Abstract
The sodium–glucose cotransporter type 1 (SGLT1) is the primary transporter for absorption of glucose and galactose in the gastrointestinal tract. Inhibition blunts and delays postprandial glucose (PPG) excursion. Sodium–glucose cotransporter type 2 (SGLT2) is expressed in the kidney, where it reabsorbs 90% of filtered glucose. Thus, a dual SGLT1 and SGLT2 inhibition (compared with selective SGLT2 inhibition) could result in lower PPG and robust A1c reduction even in patients with reduced kidney function. Sotagliflozin is an oral potent dual inhibitor of the insulin-independent SGLT1 and SGLT2. Preliminary data released from phase 2 and 3 clinical studies in adults with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) showed improved glycemic control, and met efficacy endpoints beyond A1c with a safety profile consistent with the SGLT class: significant reduction in body weight, systolic blood pressure, and efficacy maintained in lower estimated glomerular filtration rate levels with no increased hypoglycemia. Increased risk of diabetic ketoacidosis (DKA) with uncharacteristically mild-to-moderate glucose elevations (euglycemic DKA) is associated with the use of all the approved SGLT2 inhibitors. Factors that trigger DKA include insulin reductions, low caloric and fluid intake, intercurrent illness, and alcohol use. However, DKA is detectable and manageable with proper patient education. With sotagliflozin, DKA rates were not higher than the expected background rate in T1DM, but numerically higher than placebo. Sotagliflozin is the first oral SGLT1 and SGLT2 inhibitor developed for the treatment of adult patients with T1DM, in adjunct with insulin, and has the potential to address unmet needs for patients with T1DM and possibly T2DM, with a favorable benefit/risk profile.
Introduction
T
It was demonstrated nearly four decades ago that in patients with poorly controlled T1DM, the maximum tubular reabsorption capacity for glucose was significantly increased although one would have suspected that during hyperglycemia, with increased interstitial and intracellular glucose concentrations, the reduced glucose concentration gradient across the basolateral membrane of the proximal renal epithelial cell would attenuate glucose efflux. The paradoxically increased glucose reabsorption in T1DM could be explained by an upregulation of glucose transporters, which was shown in tubular cells cultured from patients with T2DM.
Several SGTL2 inhibitors are approved for the treatment of patients with T2DM and are now under investigation in people with T1DM as well. In type 1, agents from these groups showed benefits in A1c after regular use as well as in weight, blood pressure, and hypoglycemia. 8 In the younger population, dapagliflozin as one substance of this group appears to be well tolerated with the same pharmacokinetic and pharmacodynamic profile in pediatric patients with type 1 diabetes. 9 Thus, this class of drugs represents a possible approach for combination therapy in a wide range of diabetes patients. These benefits need to be balanced against possible side effects of this therapy as diabetic ketoacidosis (DKA), urinary tract infections (UTIs), and genital mycoses are more common. This review focuses on the novel approach of dual SGLT1 and SGLT2 inhibition as potential adjunctive treatment for T1DM.
Unmet Needs in Type 1 Diabetes
Insulin therapy is the mainstay of treatment for T1DM. The goal of insulin therapy is to replace the deficient hormone and to attain normoglycemia. However, this goal remains elusive because of the difficulty in replicating the minute-to-minute variations of physiologic insulin secretion and the difference in delivery of exogenous insulin action compared with normal secretion of endogenous insulin directly into the portal vein. The acute and chronic complications of diabetes are attributable to the failure of exogenous insulin to completely mimic physiologic insulin secretion. DKA and severe hypoglycemia remain two of the most common serious complications for patients with T1DM. 10 Intensified insulin regimens are associated with weight gain during T1DM treatment, especially in females. 11 Taken together, treatment goals and patient-related outcomes beyond A1c such as time in range assessed by continuous glucose monitoring, hypoglycemia, emotional well-being, and quality of life are not reached in nearly half of the patients with T1DM, particularly adolescents, and it is unlikely that technological advances such as pumps or even closed-loop approaches will be able to solve the issues in the near future 12,13 as postprandial regulation remains a challenge particularly in type 1 diabetes.
