Abstract
Objective:
Accuracy of continuous glucose monitoring (CGM) devices in hypoglycemia has been a widely reported shortcoming of this technology. We report the accuracy in hypoglycemia of a new version of the Dexcom (San Diego, CA) G4 Platinum CGM system (software 505) and present results regarding the optimum setting of CGM hypoglycemic alerts.
Materials and Methods:
CGM values were compared with YSI analyzer (YSI Life Sciences, Yellow Springs, OH) measurements every 15 min. We reviewed the accuracy of the CGM system in the hypoglycemic range using standard metrics. We analyzed the time required for the CGM system to detect biochemical hypoglycemia (70 mg/dL) compared with the YSI with alert settings at 70 mg/dL and 80 mg/dL. We also analyzed the time between the YSI value crossing 55 mg/dL, defined as the threshold for cognitive impairment due to hypoglycemia, and when the CGM system alerted for hypoglycemia.
Results:
The mean absolute difference for a glucose level of less than 70 mg/dL was 6 mg/dL. Ninety-six percent of CGM values were within 20 mg/dL of the YSI values between 40 and 80 mg/dL. When the CGM hypoglycemic alert was set at 80 mg/dL, the device provided an alert for biochemical hypoglycemia within 10 min in 95% of instances and at least a 10-min advance warning before the cognitive impairment threshold in 91% of instances in the study.
Conclusions:
Use of an 80 mg/dL threshold setting for hypoglycemic alerts on the G4 Platinum (software 505) may provide patients with timely warning of hypoglycemia before the onset of cognitive impairment, enabling them to treat themselves for hypoglycemia with fast-acting carbohydrates and prevent neuroglycopenia associated with very low glucose levels.
Introduction
T
The poor accuracy of previous generations of CGM devices in the hypoglycemic range may be an important factor in explaining the lack of evidence for the benefit of CGM in reducing the incidence of hypoglycemia. The recent American Diabetes Association (ADA) and Endocrine Society Workgroup on Hypoglycemia report discussed the use of CGM in patients at risk for hypoglycemia: “Recent technological developments have provided patients with new tools for glucose monitoring. Real-time CGM, by virtue of its ability to display the direction and rate of change, provides helpful information to the wearer leading to proactive measures to avoid hypoglycemia, e.g., when to think about having a snack or suspending insulin delivery on a pump. The CGM's audible and/or vibratory alarms may be particularly helpful in avoiding severe hypoglycemia at night and restoring hypoglycemic awareness.” 7 The ADA and Endocrine Society Workgroup on Hypoglycemia explicitly identified the potential problem associated with poor CGM accuracy in the hypoglycemic range: “Retrospective and real-time CGMs represent an evolving technology that has made considerable progress in overall accuracy. However, the accuracy of CGMs in the hypoglycemic range is poor.” 7
The ADA has recommended the use of 70 mg/dL for hypoglycemic alerts in order to give the patient adequate time to treat an impending hypoglycemic event and prevent a further decrease of the blood glucose level into more pronounced hypoglycemia. 8 As noted by Cryer 9 in an article on the appropriate glucose level for hypoglycemic alerts, 70 mg/dL approximates the lower end of fasting glucose and the threshold for the counterregulatory response in individuals without diabetes. In particular, he noted that “the recommended glucose alert level of 70 mg/dL…generally gives the patient time to take action to prevent a clinical hypoglycemic episode.” 9 A guidance document on CGM published by the Clinical and Laboratory Standards Institute noted the clinical motivation for the characterization of CGM alerts with following question on behalf of patients using these devices: “If my blood glucose levels go below my hypoglycemic alert threshold…, what is the likelihood that the CGM will alert me to the situation?” 10
The goal of a CGM hypoglycemia alert is to detect a hypoglycemic state before it progresses to a level of neuroglycopenia and cognitive impairment that would jeopardize the patient's ability to take action and recover without assistance from others. The clinical threshold for cognitive impairment may vary from individual to individual, depending on a variety of factors. 11 Nonetheless, there is a general consensus that neuroglycopenic symptoms are commonly observed at approximately 55 mg/dL. 12 Blood glucose measurements of less than 55 mg/dL are associated with a high risk of severe hypoglycemia requiring the need for assistance from others, cognitive impairment, seizures, or comas. 13 In this article, we examine the time between the CGM hypoglycemic alert and the actual blood glucose value crossing the threshold for biochemical hypoglycemia, defined as 70 mg/dL. We also examine the time between the CGM hypoglycemic alert and the actual blood glucose value crossing the threshold for cognitive impairment due to hypoglycemia, defined as 55 mg/dL.
