Abstract
Controlling glycemia in diabetes remains key to prevent complications in this condition. However, glucose levels can undergo large fluctuations secondary to daily activities, consequently creating management difficulties. The current review summarizes the basics of glucose management in diabetes by addressing the main glycemic parameters. The advantages and limitation of HbA1c, the gold standard measure of glucose control, are discussed together with the clinical importance of hypoglycemia and glycemic variability. The review subsequently moves focus to glucose monitoring techniques in diabetes, assessing advantages and limitations. Monitoring glucose levels is crucial for effective and safe adjustment of hypoglycemic therapy, particularly in insulin users. Self-monitoring of blood glucose (SMBG), based on capillary glucose testing, remains one of the most widely used methods to monitor glucose levels, given the relative accuracy, familiarity, and manageable costs. However, patient inconvenience and the sporadic nature of SMBG limit clinical effectiveness of this approach. In contrast, continuous glucose monitoring (CGM) provides a more comprehensive picture of glucose levels, but these systems are expensive and require constant calibration which, together with concerns over accuracy of earlier devices, restrict CGM use to special groups of patients. The newer flash continuous glucose monitoring (FCGM) system, which is more affordable than conventional CGM devices and does not require calibration, offers an alternative glucose monitoring strategy that comprehensively analyzes glucose profile while sparing patients the inconvenience of capillary glucose testing for therapy adjustment or CGM calibration. The fast development of new CGM devices will gradually displace SMBG as the main glucose testing method. Avoiding the inconvenience of SMBG and optimizing glycemia through alternative glucose testing strategies will help to reduce the risk of complications and improve quality of life in patients with diabetes.
Introduction
T
The current review will cover glycemic management in diabetes by addressing clinically relevant parameters together with the advantages and drawbacks of current glucose testing methods. Special emphasis will be placed on the potential clinical benefit of using systems other than SMBG for measuring glucose in individuals with diabetes.
Glycemic Parameters and Clinical Outcome
Hyperglycemia, hypoglycemia, and glycemic variability (GV) have been linked to adverse clinical outcomes, which will be briefly discussed below.
Hyperglycemia
It is well accepted that hyperglycemia in diabetes directly contributes to short/long-term microvascular complications and has a long-term role in the development of macrovascular disease. 5,7 Several mechanisms have been proposed for the adverse vascular effects of hyperglycemia, including increased oxidative stress and enhanced mitochondrial superoxide production, 8 as well as endothelial dysfunction leading to the development of an inflammatory/thrombotic environment. 9
The role of improving glycemia in preventing vascular complications has been extensively studied in various diabetes populations. The Diabetes Control and Complications Trial (DCCT), conducted on 1441 patients with T1DM, has shown that a reduction in HbA1c by 1.7% decreases microvascular complications over a median follow-up of 6.5 years. 4 Interestingly, some of these benefits were observed early such as nephropathy where albuminuria was reduced within 1 year of maintaining good glycemic control. At the end of DCCT, more than 95% of patients agreed to participate in an extension to the study using an observational design in the Epidemiology of Diabetes Interventions and Complications (EDIC). In EDIC, HbA1c levels became similar in intervention and standard groups, but a reduction in macrovascular complications over a follow-up period of 10 years was evident in those who previously received intensive intervention for glycemia, 5 stressing the importance of controlling glucose levels early in the disease process. More recently, 30-year follow-up data of DCCT have been published, which further demonstrate a difference between two study arms in relation to macrovascular complications. 10
The UK Prospective Diabetes Study (UKPDS) randomized 4209 newly diagnosed T2DM patients to intensive and standard glycemic control and included an extension observational period mimicking the design of DCCT-EDIC. The findings in UKPDS were largely similar to DCCT-EDIC with early and late reduction in microvascular and macrovascular complications, respectively, although this was observed with a more modest 0.9% difference in HbA1c comparing study arms. 11 Results from DCCT-EDIC and UKPDS led to the concept of metabolic memory or legacy effect calling for early control of glycemia to reduce long-term vascular complications.
However, studies on intensive glycemic management in diabetes patients with established macrovascular disease have raised concerns. Although a reduction in microvascular disease was demonstrated with tight glycemic control, 12,13 this was associated with increased mortality in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study, involving 10,251 high vascular risk patients. 13 It is worth noting that target HbA1c in the intervention arm was an ambitious 6.0% (reaching 6.5% in the study), whereas the standard arm had a respectable HbA1c of 7.4%. No clear reasons have been identified for the increased mortality in the intervention arm of the ACCORD trial, although various hypotheses have been put forward, including increased incidence of severe hypoglycemia. 14
A general criticism of glycemic outcome studies is the use of HbA1c as the main assessor of glycemia, an approach that has a number of limitations. First, HbA1c does not always give an accurate measure of average glucose levels. For example, an HbA1c of 7% (53 mmol/mol), indicating good glycemic control, reflects an average glucose of anything between 123 and 185 mg/dL, a range that becomes even wider with higher HbA1c of 10% (range 192–282 mg/dL); therefore, HbA1c represents only a rough estimation of average glucose levels. 15 Second, HbA1c does not address hypoglycemia or GV, both of which are associated with adverse clinical outcomes (detailed below). Third, HbA1c can be misleading in patients with blood abnormalities, including the common iron deficiency anemia, or the presence of advanced diabetic complications, such as end-stage renal disease. 16 –19
Therefore, it is important to understand the limitations of HbA1c and supplement glycemic assessment by glucose checks, a view supported by the recent ADA recommendation. 20 Figure 1 summarizes the advantages and limitations of HbA1c as a measure of glycemia.