Sotagliflozin: Mechanism of Action
The SGLT1 is the main uptake mechanism for glucose and galactose in the gut. It is responsible for 10% of renal glucose reabsorption in segment S3 of the proximal renal tubule. The SGLT1 is a high-affinity (Km = ∼0.5 mM), low-capacity transporter that incorporates Na+ and glucose into the cell in a 2:1 ratio. A more important role in diabetes is played by the SGLT2. The SGLT2 is expressed almost completely in the brush border membrane of the renal proximal tubular cells in segments S1 and S2 and accounts for 90% of total renal glucose absorption. It is a low-affinity (Km = ∼2 mM), high-capacity transporter that incorporates Na+ and glucose into the cell at a 1:1 ratio. 14 Sotagliflozin (LX4211) is a novel, orally delivered, small-molecule dual inhibitor of SGLT1 and SGLT2. 15 Inhibition of SGLT1 delays and reduces glucose absorption in the proximal intestine, improving postprandial glycemic control. 16 Inhibition of SGLT2 reduces renal glucose reabsorption. 17 Sotagliflozin-mediated SGLT1 inhibition in the intestine is local and not systemic. Sotagliflozin is extensively plasma protein bound (>93%), resulting in plasma-free concentration of <6 ng/mL, below the human SGLT1 IC50 of 15.4 ng/mL (i.e., 36.3 nM). Taken together, sotagliflozin at doses up to 400 mg is unlikely to exhibit clinically significant inhibition of systemic SGLT1.
Rationale for Dual SGLT1 and SGLT2 Inhibitions
Sotagliflozin has the potential to significantly alter dietary glucose load in patients with T1DM by both reducing glucose absorption in the intestine (through its SGLT1 inhibition) and enhancing glucose excretion in the urine (although its SGLT2 inhibition) (Fig. 1). Because SGLT1 and SGLT2 inhibition is independent of insulin activity, sotagliflozin provides a potentially unique, insulin-independent, therapeutic activity. The anticipated therapeutic effects of SGLT1 inhibition with a delay and reduction of glucose absorption in the proximal intestine would be improving postprandial glycemic control. 18 This should result in clinical benefits of lower and less variable mealtime bolus insulin requirements, lower mealtime bolus requirements resulting in lower postprandial insulin levels, which should translate to lower risk of postprandial hypoglycemia. 