Abstract
Achievement of well-controlled blood glucose is essential for preventing complications in patients with type 1 diabetes. Since the inception of continuous subcutaneous insulin infusion, the aim has been to develop an artificial pancreas, with the ability to use an automated algorithm to deliver one or more hormones in response to blood glucose with the intent to keep blood sugar as close to a prespecified target as possible. Development and rapid improvement of continuous glucose sensor technology has recently allowed swift progress toward a fully closed-loop insulin delivery system. In 2017, Medtronic began marketing the 670G insulin pump with Guardian 3 sensor. When in auto mode, this is a hybrid closed-loop insulin delivery system that automatically adjusts basal insulin delivery every 5 min based on sensor glucose to maintain blood glucose levels as close to a specific target as possible. Patients receive prandial insulin by entering carbohydrate amount into the bolus calculator. Early studies show improvement in HbA1c in both adults and adolescents with this technology. Initial safety trials showed no occurrence of diabetic ketoacidosis or hypoglycemia. The utility of this device is limited by blood glucose targets of 120 and 150 mg/dL that are unacceptably high for some patients. Notwithstanding recent advances, we are far from a system that is able to replicate islet function in the form of a fully automated, multihormonal blood glucose control device.
Introduction
P
Our objective is to describe the history of continuous subcutaneous insulin infusion (CSII) and continuous glucose monitor (CGM) and how these components allowed the development of the first commercially available “artificial pancreas” (AP), although many would prefer the nomenclature of “closed-loop insulin delivery.” We then go on to describe practicalities of the initial hybrid closed-loop (HCL) insulin delivery system released by Medtronic.
Since the first use of CSII in the late 1970s, real-time CGM in the early 2000s, and the eventual sensor-augmented pump and “low-glucose suspend” after that, the obvious next step was further integration between the two for a closed-loop system, which ideally would require minimal interaction from the patient. The accuracy of the sensors has only recently become adequate to safely move this technology forward.
Devices designed to mimic pancreatic endocrine function have been under development since the 1970s. Initial systems 1,2 were plagued by mixed results and overly complex devices requiring intravenous insulin infusions. Tamborlane et al. 3 further refined the use of CSII by providing flexibility in the dosing of bolus insulin (independent of basal infusion rate). In 1983, MiniMed commercialized the first insulin pump, the 502. Soon after development of CSII, the vision has been for development of the “artificial pancreas.” 4
The development of CGM was the next major step in creating the AP. In 1999, the FDA approved the first physician-use CGM system. CGM technology progressed rapidly, with five devices on the market internationally by 2005. 5
In 2006, the Juvenile Diabetes Research Foundation (JDRF) launched the AP Project and has been funding development in this area since that time. Also in 2006, Medtronic (purchased MiniMed in 2001) introduced the 530G with integrated insulin pump and CGM. This is the system on which the AP system has been built. In 2013, the threshold suspend feature was approved by the FDA as part of the MiniMed 530G and Enlite sensor combination. This combined insulin pump-CGM was the first in the United States to allow the blood glucose readings on the sensor to control the function of the insulin infusion in the form of the low-glucose suspend feature. This CGM-CSII combination was promoted as a “first-generation artificial pancreas device.” 6
Before the recent pivotal trial 7 evaluating safety of the Medtronic HCL system, several groups have demonstrated feasibility, safety, and effectiveness of closed-loop technology. In 2008, Weinzimer et al. 8 showed for the first time that HCL insulin delivery could improve glucose control at night. This group used a device that was the precursor to the Medtronic 670G. Time in range (TIR, 70–180 mg/dL) improved significantly from 58% to 85% (P < 0.002) in the open-loop versus closed-loop groups. In 2010, Kovatchev et al. 9 published results of a multicenter, international study of open- versus closed-loop insulin delivery with the use of OmniPod Insulin Management System (Insulet Corp.) and FreeStyle Navigator CGM (Abbott Diabetes Care). TIR improved (64% vs. 78%, P = 0.029) and hypoglycemia decreased (23 vs. 5, P = 0.01) with closed-loop insulin delivery. In 2013, Phillip et al. 10 showed that use of an AP system reduced hypoglycemia in pediatric patients. In 2014, Kovatchev et al. published results 11 of hypoglycemia risk in open-loop versus closed-loop insulin delivery and demonstrated significantly less hypoglycemia in adult patients using the closed-loop system. In 2014, Russell et al. released data 12 evaluating the safety and effectiveness of a bihormonal AP and showed improved mean glucose with less hypoglycemia in adults and adolescents. In 2015, Thabit et al. 13 did a prolonged (12-week) study of a closed-loop system in adults and children, compared with sensor-augmented pump, and showed improved glucose control, decreased hypoglycemia, and a decreased HbA1c in the adults.
