG supports optimization of glucose by enabling real-time insulin and carbohydrate decisions. International clinical guidelines for the management of T1DM recommend SMBG testing 4–10 times a day. 1,2
A device for intermittently scanned continuous glucose monitoring (isCGM) (FreeStyle Libre; Abbott Diabetes Care), also known as flash glucose monitoring, obtained CE marking and FDA approval in 2014 and 2017, respectively. The device does not allow for calibration and has been marketed primarily as a replacement for SMBG for people with diabetes. The criteria for reimbursement are variable from country to country but favor those with T1DM on intensive insulin treatment.
In the United Kingdom, NHS England has implemented national clinical criteria for NHS reimbursement of isCGM for people with T1DM, and those with any type of diabetes on hemodialysis, with a predicted estimate that 20% of the total U.K. population with T1DM will meet the reimbursement criteria. 3
The IMPACT trial, the only randomized controlled trial to date comparing isCGM with SMBG for 6 months in adults with well-controlled T1DM, showed a significant reduction in hypoglycemia (<3.9 mmol/L) with isCGM with no impact on HbA1c. 4 The control group in the IMPACT trial SMBG 5.4 times per day versus 0.5 times per day for isCGM users. 4 A cost-effectiveness analysis for isCGM for the U.K. T1DM population showed that the additional cost of isCGM compared with SMBG may be offset by the reduction in hypoglycemia observed with isCGM. 5 It is important to note that the model assumed SMBG 0.5 times per day for isCGM use, based on the IMPACT data, and the cost improvement was for severe hypoglycemia making it applicable to a high-risk T1DM population. Similarly, in Sweden, isCGM has been shown to be potentially cost-effective compared with SMBG in T1DM management. 6
However, although it does not require calibration, isCGM does not wholly replace SMBG – the manufacturer's regulatory-approved U.K. label for isCGM (Freestyle Libre) outlines a set of criteria for when confirmatory capillary blood glucose test is indicated (
In the Impact on Hypoglycaemia Awareness of Real Time CGM and Intermittent Continuous Glucose Data (IHARTCGM) randomized controlled study, we evaluated the impact of isCGM (Freestyle Libre) compared with real-time CGM (Dexcom G5) in people with T1DM at high risk of hypoglycemia for 8 weeks. 7 In this short report, we present the time spent in an SMBG-indicated state and the daily number of transitions to an SMBG-indicated state during isCGM use in this high-risk population.
Methods
The IHARTCGM study protocol has been described in detail elsewhere. 7 Adults with T1DM and impaired awareness of hypoglycemia or a recent history of severe hypoglycemia were randomized to isCGM or real-time CGM for 8 weeks after a short blinded CGM run-in period. For the purpose of this post hoc analysis, we extracted the complete isCGM data set of 8 weeks of glucose data for all 20 participants randomized to isCGM. All participants were given education in line with the device manufacturer's guidance at the randomization visit and used the device for 8 weeks. Participants were not asked to keep a record of SMBG monitoring to minimize study burden.
The percentage of values meeting one or more of the criteria for SMBG testing was calculated for each participant, expressed as hours per day. A value met one of the SMBG testing criteria if (1) it was <70 mg/dL, (2) the rate of change was >2 mg/(dL·min), or (3) the estimated glucose value in the next 15 min was predicted to be <70 mg/dL, based on the current rate-of-change (impending hypoglycemia). The number of transitions to an SMBG-indicated state per day was calculated for each participant, assuming 96 readings per day.
Results
Complete data sets for 20 participants on isCGM for 8 weeks were included in the analysis [12 male; median interquartile range (IQR): age 48.5 years (34–63); median (IQR): duration of diabetes 28.0 (16.5–36.5); median (IQR): HbA1c 55 (48–65) mmol/mol, 7.2% (6.5%–8.1%)]. The time spent in SMBG-indicated state based on the participants meeting one or more criteria and the total number of transitions to an SMBG-indicated state are summarized in Table 1. The mean total number of transitions to an SMBG-indicated state per day, excluding the rapidly rising criteria, equates to 3.89 (1.15) events, suggesting that the majority of SMBG-indicated events are associated with rapidly falling glucose, hypoglycemia (<70 mg/dL), or impending hypoglycemia. The number of SMBG-indicated events per day corresponding to each of the main criteria for capillary blood glucose testing is illustrated in Figure 1.