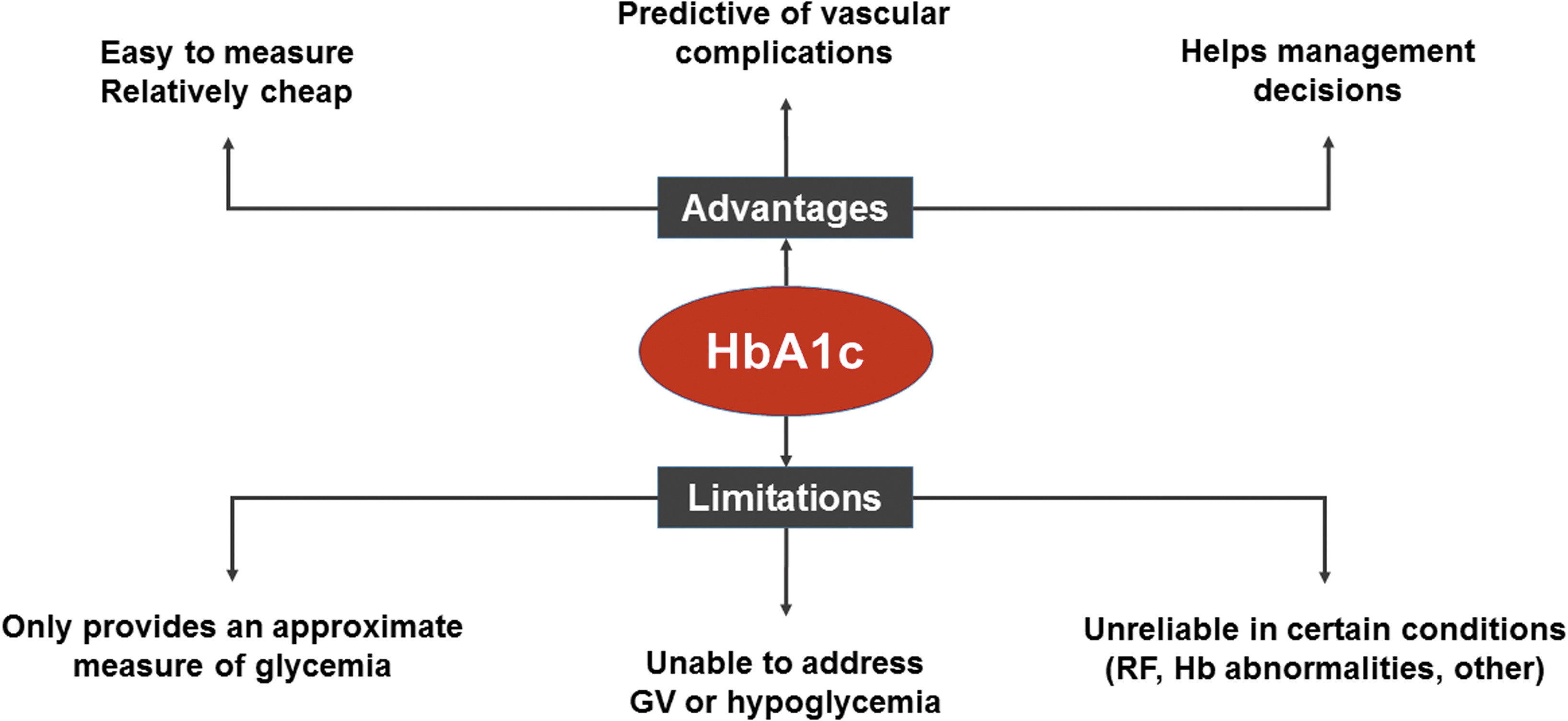
Advantages and limitations of HbA1c. The most widely used measure of glycemia, HbA1c, is easy to measure and has a clear role in optimizing therapy. However, HbA1c has a number of limitations that should be taken into account in clinical practice. GV, glycemic variability; Hb, hemoglobin; RF, renal failure.