The use of 70 mg/dL as the recommended threshold for hypoglycemic alerts is based on measurement of blood glucose and not interstitial fluid glucose as measured by CGM devices. The physiological delay between blood glucose and interstitial glucose may impact the time for the CGM system to indicate the presence of biochemical hypoglycemia. Although recent radiotracer studies have shown that the true physiological lag between blood glucose and interstitial fluid glucose is small, the lag time is not zero. 14 Interstitial fluid glucose may lag blood glucose by up to 10 min. 15 In cases of rapidly falling blood glucose levels, CGM devices with threshold alerts set at 70 mg/dL may not alert the patient for biochemical hypoglycemia until many minutes after the blood glucose value has crossed the threshold. In this article, we examined whether using a higher CGM alert threshold for hypoglycemia (80 mg/dL instead of 70 mg/dL) could mitigate the effect of interstitial fluid glucose delay relative to blood glucose during periods of rapidly falling glucose levels.
Materials and Methods
Data for the analysis included in this article were obtained from a pivotal study submitted for regulatory approval of the Dexcom (San Diego, CA) G4 Platinum (software 505) CGM system with a modified algorithm for converting the raw electrochemical sensor signal into calibrated glucose values. Details of the new algorithm have been reported previously as applied retrospectively to the original G4 Platinum pivotal study data. 16 In this article, the new algorithm was applied prospectively in an open-label, single-arm, multicenter, pivotal study. Study design, demographics, and overall results of the study for the new algorithm have been reported by Bailey et al. 17
Following screening, 51 subjects with diabetes mellitus were enrolled at three clinical centers in the United States. The G4 Platinum (software 505) reported by Bailey et al. 17 and discussed further here was approved by the U.S. Food and Drug Administration in November 2014.
All subjects wore one sensor for up to 7 days. Subjects participated in one clinic session lasting up to 12 h in order to obtain blood glucose measurements with a laboratory reference to assess the CGM accuracy. Venous samples were drawn approximately once every 15±5 min to allow for measurement of blood glucose level using a YSI 2300 STAT Plus™ glucose analyzer (YSI Life Sciences, Yellow Springs, OH). Glucose was safely manipulated to obtain values in the low glucose range and across the entire glycemic spectrum. Subjects and clinicians were blinded to CGM values and alerts during the clinic session. All subjects provided witnessed, written informed consent prior to enrollment.
Analysis methods
We report on the accuracy of the new version of the G4 Platinum CGM in the hypoglycemic range using standard methods of analysis such as the mean absolute difference and the percentage of points within (±) 10 mg/dL, 15 mg/dL, and 20 mg/dL of the reference value in the hypoglycemic range (40–80 mg/dL). Density bias or Bland–Altman plots are an accepted method for assessing performance of a novel measurement technology compared with an established reference measurement method. 18 In CGM, the y-axis of the Bland–Altman plot is typically the difference between the sensor and the reference, and the x-axis is the reference measurement alone. In order to assess whether the accuracy of the new CGM system improved relative to previous generations of CGM devices, we compare Bland–Altman plots in the hypoglycemic range for the original G4 Platinum and the new G4 Platinum (software 505).