19 In addition, lower postprandial glucose (PPG) led to longer time in range, fewer unexpected blood glucose numbers, and improved A1c, while lower peak PPG should translate to less need for “correction bolus” to treat elevated PPG. Furthermore, reduced glucose absorption in the proximal intestine results in more glucose delivery to the distal intestine leading to bacterial fermentation, which produces short-chain fatty acids that cause vasodilation, resulting in decreased vascular resistance and reduction of systolic blood pressure (SBP). 20 Moreover, release of incretins such as glucagon-like peptide 1 (GLP-1) and peptide YY (PYY) by L-cells 21 may lead to weight loss by increasing satiety. 22 –24 PPG is the predominant contributor to A1c in patients with satisfactory to good control of diabetes. In a review, 25 the relative contribution of PPG was high (70%) in patients with good glycemic control (A1c <7.3%) and decreased progressively (30%) in patients with poor glycemic control (A1c >10.2%). Therefore, the sotagliflozin-mediated PPG reduction is expected to have the greatest effect on A1c in those with good glycemic control. In contrast, SGLT2 inhibition results in glucose reduction independent of meals, with fasting plasma glucose (FPG) as the best example. In the same review, 25 the authors found that the contribution of FPG showed a gradual increase with increasing A1c. Therefore, the sotagliflozin-mediated FPG reduction is expected to have the greatest effect on A1c in those with poor glycemic control. Evidence that the SGTL1 inhibition contributes substantially to the attenuated PPG rise comes from research in patients with T2DM with reduced Glomerular Filtration Rate (GFR) when no drug-related increase in urinary glucose excretion (UGE) is possible. The PPG effects of SGLT1 and SGLT12 inhibition with sotagliflozin were maintained in patients with an estimated glomerular filtration rate (eGFR) <45 mL/min/1.73 m2 despite the expected reduction in UGE, suggesting that dual SGLT1 and SGLT2 inhibition with sotagliflozin could prove useful for the treatment of patients with diabetes beyond what can be achieved with SGLT2 inhibition alone. 3
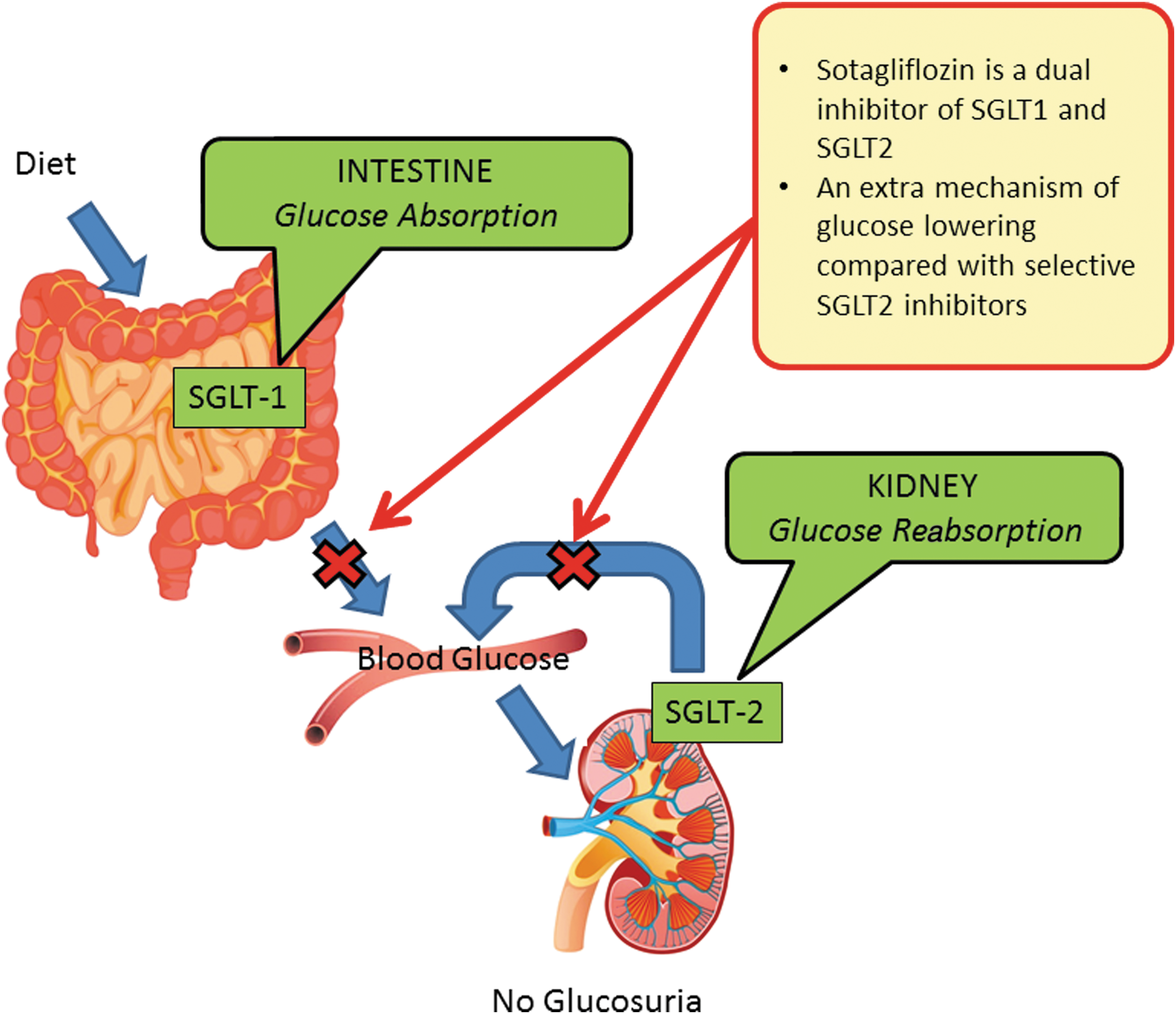
Dual SGLT1 and SGLT2 inhibitions with SOTAGLIFLOZIN (compared with selective SGLT2 inhibition) should result in lower postprandial glucose, and robust HbA1c reduction achieved with less renal glucose excretion, that is, maintained with reduced kidney function. SGLT1, sodium–glucose cotransporter type 1; SGLT2, sodium–glucose cotransporter type 2.
