Abstract
Background:
Albuminuria characterizes the progression of kidney injury. The effect of canagliflozin on the excretion of microalbumin was assessed for investigating its renoprotective potential in Japanese patients with type 2 diabetes mellitus (T2DM).
Patients and Methods:
Twenty Japanese patients with T2DM and microalbuminuria were enrolled and administered with 100 mg of canagliflozin once a day for 12 weeks. These subjects were admitted to the clinic at the start and end of the treatment period for 24-h urine collection. The primary endpoint was the percentage change in geometric mean 24-h urinary albumin excretion from baseline to week 12.
Results:
The urinary albumin level decreased by 42.0% (95% confidence interval: 21.9–57.0;
Conclusions:
The results of this study indicate that canagliflozin decreases microalbuminuria in Japanese patients with T2DM. Albuminuria could be reduced as a result of changes in various physiological pathways; therefore, it is imperative that future, large-scale, studies attempt to determine the detailed mechanisms involved. Canagliflozin may offer a novel therapeutic option for Japanese patients with T2DM and incipient nephropathy.
Introduction
D
Canagliflozin is a sodium glucose cotransporter 2 (SGLT2) inhibitor that increases the urinary excretion of glucose by inhibiting urinary glucose reabsorption in the proximal tubule.
11
Several clinical studies have demonstrated that canagliflozin can control hyperglycemia and reduce body weight and blood pressure in patients with T2DM.
12
–15
In addition, SGLT2 inhibitors are thought to exert renoprotective effects by enhanced tubuloglomerular feedback, leading to a reduction in hyperfiltration,
16
–18
and by reducing inflammatory, fibrotic, and hyperplastic responses of proximal tubular cells via blocking glucose flow into the proximal tubule cells.
19
In a phase 3 trial, compared with glimepiride, canagliflozin reduced urinary albumin excretion and caused an initial reduction in estimated glomerular filtration rate (eGFR), followed by a stabilization of kidney function.
20
In the
In the present study, we aimed to examine the renoprotective effect of canagliflozin in Japanese patients with T2DM and microalbuminuria via 24-h urine collection. For further characterizing the effect of this treatment on kidney, we also investigated changes in biomarkers for kidney injury, inflammation, and oxidative stress.
Patients and Methods
Patients
Male and female patients (age 20–74 years) with T2DM exhibiting an albumin-to-creatinine ratio of 30.0–299.9 mg/g creatinine in a first-morning-void urinary test were selected. Subjects were treated using diet and exercise therapy alone, or in combination with antidiabetic drug therapy other than SGLT2 inhibitors, insulin, and glucagon-like peptide-1 receptor agonists. Other inclusion criteria were as follows: hemoglobin A1c (HbA1c) ≤10%, systolic blood pressure (SBP)/diastolic blood pressure (DBP) <140/90 mmHg, and eGFR ≥45 mL/min/1.73 m2. eGFR was calculated according to the Japanese eGFR equation on the basis of serum creatinine (eGFRcreat). 23
Study design
This study was a single-site, single-arm, open-label study aimed at evaluating the effect of canagliflozin on urinary microalbumin levels in patients with T2DM. We set our exploratory sample size at 20 subjects for feasibility of the study with an estimation accuracy of 95% confidence interval (CI), equating to ∼1/2 of the standard deviation (SD). Canagliflozin (100 mg) was administered once a day before or after breakfast for 12 weeks. Subjects were instructed not to change their diet and exercise regimen during the study and keep the dosage and administration of all concomitant medications constant. Subjects were admitted to the Naka Kinen Clinic 1 day before the start and end of the 12-week treatment period for 24-h urine collection. Spontaneous reports of adverse events (AEs) were collected from patients at the hospital visits. AEs were classified according to the Medical Dictionary for Regulatory Activities/Japanese Edition (MedDRA/J) version 20.0. This study was conducted between August 2016 and December 2017 in accordance with the Declaration of Helsinki (revised in October 2013). Written informed consent was obtained from all individuals before participation in this study. The protocol was registered in the UMIN Clinical Trial Registry as UMIN000023959 and was approved by the Institutional Ethics Review Committee of Naka Kinen Clinic. Members of the Clinical Operations Department, DOT World Co., Ltd. (Tokyo, Japan) performed monitoring and auditing in accordance with the Japanese governmental ethical guidelines and the study protocol.