We examined the impact of setting the CGM hypoglycemic alert at either 70 mg/dL or 80 mg/dL on two new metrics for assessing the clinical utility of CGM to prevent hypoglycemia. The first new metric is the biochemical hypoglycemia alert time (BHAT), defined as the time for the CGM system to alert to indicate hypoglycemia compared with the estimated time the YSI value crossed the 70 mg/dL threshold. The second new metric is the hypoglycemic cognitive impairment threshold alert (HCITA) time, defined as the time for the CGM system to alert compared with the estimated time that the YSI value crossed 55 mg/dL. The estimated time for the YSI value to cross a threshold was obtained from a linear interpolation between the two sequential decreasing YSI values on each side of the threshold value. The threshold for biochemical hypoglycemia and the threshold for hypoglycemic cognitive impairment are shown graphically in Figure 1.
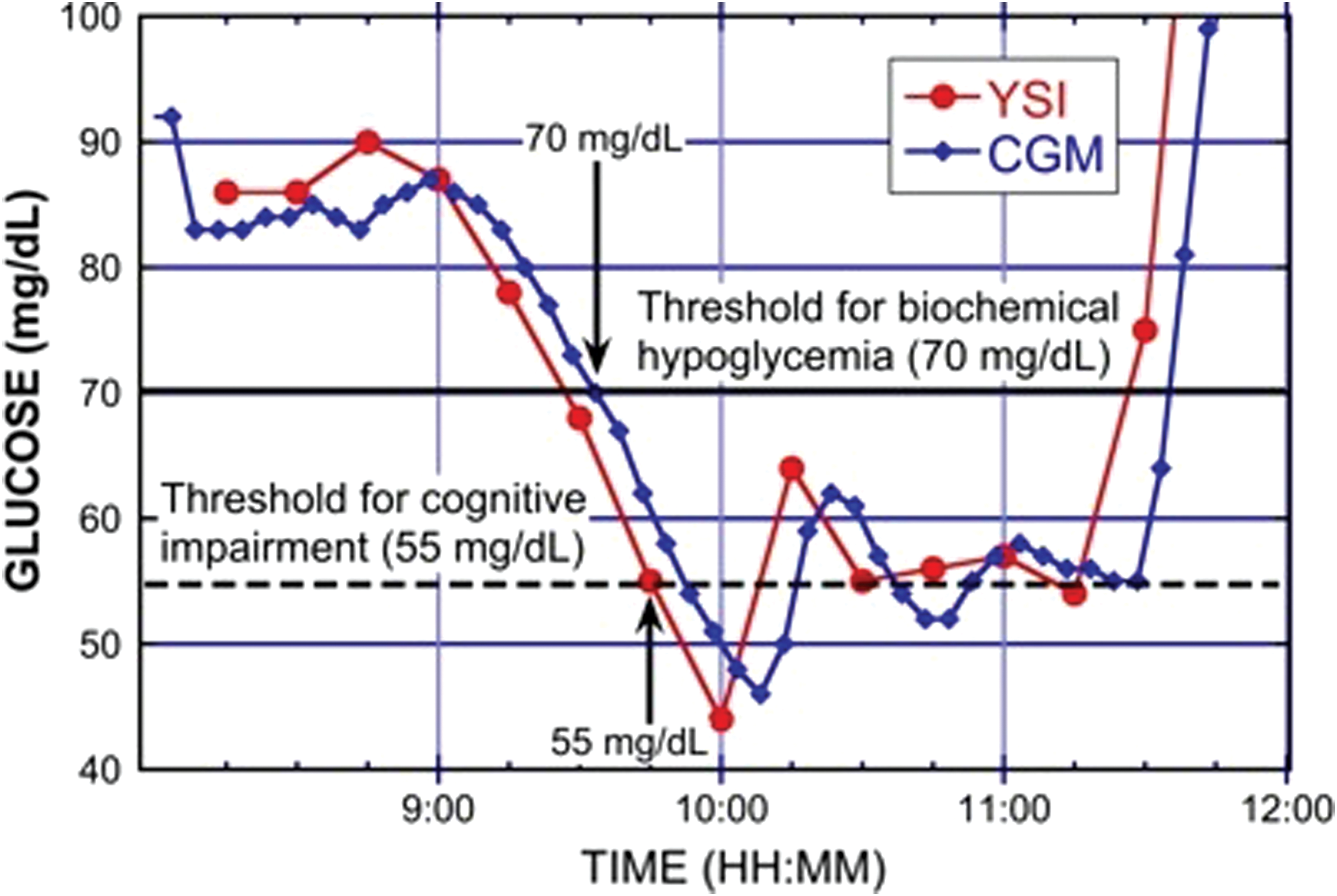
Temporal trace showing descent into hypoglycemia as measured from venous samples with the YSI (circles) and interstitial glucose values from the new G4 Platinum continuous glucose monitoring (CGM) system (diamonds). In this example, the CGM provided an alert to the onset of biochemical hypoglycemia (70 mg/dL) 5 min after the YSI analyzer value crossed the threshold and provided a warning 12 min before the YSI analyzer value crossed 55 mg/dL, defined here as the cognitive impairment threshold. Color graphics are available at
In our analysis, we provide data on the difference in time between when the YSI measurements were estimated to cross 70 mg/dL and the time the CGM system alerted for hypoglycemia for each episode of hypoglycemia observed during the in-clinic portion of the study as determined by YSI measurements. The data are plotted in a cumulative distribution function showing the percentage of two different CGM hypoglycemia alert settings compared with the time for the estimated YSI value crossing of the biochemical hypoglycemia threshold. Cumulative distribution functions are also used to show the percentage of two different CGM hypoglycemia alert settings compared with the time for the estimated YSI value crossing of the threshold for cognitive impairment.
