Abstract
Hypoglycemia is the limiting factor in controlling glucose levels in Diabetes. Rather than being a side effect, hypoglycemia is the mechanism of action for insulin therapy, with a very narrow therapeutic window. Until recently, regulatory bodies listed hypoglycemia only as an adverse effect of therapy; however, one insulin preparation is now recognized and labelled as reducing the risk of severe hypoglycemia. This paper describes internationally agreed upon definitions for hypoglycemia and proposed regulatory approaches for recognition and labeling of diabetes therapies to facilitate personalized care.
S
The FDA recognizes severe hypoglycemia, as defined by the DCCT, as an adverse event in regulatory trials. As most trials exclude individuals at high risk of severe hypoglycemia, its occurrence in clinical trials is unusual. Despite its rarity in clinical trials, severe hypoglycemia has been increasing since the publication of the DCCT. Severe hypoglycemia was more than threefold higher in the intensively treated group of DCCT than in the conventionally treated participants. However, during the long-term observational phase of EDIC, where the HbA1c levels were similar between groups, severe hypoglycemia was no different between groups. 3 This has led many to believe that avoidance of severe hypoglycemia can be achieved by simply elevating the target HbA1c. This has not been borne out, however, as data show comparable prevalence of severe hypoglycemia across a broad HbA1c range in both adolescents and adults, 4 as well as in the elderly. 5 Instead, they demonstrate a strong relationship between duration of diabetes and hypoglycemic risk, with a progressive increased prevalence from <20 years to >40 years duration, regardless of age. 4 Given the decreased mortality in diabetes 6 and subsequent longer longevity, it is not surprising that the incidence of hypoglycemia hospitalization has significantly risen for both type 1 and 2 diabetes. 7 In fact, hypoglycemia now exceeds hyperglycemia as a cause for hospitalization in the United States. 8
The relationship between hypoglycemia and mortality remains controversial. In the advance trial, severe hypoglycemia was associated with a two- to fourfold increase in macrovascular events and mortality. 9 However, many have argued that hypoglycemia is a marker for frailty, as temporal causal relationships are difficult to demonstrate. Nevertheless, hypoglycemic clamp trials demonstrate acute QT interval prolongation, ST segment depression and T wave flattening in normal individuals exposed to hypoglycemia. 10 Individuals with type 2 diabetes and cardiovascular disease demonstrated a variety of arrhythmias in association with hypoglycemia, including bradycardia, atrial, and ventricular ectopy. 11
It is very apparent, however, that hypoglycemic events short of loss of consciousness or requiring the assistance of a third party have significant impact on patient and family quality of life. These are not uncommon events. Individuals with type 1 diabetes are at increased risk, with >60% experiencing daily to weekly episodes. 12 Although people with type 2 diabetes experience less hypoglycemia (25% with weekly episodes), by virtue of their significant increased number, more type 2 diabetes individuals suffer hypoglycemia than do type 1 diabetes individuals. There are now adequate data to support the fact that hypoglycemia with maintenance of consciousness significantly impacts morbidity and mortality in individuals with diabetes.
Approximately 20% of nonsevere hypoglycemia occurs during the night, with disruption of sleep and significant patient and family anxiety. 13 Two separate studies, utilizing different instruments, confirm a linear relationship between hypoglycemia severity and decrements in quality of life among subjects with type 2 diabetes. 14,15 Subjects report poorer adherence, more resource utilization, and diminished treatment satisfaction after hypoglycemia. 16 Fear of hypoglycemia has been recognized clinically 17 and impacts both patient and healthcare provider behaviors. 16
As a result, the European Association for the Study of Diabetes (EASD), together with the ADA, produced recommendations for classifying hypoglycemia in regulatory clinical trials. 18 (Table 1) They recognize the importance of a plasma glucose <70 mg/dL (Level 1) as an alert value to assist patients, their families, and their caregivers that there may be a need to adjust insulin dosing or provide rapid acting carbohydrate. A threshold value of <55 mg/dL independent of symptoms was established as level 2 hypoglycemia, representing a level at which cognitive function is usually impaired, repeated episodes may result in reduced awareness of hypoglycemia and predict severe episodes, and as a predictor of morbidity and mortality. To maintain the relevance and importance of severe hypoglycemia, the International Hypoglycemia Study Group maintained a level 3 hypoglycemia independent of measured glucose but requiring external assistance due to severe cognitive impairment. Until recently, the FDA had not included hypoglycemia as an outcome or as an advantage for any diabetes-related product. Hypoglycemia only appeared in the package insert as part of the adverse event profile for these drugs.