As more groups have demonstrated safety and improved glycemic control with closed-loop systems, device development has been rapid, with several companies designing promising new technologies and/or infusion algorithms. Notably, Buckingham et al. recently published data
14
on the safety and feasibility of an HCL system with OmniPod, DexCom G4 CGM, and a model predictive control (MPC) algorithm. The tslim insulin pump (Tandem Diabetes Care, Inc.) was previously shown to improve TIR with the Diabetes Assistant USS algorithm and DexCom CGM by Ly et al.
15
Currently, Tandem and Type Zero are collaborating on a trial (
Regarding the current commercially available HCL by Medtronic, Bergenstal et al. 7 released a Research Letter summarizing results of the pivotal trial, in October 2016, evaluating safety of the HCL insulin delivery system. One hundred twenty-four patients were studied on the HCL system for 3 months after a 2-week run-in period. There was no control group, and thus, no claims regarding effectiveness can be made. There were few serious or device-related adverse events. Garg et al. 17 expanded on these data for adults. Sensor glucose average between the run-in period (146.1 mg/dL) and the study period (148.3 mg/dL) was borderline significant (P = 0.04203). Hypoglycemia was reduced below 50 mg/dL (<0.001) and below 70 mg/dL (P < 0.001). Other notable findings for adults in this trial were an increase in weight (79.9 kg vs. 81.3 kg, P < 0.001), an increase in total daily insulin (44.9 U vs. 47.9 U, P < 0.001), and a decrease in the within-day coefficient of variation of glucose (33.1% vs. 30.3%, P < 0.001). Although HbA1c decreased, this trial was not designed to evaluate effectiveness because of lack of a control arm. Based on the established safety data, the FDA approved the Medtronic 670G automated insulin delivery system in the United States. In 2017, Medtronic began to market this system.
It is widely agreed that success of a closed-loop system is based equally on the quality and efficacy of the infusion algorithm and accuracy of the sensor. CGM quality, as measured with mean absolute relative difference (MARD), has evolved since initial release of glucose sensors. The MARD is the difference between laboratory (or occasionally fingerstick) glucose and sensor glucose. The lower the MARD, the more accurate the sensor. Medtronic has released several sensors over the years: the Guardian Real-Time, the Sof-Sensor, the Enlite Sensor, and the Guardian 3 sensor (known as the Enlite 3 in Europe and Asia), which is currently part of the 670G HCL system.
MARD data reported are highly variable and differ where the study is conducted, especially when comparing the clinical research unit versus ambulatory setting. 18 The reported values also may be influenced by the reporter and funder of the study. For example, Calhoun et al. reported the MARDs for the Medtronic Sof-Sensor and the Enlite to be 16% and 18%, respectively. 19 However, in 2012, a group from Medtronic wrote a letter to the editor that reported the MARD for the Sof-Sensor was 9.9%. 20 Similarly, a Medtronic-funded study published in 2014 reported the MARD for the Enlite sensor to be 13.6%. 21 This is the figure that is quoted in the patient and provider information for the Enlite sensor. In 2017, another group reported the MARD for Enlite sensor to be 19%. 22 The new Guardian 3 sensor, which is used in the new HCL insulin delivery system, is less well studied. In 2017, Christiansen 23 evaluated the accuracy and performance of the Guardian 3 CGM and reported an MARD of 10.3% when calibrating every 12 h and 9.6% when calibrating 3 to 4 times per day. The MARD reported in promotional materials by Medtronic is 9.64% for the new Guardian 3 sensor.