We analyzed missed hypoglycemic alerts (or false-negatives) by defining a missed alert as the absence of a CGM hypoglycemia alert within 30 min of the YSI reading of 70 mg/dL. Similarly, we analyzed spurious hypoglycemic alerts (or false-positives) by defining a false alarm as a CGM alert for hypoglycemia without an estimated YSI reading of 70 mg/dL within 30 min of the CGM alert. This is consistent with the DirecNet consortium definitions, in which missed hypoglycemic alerts were defined as the absence of a CGM hypoglycemia alert within 30 min of the YSI interpolation crossing the 70 mg/dL threshold. 19 Similarly, we analyzed spurious hypoglycemic alerts as a CGM alert for hypoglycemia without an estimated YSI reading of 70 mg/dL within 30 min of the CGM alert. We analyzed the time difference between the CGM alert indicating hypoglycemia and the estimated YSI value crossing of the 55 mg/dL threshold. The data are plotted in a cumulative distribution function showing the percentage of CGM alerts for biochemical hypoglycemia compared with the time for the estimated YSI value crossing of the threshold for cognitive impairment due to hypoglycemia.
Results
Over the measurement range of 40–400 mg/dL, there were in total 2,263 matched pairs, 346 matched pairs with YSI reference data between 40 and 80 mg/dL, and a total of 288 independent YSI measurements of biochemical hypoglycemia defined as 70 mg/dL or less. There were 40 temporally distinct episodes of biochemical hypoglycemia based on venous reference sample measurements made with the YSI analyzer. There were 34 episodes of more pronounced hypoglycemia based on YSI measurements crossing the cognitive impairment threshold of 55 mg/dL.