Hypoglycemia
Hypoglycemia is increasingly seen in diabetes given the ever tighter glycemic targets and this complication of therapy can have detrimental effects through a number of mechanisms, including cardiac dysrhythmias, increased production of vascular inflammatory molecules, and an enhanced thrombotic environment (reviewed in Frier et al. 21 and King and Ajjan 22 ). Therefore, it is not surprising that a number of studies have shown associations between hypoglycemia and high mortality in patients with both T1DM and T2DM. 23 –29 Furthermore, accumulating evidence suggests that hypoglycemia is more frequent than initially envisaged, 30,31 even in those classified as having “preserved hypoglycemic awareness”; therefore, the size of problem may be even bigger than previously projected. HbA1c fails to address hypoglycemia and therefore frequent SMBG, which is impractical, or CGM, which can be costly, is required to accurately identify periods of hypoglycemia. The advent of new glucose monitoring devices that are affordable, convenient, and accurate will help to further address the role of hypoglycemia in adverse clinical outcomes (discussed below).
Glycemic variability
Emerging evidence suggests that GV, both medium and short term, contributes to adverse clinical outcome in diabetes. A recent meta-analysis assessing medium-term GV, measured as fluctuation in HbA1c, has shown associations with microvascular and macrovascular complications in T1DM and T2DM as well as mortality in T2DM. 32 Fluctuation in glucose levels has also demonstrated associations with increased mortality and longer hospital stay, which was independent of HbA1c levels. 33
Proposed mechanisms for the association between GV and adverse vascular outcomes include increased oxidative stress and enhanced expression of proteins involved in vascular pathology. 34 This perhaps explains the association between GV and plaque characteristics as well as atherosclerotic cap thickness in 70 patients with established coronary artery disease. 35 Moreover, GV may directly predispose to myocardial infarction through the documented association with coronary plaque instability as shown in 76 individuals with acute coronary syndrome and T2DM. 36
While GV is potentially an important glycemic parameter, we should be cautious in our conclusions as current evidence linking GV with adverse clinical outcomes is relatively weak. Studies have mainly relied on SMBG or HbA1c measurements for estimation of GV and neither gives a full glycemic picture. Moreover, there is no standardized definition for GV at present as a large number of markers have been used without clear indication as to which one is the most relevant clinically. 37 Also, the magnitude of GV is dependent on average glucose levels, and therefore, it can be argued that correction for HbA1c (or mean glucose) is required when assessing variability, which further complicates matters. 38 Finally, the association between GV and low glucose levels, including nocturnal hypoglycemia, 39 –41 raises the possibility that GV is not an independent predictor of adverse clinical outcome. Therefore, further longitudinal glycemic studies evaluating the relationship between a standardized measure of GV and clinical outcome are needed.
The Ideal Glycemic Marker
HbA1c has been used for decades as the main glycemic marker, which is understandable given the ease of testing and the documented association between HbA1c and complications. However, HbA1c is one glycemic marker that displays only an approximate measure of glucose levels and fails to address other parameters such as hypoglycemia and GV, both of which may influence clinical outcome. Therefore, for a comprehensive assessment of glycemia, a combination of glycemic parameters is required that includes hyperglycemia (assessed as HbA1c as well as glucose levels), hypoglycemia, and GV, which led to the concept of “The Triangle of Diabetes Care.” 42 Glycemic assessment should perhaps follow this triple approach, although the definition of GV is in need of refinement.
It should be noted that these glycemic parameters are not necessarily independent. For example, GV increases in individuals with repeated hypoglycemia, whereas large fluctuation in glucose levels can also increase HbA1c.
Glucose Testing Methods
Checking glucose levels becomes important when treating with oral agents that cause hypoglycemia, such as sulfonylureas, and a necessity in insulin-treated individuals. Glucose testing has been performed using three methods: SMBG, CGM, and, more recently, flash continuous glucose monitoring (FCGM).
Self-monitoring of blood glucose
This is the most widely used glucose testing method and generally enjoys good accuracy but is not devoid of drawbacks. SMBG is inconvenient to patients as testing can be painful, many describe it as antisocial, and is affected by daily life activities, which can pose a problem, particularly in younger patients. Also, SMBG misreporting remains an issue, 43 which may have serious clinical implications. Importantly, frequent testing is required to effectively alter management and achieve good glycemic control. In support of this, an analysis of 20,555 patients with T1DM has shown an inverse correlation between frequency of glucose testing and HbA1c levels. T1DM individuals testing three to four times/day had an HbA1c of 8.6%, dropping to 7.6% in individuals performing ≥10 tests/day, 44 with similar findings documented in patients with T2DM. 45 This indicates that frequent glucose testing is required to improve glycemic control, which can be difficult to maintain long term. Finally, the quality of the glucose testing strips can be variable and some versions do not necessarily provide high accuracy, emphasizing the importance of ongoing quality control studies. 46
Continuous glucose monitoring
CGM devices, recording interstitial glucose every 5 min, have the advantage of giving a more comprehensive picture of glycemia by providing a large number of glucose readings. Sensor life is limited to 1 week at best, although this is extended to 2 weeks with FCGM that offers an alternative form of CGM (detailed below). A new generation of implantable sensors further extends sensor life to 6 months, 47 but the large interindividual variability observed (100%, 82%, and 40% of sensors were functional at 45, 90 and 180 days, respectively) and the limited accuracy at lower glucose levels indicate that further refinement is needed before implantable sensors are available to use in clinical practice.