670G Pump Basics
The function of the “artificial pancreas” in its current state is threefold: CGM monitoring, insulin delivery, and hormone delivery control. The final component, considered the brain of the system, is evolving. As described above, its first form came as the threshold suspend feature. The 670G provides more sophisticated automation. When in “auto mode,” the Medtronic 670G automatically adjusts basal insulin delivery every 5 min based on sensor glucose to maintain blood glucose levels at a specific target. In addition, the suspend before low function stops insulin delivery before hypoglycemia is predicted to occur, and restarts insulin once blood glucose levels normalize.
The 670G insulin pump functions in two different modes: auto mode and manual mode. Manual mode is similar to previous CSII with linked CGM, with the added feature of stopping insulin before hypoglycemia occurs. Basal rates are adjusted in manual mode just as they are in other insulin pumps: with a provider-set basal rate during a specific time block. Both modes require manual bolus administration by entering carbohydrate information into the insulin pump, from which the bolus calculator determines a recommended bolus.
The insulin pump becomes quite different when in “auto mode.” While in this mode, the technology uses proportional integral derivative (PID) algorithms that respond to measured glucose levels. 24 With this algorithm, small adjustments are made in the basal insulin rate. The PID algorithm is just one of several control algorithms that have been used in automated insulin delivery. Other algorithms that have previously been used are MPC, described by Hovorka et al. in 2004, 25 and MD-Logic. 26 The basal insulin can be thought of more as microboluses infused every 5 min. It is possible, and typical, for auto mode users to exit to manual mode. Automatic push to manual mode occurs for a variety of reasons, such as persistent hyperglycemia, sensor problems, and user problems (e.g., failure to calibrate).
Because of automatic adjustment of the basal insulin delivery based on sensor glucose, there are fewer modifiable parameters when in auto mode. The only settings that can be changed are active insulin time (AIT), insulin to carbohydrate ratio (ICR), and basal target glucose. There are two prespecified, nonmodifiable blood glucose targets for basal insulin: 120 and 150 mg/dL. Typically, patients are advised to use the 120 mg/dL target. During periods of exercise, it is recommended to use the 150 mg/dL target. Mealtime insulin is given as calculated by the bolus calculator after the user enters the amount of carbohydrates to be consumed. It is not possible to give a manual bolus without entering carbohydrates consumed while in auto mode. For hyperglycemia, the insulin pump will suggest a correction based on a blood glucose target of 150 mg/dL with an insulin sensitivity factor that is calculated by the algorithm every 24 h. Although the AIT can be changed while in auto mode, the modification does not feedback to the algorithm. In other words, a change in AIT from 3 to 4 h will not lead to less insulin delivered while in auto mode. The AIT remains necessary in manual mode, where it does impact the amount of insulin given through the bolus calculator.
Protocol for patient initiation onto HCL varies among clinics. At our academic center, we have found it most effective to standardize the process. Appropriate training, even for experienced CSII users, can be time-intensive, which is why we now accept assistance from the Medtronic trainers. Our preference was to do the training independently, but the intensity of the training for our large number of patients makes this impossible. All patients who receive the 670G insulin pump are transitioned from the previous pump to the 670G (in manual mode) with settings from the previous pump. The new insulin pump needs a minimum of 48 h of data to function in auto mode. Ideal amount of time is unknown, but we have found 48 h to be inadequate. In our practice, we target 1 week in manual mode before entering auto mode. Before entering auto mode, it is required that patients upload their data to the CareLink® personal account and allow either our certified diabetes educator or Medtronic representative to review the data. In our clinic, patients who are ready to enter into auto mode are those who are blousing through the bolus calculator, calibrating the sensor 2.5 times per day, and measuring fingerstick glucose levels four times per day.
Once readiness has been demonstrated, the patients participate in a short tutorial about auto mode. Before entering auto mode, adjustments are made to the insulin pump settings. Typically, basal insulin is decreased by about 10% (for when patients enter manual mode), ICR is strengthened (by about 10%), and the AIT is set between 3.5 to 4.0 h.
Anecdotally, most patients require significantly less basal insulin in auto mode than they receive while in manual mode. There may be many reasons for this observation, but we find most patients when doing well only require about 40% of their insulin as basal insulin, not the typical 50% historically recommended. The higher carbohydrate ratios indicate that many patients in manual mode (or even on multiple injections) receive too much basal insulin, which helps to mitigate the postprandial spikes. Patients in auto mode, now on a more appropriate basal dose, need to appreciate they may spike higher without the excess basal insulin, and thus waiting a longer time between the bolus and eating can be useful.