The pivotal clinical study of the original Dexcom G4 Platinum CGM system found improved accuracy compared with previous generations of CGM devices over the entire physiological range (40–400 mg/dL). 20 Over the measurement range of 40–400 mg/dL, there were in total 9,093 matched pairs, 1,351 matched pairs with YSI reference data between 40 and 80 mg/dL, and a total of 1,057 independent YSI measurements of biochemical hypoglycemia defined as 70 mg/dL or less. The overall mean absolute relative difference was 13%, and the overall percentage of matched pairs between the CGM reading and venous reference measurement within 20% or 20 mg/dL was 82%. In the hypoglycemic range defined as 70 mg/dL, the mean absolute difference was 11 mg/dL. Independent studies of the original G4 Platinum CGM system have found similar high levels of accuracy. 21 –23 The pivotal clinical study of the new G4 Platinum CGM system (software 505) reported here gave an overall mean absolute relative difference of 9%, with 93% of all points either within 20% or 20 mg/dL of the reference measurement. 17 In the hypoglycemic range defined as 70 mg/dL, the mean absolute difference of the new G4 Platinum (software 505) was 6 mg/dL.
Figure 2A shows an expanded Bland–Altman plot for the hypoglycemic range (40–80 mg/dL) from the original G4 Platinum data with 52%, 71%, and 83% of all points falling within (±) 10 mg/dL, 15 mg/dL, and 20 mg/dL, respectively, of the laboratory reference YSI measurement. The frequency of measurements with a given bias is color-coded in the second y-axis, giving a density contour map of the distribution. The expanded Bland–Altman plot for the original G4 Platinum data shows a large number of points outside of 20 mg/dL in the hypoglycemic range. Figure 2B shows an expanded Bland–Altman plot for the hypoglycemic range (40–80 mg/dL) for the new G4 Platinum (software 505) with 76%, 90%, and 95% of all points falling within (±) 10 mg/dL, 15 mg/dL, and 20 mg/dL, respectively, of the laboratory reference YSI measurement.