CGM has been used retrospectively and in real time with the latter having the advantage of responding quickly to unwanted changes in glucose levels. CGM has also been used linked to insulin pumps allowing the development of sensor-augmented pumps that suspend insulin infusion when hypoglycemia is detected or predicted, 48,49 with more sophisticated systems even adjusting for hyperglycemia. 50
In general, CGM studies have shown an improvement in glycemia, measured as HbA1c, and/or reduction in hypoglycemia, rendering them cost-effective in some diabetes populations despite the high outlay compared with SMBG (reviewed in Vigersky and Rodbard 51,52 ). However, CGM is still not widely used, secondary to inertia of physicians, concerns over accuracy, and the general high costs, as well as patient-related factors, including the need for constant calibration and difficulties encountered in data interpretation.
What are the key glycemic measures generated by CGM?
Interpreting CGM data can pose a challenge to patients and healthcare professionals (HCP) alike. Therefore, attempts have been made to standardize CGM glycemic measures. A recent consensus conference suggested that relevant CGM data should include current glucose levels, glucose trend arrows, default alert for hypoglycemia set at <70 mg/dL, and the inclusion of a standardized report that includes various glucose metrics such as time in range, GV, and patterns of hypoglycemia and hyperglycemia. 53 However, a clear process for analyzing long-term glucose data is still lacking and this is currently left at the discretion of the HCP. Ambulatory glucose profile (AGP), generated by CGM, has been proposed as a good platform to standardize interpreting glucose data and draw up effective management decisions. 54
Retrospective or real-time CGM?
Retrospective CGM, also referred to as professional CGM, is usually used intermittently and mainly for diagnostic purposes with the patient blinded to glucose data. In contrast, real-time CGM, usually used longer term, enables patients to see glucose readings and make management changes as appropriate. 55 The latter is equipped with an alarm system to warn of hypoglycemia that can be particularly helpful in those with hypoglycemic unawareness. Using retrospective or real-time CGM depends on the type of patients and the reasons for requesting CGM. Younger patients, majority of whom have T1DM, are generally more comfortable at making changes to their treatment and usually benefit from real-time CGM. Some of the older patients with insulin-treated T2DM are less likely to make changes to their treatment without input from the HCP, and therefore, retrospective CGM may be more appropriate, although this cannot be generalized. However, this view is challenged by others who advocate replacing blinded with real-time CGM for both T1DM and T2DM patients. 56
Is CGM appropriate for all patients with diabetes?
The benefits of CGM in children and younger adults with T1DM are beyond doubt with a number of studies showing improvement in glycemic control and reduction in hypoglycemia. The landmark Juvenile Diabetes Research Foundation (JDRF) trials, using the three main CGM devices (DexCom, Medtronic, and FreeStyle Navigator) have shown that CGM improves HbA1c by 0.5%–0.8% without an increase in hypoglycemia in 322 individuals with T1DM and starting HbA1c >7.0%. 57 A secondary study evaluated 129 T1DM patients with HbA1c <7.0% and demonstrated reduction in hypoglycemia by 30%–50% using real-time CGM, indicating a benefit even in those who are labeled as having good glucose control. A meta-analysis of 10 studies comparing real-time CGM with SMBG in T1DM showed a 0.26% reduction in HbA1c without an increase in hypoglycemia further supporting the use of CGM in this population. 58 It should be noted that these glycemic benefits were evident with early CGM devices, which had inferior accuracy compared with current sensors. Therefore, more studies are required with the new sensors and some are already underway. One recent multicenter CGM study, including a modest number of 25 patients with T1DM and using AGP for glycemic assessment, has shown a marginal reduction in HbA1c of 0.3% (P = 0.05) comparing baseline with 3 months of sensor use, associated with a significant reduction in hypoglycemia from 1.4 ± 1.7 to 0.8 ± 0.8 h/day. 31 One advantage of this work is a real-life study design where treatment changes were left at the discretion of the HCP, but larger trials are needed before concrete conclusions can be drawn. Two more recent high-quality multicenter studies have been conducted to further investigate the role of CGM in T1DM patients treated with multiple daily insulin (MDI) injections. 