Abstract
Background:
Computer clinical decision support (CDS) systems are uncommon in the pediatric intensive care unit (PICU), despite evidence suggesting they improve outcomes in adult ICUs. We reasoned that a bedside CDS protocol for intravenous insulin titration, eProtocol-insulin, would be feasible and safe in critically ill children.
Methods:
We retrospectively reviewed data from non-diabetic children admitted to the PICU with blood glucose (BG) ≥140 mg/dL who were managed with intravenous insulin by either unaided clinician titration or eProtocol-insulin. Primary outcomes were BG measurements in target range (80–110 mg/dL) and severe hypoglycemia (BG ≤40 mg/dL); secondary outcomes were 60-day mortality and PICU length of stay. We assessed bedside nurse satisfaction with the eProtocol-insulin protocol by using a 5-point Likert scale and measured clinician compliance with eProtocol-insulin recommendations.
Results:
Over 5 years, 69 children were titrated with eProtocol-insulin versus 104 by unaided clinicians. eProtocol-insulin achieved target range more frequently than clinician titration (41% vs. 32%, P < 0.001). Severe hypoglycemia was uncommon in both groups (4.3% of patients in eProtocol-insulin, 8.7% in clinician titration, P = 0.37). There were no differences in mean time to BG target or median BG between the groups. Mortality was 23% in both groups. Clinician compliance with eProtocol-insulin recommendations was 89%. Nurses believed that eProtocol-insulin was easy to understand and safer than clinician titration.
Conclusions:
eProtocol-insulin is safe for titration of intravenous insulin in critically ill children. Clinical research protocols and quality improvement initiatives aimed at optimizing BG control should utilize detailed computer protocols that enable replicable clinician decisions.
Introduction
S
In the adult intensive care unit (ICU), the use of computer clinical decision support (CDS) for BG control results in more consistent BG target levels and fewer adverse events than do paper protocols. 14 –16 To optimize BG control, investigators have implemented bedside paper protocols or guidelines for intravenous insulin titration in the pediatric ICU (PICU). 17 –19 However, CDS protocol use in the PICU is not widely accepted. 20 –22 Clinicians express concern about both the introduction of unseen risk and alienation from bedside decision making. 23 –25 Workflow burden and excessive time consumption are other stated barriers to bedside CDS implementation. 26 Ironically, it is excessive physician burden, and the associated information overload, that justifies CDS use. 26 –28