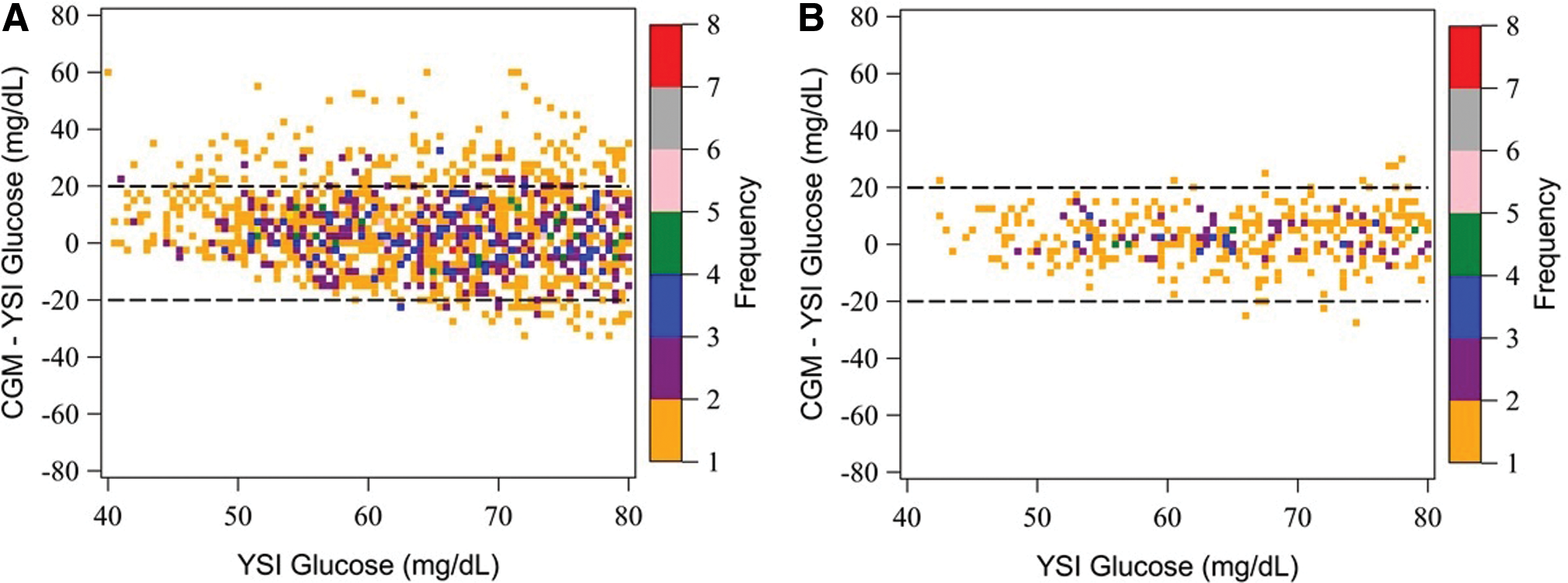
Density bias (Bland–Altman) plot of
Using the definition for a spurious alert given above of no greater than a 30-min temporal delay between the CGM hypoglycemia alert and an actual reference measurement indicating biochemical hypoglycemia, there were two instances of spurious alerts out of 41 episodes (5%) using the 70 mg/dL alert setting and six instances using the 80 mg/dL alert setting (15%). However, all six cases did involve actual instances of hypoglycemia. In two cases, common to both settings, the CGM device exhibited a negative bias of 20 mg/dL, possibly due to a calibration error, and provided a hypoglycemic alert more than 30 min in advance of the YSI value crossing the threshold for biochemical hypoglycemia. In the other four cases associated with the 80 mg/dL CGM hypoglycemia alert setting, the YSI value crossed the threshold for biochemical hypoglycemia more than 30 min after the CGM device alert not due to a calibration error but rather due to a slow descent into hypoglycemia captured equivalently on both the CGM and YSI measurements. There were five instances of missed alerts for the CGM alert setting of 70 mg/dL (12.5%) in which the YSI value crossed the threshold for biochemical hypoglycemia without a CGM hypoglycemic alert. There were, however, only two cases of missed alerts for the CGM hypoglycemia alert setting at 80 mg/dL (5%). In both these cases, the CGM system read high because of a calibration error and therefore did not alert for true hypoglycemic events.
Cumulative distribution functions for the BHAT at the CGM hypoglycemia alert settings of 70 mg/dL and 80 mg/dL are shown in Figure 3A. Cumulative distribution functions for the HCITAT are shown in Figure 3B.
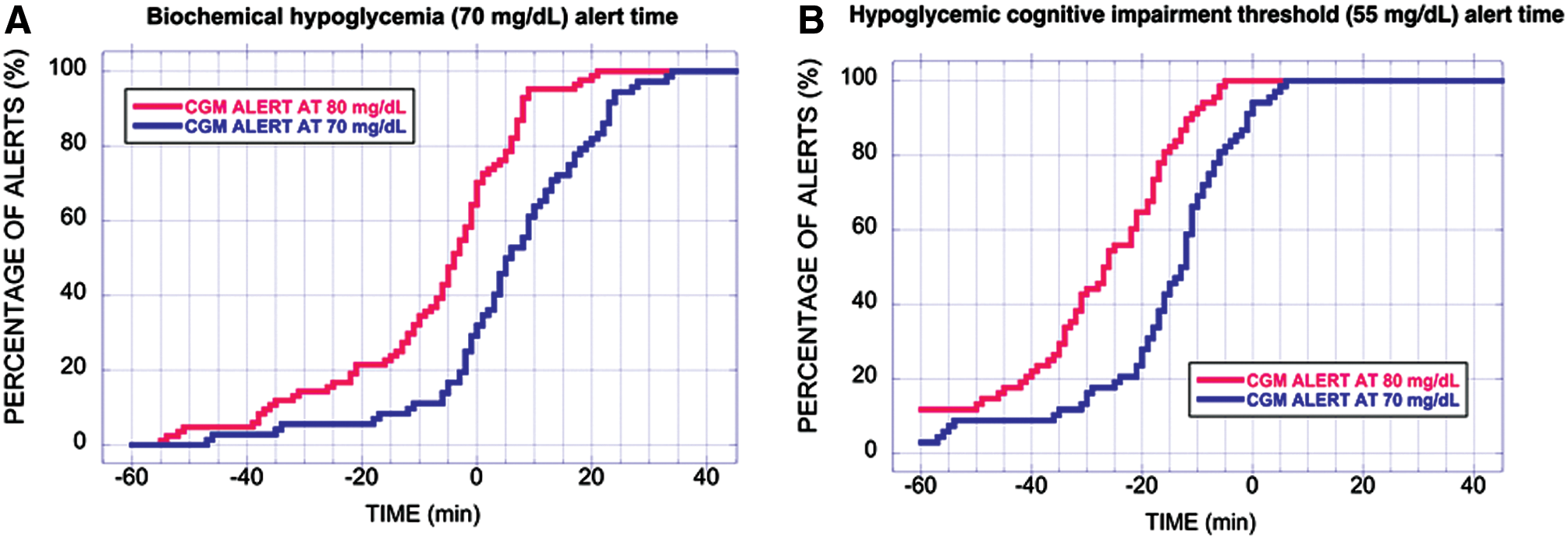
Cumulative distribution functions.