59,60 The first is the DIAMOND trial, a randomized multicenter study conducted in the United States involving 24 endocrine centers and 158 patients with T1DM. Inclusion criteria included age >25 years, HbA1c between 7.5% and 10.0%, and treatment with MDI injections. Randomization took a 2:1 assignment to intervention (n = 105), using Dexcom Platinum 4 with an enhanced algorithm (software 505) or control group (n = 53) relying on SMBG for measurement of glucose levels (at least four times/day). The intervention group was instructed to test blood glucose twice/day (to calibrate the device) as well as before insulin dosing. A total of 155 individuals completed the study (102 and 53 in the intervention and control groups, respectively), indicating excellent retention rate. HbA1c at 24 weeks, the primary endpoint, was 0.6% lower in intervention compared with the control group. Moreover, this was associated with a significant reduction in hypoglycemia in the intervention compared with the standard arm of the study, indicating that CGM reduces both hyper- and hypoglycemia in T1DM on MDI therapy. However, important limitations of the study are exclusion of individuals younger than 25 years of age and those with HbA1c >10%, making general applicability of the results to all MDI-treated T1DM patients uncertain. Furthermore, individuals in the intervention arm continued to perform SMBG to calibrate the device and to test before meals, practically making the intervention arm a combination of SMBG and CGM. Finally, it is unclear whether these glycemic improvements are sustained and observational studies using real-life setting will help to clarify the effects of this glucose monitoring strategy on long-term glycemic control. The second study is the GOLD trial, conducted in Sweden on MDI-treated T1DM patients. A total of 161 patients were recruited with inadequate glycemic control, defined as HbA1c >7.5%, and without having an upper limit. Patients were randomized to Dexcom Platinum G4 or standard capillary glucose monitoring. This was an ambitious crossover trial with data analyzed at 26 and 69 weeks, which included 17 weeks of wash-out period. A total of 142 patients completed the trial, demonstrating a lower retention rate compared with DIAMOND. There was 0.4% improvement in HbA1c in those using CGM compared with SMBG, associated with higher treatment satisfaction and lower fear of hypoglycemia, indicating improvement in quality-of-life measures in these patients. Of note, the crossover design showed that CGM not only improves glycemia at both intervention periods but discontinuation of this glycemic monitoring strategy also results in worsening of glucose control.
Taken together, randomized controlled trials (RCTs) in T1DM patients show that CGM improves glycemic control regardless whether patients are treated with an insulin pump or receiving MDI therapy. Naturally, there is always the issue of selection bias with CGM studies, and therefore, real-life clinical data are required to further understand the subgroup(s) of patients who would benefit the most from this glucose monitoring strategy. Information overload has been criticized as a drawback of CGM in T1DM, but studies have shown no increase in psychosocial stress, making this glucose testing method both convenient and effective. 61,62
In contrast to T1DM, only limited studies, frequently with a retrospective design and involving a low number of patients, investigated the use of CGM in T2DM. Findings ranged from no effect on glycemia to significant reduction in HbA1c and/or hypoglycemia (summarized in Vigersky and Shrivastav 55 ). One additional multicenter study conducted over 3 months investigated the role of CGM/AGP in 28 T2DM patients on MDI regimen and has shown a reduction in HbA1c by 0.9% comparing baseline with study end without an increase in hypoglycemia, 31 suggesting this group of patients may benefit from CGM.
Clearly, more CGM studies are required in T2DM, particularly in high-risk groups such as older individuals, those with a history of severe hypoglycemia, advanced renal disease, or vascular complications. 53 Pregnant women with diabetes are also likely to benefit from CGM and although some work supports this view, 63 more studies in this area are warranted.
Flash Continuous Glucose Monitoring
The FreeStyle Libre is a relatively new glucose monitoring system that resembles established CGM devices as it measures interstitial glucose every minute and records readings every 15 mins.
64
FCGM has a number of advantages over conventional CGM, including factory calibration, thus offering a genuine break from capillary glucose testing, long sensor life of 2 weeks, and relatively low costs. There are two forms of the device, FreeStyle Libre and Libre Pro for real-time and retrospective glucose data analysis, respectively (
FreeStyle Libre presents glucose measures as numbers and trend arrows with longer term data presented as AGP and time spent in euglycemia, hypoglycemia, and hyperglycemia allowing for quick data interpretation. A systematic approach to AGP, modified according to local need, facilitates patient education on glucose patterns and provides a useful tool to safely and effectively alter hypoglycemic therapy. 67 Moreover, the glucose reader can act as a capillary glucose and ketone monitor further increasing convenience and ensuring that patients do not need to carry two devices. Accuracy of FreeStyle Libre is similar to conventional CGM devices with a mean absolute relative difference (MARD) of 11.4%, but the system fails to provide an alarm to warn of hypoglycemia and current versions do not have the facilities to directly connect to insulin pumps.