Potential Risks of Dual SGLT1 and SGLT2 Inhibitions
SGLT2 inhibition is associated with an increased risk for ketosis and DKA. 26 Possible contributors to this increased risk of ketosis and DKA include a reduction in insulin doses, sometimes required in patients when SGLT2 inhibitors are combined with insulin to avoid hypoglycemia. Lower insulin doses may be inadequate to suppress lipolysis and ketogenesis. Moreover, an inhibition of SGLT2 expressed in pancreatic alpha-cells promotes glucagon secretion, which promotes lipolysis and ketogenesis. In addition, inhibition of SGLT2 decreases renal clearance of ketone bodies, thereby increasing plasma ketone body levels. 27 The dual mechanism of action of sotagliflozin based on SGLT1 and SGLT2 inhibition may attenuate the increased risk of ketosis relative to selective SGLT2 inhibitors based on the following observed in preclinical and clinical trials. A reduced glucose absorption in the proximal intestine causes more glucose delivery to the distal intestine where L-cells release GLP-1 and PYY. In a preclinical model of T1DM, SGLT1 inhibition was associated with higher GLP-1 levels, reduced glucagon levels, and reduced ketogenesis. 28 In a Phase 2 proof-of-concept study of sotagliflozin patients with T1DM, compared with similar studies of selective SGLT2 inhibitors, sotagliflozin was associated with more modest reduction in basal insulin, 29 which may lead to a decreased risk of lipolysis, ketosis, and/or DKA. Also, in patients with T2DM receiving sotagliflozin, no statistically significant increases in glucagon were observed. 3
Clinical Data with Sotagliflozin
Canagliflozin, dapagliflozin, empagliflozin, and sotagliflozin are being studied in a clinical program of Phase 1, 2, and 3 studies (Table 1). So far, no head-to-head studies between any of the compounds are available. Garg et al. recently reported the results of the Phase 3, multicenter inTandem3 placebo-controlled trial in 1402 poorly controlled patients with type 1 diabetes, who were treated with sotagliflozin (400 mg/day) or placebo for 24 weeks. 6 The primary endpoint was a composite of A1c <7% at week 24, with no episodes of severe hypoglycemia or DKA. After 24 weeks, 28.6% of the sotagliflozin group achieved a glycated hemoglobin level <7% versus 15.2% of the placebo group (P < 0.001). Beyond the improvement of A1c, patients in the sotagliflozin group had significantly greater changes from baseline in glycated hemoglobin, weight, SBP, and mean daily bolus dose of insulin, compared with the placebo group. The rate of severe hypoglycemia was similar between the two groups (3.0% [21 patients] and 2.4% [17 patients] respectively), while the rate of documented hypoglycemia with glucose ≤55 mg/dL was significantly lower in the sotagliflozin group than in the placebo group. The rate of DKA was 3.0% in the sotagliflozin group versus 0.6% in the placebo group. The addition of sotagliflozin to insulin therapy in patients with type 1 diabetes significantly increased the proportion of patients who achieved a glycated hemoglobin level <7.0% without increasing the risk of severe hypoglycemia. The least-squares mean change from baseline was significantly greater in the sotagliflozin group than in the placebo group for glycated hemoglobin (difference, −0.46 percentage points), weight (−2.98 kg), SBP (−3.5 mm Hg), and mean daily bolus dose of insulin (−2.8 U/day) (P ≤ 0.002 for all comparisons). Already in the published inTandem3 trial almost twice as many patients treated with sotagliflozin were able to achieve the glycemic goal of A1c <7.0%. In a prespecified analysis of other clinical benefits, the proportion of patients with no severe hypoglycemia, no DKA, and no weight gain at Week 24 was higher with sotagliflozin. However, overall only less than 30% of patients on sotagliflozin achieved this primary endpoint. In particular, rather high baseline HbA1c-values may have contributed to this but also the lack of prior insulin optimization could have contributed to the rather low success rate.