In the data shown in Figure 3A, the BHAT values are primarily positive numbers, indicating that CGM often alerts after the YSI has crossed the threshold for biochemical hypoglycemia. The 70 mg/dL CGM hypoglycemic alert setting provided an alert to the patient of a hypoglycemic state within 10 min of actual biochemical hypoglycemia in 62% of the instances observed in the study. By contrast, the 80 mg/dL CGM hypoglycemic alert setting provided an alert to the patient of a hypoglycemic state within 10 min of actual biochemical hypoglycemia in 95% of the instances observed in the study. In the data shown in Figure 3B, the HCITAT values are primarily negative numbers, indicating that the alert would provide the patient with warning of hypoglycemia before the YSI value has crossed the threshold for cognitive impairment and before patients experience an elevated risk for requiring the assistance of others for proper treatment of hypoglycemia. The 70 mg/dL CGM hypoglycemic alert setting provided an alert to the patient 10 min before the actual blood glucose level crossed the threshold for cognitive impairment in 68% of the instances observed in the study. By contrast, the 80 mg/dL CGM hypoglycemic alert setting provided an alert to the patient 10 min before the actual blood glucose level crossed the threshold for cognitive impairment in 91% of the instances observed in the study. The differences in BHAT and HCITAT for the two CGM hypoglycemia alert settings are given in Table 1.
The biochemical alert time is the time for the continuous glucose monitoring (CGM) value to indicate hypoglycemia compared with when the YSI analyzer value has crossed the 70 mg/dL threshold. The hypoglycemic cognitive impairment threshold alert time is the time for the CGM value to indicate hypoglycemia compared with the time when the YSI analyzer value crosses the threshold of 55 mg/dL.
Data in the top half of Table 1 show that the CGM alert setting of 70 mg/dL provided an alert for hypoglycemia within 10 min 62% of the time that the YSI value crossed the threshold for biochemical hypoglycemia. The CGM alert setting of 80 mg/dL provided an alert for hypoglycemia within 10 min 95% of the time that the YSI value crossed the threshold. Data in the bottom half of Table 1 show that the CGM alert setting of 70 mg/dL provided an alert for hypoglycemia 15 min before the YSI value crossed the 55 mg/dL threshold for cognitive impairment in 45% of the cases, 10 min before the YSI value crossed the 55 mg/dL threshold in 68% of the cases, and 5 min before the YSI value crossed the threshold in 82% of the cases. The CGM alert setting of 80 mg/dL provided an alert for hypoglycemia 15 min before the YSI value crossed the 55 mg/dL threshold for cognitive impairment in 82% of the cases, 10 min before the YSI value crossed the 55 mg/dL threshold in 91% of the cases, and 5 min before the YSI value crossed the threshold in 100% of the cases.
