Abstract
FlexTouch® (Novo Nordisk A/S, Bagsvaerd, Denmark) is a pen-injector with a torque spring mechanism requiring a low activation force. This laboratory-based study compared the activation force of FlexTouch during the injection of insulin with different needles and at temperature conditions within the range at which the device is recommended for use. Using a tensile tester, activation force was measured at maximum dose settings for insulin detemir (100 U/mL) and insulin degludec (100 and 200 U/mL) at standard (23°C ± 5°C), cool (5°C ± 3°C), and warm (30°C ± 2°C) conditions. Activation force was measured with two 32-gauge needles differing in internal diameter at standard conditions. At standard, cool, and warm conditions, estimated mean activation forces with 95% confidence interval were 5.71 newtons (N) (5.63–5.79), 5.94 N (5.83–6.06), and 5.69 N (5.58–5.80) with insulin detemir, 5.53 N (5.45–5.62), 5.56 N (5.44–5.67), and 5.33 N (5.22–5.44) with 100 U/mL insulin degludec, and 5.53 N (5.45–5.61), 5.83 N (5.71–5.94), and 5.56 N (5.45–5.68) with 200 U/mL insulin degludec, respectively. Mean activation forces were observed to be low with very small variability between measurements; however, the differences between insulins and temperature conditions were statistically significant. The activation force required by FlexTouch remained low across all situations tested. The differences between activation force needed with different insulins and temperature conditions were small and unlikely to be clinically meaningful.
Introduction
I
Notwithstanding these benefits, current limitations associated with pen-injectors include the extension of the dose button (especially as the dose increases), handling during injection, and the manual force needed to deliver the insulin.
Most pen-injectors rely on thumb pressure to depress an extended dose button (termed dose force). This force needs to be applied throughout the entire injection and directly influences the rate of insulin delivery. Depressing the dose button can be particularly difficult for younger patients who cannot reach over the extension of the dose button and patients who may have impaired manual dexterity, strength, and nerve function, for example, elderly patients. 9,10
FlexTouch® (Novo Nordisk A/S, Denmark) is a pen-injector with a torque spring injection mechanism and no dose button extension. 11 The activation force (equivalent to the dose force for pen-injectors with extending dose button) is loaded into the internal spring when the dose is set, and the spring's energy is activated when the dose button is pressed. As a consequence, the injection force of FlexTouch is significantly lower than other widely used prefilled pens. 12,13
It is important that FlexTouch retains a low and consistent activation force during everyday use. Variables that may affect the functioning of FlexTouch include the inner diameter of the needle being used and the external temperature. 14,15 The aim of this study was to determine the activation force of FlexTouch with maximum doses of commonly used insulins, two different needles, and at temperature conditions within the range at which the device is recommended for use.
Materials and Methods
Materials
The insulin formulations tested were insulin detemir (100 U/mL; batch EP50929), insulin degludec (100 U/mL; batch EP50570), and insulin degludec (200 U/mL; batch EP50955). Two 32-gauge needles with different internal diameters were used (NovoFine Plus®, Novo Nordisk, 4 mm, extra thin wall [ETW], batch 14F04N; SureComfort®, Allison Medical, Inc., USA, 6 mm, thin wall [TW], batch P140712).
Equipment
A climate chamber (Vötsch WKL34; Vötsch Industrietechnik GmbH, Balingen, Germany) was used to establish three temperature conditions: standard (23°C ± 5°C with a relative humidity [RH] of 50% ± 25%), cool (5°C ± 3°C with no RH requirement), and warm (30°C ± 2°C with a 50% ± 10% RH). Activation force was measured in newtons (N) using a universal tensile tester (Lloyd LRX; Lloyd Instruments Ltd., Humlebaek, Denmark), with a 100 N load cell.
Activation force testing
Laboratory testing was performed at the SP Technical Research Institute of Sweden (Sp Sveriges Tekniska Forskningsinstitut A/S, Denmark). Activation force was measured at the maximum injectable dose setting: 80 U for insulin detemir (100 U/mL) and insulin degludec (100 U/mL), and 160 U for insulin degludec (200 U/mL) with ETW and TW needles. The insulin formulations were tested with the ETW needle at standard, cool, and warm conditions and with the TW needle at standard conditions.
Needles and pen-injectors were acclimatized for at least 4 h in the climate chamber. One pen-injector at a time was taken out of the chamber and used for testing within 3 min. A needle was attached to the pen-injector, and air expelled until liquid appeared at the tip of the needle. The dose was set and the pen-injector was then placed in the tensile tester with the needle facing downward. The tensile tester was set to a preload of 0.2 N and an approaching speed of 0.5 mm/s. After preload was reached, force measurements were performed at a pressing speed of 1 mm/s until a distance of 2.8 mm from the position where preload was reached. The activation of the injection mechanism was indicated by a peak in the force readings after pressing ∼1.4 mm and this was recorded as the activation force. For each experimental condition, a mean activation force was obtained from 25 measurements using 25 pen-injectors, with 1 measurement per pen-injector. A new needle was used for each measurement.