A1c, glycated hemoglobin; BHB, beta hydroxybutyrate; CANA, canagliflozin; DEPICT-1, dapagliflozin evaluation in patients with inadequately controlled type 1 diabetes; DKA, diabetic ketoacidosis; EU, European Union; JDRF, Juvenile Diabetes Research Foundation; LTE, long-term extension; SOTA, sotagliflozin; RCT, randomized controlled trial; U.S., United States.
The endpoint of the hitherto unpublished European and U.S./Canadian inTandem1 and 2 trials with insulin optimization before starting sotagliflozin will be able to answer how much added benefit of adjunct therapy can be expected. Weight gain with intensive insulin therapy remains to be a big challenge. Body weight was a secondary endpoint and was chosen because intensive insulin therapy in T1DM is associated with weight gain. It was expected that optimized therapy could be associated with weight gain, while local (gastrointestinal) SGLT1 inhibition and systemic (renal) SGLT2 inhibition could result in weight reduction even in the setting of optimized insulin therapy. Body weight reduction (absolute) over time in both sotagliflozin groups was gradual and progressive over the entire treatment period without evidence of reaching a plateau before the end of the study. Considering a mean BMI of 30 kg/m2 at baseline, achieving a weight loss of 3% to 5% is clinically relevant and associated with significant benefits. 30,31 These results suggest that adjunct therapy with sotagliflozin was perceived as helpful in addressing the challenges of living with T1DM.
Continuous Glucose Monitoring (CGM) percent time in range (70 to 180 mg/dL) is the primary endpoint specified for the CGM substudy meta-analysis. Also, the exclusive SGLT2-inhibitors empagliflozin, 32 dapagliflozin, 33 and canagliflozin 34 have reported significant improvements of time in range in CGM studies. Because enrollment targets were not met in the inTandem1 and 2 studies, patients enrolled in this substudy were pooled for analyses with those enrolled in both studies and the primary analyses were prespecified to be performed on the combined data set. It is likely that a placebo-corrected increase of time in range (70 to 180 mg/dL) may best reflect the glycemic improvement for an individual patient beyond A1c. 35 In the Phase 2 proof-of-concept study, treatment with sotagliflozin increased% time in target range from 56% at baseline to 68%, corresponding to nearly 3 h/day with no increase in% time <70 mg/dL. 36 The increase in CGM% time in target range observed for the sotagliflozin group translates to less time spent in hypoglycemia and hyperglycemia. Inasmuch as time in range is considered very relevant for patient well-being and adjustment of insulin therapy, long-term studies relating this parameter to long-term outcomes are currently lacking.