ADA, American Diabetes Association.
IHSG, International Hypoglycaemia Study Group.
The peer-reviewed literature, however, reveals several comparative trials that demonstrate a statistically and clinically significant difference in hypoglycemic events. Head-to-head comparisons of dipeptidyl peptidase-4 (DPP-4) inhibitors, glucagon-like peptide-1 (GLP-1) receptor agonists, and sodium-glucose transporter-2 (SGLT-2) inhibitors against sulfonylureas all demonstrate clinically meaningful reductions in hypoglycemic events. 19 –21 Comparisons of insulin analogues with human insulin have also demonstrated a reduction in events, but in small numbers. 22 The combination of a GLP-1 receptor agonist with basal insulin demonstrates a reduced risk of hypoglycemia, with equivalent HbA1c, compared with basal-bolus insulin therapy. 23 All of these studies excluded individuals at high risk for severe hypoglycemia, resulting in a very low event rate for this complication. Furthermore, the studies frequently used different criteria for the diagnosis of nonsevere hypoglycemia and a variety of ascertainment methods rendering it impossible to compare outcomes.
Recently, studies have been undertaken with prespecified outcomes related to hypoglycemia. As part of an FDA-mandated cardiovascular outcome trial, insulin degludec was compared with insulin glargine in a double-blind, treat-to-target design. With equivalent HbA1c levels, there was no difference in cardiovascular event rates, but a significant 40% reduction in adjudicated severe hypoglycemia with degludec. 24 (Fig. 1) A statistically significant 36% reduction in documented hypoglycemia <56 mg/dL was also shown during the night. Blinded, crossover studies of insulin glargine and insulin degludec in both type 1 and 2 subjects confirmed reductions in severe and/or documented hypoglycemia <56 mg/dL. In type 1 diabetes, an 11% reduction was seen in overall symptomatic hypoglycemia with degludec, with a 36% reduction in nocturnal events. 25 In patients with type 2 diabetes, the overall event rate was lower, but overall hypoglycemia was reduced by 30% with degludec and nocturnal events were reduced by 42%. 26
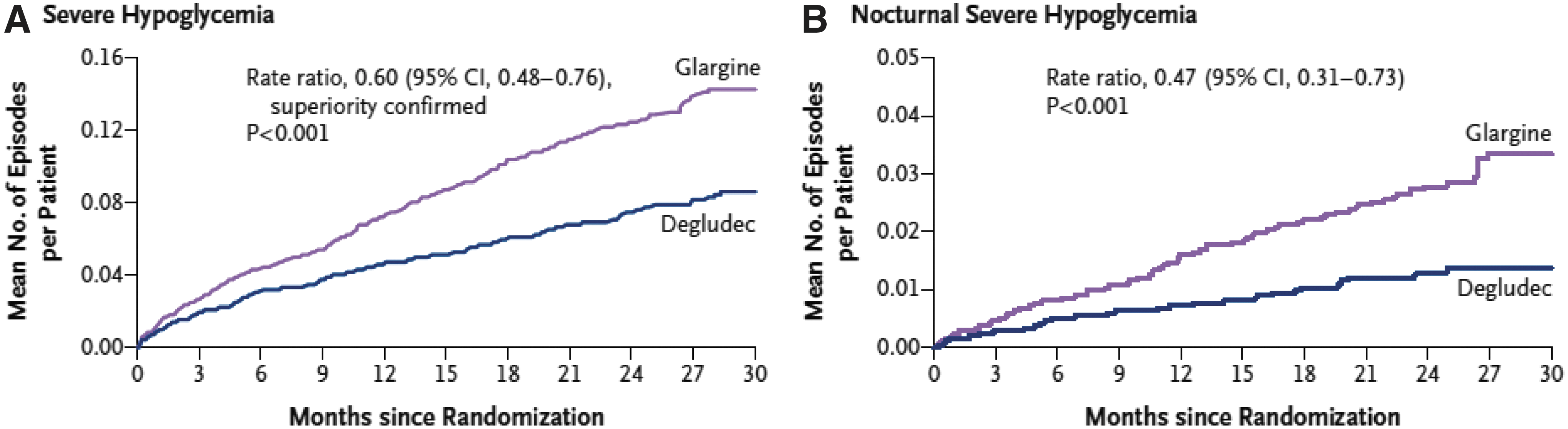
Cumulative events of severe hypoglycemia in a randomized, double-blind clinical trial of insulin degludec versus insulin glargine (The DEVOTE Trial). 24
The FDA following the lead of the European Medicines Agency (EMA) in March 2017 recognized the profound difference in severe hypoglycemia between insulin glargine and insulin degludec by including the data from the DEVOTE Trial in the degludec label. However, no mention in the package insert reflects the findings in type 1 diabetes or for documented hypoglycemia short of loss of consciousness or need for assistance.