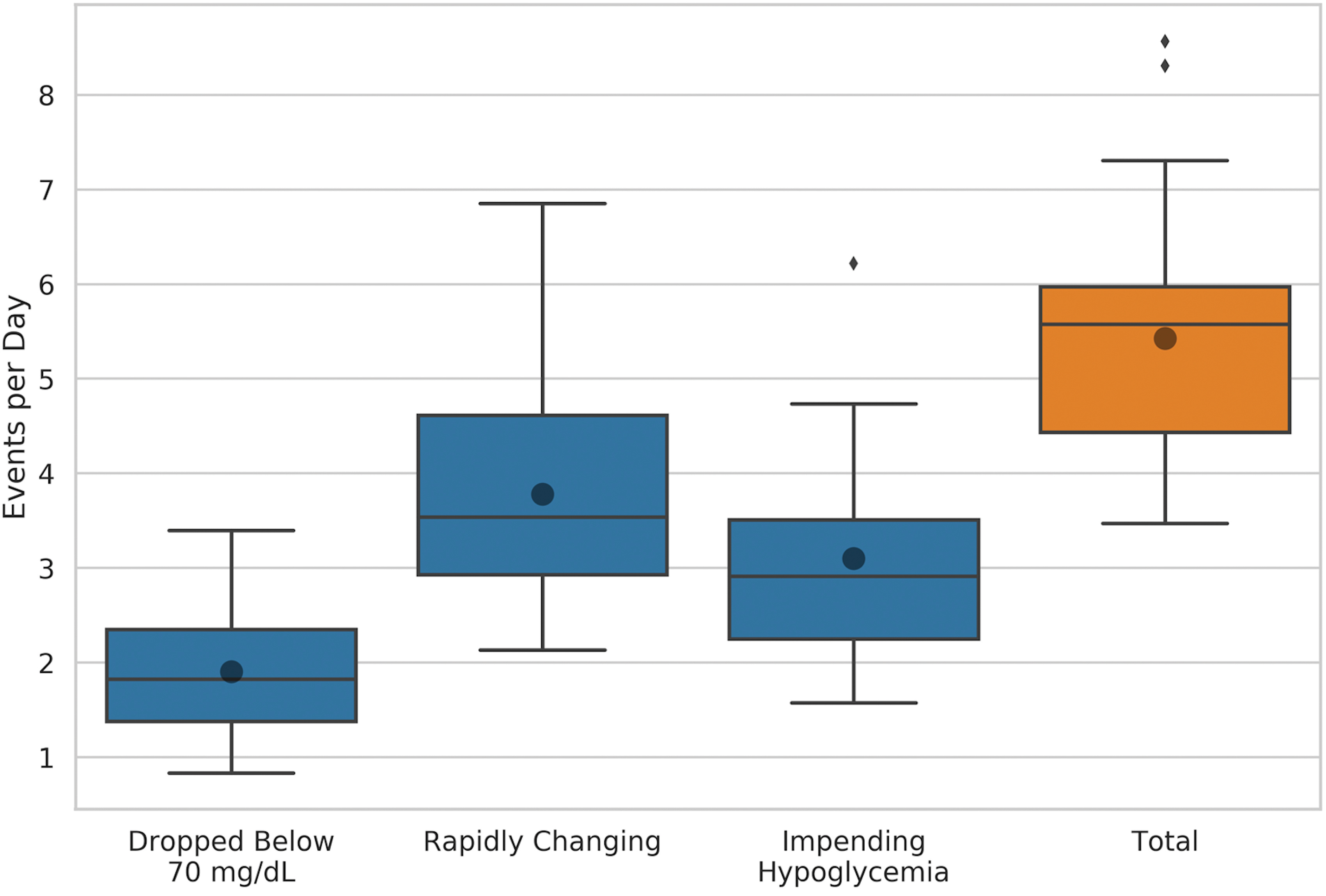
Median (IQR) number of transitions to an SMBG-indicated state per day corresponding to the SMBG-indicated criteria. The small filled diamonds correspond to outliers. The dots correspond to the mean number. IQR, interquartile range; SMBG, self-monitored blood glucose. Color images are available online.