Mitigating the Risk for DKA in SGLT2 Inhibitors
As noted in the literature and labeling regarding approved SGLT2 inhibitors, these agents increase the risk of DKA. DKA has been reported with SGLT2 inhibitors in both type 1 and type 2 diabetes. In T1D, DKA generally occurs when not enough insulin is administered. This may happen, for example, when an insulin pump fails, insulin doses are missed, the dose of insulin is substantially reduced, or the metabolism receives metabolic stress-like illnesses or operation. For example, in an 18-week study of canagliflozin in T1DM, the incidence of DKA was 6.0% on canagliflozin 300 mg compared with 0% on placebo. The placebo-subtracted mean reduction in insulin dose was 22.4% for basal insulin and 12.1% for bolus insulin. UGE was not reported in this study, 37 but relatively high levels of ∼100 g/day are described for canagliflozin in type 2 diabetes at both the 100 and 300 mg doses. 38 Of note, rates of DKA in the dapagliflozin evaluation in patients with inadequately controlled type 1 diabetes (DEPICT-1) trial 33 appear to be lower than those observed with sotagliflozin, a difference that resulted mainly from different adjudication criteria for DKA. In DEPICT-1, suspected DKA was adjudicated as “definite,” “possible,” or “unlikely,” and only cases that met the criteria for “definite” were positively adjudicated, while this was handled differently in the publication of the inTandem3 trial. 6 The rates of DKA would have been similar if the investigators in the two trials had included all but the “unlikely” results as being positive. 39
Recognizing the risk of DKA, experts in T1DM have still recommended more clinical research on SGLT2 inhibition, with the view that DKA risk factors can be identified, monitoring can be implemented, and education can be provided to ensure that DKA is recognized in a timely manner and treated effectively. 36 Overall, it may be difficult to compare DKA rates in studies as there is currently no uniform strategy for risk mitigation and the vigilance of this class effect on ketone production may have affected the various studies in different ways.
In case of the sotagliflozin program, the sponsor enhanced DKA risk mitigation activities, including distribution of point of care testing beta hydroxy butyrate meters to all patients and sites, revision of patient wallet cards with instructions on ketone identification and correction, a protocol amendment to emphasize the importance of patient adherence with urine and blood ketone assessment, and the need for early investigator notification of BHB elevation so that appropriate action could be taken.
As would be expected from what we know so far, the incidence of DKA was highest in patients using insulin pumps. Among the cases of DKA, there were several associated with insulin pump failures. BHB monitoring should be performed when there is a suspicion of insulin pump failure and may be recommended after change in insulin pump materials. Patient selection for adherence and clinical risk factors, as well as a transition period with establishing regular blood ketone measurement before initiation of adjunct therapy, may be useful for further mitigating the risk (Table 2). Patients should be advised that also in case of illness with increased insulin need, SGLT inhibitors should not be taken and ketones tested regularly. The importance of raising blood glucose levels so insulin can be given needs to be reiterated. In a nutshell: “When in doubt, don't take the medication. If you have ketones, drink fluid, eat carbs, give insulin.”
CGM, continuous glucose monitoring; SMBK, self monitoring of blood ketones.
Among other events of special interest, genital yeast infections were the most common. However, they were generally mild and led to study drug discontinuations in only ≤1% of patients. The incidence of UTIs was similar on placebo and sotagliflozin. Diarrhea was more common on sotagliflozin than placebo, reflecting clinical SGLT1 inhibition in the gastrointestinal tract with an osmotic effect. It was generally mild. As far as the data are available, less than 2% of patients discontinue study drug due to diarrhea. Overall, key safety results included an increase in DKA and a low incidence of hypoglycemia with sotagliflozin compared with placebo. Education and monitoring initiated after the DKA risk became apparent and may be able to limit the incidence and severity of DKA, supporting the use of dual SGT1 and SGT2 inhibitions as an adjunct to insulin.
Noninsulin Adjunctive Therapy: Balancing Risk and Benefit
Registry data indicate that adjunct therapy for type 1 diabetes is widespread already today although it presently is still “off-label.” 40,41 Metformin so far has been used most for this purpose potentially reducing insulin need and with known benefits with regard to obesity in patients with type 2 diabetes. New publications show no benefit to this approach with an increase of side effects, 42,43 although discussions are ongoing if it might have a wider role in cardiovascular risk management. A recent meta-analysis of 14 studies of 4591 T1DM patients treated with SGTL2 inhibitors revealed that such therapy significantly reduced HbA1c by 0.4% (95% confidence interval [CI]: 0.35–0.46; P < 0.001; I2 = 0%), fasting plasma glucose by 1.14 mmol/L (0.8–1.47), body weight by 2.68 kg (2.0–3.36), and SBP by 3.37 mmHg (1.46–5.28). In addition, bolus insulin decreased by 3.6 U/day (2.0–5.3) and basal insulin decreased by 4.2 U/day (2.2–6.3). 8 Continuous glucose monitoring showed a decrease of glucose excursions compared to placebo, with reduced variation of mean blood glucose, the glucose standard deviation, and the mean amplitude of glucose excursion. 8 Noninsulin-based add-on therapies particularly with the class SGLT2 inhibitors offer a potential added value to established therapies (multiple daily injections (MDI) and continuous subcutaneous insulin infusion (CSII)) as well as future approaches (sensor-augmented therapy and closed loop) for type 1 diabetes.