Recent multicenter trials have shown that FreeStyle Libre use is associated with improved glycemic parameters in insulin-treated patients with diabetes. The IMPACT trial, involving 23 European sites and 241 T1DM patients with good glycemic control (HbA1c 6.7%) and no history of hypoglycemic unawareness, randomized 120 patients to FreeStyle Libre and 121 to SMBG. The study met the primary endpoint showing 38% reduction in time spent in hypoglycemia at 6 months with FreeStyle Libre while maintaining HbA1c (hypoglycemia reduction by 1.39 and 0.14 h/day in intervention and control arms, respectively; P < 0.001). 65 Interestingly, the reduction in hypoglycemia was evident within the first month of starting the trial and was sustained throughout. The REPLACE multicenter study, conducted in 26 European centers, investigated the use of FreeStyle Libre in 224 patients with T2DM on multiple daily injections of insulin. Individuals were randomized to intervention with FreeStyle Libre, while the control group continued to use SMBG (2:1 randomization). The primary endpoint was difference in HbA1c between study arms at 6 months. Although no significant difference in HbA1c was shown, individuals younger than 65 years demonstrated a reduction in HbA1c by 0.5% in the intervention arm compared with 0.2% in controls (P < 0.05). 66 Moreover, hypoglycemia was reduced by 43% in intervention compared with control arm (P < 0.001), associated with improvement in quality-of-life measures (P < 0.001). The reduction of hypoglycemia in both IMPACT and REPLACE, including nocturnal hypoglycemia, occurred despite the absence of an alarm in the FreeStyle Libre system.
In summary, CGM and FCGM use improves HbA1c and/or reduces hypoglycemia, although more studies in T2DM patients are needed to identify subgroups that would benefit the most from non-SMBG methods for glucose testing. The advantages and limitations of glucose testing methods are summarized in Figure 2.
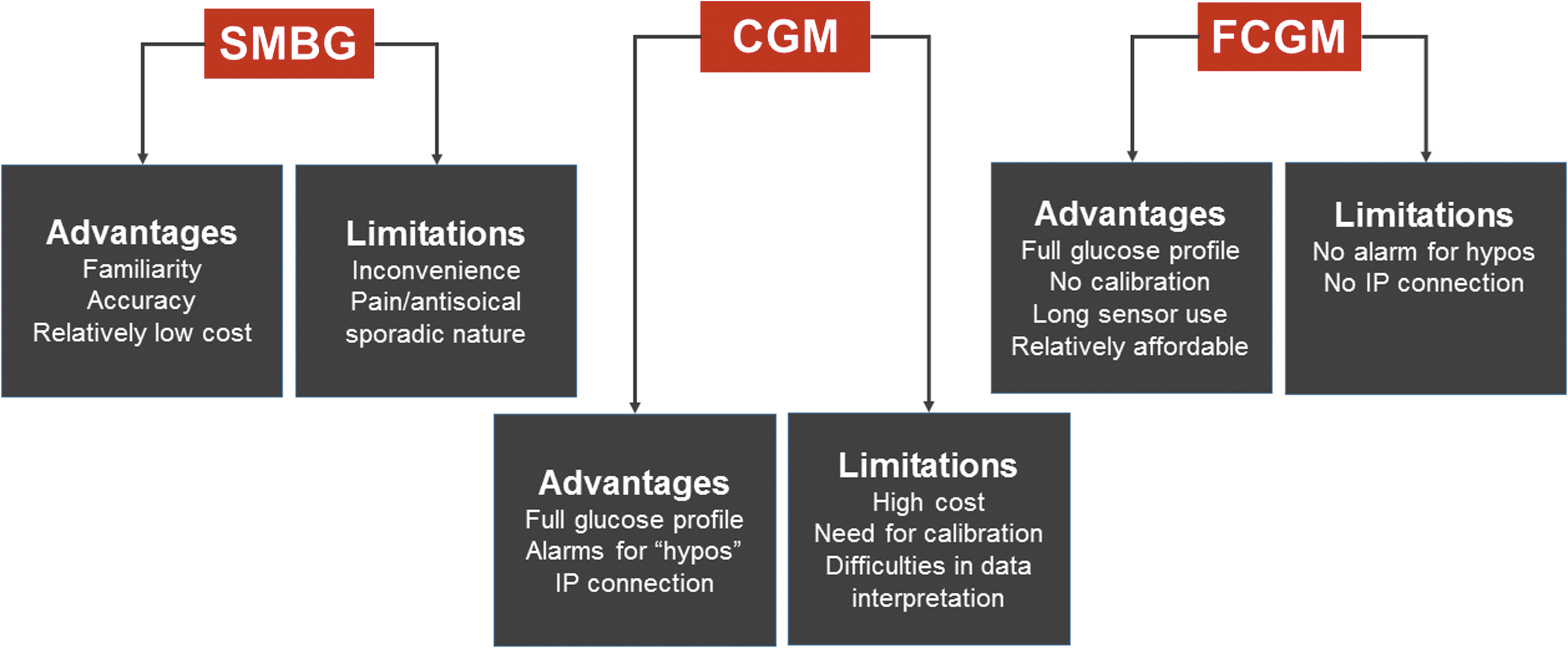
Advantages and limitations of SMBG, CGM, and FCGM. SMBG is inconvenient and fails to provide a complete set of glucose data. In contrast, both CGM and FCGM provide a comprehensive picture of glucose levels with the former having an alarm function and the ability to link with IP. CGM devices require constant calibration and current systems are expensive, whereas FCGM does not require calibration, enjoys long sensor life, and is relatively affordable. CGM, continuous glucose monitoring; FCGM, flash continuous glucose monitoring; IP, insulin pumps; SMBG, self-monitoring of blood glucose.