Along with the new pump, algorithm, sensor, and transmitter, the CareLink reports that give details about pump and sensor data have changed. CareLink is a cloud-based program that was created by Medtronic, is available to both patients and providers, and gathers information from the Medtronic insulin pumps and CGMs. Reports can be generated for and by patients and providers. The CareLink reports on the new platform have changed significantly compared with previous versions.
Before discussing the new CareLink reports from the HCL, it is prudent to describe the features from older reports that included the following sections: Adherence, Sensor and Meter Overview, Logbook, Device Settings Snapshot, and Daily Detail (Fig. 1). The Daily Detail section shows blood glucose average, carbohydrates consumed, and daily insulin. The format in which these data are provided allows the provider to view the patient's response to insulin delivery for a meal and/or a correction. Access to these data allows the provider to assess the appropriateness of settings for bolus and correction.

Meal bolus information from insulin pumps 530G and 670G.
The sections on the updated CareLink reports are as follows: Meal Bolus Wizard (Fig. 1), Assessment and Progress (Fig. 2), Weekly Review, Logbook, and Device Settings.
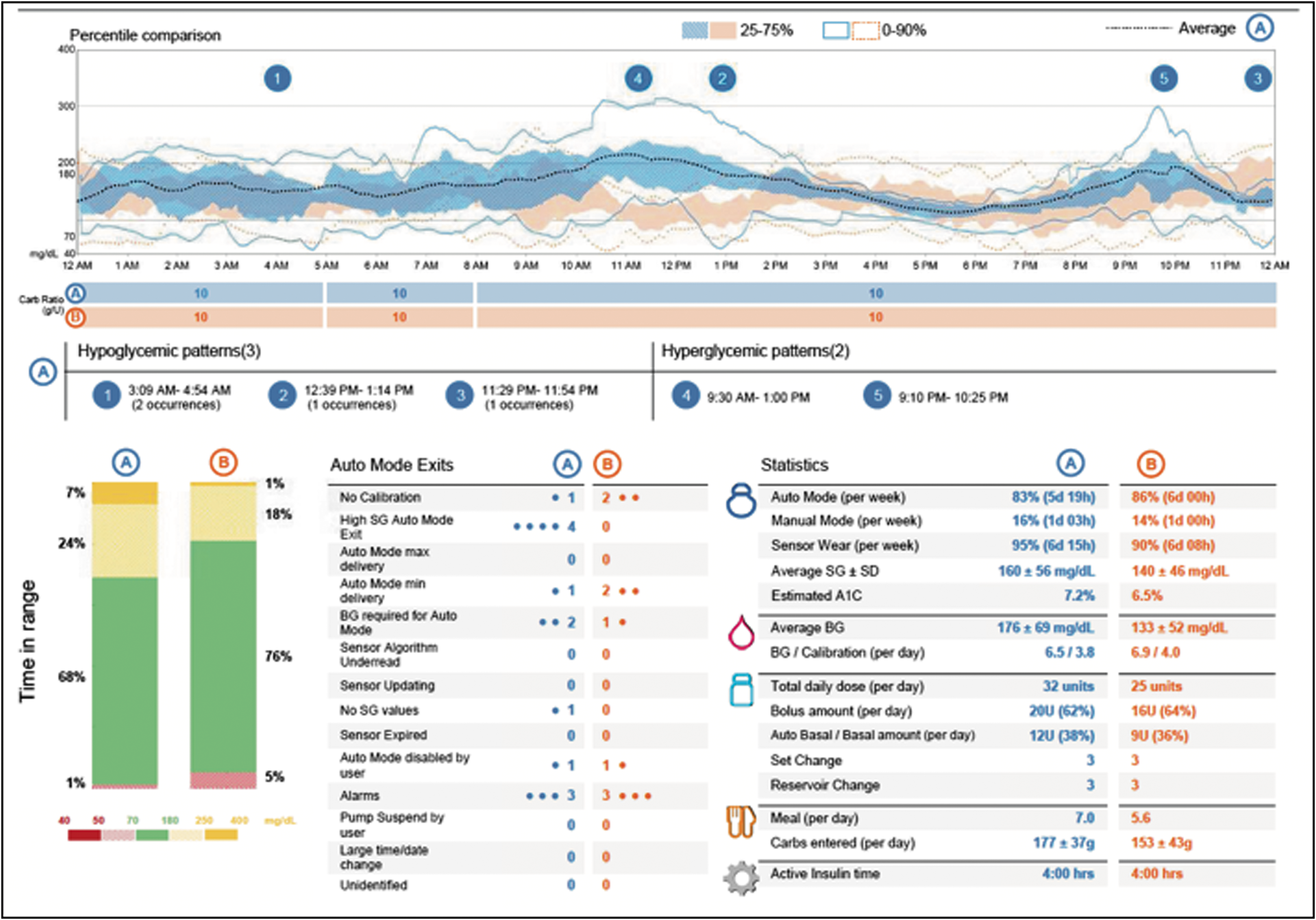
Assessment and Progress portion of CareLink reports from HCL system. Information includes AGP, TIR, data on auto mode exits, and statistics (Fig. 3). AGP, ambulatory glucose profile; TIR, time in range.