Detailed CDS protocols enable replicable physician decisions, can provide personalized patient care recommendations, and can lessen physician cognitive burden. 26 –31 In the adult ICU, eProtocol-insulin use resulted in superior achievement of the BG target with excellent clinician compliance (95%). 16,32 We modified eProtocol-insulin interface and insulin dose display recommendations for use in children and implemented eProtocol-insulin in the PICU. We used the same rules as used in the adult ICU. 16,32 We compared patients who received IV insulin therapy by unaided clinician titration (clinician titration) with those managed with eProtocol-insulin (eProtocol-insulin) to evaluate safety and feasibility of this CDS tool in the PICU.
Methods
We performed a retrospective descriptive cohort analysis of hyperglycemic (BG ≥140 mg/dL [7.8 mmol/L]) PICU patients who received intravenous insulin and were admitted between January 2003 and January 2008 (Fig. 1). Institutional review boards approved the study with a waiver of informed consent (University of Utah 00013681; Intermountain Healthcare 1008548). We compared treatment without a protocol or unaided clinician titration (clinician titration) with titration with a detailed, context-sensitive computer protocol that generated personalized care recommendations (eProtocol-insulin) (NIH-roadmap contract No. HHSN268200425210C); the rules and algorithm for eProtocol-insulin are published in the Supplementary Appendix A (Supplementary Data are available online at
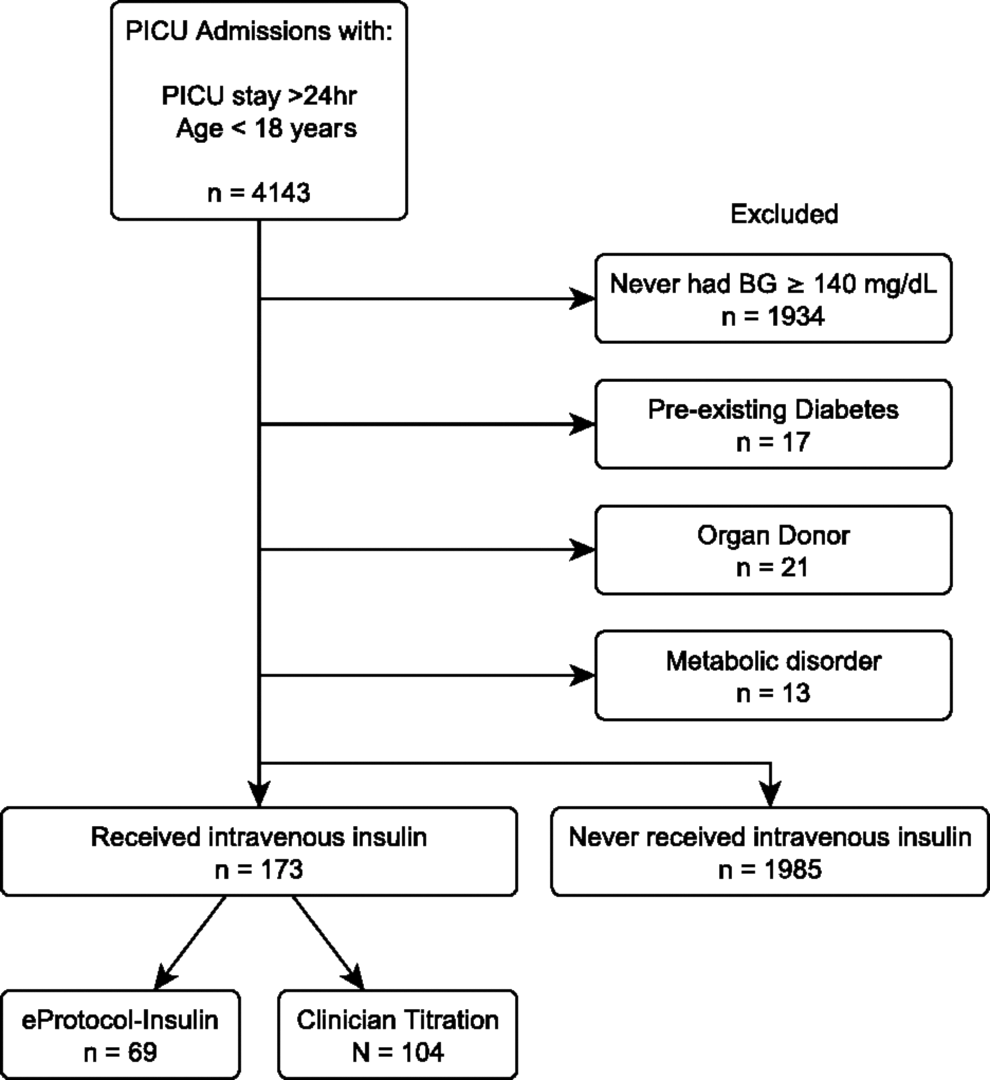
Inclusion diagram for comparison cohort.
Study site and subject selection
We included children <18 years old who remained in the PICU for ≥24 h, had at least one BG ≥140 mg/dL, and were treated with intravenous insulin. All patients were admitted to a 26–32 bed PICU (2000 admissions/year) within a 242–248 bed, university-affiliated, tertiary care pediatric hospital. PICU clinicians treated hyperglycemia with a BG target range of their choice for the entire study period. At the time of the study, there was not only enthusiasm for an 80–110 mg/dL target range but also concern about hypoglycemia, and, thus, a target range of 80–140 mg/dL was sometimes chosen. For the first half of the study period, insulin titration was directed by unaided clinician titration; for the second half of the study period, eProtocol-insulin was used. We focus the discussion on the 80–110 mg/dL BG target range but report relevant outcomes for both BG target ranges, since the BG target range chosen by the clinician was not recorded in the clinician titration group. For patients with multiple PICU admissions during the study period, we included only the first admission. We excluded patients who might have received insulin for diagnoses other than stress hyperglycemia (patients with diagnoses of diabetes, primary hypoglycemia, or medium-chain acyl-CoA dehydrogenase deficiency [ICD-9 codes: 250.00–250.03, 250.10–250.13, 250.31–250.33, 251.0–251.2, 775.6, 775.0, 270.3]).