Endpoints
The primary endpoint was the percentage change in geometric mean 24-h urinary albumin excretion from baseline to week 12. Secondary endpoints included changes in laboratory data. Any AEs that occurred during the period of ongoing canagliflozin treatment were recorded.
The following parameters were measured in 24-h urine samples: albumin, transferrin,
Statistical analyses
Data are presented as mean ± SD or mean with 95% CI. Considering the skewed distribution of data, 24-h urinary albumin and the albumin-to-creatinine-ratio in first-morning-void urine samples were log transformed for analysis. The results of the mean changes and 95% CI in the log transformed data from baseline to 12 weeks were back transformed (anti-log) to obtain geometric means of the ratios of the 12-week value to the baseline value and corresponding 95% CI. The values were then expressed as the percentage change from the respective baseline values. Changes from baseline were analyzed using two-tailed paired
Results
Patients
A total of 20 subjects (15 males and 5 females) were treated with 100 mg of canagliflozin for 12 weeks. None of the subjects withdrew after consenting to the study. At baseline, these subjects had an HbA1c level of 7.30 ± 0.91% and were 62.9 ± 8.6 years old. Mean body weight was 67.3 ± 11.1 kg, mean body mass index was 26.2 ± 4.4 kg/m2, and the mean duration of type 2 diabetes was 13.6 ± 9.3 years. Twelve patients (60%) were taking an angiotensin receptor blocker.
Primary endpoint
As shown in Figure 1, patients had a geometric mean 24-h urinary albumin of 142.4 mg/day at baseline and 82.6 mg/day after 12 weeks of treatment with canagliflozin. The geometric mean 24-h urinary albumin significantly decreased by 42.0% (
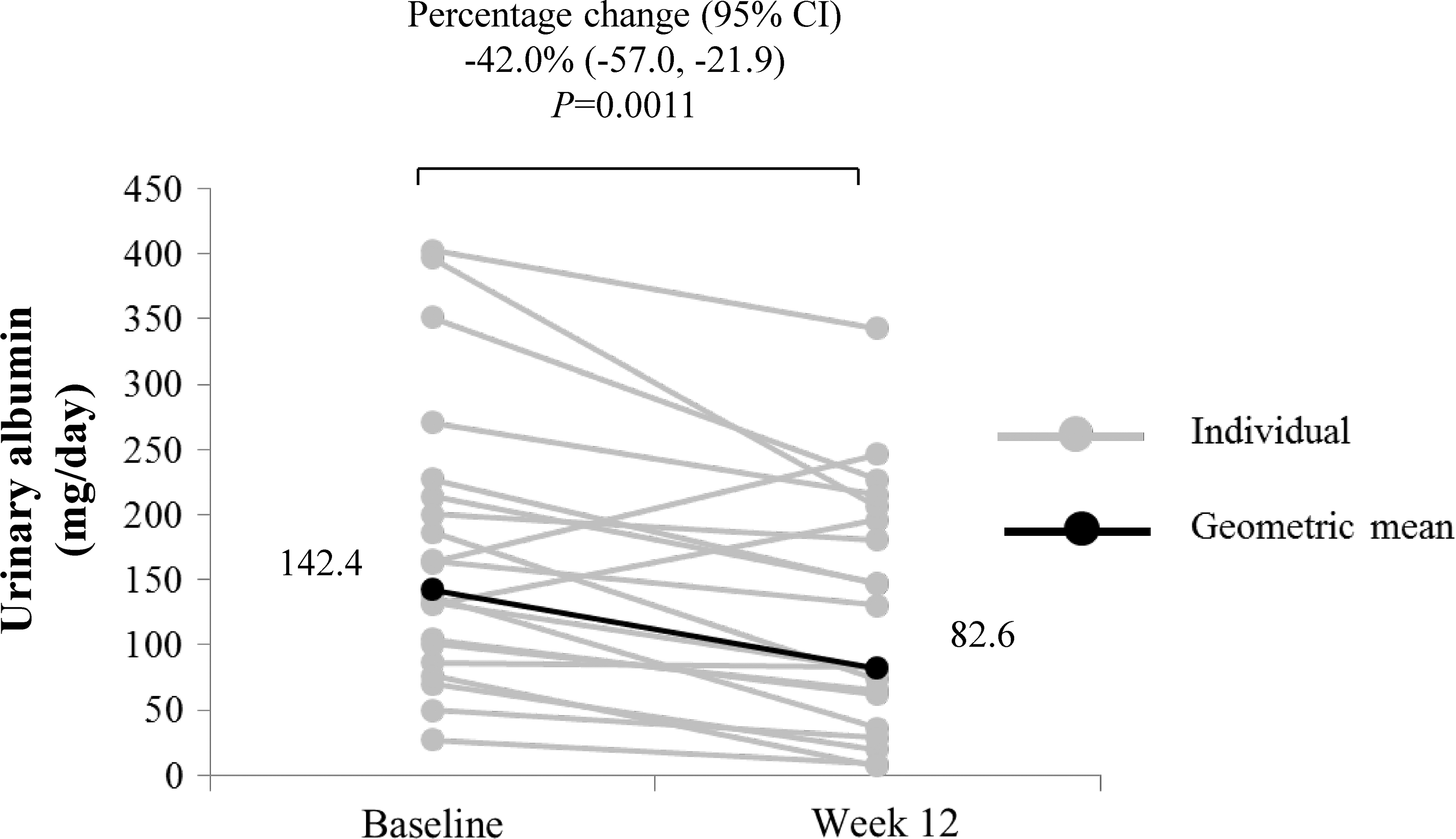
Change in 24-h urinary albumin from baseline to week 12. Data are expressed as individual measurements (gray circles) and geometric mean (black circles and values in the figure). Statistical comparison for the change in geometric mean from baseline to week 12 was conducted using a paired
Data are expressed as geometric mean (95% CI) and
CI, confidence interval; SD, standard deviation.
Fasting plasma glucose, body weight, HbA1c, and blood pressure
Twelve weeks of treatment with 100 mg canagliflozin significantly reduced fasting plasma glucose and HbA1c by 24 mg/dL and 0.24%, respectively (Table 2). Body weight also significantly decreased by 1.7 kg, whereas no significant differences were detected in SBP and DBP (Table 2).