The Meal Bolus Wizard portion provides information on meal boluses timed by the meal. Similar to the Daily Details described above, details are provided on average carbohydrates consumed and average bolus amount. The Meal Bolus Wizard data are unfortunately aggregated, meaning individual boluses are not reported, which is a departure from earlier versions of the reports. The removal of this valuable tool has thwarted the provider's ability to evaluate in an effective manner blood glucose response to boluses in manual mode.
The Assessment and Progress page is arguably the most useful. It provides a high-level view of blood glucose control with the aggregate sensor data. It contains an ambulatory glucose profile (AGP), similar to those provided by DexCom and FreeStyle Libre (Abbott Diabetes Care) CGMs. Compared with older CareLink report versions that provided a 24-h overlay of CGM data, the updated reports present sensor data as a percentile comparison with three curves: average glucose, 25% to 75% percentile blood glucose, and 0% to 90% blood glucose. TIR is included, showing a snapshot of blood glucose control and frequency of hypoglycemia. Also included in this section is the statistics (Fig. 3), which provides the time the patient was in auto mode, the sensor wear, sensor glucose, total daily insulin, bolus insulin amount, and carbohydrates entered. One-month data are collected and compared in 2-week blocks.
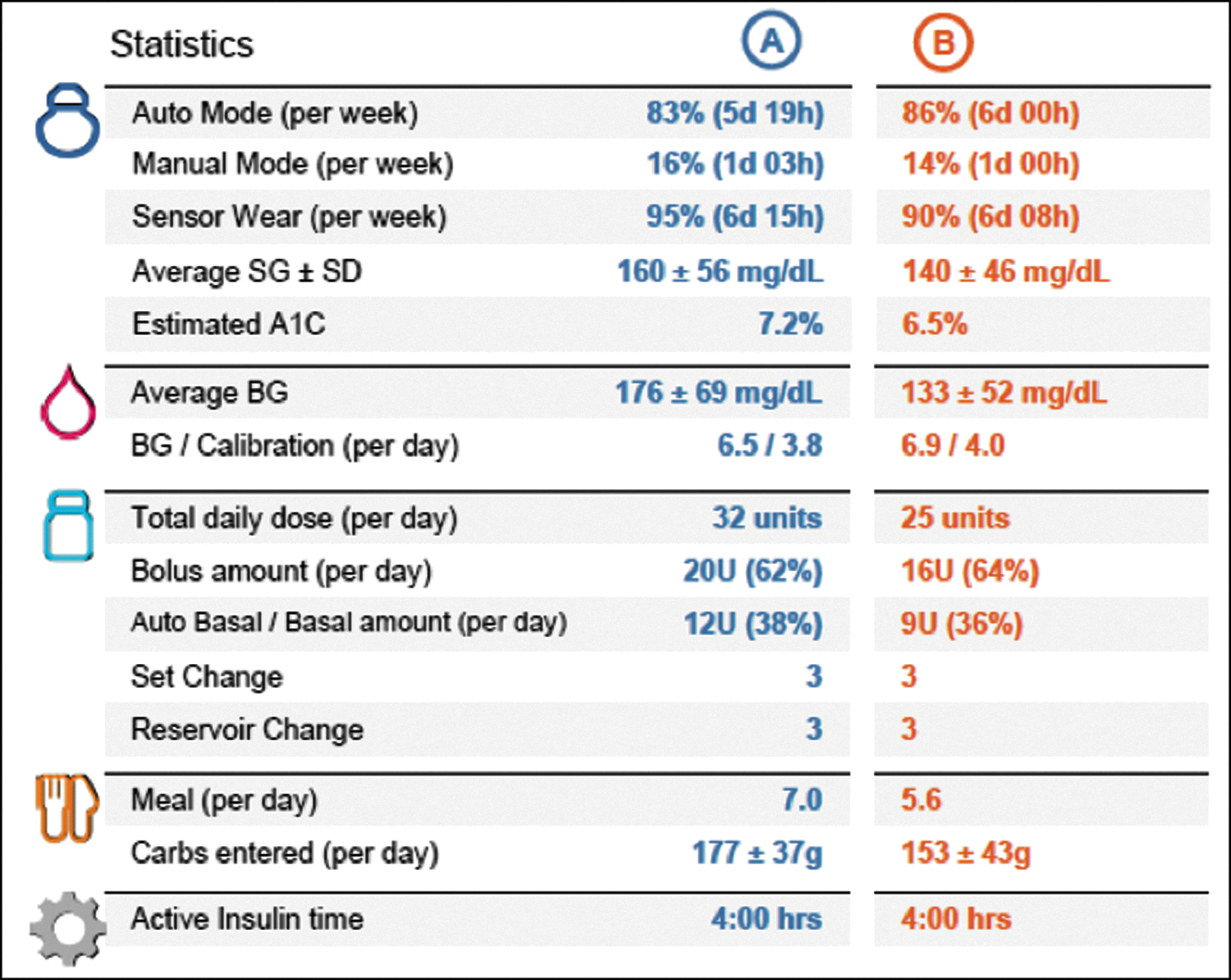
Statistics portion, found in the Assessment and Progress section of updated CareLink reports, provides information about time in auto mode, sensor wear, average glucose, insulin use, and carbohydrates consumed. “A” and “B” represent 2-week blocks within the last month with “A” being the most recent 2 weeks.
The Adherence section from older CareLink reports is eliminated in the new reports. When in auto mode, the details are admittedly obsolete. However, for patients who use the pump primarily in manual mode, the Adherence portion could be useful. The Adherence section provides data on bolus events, specifically about the number of manual boluses, bolus calculator events, and overrides (when patient gives more or less insulin than recommended by bolus calculator).