Data collection
Data were obtained from the highly detailed Intermountain Healthcare Electronic Medical Record. Receipt of intravenous insulin was confirmed by a manual review of the chart. In addition to demographic data, we calculated the PRISM III score based on the first 12 h of PICU admission to determine illness severity. 33 We included BG values obtained from blood gas labs, bedside point of care, and clinical labs. If two or more BG values were obtained within 15 min of one another, only the first value was retained for the analysis. We calculated how long it took from the initiation of insulin to achieve target range BG (80–110 and 80–140 mg/dL) and what percent of readings during the first 120 h (5 days) were within target range. We assessed how many patients had at least one BG value ≤70 mg/dL (“mild hypoglycemia”) or ≤40 mg/dL (“severe hypoglycemia”). Because of known inaccuracies of low point-of-care BG values, we verified that all BG values ≤40 mg/dL were confirmed with a serum measurement at the same time. We calculated 60-day mortality and hospital- and PICU lengths of stay. We assessed bedside nurse satisfaction with a short survey of eProtocol-insulin based on a 5-point Likert scale (Supplementary Appendix B).
Statistical analysis
We compared the descriptive statistics and clinical outcome rates of the eProtocol-insulin and clinician titration groups. We compared central tendency with Wilcoxon rank-sum tests and proportions with chi-square and Fisher's exact tests. We adjusted the alpha level of statistical significance (0.05) for multiple comparisons (Bonferroni). We also fit a univariate logistic regression to assess the overall association between severe hypoglycemia and 60-day mortality.
Results
Demographics
Of the 2158 PICU patients, 173 (8%) were treated with intravenous insulin. We observed no differences in baseline characteristics or in antecedent hyperglycemia between eProtocol-insulin (n = 69) and clinician titration (n = 104) groups (Table 1).
Nine patients did not achieve glucose control (80–110 mg/dL): eight from the clinician titration group and one from the eProtocol-insulin group.
Three patients did not achieve glucose control (80–140 mg/dL): two from the clinician titration group and one from the eProtocol-insulin group.
PICU, pediatric intensive care unit.
Both eProtocol-insulin and clinician titration groups had similar BG values (median 116 vs. 118 mg/dL, respectively, P = 0.34), and time to the first BG value within target (10 vs. 12 h, P = 0.22 for 80–110 mg/dL; 7 vs. 6 h, P = 0.74 for 80–140 mg/dL). However, during the first 120 h of intravenous insulin administration, we observed a significant difference in the percentage of BG values within both target ranges (for 80–110 mg/dL: 41% of eProtocol-insulin group, 32% of clinician titration group, P < 0.001; for 80–140 mg/dL: 68% of eProtocol-insulin group, 62% of clinician titration group, P < 0.001). A total of nine patients did not achieve a BG value within the 80–110 mg/dL target range (one eProtocol-insulin group, eight clinician titration groups).
The distribution of BG values in both eProtocol-insulin and clinician titration groups is shown in Figure 2. We observed no statistically significant difference between the eProtocol-insulin and clinician titration groups for mild hypoglycemia (64% vs. 56%, P = 0.34). We observed no significant difference in occurrence of severe hypoglycemic BG values (0.08% in eProtocol-insulin vs. 0.20% in clinician titrated, P = 0.13), nor the percentage of patients who experienced severe hypoglycemia (4.3% vs. 8.7%, P = 0.37). For comparison, 3.4% of the patients who did not receive intravenous insulin had a severely hypoglycemic BG value. In the eProtocol-insulin group, clinicians chose the 80–110 mg/dL target 90% of the time, and patients received an average (SD) of 0.07 (0.06) U/(kg·h) of insulin with a mean duration of insulin infusion of 4.1 days.