Statistics
Differences in activation force between insulin detemir (100 U/mL) and insulin degludec (100 and 200 U/mL), temperature condition, and needle were investigated by a linear normal model (analysis of variance) with the needle and the interaction between insulin and temperature condition as fixed effects. The estimated means and estimated differences between means with 95% confidence intervals (CI) were calculated.
Results
The estimated mean activation forces measured with insulin detemir (100 U/mL) and insulin degludec (100 and 200 U/mL) under different temperature conditions are shown in Figure 1. Although the differences in the mean activation forces for each combination of insulin and temperature condition were statistically significant (P < 0.0001), these differences were small and are unlikely to be of clinical relevance. Mean activation forces were between 5 and 6 N with an ∼5% decrease in the estimated mean activation force across the cool, standard, and warm temperature conditions (estimated differences between temperatures [95% CI]; insulin detemir: cool vs. standard 0.23 N [0.09–0.37], standard vs. warm 0.02 N [−0.12 to 0.16]; insulin degludec [100 U/mL]: cool vs. standard 0.02 N [−0.12 to 0.16], standard vs. warm 0.20 N [0.06–0.34]; insulin degludec [200 U/mL]: cool vs. standard 0.30 N [0.16–0.44], standard vs. warm −0.03 N [−0.17 to 0.11]). The mean activation forces were similar between the 32-gauge ETW needles (5.62 N [95% CI: 5.58–5.66]) and TW needles (5.70 N [95% CI: 5.61–5.78]) when accounting for differences with temperature condition and insulin (estimated difference between needles [95% CI]: −0.08 N [−0.17–0.01]; P = 0.10).
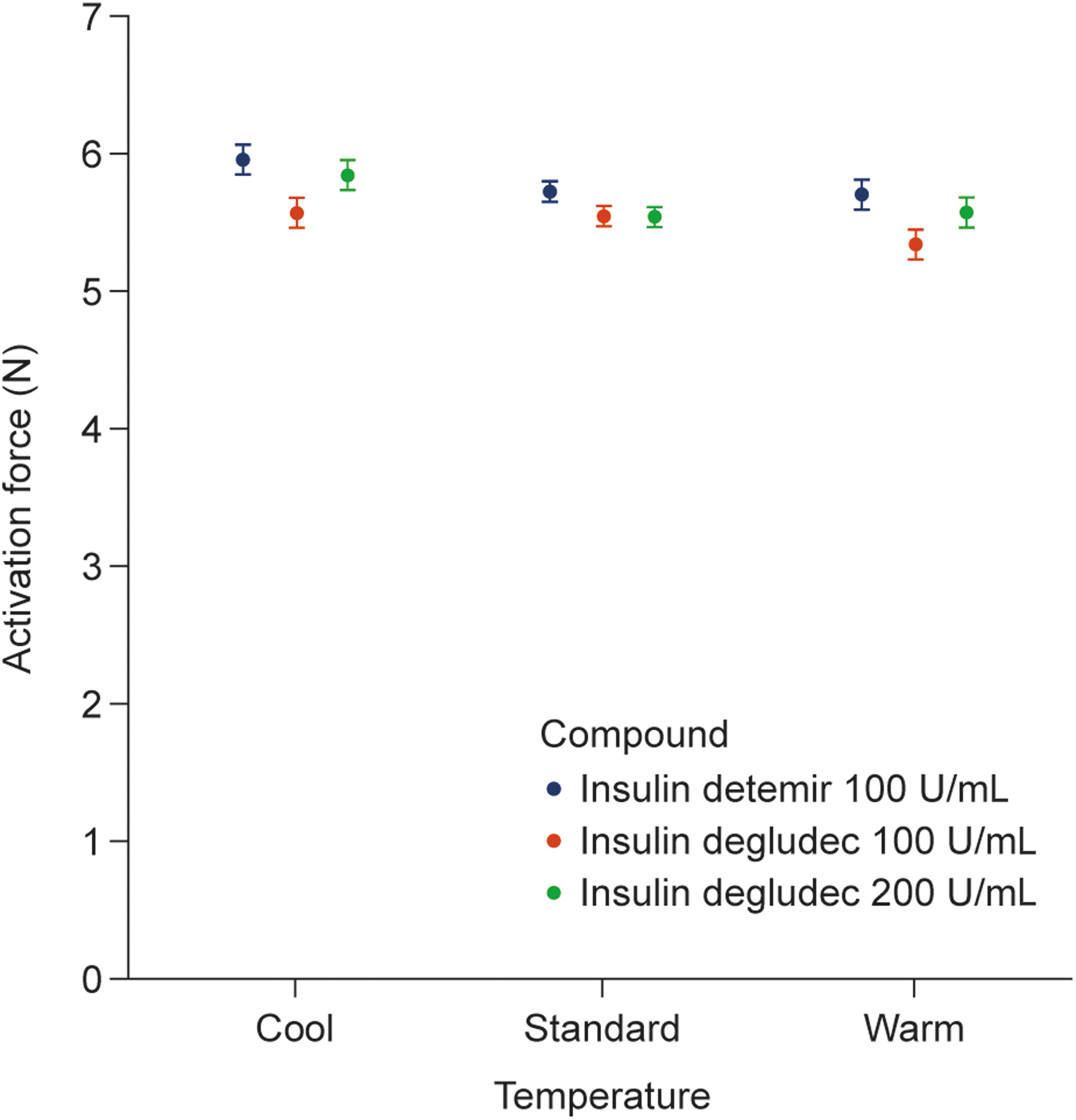
Estimated activation force by compound and temperature. Activation force required by FlexTouch® to inject maximum doses of insulin detemir (100 U/mL) and insulin degludec (100 and 200 U/mL) under different temperature conditions. Data are shown as least square means (95% confidence intervals) estimated from a linear normal model. N, newtons; U, units.