Data are expressed as mean ± SD, mean ± SD (95% CI), and
DBP, diastolic blood pressure; HbA1c, hemoglobin A1c; SBP, systolic blood pressure.
eGFR and creatinine clearance
eGFRcreat and eGFRcys significantly decreased by 8.9 mL/min/1.73 m2 and 8.0 mL/min/1.73 m2, respectively (Table 3). Creatinine clearance also decreased by 16.9 mL/min/1.73m2 (Table 3).
Data are expressed as mean ± SD, mean ± SD (95% CI), and
eGFRcreat, estimated glomerular filtration rate based on creatinine; eGFRcys, estimated glomerular filtration rate based on serum cystatin C.
Biomarkers for tissue damage, oxidative stress, inflammation, and the renin–angiotensin–aldosterone system
Next, we investigated a range of markers for glomerular damage. While the urinary transferrin level was significantly decreased after 12 weeks of treatment with canagliflozin (Table 4), the level of urinary type IV collagen did not change significantly. Regarding urinary markers for tubular injury, L-FABP and NGAL significantly decreased and increased, respectively, after 12 weeks of canagliflozin treatment (Table 4). Urinary NAG, β2-microglobulin, and KIM-1 remained unchanged, while urinary 8-OHdG, a marker of oxidative stress, was significantly decreased after 12 weeks of treatment with canagliflozin. Levels of inflammatory markers, such as plasma IL-18 and serum high-sensitivity CRP, did not significantly change. Plasma TNFR2 levels significantly increased, while TNFR1 levels did not significantly change. Plasma aldosterone levels and renin activity were both significantly increased, while urinary aldosterone did not significantly change.
Data are expressed as mean ± SD, mean ± SD (95% CI), and
CRP, C-reactive protein; 8-OHdG, 8-hydroxy-2′-deoxyguanosine; IL-18, interleukin-18; KIM-1, kidney injury molecule-1; L-FABP, liver-type fatty acid binding protein; NAG,
Hematocrit, water intake, urinary volume, serum uric acid, and erythropoietin
Hematocrit significantly increased by 3.0%, whereas erythropoietin and serum uric acid levels did not significantly change (Table 5). Water intake and urinary volume significantly increased by 432 and 345 mL/day, respectively (Table 5). There was a positive correlation noted between changes in water intake and urinary volume.