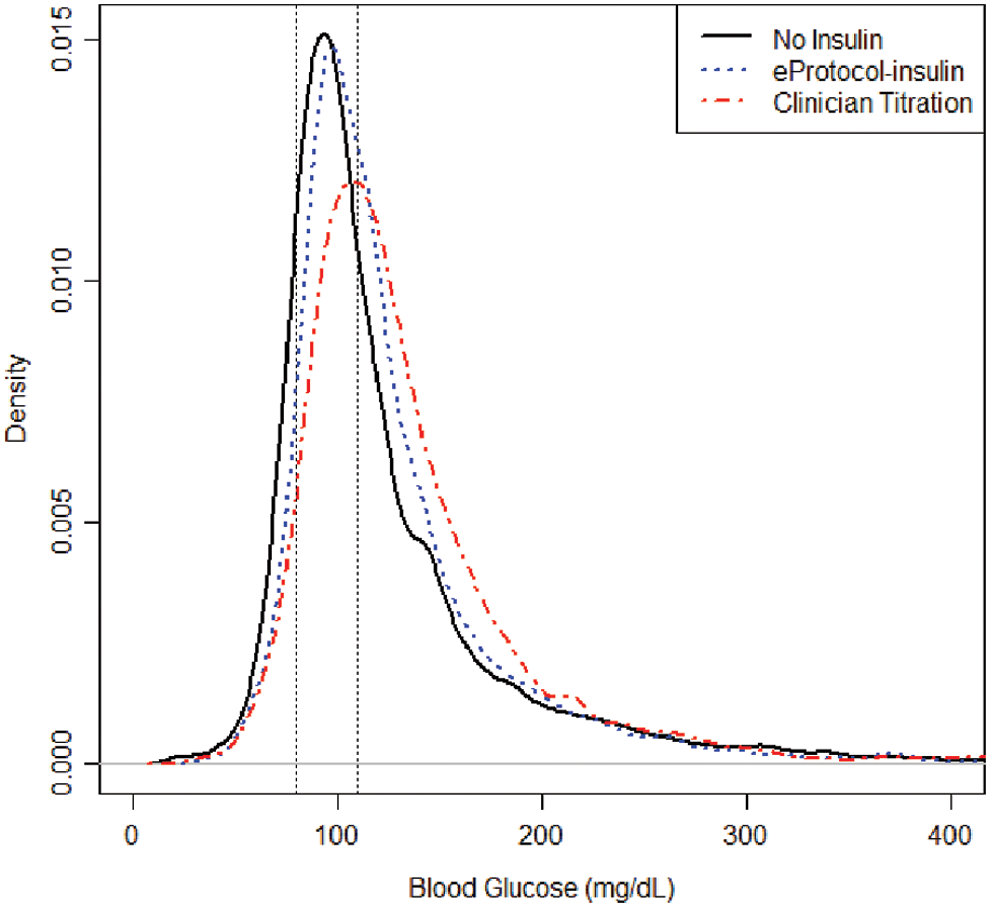
Distribution of all blood glucose values in pediatric intensive care unit for eProtocol-insulin versus clinician titration (values from patients who met inclusion criteria but did not receive insulin are also displayed for reference).
We observed no difference in mortality or PICU length of stay between eProtocol-insulin and clinician titration groups (Table 1). BG values were rarely ≤40 mg/dL, and this was not associated with mortality (univariate logistic regression Odds Ratio 0.468, P = 0.49).
eProtocol-insulin compliance
Clinician compliance with overall eProtocol-insulin recommendations was high, with 89% of recommendations accepted. Compliance with eProtocol-insulin recommendations was highest (92%) when the BG value was between 80 and 110 mg/dL and lowest (71%) when the BG value was below 60 mg/dL. Common reasons for non-compliance with eProtocol-insulin included administration of medications that interfere with insulin and glucose delivery, as well as physician disagreement with the recommendation. Technical problems with the computer were listed infrequently. A complete list of reasons for non-compliance are provided in the Supplementary Appendix C.
Nurse-rated feasibility and satisfaction
In general, nurses rated eProtocol-insulin on a Likert scale of 1 (disagree) to 5 (agree) as easy to understand (mean [SD]: 4.27 [0.59]) with clear recommendations (4.21 [0.89]). Nurses perceived eProtocol-insulin as slightly safer (3.85 [0.69]) than clinician titration and perceived it to be associated with less hypoglycemia (3.57 [0.85]).
Discussion
eProtocol-insulin, a bedside CDS protocol for insulin titration in the PICU, performs similarly, and more favorably in some regards, than clinician titration of insulin for the management of hyperglycemia in the PICU. eProtocol-insulin with a target of 80–110 or 80–140 mg/dL was safe and associated with a hypoglycemia rate similar to the rate in patients not receiving intravenous insulin. eProtocol-insulin facilitated collection of granular data (insulin dose, clinician chosen BG target) and enabled calculation of clinician compliance with protocol recommendations.