Discussion
FlexTouch pen-injectors are currently available with a range of insulins and different needles. When not being used, pen-injectors should be kept in a refrigerator at a temperature between 2°C and 8°C and away from the freezing element. When in use, it is recommended that the FlexTouch pen-injector is operated at room temperature, below 30°C, and out of direct sunlight. However, diverse climatic conditions can result in pen-injectors being exposed to a range of temperatures during everyday use and it is important that performance is not adversely affected by these variables. This is the first study to demonstrate that the low activation force of FlexTouch is only minimally affected by temperature conditions representing the extremes of the recommended range for use. The small differences observed between conditions in these laboratory tests are unlikely to be of clinical relevance.
The torque spring injection mechanism of FlexTouch results in a lower activation force compared with the dose force of other pen-injectors (Table 1). Previous technical studies show the mean (standard deviation) activation force of FlexTouch to be 5.1 (0.5) N at maximum dose settings; this is between 56% and 86% lower than the force required by SoloSTAR® (Sanofi-Aventis, Paris, France) and KwikPen® (Eli Lilly and Co., Indianapolis) 12,13 and is consistent with the activation forces measured under standard conditions in this study. There was also very little variation in the repeated force measurements, as shown by the narrow CIs and the small differences between the temperature conditions, highlighting that the activation force of FlexTouch remains consistently low.
FT, FlexTouch®; HCP, healthcare professional; N, newtons; s, second; U, unit.
In a clinical context, the activation/dose force required to deliver insulin by a pen-injector can be a barrier to insulin therapy and the low activation force of FlexTouch aims to reduce the difficulties of injecting insulin. Usability studies show that FlexTouch is preferred to vials and syringes and other widely used pen-injectors by people with T1D and T2D, caregivers, and healthcare professionals (HCPs) (Table 1). As an example, in a study in which 61% of 90 participants with T2D had impaired dexterity, FlexTouch was considered superior to InnoLet® (Novo Nordisk A/S, Denmark) with respect to injection procedure, handling, and confidence in correct dosing. 16
The low activation force of FlexTouch is reflected in a high maximum flow rate (Table 1). While injection speed has often been considered to impact upon a patient's perception of injection pain, evidence from a randomized controlled trial demonstrates that subcutaneous injection speed had no effect on injection pain. 17 Indeed, a higher injection speed may contribute to a shorter dwell time of the needle in the injection site. 18 For most prefilled pen-injectors, needle choice has an impact on the force required to depress the dose button, with a larger diameter associated with reduced force. 19 However, thinner needles tend to be preferred by patients as they are associated with less pain, bleeding, and bruising than thicker needles. 20,21 While the outer diameters of the needles used in this study were consistent, the ETW technology allows for a larger inner diameter, and so an increased flow rate, compared with the TW needles. Nonetheless, the activation force was similar with the two 32-gauge needles tested when accounting for the differences with insulins and temperature conditions, demonstrating that a patients' needle choice (ETW vs. TW technology) does not impact upon the activation force of FlexTouch. This is not unexpected as, due to the torque spring mechanism, the activation force of FlexTouch is unlikely to be affected by needle diameter.
A limitation of this study was that measurements were performed in a controlled, laboratory environment and other factors, such as the tissue being injected into, may affect injection force when performed by patients or HCPs. However, accurately measuring force in a clinical setting would be very challenging. Clinical studies are needed to assess the range over which injection force influences patient perception and preference, and if injection force is an important factor in insulin therapy adherence and, ultimately, in improved glycemic control.
In conclusion, the low activation force of FlexTouch with commonly used insulins is maintained across needle technologies and temperature conditions (within the recommended range).
Footnotes
Acknowledgments
The authors thank the SP Technical Research Institute of Sweden for performing the measurements. The authors also acknowledge Joakim Petersen at Novo Nordisk for his technical assistance. Medical writing and submission support were provided by Helen Parker and Beverly La Ferla of Watermeadow Medical, an Ashfield Company, part of UDG Healthcare plc, funded by Novo Nordisk. Funding sources: This study was funded by Novo Nordisk A/S.
Author Disclosure Statement
B. R., N.G., T.H, T.R., and T.S. are employees of Novo Nordisk. T.S and T.H. are shareholders in Novo Nordisk.