Mean (SD) Time Spent in an SMBG-Indicated State and Mean (SD) Number of Transitions to an SMBG-Indicated State Per Day
SMBG, self-monitored blood glucose.
Discussion
The results from this post hoc analysis demonstrate that participants randomized to isCGM in the IHARTCGM trial spent >5 h a day in an SMBG-indicated state and, on average, more than five transitions to an SMBG-indicated state occurred per day while using isCGM. These findings are important for the cost-effectiveness of isCGM and suggest that previous models for reductions in hypoglycemia assuming 0.5 SMBG per day may require revision especially as, in the high-risk cohort recruited to the IHARTCGM study, hypoglycemia was not reduced at the study endpoint in the isCGM group. Furthermore, a recent study published showed that isCGM does not appear to have a role in preventing severe hypoglycemia in at-risk individuals with diabetes. 8
These data have additional important implications for decision-making around insulin dose calculations and driving, with potential adverse outcomes arising from omitting confirmatory tests in line with the label. In the IMPACT study, the participants in the SMBG group (n = 120) tested 0.5 times per day for 6 months. 4 It is well known that adhering to SMBG guidelines is challenging for people with T1DM due to multiple barriers. 9
The number of transitions to an SMBG-indicated state does not automatically equate to the number of required SMBG tests. Users are generally advised to scan their isCGM sensor a minimum of eight times per day, and if a transition to an SMBG state occurs outside of when they scan (i.e., when they have no reason to scan), then it may have no adverse impact.
This analysis is limited by the short duration and relatively small study population of the IHARTCGM study, and we do not have records of SMBG frequency as it was not part of the study protocol. However, this is the first analysis of this kind and is important in selecting a high-risk homogeneous group.
Although randomized controlled data evaluating isCGM versus SMBG in T1DM are limited to the IMPACT study, there is a growing evidence base, derived from observational studies in T1DM, implying that isCGM has a favorable impact on glycemic control and user satisfaction. 10 Our findings suggest that all studies of isCGM should report both the predicted and actual SMBG frequencies. With the advent of isCGM with alarms, a qualitative alarm to require confirmatory SMBG should be considered by manufacturers and regulators. With improving accuracy and clearer labels, this alarm would become infrequent but is potentially critical for driving and insulin dosing decisions.
In conclusion, our findings emphasize the need for continued SMBG testing among people at high risk of hypoglycemia who use isCGM. Cost-effectiveness analyses for isCGM may require revision.
Footnotes
Acknowledgments
The authors thank all the study participants for their valuable time and commitment to the IHARTCGM study. The authors also thank Mark Derdzinski (Senior Data Scientist, Dexcom) for his contribution toward the data analysis for this article.
Authors' Contributions
M.R. analyzed the initial data set and wrote the article. N.O. reviewed and edited the article. N.O. is the guarantor of this study and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Disclaimer
The views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health. Imperial College London is supported by the NIHR Local Clinical Research Network and the Imperial NIHR Biomedical Research Centre.
Author Disclosure Statement
M.R. has received research funding toward an investigator-initiated study from Dexcom and has participated in advisory boards for Roche Diabetes. N.O. has received research funding toward investigator-initiated studies from Dexcom, and has participated in advisory boards for Roche Diabetes, Dexcom, and Medtronic Diabetes.
Funding Information
The IHARTCGM study was funded by Dexcom. The funder was not involved in the design of the study, collection of data, or publication decisions. The IHARTCGM study was supported by the NIHR CRF at Imperial College Healthcare NHS Trust.