Data are expressed as mean ± SD, mean ± SD (95% CI), and
Correlation between changes in 24-h urinary albumin and other parameters
The change in 24-h urinary albumin was significantly correlated with those in urinary transferrin and plasma renin activity (
Safety
Over the 12 weeks of canagliflozin treatment, there were no reports of serious AEs. There was one event involving upper respiratory infection in one subject and one event of muscle pain in another subject; both these patients recovered during the treatment period. In addition, there were no notable changes in laboratory test values associated with the safety of subjects.
Discussion
The development of albuminuria not only characterizes the progression of kidney injury 3,4 but it can also predict cardiovascular risk in patients with T2DM. 25 Therefore, the guidelines recommended the regular assessment of albuminuria and reducing albuminuria in patients with T2DM. 26,27 However, many patients with T2DM worldwide, including Japan, have substantial residual albuminuria and continue to progress toward ESKD. Thus, additional treatment complementing the existing therapies remains an important unmet medical need. The present pilot study showed that 12 weeks of treatment with canagliflozin significantly reduced 24-h urinary albumin excretion in Japanese patients with T2DM and microalbuminuria.
In the present study, canagliflozin reduced 24-h urinary albumin excretion by 42%. A similar magnitude of reduction in albuminuria was previously reported following canagliflozin treatment in Western patients with T2DM. 20 Furthermore, consistent with the results of previous non-Japanese studies, 28 –30 the changes in HbA1c, fasting plasma glucose, blood pressure, and body weight did not correlate with the albuminuria response during canagliflozin treatment. Thus, canagliflozin appears to reduce urinary albumin excretion in patients with T2DM without racial differences.
We also observed that the levels of urinary transferrin were reduced after 12 weeks of treatment with canagliflozin. Transferrin is a biomarker for glomerular damage. Furthermore, it is well known that urinary transferrin showed a high correlation with urinary albumin. 31 Taken together, the amelioration of damaged glomerular function in these patients is suggested. In addition to glomerular damage, recent studies have also shown the involvement of tubular damage during the pathogenesis of diabetic nephropathy. 32,33 However, the tubular damage markers evaluated in this study were all within the reference ranges both at baseline and at week 12. There was a small decrease in urinary L-FABP level, which reflected the mitigation of proximal tubular stress presumably via reducing the amount of work required following canagliflozin treatment. 34 –36 Even though this could increase albumin reabsorption, it is unlikely that increased tubular albumin reabsorption plays a major role in the reduced level of urinary albumin excretion.
In this study, eGFR was lower following 12 weeks of canagliflozin treatment. Previous investigations on SGLT2 inhibitors, including canagliflozin, putatively by activated tubuloglomerular feedback, 37 showed an acute decline in eGFR followed by a period of maintained kidney function. 20,29,38,39 A similar reduction in eGFR was reported with renin–angiotensin–aldosterone system inhibitors, 40 –42 and this would suggest that an intraglomerular hemodynamic effect, including the alleviation of glomerular hypertension, contributes to the reduction of albuminuria. 43,44 In addition, the activation of inflammatory pathways and oxidative stress has been shown in the kidneys of patients with diabetic nephropathy, 45,46 and is estimated to be a factor that initiates and sustains disease progression. However, the inflammatory biomarkers measured in this study were not suppressed and circulating TNFR2 levels slightly increased after canagliflozin treatment. Intriguingly, urinary levels of the oxidative stress marker 8-OHdG were reduced after treatment with canagliflozin. Moreover, hematocrit was elevated after treatment with canagliflozin, which is expected to deliver more oxygen to tissues to reduce ischemic stress. 34,35 However, we did not observe any association between changes in 24-h urinary albumin excretion and either of the abovementioned variables, including eGFR, 8-OHdG, and hematocrit. Although the change in plasma renin activity was significantly correlated with change in urinary albumin, we could not conclude that it was a real finding, because the correlation was driven almost exclusively by a single subject with a very large decrease in 24-h urinary albumin and no clear trend was observed in the rest of the subjects. As the present study cannot provide conclusive evidence for the precise mechanism involved, we must assume that multiple characteristics of the drug could be involved in reducing albuminuria.