It remains to be elucidated if the combined inhibition of SGLT1 and SGLT12 holds any particular benefit in type 1 or type 2 diabetes compared with the class SGLT2 inhibitors or individual drugs. Overall, this group represents attractive candidates for a combination therapy, and intended regulatory submission for the indication of T1DM has been announced for both dapagliflozin and sotagliflozin. The insulin-independent mode of action, the beneficial effects on body weight even potential for the preservation of beta-cell function through reduction of glucose toxicity, or potential protective effects regarding nephropathy due to improved renal hemodynamics delineate potential benefits. On the background of the beneficial results on cardiovascular outcomes in T2DM so far described for empagliflozin, 44 it is attractive to speculate that a similar benefit could be possible for patients affected from T1DM, which is a major risk factor for premature cardiovascular death. 45
The appropriate use of such drugs by pediatric patients to meet their unmet medical needs has to include a single, simple dose per age or weight category, and clinical trials indicating meaningful benefits in A1c reduction, no untoward weight gain, and no consequences of increased UGE regarding growth and development decreased daily insulin requirement, decreased daily glucose variability as well as a well-described and manageable safety profile, particularly a potentially decreased hypoglycemia rate, and a low incidence of DKA. Particular challenges in the very young (patients <6 years of age) could include potential toxicity of these drugs on developing nephrons and that the method of action makes use of such drugs in patients using diapers undesirable and unsafe (UTIs). However, as glycemic variability is a particular challenge in this age group such use should not be generally ruled out provided no additional safety signal becomes apparent when studying older kids.
Special attention will be paid to prevent DKA. The risk of DKA must be considered in the event of nonspecific symptoms such as nausea, vomiting, anorexia, abdominal pain, excessive thirst, difficulty in breathing, confusion, and unusual fatigue or sleepiness nevertheless in normoglycemic state. Both patients and healthcare providers need to be educated in the principle of euglycemic DKA. They need to understand how—due to the insulin-independent mode of action—there is a possibility that ketoacidosis in patients treated with SGLT1 and SGLT2 inhibitors is not accompanied by typical hyperglycemia as usually expected for DKA. Besides a clear scheme for assessment of ketones (preferably as BHB in capillary blood), a treatment algorithm on how to act according to BHB and glucose values should be available for patients using this new therapeutic approach.
Patients who may be at higher risk of DKA while taking SGLT1 and SGLT2 inhibitors include patients on a very low carbohydrate diet (as the combination may further increase ketone body production), severely dehydrated patients, and patients with a history of ketoacidosis or known to have a low beta-cell function reserve. Insulin deficiency might lead to ketoacidosis, which could be life threatening if not recognized and appropriately treated. Patients with insulin background therapy will be made aware of this risk and be instructed not to reduce their insulin dose below physicians' recommendations. With this in mind, the study data so far indicate that
the overall risk/benefit profile was acceptable for dual SGLT1 and SGLT2 inhibition with sotagliflozin as an adjunct to insulin in patients with T1DM and warrants further study for T2DM, particularly but not limited to those with renal impairment as well.
Footnotes
Author Disclosure Statement
T.D. received honoraria and research support from Abbott, Bayer, BMS/AstraZeneca, Boehringer Ingelheim, DexCom, Eli Lilly, Lexicon, Medtronic, Novo Nordisk, Sanofi, and Roche. He is a shareholder of DreaMed Ltd. T.B. received honoraria and research support from Medtronic, Ypsomed, DexCom travel support, and Novo Nordisk. O.K. received honoraria and research support from Abbott, Eli Lilly, Novo Nordisk, and Sanofi. She is a shareholder of DreaMed Ltd.