Accuracy of CGM Sensors in Clinical Practice
Accuracy of earlier sensors such as GlucoWatch G2 was disappointing with a MARD of 22%. However, this has significantly improved over the past decade with MARD gradually dropping to 13.8% with Enlite, 13.0% with Dexcom G4 platinum, 12.8% with FreeStyle Navigator, and an impressive 9.0% with Dexcom G4 platinum equipped with an advanced software. 68 Although of great clinical interest, head to head studies comparing different sensors are relatively scarce. In 24 patients with T1DM, Dexcom showed superior accuracy to Enlite sensor (Medtronic), demonstrating an MARD ± SD of 12.2% ± 12.0% and 19.9% ± 20.5%, respectively (P < 0.001). Accuracy suffered in the hypoglycemic range, but a difference between the two sensors was still evident (21.2% ± 21.8% and 36.2% ± 42.6%, respectively; P < 0.001). 69 Funding of the work by Dexcom and the direct supply of the sensors by the company were criticized, but another study, independent of either of the two companies, showed similar results with greater accuracy of Dexcom compared with Enlite in 38 subjects with T1DM (MARD of 13.9% and 17.8%, respectively; P < 0.001). 70
Despite not needing calibration, the new FreeStyle Libre shows good accuracy with a MARD of 11.4%, which is not affected by body mass index, age, type of diabetes, or treatment with insulin. 64 In a head to head comparison of Libre with Dexcom G4 in patients with T1DM, a good agreement was detected with a MARD in euglycemic, hyperglycemic, and hypoglycemic range of 15.8%, 13.4%, and 18.7%. 71 Subtle differences were detected such as lower glucose reading with Libre overnight, but this was minor and did not have management implications. 71 Compared with SMBG, both sensors showed a MARD <15% but it is worth stressing that Dexcom users had to calibrate their device twice daily, whereas those on Libre did not require calibration.
A key issue remains in relation to nonadjunctive use of CGM to make treatment decisions. The reluctance to rely only on CGM data is related to sensor delay and concerns over accuracy, particularly in certain situations, such as incorrect calibration, compression of sensor site, and sensor interfering substances (except for the Libre sensor) such as acetaminophen. One of the proposed MARD safety thresholds of 10% appears to be the current cutoff for nonadjunctive CGM use, which is met by Dexcom G4 platinum coupled with 505 software. 72 Indeed, a recent study using this device, REPLACE-BG, randomized 226 T1DM patients from 14 centers to CGM-only group (n = 149) or control group (CGM+SMBG, n = 77). Patients were adults, on insulin pumps, had HbA1c <9.0% (<75 mmol/mol) with almost half being CGM users before the study. Mean time in euglycemia, defined as glucose between 70 and 180 mg/dL, remained constant in both groups at 26 weeks compared with baseline, indicating that CGM can be used solely to monitor glycemia and adjust therapy. It should be noted, however, that SMBG was still needed in the CGM-only group at a mean of 2.8 tests/day (mainly to calibrate the device) as opposed to 5.4 tests conducted in the CGM+SMBG group. 73
Recommendations for CGM Use in Clinical Practice
Various guidelines have been developed for CGM use in patients with diabetes. However, given the general paucity of data, these guidelines have generally adapted a pragmatic approach by trying to address subgroups of patients who would most benefit from this glycemic monitoring strategy. The National Institute for Health and Care Excellence recommended CGM use for those whose HbA1c is poor despite at least 10 SMBG tests/day. 74 Moreover, patients with more than one episode of severe hypoglycemia/year, complete unawareness of hypoglycemia, frequent asymptomatic hypoglycemia compromising daily activities, and extreme fear of hypoglycemia were also recommended as candidates for CGM. Long-term CGM use, however, required demonstrating clinical benefit, including a significant drop in HbA1c.