Ascertainment of nonsevere hypoglycemia is poor in most clinical trials, relying predominantly on symptoms and/or documentation with self-monitored blood glucose (SMBG). Many subjects with longstanding diabetes and those with recurrent hypoglycemia may have blunted symptoms of hypoglycemia, resulting in decreased recognition. 27 The FDA has reviewed several continuous glucose monitoring (CGM) systems for clinical use recently with greater accuracy on the hypoglycemic end and approved them. There remains an internal inconsistency within the FDA, as the device branch fully supports the use of CGM data in the assessment of hybrid and closed loop devices, whereas the drug branch has yet to recommend CGM use in approval or labeling. Earlier studies with CGM suggested that as much as 63% of type 1 diabetes and 47% of type 2 diabetes subjects had asymptomatic episodes of hypoglycemia. 28 More recently, a presentation at the 77th Scientific Sessions of the ADA showed that individuals with type 1 diabetes had an overall rate of hypoglycemia of 5.8 events/patient-week corresponding to 10.1 h with hypoglycemia/patient-week. 29
Controlled clinical trials in type 1 diabetes demonstrated a significant reduction in time <70 mg/dL, as well as <50 mg/dL in those using CGM compared with the control SMBG. 30 This was substantiated with a 72% reduction in hypoglycemic events in a high-risk population with a history of severe hypoglycemia or hypoglycemia unawareness. 31 Real-world use of CGM in a prospective observational trial of type 1 diabetes showed an almost 75% reduction in hospitalization for hypoglycemia and a reduction of 68% in days of hospitalization per 100 patient-years. 32 Results in type 2 diabetes are less dramatic, due to the decreased frequency of hypoglycemia.
Nevertheless, an international consensus group recommends hypoglycemia in clinical trials be defined as the number of minutes or hours below the established threshold, with an event defined as at least 15 min below the threshold. 33 The ADA and EASD recommend utilization of CGM, together with patient-reported outcomes, as an inherent component of regulatory trials for drug approval. 34 This is consistent with the EMA guidance for use of CGM in clinical trials. Clinical determination of which therapeutic approach may maximize metabolic control while minimizing hypoglycemia requires comparative data using equivalent definitions, and methods of ascertainment that increase the sensitivity and specificity of hypoglycemia. As we identify patient populations at risk and therapies with documented reductions in hypoglycemia, we can truly move into the era of personalized diabetes management.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