The use of the downloaded data in care of patients with diabetes mellitus using insulin is essential. It becomes even more important when these people are using CSII and CGM. However, the use of downloaded data is not done uniformly in clinics. Extracting data from the devices (meters, pumps, and sensors) can be costly and time-intensive. As multiple different pump and sensor platforms are developed, the ability to quickly download and interpret data will become more difficult. Innovation is ongoing in this area, with private groups working to develop platforms that standardize the data.
It is possible, and expected, for patients to upload data to CareLink. By uploading data to the cloud, information can be shared with device representatives, providers, and nurses outside of the visit. It is unreasonable to assume, however, that patients will reliably upload data to the cloud on a regular basis, 27 still leaving the burden of data gathering on the clinician and the team during the office visit.
The Reality Check
Despite the advances in technology, the current artificial insulin delivery system requires significant patient and provider involvement in addition to high-resource utilization on initialization. The novelty of this system demands ongoing involvement from device representatives to help users and providers learn the new system. We have undoubtedly increased utilization of the Medtronic representatives in our practice. With this increased demand on the manufacturer for patient care and education, our concern is that total costs increase.
In addition to use of company representatives, hospital and clinic diabetes educators and nurses are spending more time interfacing with patients and product agents. Data must be downloaded to provide optimal patient care, an additional point of staff involvement and resource utilization. Nonreimbursed time from our nonphysicians before the 670G was ∼$100,000 per year in our clinic. 28 The physician involvement of learning the technology, interpreting the download, and troubleshooting questions is impactful and of questionable sustainability in our productivity-centered system.