Discussion
Insulin-induced hypoglycemia in type 1 diabetes remains a major problem in the treatment and management of the disease. Weinstock et al. 24 reported from the T1D Exchange Registry that in all adults with available data (n=4,973), there was an 11.8% incidence in the previous 12 months of seizure or coma due to hypoglycemia. In subjects with a duration of diabetes of less than 20 years, 9% have had an episode of seizure or coma in the previous 12 months, whereas for those with a duration of diabetes of over 40 years, that number increased to 19%. These numbers are unacceptably high, so clearly new strategies to reduce the frequency of severe hypoglycemia are required. The routine use of CGM to date, however, for the prevention of hypoglycemia has been limited by persistent concerns about poor accuracy in the hypoglycemic range. The system reported here—the new Dexcom G4 Platinum CGM (software 505)—has improved accuracy in the hypoglycemic range compared with previous CGM devices. 25,26
The new alert metrics proposed here, assessing the time relationship between the CGM alert and glucose crossing the biochemical hypoglycemia and cognitive impairment thresholds, are clinically relevant because they provide a measure for the real-time, instantaneous potential benefit, or lack thereof, of CGM devices to identify significant hypoglycemia from the perspective of the patient. Use of the 80 mg/dL setting for the CGM hypoglycemia alert gave substantially more time than the 70 mg/dL alert setting for the patient to take corrective action to reverse hypoglycemia at the threshold for biochemical hypoglycemia and at the threshold for cognitive impairment due to hypoglycemia. However, using a higher threshold did result in a modest increase in spurious alerts.
Because most patients are trained to use the CGM hypoglycemia alert to treat themselves for hypoglycemia with fast-acting carbohydrates, the data presented above for the new G4 Platinum CGM provide patients with the opportunity to optimize the setting of the CGM hypoglycemic alerts for their personal preferences. The 80 mg/dL setting for CGM hypoglycemic alerts resulted in 5% missed alerts compared with 12.5% for the 70 mg/dL alert setting. Conversely, there were 15% spurious alerts at the 80 mg/dL CGM hypoglycemic alert setting compared with only 5% for the 70 mg/dL alert setting. Patients who want to minimize missed alerts might prefer the higher CGM hypoglycemic alert setting, whereas those who want to minimize spurious alerts might prefer the lower alert setting.
If the CGM can provide reliable and timely warning of hypoglycemia before patients have spent too much time below the cognitive impairment threshold of 55 mg/dL, there is a greater likelihood that they can follow current ADA treatment guidelines and treat themselves with short-acting carbohydrates without requiring the assistance of others. 27 In an article on severe complications of nocturnal hypoglycemia, Buckingham et al. 28 noted the “likelihood that prolonged or severe hypoglycemia is needed before there is a seizure.” This further underscores the clinical importance of the data presented above showing that setting the CGM hypoglycemia alert at 80 mg/dL provided advanced warning in all cases before the YSI value crossed the 55 mg/dL threshold.
The improved accuracy in the hypoglycemic range and the enhanced hypoglycemia alert performance reported here address a historic problem with CGM devices. Clinical studies are needed to determine whether this will lead to improved patient confidence and improved glycemic control in patients using this technology.
Footnotes
Disclosure Statement
T.A.P. has received funds for consulting from Dexcom, Tandem Diabetes Care, Insulet, and Profusa. K.N., D.P., L.C.B., and A.B. are employees of Dexcom and own stock of the company. I.B.H. has received research grants from Sanofi and Halozyme and consulting fees from Abbott and Roche.
T.A.P. was the primary author on the manuscript and was responsible for data analysis. T.A.P. is the guarantor of the contents of the article. L.C.B. assisted with data analysis and preparation of the manuscript. K.N., D.P., and A.B. were responsible for design and execution of the clinical trial and assisted with review and editing of the manuscript. A.B. and I.B.H. assisted with the development of the new metrics for hypoglycemia accuracy and with review and editing of the manuscript.