Our experience with a CDS protocol in the PICU reinforces objectives and goals outlined by Timbie et al. These include prioritizing clinician specialists' feedback in design and addressing important performance gaps. 31 eProtocol-insulin use resulted in more BG values in target range; furthermore, all but one patient in the eProtocol-insulin group were able to reach the 80–110 mg/dL BG target range. Nurse satisfaction with eProtocol-insulin was high, which was consistent with a previous observation that a bedside rounding tool enhanced overall PICU staff satisfaction. 34
PICU nurses perceived eProtocol-insulin as easy to use, with clear recommendations, safer, and associated with a decreased frequency of hypoglycemia. The latter perception is supported by our results, although the difference in BG value ≤40 mg/dL is not statistically significant. Interestingly, although overall clinician compliance with eProtocol-insulin was high (89%), compliance with eProtocol-insulin changed based on BG level. This finding provides valuable insight into complex elements surrounding clinician decision making regarding intravenous insulin titration and can help inform future insulin titration protocol development. 16,30,32 The ability to quantify bedside clinician compliance with an electronic protocol's recommendation is an important benefit of CDS protocols. 27,28
Compliance with eProtocol-insulin was higher than that noted by Tehrani et al. for a CDS protocol for mechanical ventilation in infants. 35 Unique features of eProtocol-insulin are its use as both a clinical management protocol for insulin titration and a potential research protocol. eProtocol-insulin facilitates collection of data that might better clarify the clinical and treatment circumstances that contribute to hypoglycemia, rendering a CDS protocol important for scientific rigor. 30,32 The data from the work we report in this publication were used to inform the design and study procedures for a large prospective multicenter interventional pediatric critical care trial—the National Institutes of Health Heart and Lung Failure Pediatric Insulin Titration Trial (HALF-PINT; U01-HL1-7681). HALF-PINT uses a similar CDS insulin titration protocol based on the proportional integral derivative algorithm. 6,36 –38
Reasons for different compliance with eProtocol-insulin recommendations across BG values are likely multifactorial. Inferences based on reasons given by the clinician for non-compliance include a desire not to change insulin when the patient is within the BG target. This could be due to the perceived nurse workload or from excessive cognitive burden that limits the ability of clinicians to anticipate the BG if he or she continued the current insulin dose or to predict the appropriate insulin dose if change is indicated. 26 Bedside clinician patient-specific judgment based on history and the administration of other medications may also contribute. More expansive future analysis of which recommendations were accepted and rejected is likely to be revealing. eProtocol-insulin enabled a preliminary understanding of the interaction between the protocol and the bedside clinician. 27,28 Our results argue against the claim that CDS protocols alienate the clinician from the patient interaction. Our results support the role of CDS tools to augment, not to replace, clinical judgment and to reduce cognitive workload. 26,39
Our study has several limitations. Our cohort samples are small, and we may have failed to detect clinically meaningful differences in hypoglycemia rates. Detected rates of hypoglycemia will also be dependent on the overall number of BG measurements per day. The clinician titrated group did not have a prescribed protocol for BG measurements, whereas eProtocol-insulin had explicit rules and recommendations. During the first half of the study, patients were treated by unaided clinician titration; during the second half, they were treated with use of ePrtoocol-insulin and we were unable to account for secular changes that may have influenced the treatment of hyperglycemia with insulin. Clinical perspectives regarding the best BG target also may have changed during the study period. eProtocol-insulin allowed the BG target range to be specified, whereas the clinician titration group did not. eProtocol-insulin also employed the same calculations and rules for insulin titration regardless of BG target range.
Despite the limitations cited earlier, the use of eProtocol-insulin for the management of hyperglycemia in the PICU is feasible, safe, and easily integrated into clinician work flow as a stand-alone bedside CDS system.
Conclusions
The eProtocol-insulin CDS protocol was safe, performed similarly to, and offered data collection advantages over clinician titration of IV insulin. Compliance with eProtocol-insulin recommendations was high, and bedside clinicians perceived it favorably. Clinical research protocols and quality improvement initiatives designed to determine preferable BG targets should utilize detailed CDS protocols that enable replicable clinician decisions.
Footnotes
Acknowledgments
Primary Children's Foundation Award; Intermountain Research and Medical Foundation Grant. Dean Sorenson PhD. Creator/programmer of eProtocol-insulin software.
Authors' Contributions
Conception (E.L.H., G.Y.L., K.A.S., A.H.M.), data acquisition (E.L.H., A.J., E.W., K.A.S.), data analysis (E.L.H., M.J.L., E.W.), writing the article (E.L.H., M.J.L., A.H.M.), revising the article for important intellectual content (all authors), and approval of final copy (all authors).
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