However, as our experience has increased, the learning curve has flattened, and patients and providers are more comfortable with this new technology. We are seeing that a subset of patients demands less from the healthcare system. It is our hope that this trend continues, especially as the development of a fully closed-loop system progresses.
The HCL insulin pump will of course not work on all types of patients. The preset blood glucose target of 120 mg/dL is simply too high for many patients who prefer (and can achieve) much tighter control. In addition, pregnant women (or women planning pregnancy) cannot be in auto-mode, as the factory-set blood sugar target is too high to achieve necessary goals for pregnancy. Another obvious limitation in the usefulness of auto mode are those patients on high-dose steroids, especially during tapers, as insulin resistance changes daily and it is unclear if the algorithm would be able to adapt. Patients who are very ill and/or have dynamic health challenges (cancer, infection, etc.) should wait until these health problems are stable before starting a new diabetes technology. Finally, people whose diabetes is not well controlled have major depression or other psychiatric disease, are not adherent, or who have trouble coming to clinic visits should not be started on the HCL system due to the initial high involvement for the patient.
The number of fingersticks required per day for calibration is four. As other continuous glucose sensors completely move away from requirement or capacity for calibration, the need to check blood sugar four times per day is burdensome for many. Many patients in our practice have become frustrated with the increased workload with the Medtronic 670G insulin pump, as the perception of and goal for HCL insulin delivery are to decrease patient responsibility.
Despite the terminology of “auto mode” and the nonmodifiable blood glucose targets of 120 and 150 mg/dL, people have found ways to consistently decrease blood glucose levels below the target. This is achieved by the administration of “phantom carbohydrates,” where the users inform the insulin pump that they are going to eat carbohydrates, when they are not going to eat anything. We have affectionately termed this “fake carbs” in our practice. Phantom carbohydrates can lead to hypoglycemia. It is in the patients using this strategy who are those with hypoglycemia on the HCL system. Behaviors such as this are potentially dangerous and highlight that this HCL system is not for all patients, especially those who find the blood sugar target of 120 mg/dL too high.
Conclusion
The development of the “artificial pancreas” is underway. Despite recent advances, we are far from a system that is able to replicate islet function in the form of a fully automated, multihormonal blood glucose control device. The current HCL system by Medtronic relies on a PID algorithm to make constant small adjustments in basal insulin delivery to target a blood glucose of 120 mg/dL. Patients must continue to use the bolus calculator by entering a carbohydrate amount to deliver bolus insulin.
Limitations of this device include the limited number of blood glucose targets: 120 and 150 mg/dL, making it an unacceptable device for many patients. Specifically, the use of 150 mg/dL as a target blood glucose during exercise is too low based on our early experience. We typically advise our patients to enter manual mode 2 to 3 h before exercise or consume small carbohydrate meals regularly during exercise to avoid hypoglycemia. The modified reports from the HCL system now use the TIR and AGP as primary data points to assess blood glucose control. The modified CareLink reports have eliminated some very useful data. Finally, patients need to be willing to measure blood glucose more frequently than previously when using CGM, often four times per day. Currently, much of our practice refinement is through trial and error. It will take long-term observations and clinical trials to determine how robust this technology is.
The increase in TIR and decreased amount of hypoglycemia, especially in the fasting state, observed in patients in our practice have been notable and important improvements while using this new system. Data from the initial safety trial
7
showed no development of severe hypoglycemia or diabetic ketoacidosis. HbA1c was also shown to decrease in this trial, although this was limited by lack of a control group. Anecdotally, we have found this device most helpful in patients who struggle to keep their HbA1c below 8%. Further studies are underway (
The first HCL is an important advance in our ability to control blood glucose in type 1 diabetes. Like past developments, this likely will be seen as an incremental improvement from our previous diabetes treatments, but more importantly it is a bridge to a truly automated closed loop. Both clinicians and users need to understand how this technology may ultimately impact patient quality of life.
Footnotes
Author Disclosure Statement
K.W.W.: No competing financial interests exist; I.B.H.: Research funding from Medtronic Diabetes; Consulting: Abbott Diabetes Care, Adocia, Bigfoot, and Roche.