The recent endocrine society clinical practice guidelines have generally simplified the criteria for CGM use. 75 Of note, panel members commissioned a systematic review and meta-analysis of RCTs to acquire a solid evidence-based platform for their recommendations. Pooled data from 11 RCTs on the use of real-time CGM in T1DM demonstrated that this glucose monitoring strategy alters HbA1c (95% CI) by −0.28% (−0.47, −0.09), which is primarily seen in patients older than 15 years. Therefore, use of real-time CGM was recommended for T1DM patients who have HbA1c above targets and who are willing, as well as able, to use the device on almost a daily basis. Also, it was highlighted that CGM reduces the risk of hypoglycemia, particularly with lower HbA1c levels. Intermittent CGM was suggested for insulin-treated T2DM patients, although it was acknowledged that the evidence in this group of patients is both weak and incomplete. The guidelines stressed the importance of patient education and support during periods of CGM use. Sensor-augmented insulin pump therapy as an effective tool to reduce nocturnal hypoglycemia was also recommended.
In summary, current evidence indicates that CGM use, with appropriate education and support, is advised for T1DM patients not achieving HbA1c targets with SMBG. Also, CGM is justified in T1DM individuals in whom hypoglycemia represents a significant clinical problem. CGM can also be used in T2DM patients with suboptimal HbA1c but only intermittently and long-term use is not advised. Finally, CGM is likely to be useful in pregnant women with diabetes, 63 although more studies are required in this group of patients.
FCGM is noted by its absence from current guidelines, which is not surprising given this is a new device that requires further assessment before robust recommendations can be made. However, limited evidence from two RCTs suggests a benefit in both T1DM and T2DM patients on multiple daily injections with insulin. 65,66 The rapid increase in FCGM users will provide further crucial data on the role of this glucose monitoring strategy in optimizing glycemic control in diabetes. It should be remembered that FCGM is a calibration-free device with longer sensor life but lacks alarm function and is not currently linked to an insulin pump. Therefore, clinical indications for the two glucose monitoring strategies can be different in some individuals and further studies are required to characterize these subgroups.
Conclusions and the Way Forward
Although HbA1c has been a useful clinical marker of glycemia, it reflects an approximate measure of glucose levels, displays large interindividual variability, and only provides a unidimensional analysis as it fails to address hypoglycemia and GV. Glucose testing by SMBG has been a cornerstone in glycemic management, but it is inconvenient to patients, particularly as a large number of daily tests are required for safe and effective changes in therapy. CGM certainly provides a more comprehensive assessment of glycemia, but uptake has been limited due to the need for constant calibration, high cost, and concerns over accuracy, in addition to difficulties encountered with data interpretation. However, with the advent of new and affordable glucose monitoring devices, such as FCGM, the widespread use of non-SMBG methods for glucose testing is becoming a possibility. Development of an algorithm for data analysis will further help in interpreting glucose data and make CGM/FCGM more user friendly for both patients and HCP.
The type of diabetes patient likely to benefit from CGM/FCGM has not been fully characterized with current evidence, suggesting that both of these systems are beneficial in individuals with T1DM by maintaining good glucose control while reducing the risks of hypoglycemia. CGM is also useful in individuals with a history of severe hypoglycemia or hypoglycemic unawareness and accumulating evidence suggests that some form of CGM is warranted in those with suspected hypoglycemia, particularly nocturnal.
Other groups that may benefit from CGM/FCGM include insulin-treated patients with T2DM, although studies in this group of patients remain surprisingly scarce. The heterogeneity observed in T2DM calls for additional work to further identify subgroups that would particularly benefit from this glucose monitoring strategy. For example, non-SGBG-based glucose testing is likely to be useful in individuals with diabetes following a major vascular event, such as myocardial infarction or stroke, those with advanced renal disease or blood disorders where HbA1c is less reliable as a marker of glycemia.
In addition to RCTs, observational studies using real-life CGM/FCGM data are required to fully assess the impact of these glucose monitoring strategies on glycemic measures and clinical outcomes in patients with diabetes. Main clinical benefits of CGM/FCGM are summarized in Table 1.
Self-monitoring of blood glucose is currently the most widely used method for measuring glucose levels with CGM reserved for more complicated patients with T1DM and T2DM. FCGM, which is similar to conventional CGM, is a relatively new form of glucose testing with studies demonstrating benefits in both T1DM and T2DM.
CGM, continuous glucose monitoring; FCGM, flash continuous glucose monitoring; T1DM, type 1 diabetes; T2DM, type 2 diabetes.
Widespread use of non-SMBG testing and the development of glycemic assessment protocols will familiarize both patients and HCP with alternative glucose testing methods, allowing for maximal clinical impact. Moreover, the increase in prevalence of non-SMBG devices will help companies reduce the price and further increase investment into improving accuracy and durability of sensors.
The “glycemic future” of patients with diabetes looks promising, which in turn will help to reduce complications, consequently improving quality of life and decreasing mortality in this high-risk population.
Footnotes
Acknowledgments
The author thanks the National Institute for Health Research, Diabetes UK, British Heart Foundation, and Abbott Diabetes Care for funding his glycemic research studies.
Author Disclosure Statement
The author has received research support and consultancy fees from Abbott Diabetes Care.