There were two AEs, and no serious AEs, reported during the treatment period. In a previous study in patients with T2DM and stage 3 chronic kidney disease (eGFR: 30–59 mL/min/1.73 m2), genital mycotic infections and volume depletion-related AEs were frequently observed in the canagliflozin-treated group (number of patients/number of participants [event rate]: 51/703 [7.3%] and 48/703 [6.8%], respectively) than placebo group (6/382 [1.6%] and 10/382 [2.6%], respectively), 47 and some reports have warned that SGLT2 inhibitors could induce acute kidney injury. 48,49 However, no volume depletion- or renal-related AEs were reported in our present short-term study, and no novel safety concerns were raised.
There are some limitations to this study that should be taken into consideration. The study was performed in a single-site, single-arm, and open-label manner, sample size was relatively small, and the study follow-up period was only 12 weeks. The results generated by the present study, therefore, warrant randomized-controlled, multicenter trials with a sufficiently large number of participants and a longer treatment period for fully confirming the renoprotective effects and for delineating the mechanism of action. Definitive evidence of the renoprotective effects of canagliflozin is expected to be provided by the following ongoing studies:
Conclusions
The findings of this pilot study revealed that canagliflozin significantly decreases microalbuminuria after 12 weeks of treatment in Japanese patients with T2DM. The primary mechanism for the reduction of albuminuria was not elucidated but appears to be mediated by multiple mechanisms. Canagliflozin and other SGLT2 inhibitors may offer a novel therapeutic option for Japanese patients with T2DM and incipient nephropathy.
Footnotes
Acknowledgments
The authors thank Mr. Fujio Matsuyama (DOT World Co., Ltd.) for performing the statistical analysis, Mr. Toru Kumagai (DOT World Co., Ltd.) for providing medical writing support funded by Mitsubishi Tanabe Pharma Corporation, and Makoto Ueno, PhD (Pharmacovigilance Department, Mitsubishi Tanabe Pharma Corporation), for useful advice. This study was funded by Mitsubishi Tanabe Pharma Corporation. Canagliflozin (Japanese brand name: Canaglu) was developed, manufactured, and distributed by Mitsubishi Tanabe Pharma Corporation.
Authors' Contributions
T.O. contributed to the study design and conducted the study as the principal investigator. M.A. was the medical advisor for this study and contributed to the study design. M.G., M.K., K.A., and T.H. contributed to the study design. All authors contributed to the interpretation of data, writing, and reviewing the article and approved this article for submission.
Author Disclosure Statement
T.O. received consulting fees and/or speaker bureaus from Astellas Pharma, Inc., Mitsubishi Tanabe Pharma Corporation, Novo Nordisk Pharma Ltd., Ono Pharmaceutical Co., Ltd., Sanwa Kagaku Kenkyusho Co., Ltd., and Takeda Pharmaceutical Co., Ltd., and received research support from Astellas Pharma, Inc., Abbott Japan Co., Ltd., AbbVie GK, Bayer Yakuhin, Ltd., Daiichi Sankyo Co., Ltd., Eli Lilly Japan K.K., Japan Tobacco, Inc., Kowa Company, Ltd., Mitsubishi Tanabe Pharma Corporation, Nippon Boehringer Ingelheim Co., Ltd., Novo Nordisk Pharma Ltd., Sanofi K.K., Sanwa Kagaku Kenkyusho Co., Ltd., Taisho Pharmaceutical Co., Ltd., and Takeda Pharmaceutical Co., Ltd.
M.A. received consulting fees and/or speaker bureaus from Eli Lilly Japan K.K., Kyowa Hakko Kirin Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Otsuka Pharmaceutical Co., Ltd., and Nippon Boehringer Ingelheim Co., Ltd.; received scholarship grants from Astellas Pharma, Inc., Baxter Limited, Bayer Yakuhin, Ltd., Daiichi Sankyo Co., Ltd., Kyowa Hakko Kirin Co., Ltd., Mitsubishi Tanabe Pharma Corporation, NIPRO Corporation, Otsuka Pharmaceutical Co., Ltd., Pfizer Japan, Inc., Shionogi & Co., Ltd., and Torii Pharmaceutical Co., Ltd.; and chair courses endowed by Chugai Pharmaceutical Co., Ltd., Nikkiso Co., Ltd., NIPRO Corporation, Ono Pharmaceutical Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Terumo Corporation, and Toray Medical Co., Ltd.
M.G., M.K., K.A., and T.H. are employees of Mitsubishi Tanabe Pharma Corporation.
