Abstract
Self-monitoring of blood glucose (SMBG) is universally considered to be an integral part of type 1 diabetes management and crucial for optimizing the safety and efficacy of complex insulin regimens. This extends to type 2 diabetes patients on intensive insulin therapy, and there is also a growing body of evidence suggesting that structured SMBG is beneficial for all type 2 diabetes patients, regardless of therapy. However, access to SMBG can be limited in many countries in Central and Eastern Europe. A consensus group of diabetes experts from 10 countries in this region (with overlapping historical, political, and social environments)—Bulgaria, Croatia, Czech Republic, Hungary, Poland, Romania, Serbia, Slovakia, Slovenia, and Ukraine—was formed to discuss the role of SMBG across the spectrum of patients with diabetes. The group considered SMBG to be an essential tool that should be accessible to all patients with diabetes, including those with non–insulin-treated type 2 diabetes. The current article summarizes the evidence put forward by the consensus group and provides their recommendations for the appropriate use of SMBG as part of individualized patient management. The ultimate goal of these evidence-based recommendations is to help patients and providers in Central and Eastern Europe to make optimal use of SMBG in order to maximize the efficacy and safety of glucose-lowering therapies, to prevent complications, and to empower the patient to play a more active role in the management of their diabetes.
Self-Monitoring of Blood Glucose in Diabetes—The Evidence
Introduction to self-monitoring of blood glucose
S
The recent position statement from the American Diabetes Association and the European Association for the Study of Diabetes emphasized that individualized treatment goals and therapeutic strategies are the cornerstone of success in type 2 diabetes. 2 Knowledge of prevailing glucose trends through the use of SMBG, especially in those on insulin therapy, provides a means to tailor therapy and empower the patients to play a more active role in the control of their disease. This should involve appropriate patient SMBG skills training that is reinforced on a regular basis (e.g., yearly). Furthermore, it requires the availability of accurate, reliable SMBG devices, and there is currently scope for health authorities to improve the regulation and monitoring of such devices and to standardize the approval process. 3
Current SMBG devices are ultraportable, with some requiring measurement times of only 5 s and many allowing small sample sizes of less than 2 μL, 4,5 are able to calculate means, discern between premeal and postmeal glucose measurements, and store up to 500 results in their memory. The most recent development in glucose meters technology is embedding an automated bolus calculator with the goal to propose recommendations about insulin dosage to patients, and there is increasing evidence that these new smart devices make patients' decision-making safer. 6 At present, SMBG is widely used and widely reimbursed and is considered an integral part of treatment for people with type 1 diabetes and those with insulin-treated type 2 diabetes. 7,8 In one study, SMBG usage between 1993 and 2009 increased from 67% to 90% in insulin-treated type 2 diabetes patients and from 9% to 27% in type 2 diabetes patients on oral antihyperglycemic agent therapy. 9 However, there has been considerable controversy regarding the value of SMBG in non–insulin-treated type 2 diabetes patients, especially as the data (at least until recently) have been largely inconsistent 10 –13 —as such, this patient group is the major focus of the current article. Specific goals for performing SMBG by patients with type 1 and type 2 diabetes are summarized in Table 1.
SMBG, self-monitoring of blood glucose.
Also, SMBG use and reimbursement policies vary from country to country and from region to region. The differences are particularly visible in Europe, where countries belonging to one political and economic entity (i.e., the European Union) follow discrepant regimens in regard to recommending access to and usage of SMBG. As authors of this consensus article, we have analyzed the current evidence for SMBG and set it against the situation in 10 countries in Central and Eastern Europe. We represent the countries that have accessed the European Union in the last 10 years (Bulgaria, Croatia, Czech Republic, Hungary, Poland, Romania, Slovakia, and Slovenia) and those that aspire to be in the—even if unforeseen—future (Serbia and Ukraine). In all, we cover a large part of the territory between Germany and Russia, with a population of just over 100 million. The aim of this article is to help improve blood glucose control, prevent complications, and empower type 2 diabetes patients living in these populations via recommending evidence-based SMBG use.
What do the guidelines say on SMBG in diabetes?
Clinical practice guidelines universally recommend regular individualized SMBG as an essential tool for the optimal management of all patients with type 1 diabetes. 14 –18 The underlying rationale is based on consideration of several factors, including safety (detection and prevention of hypoglycemia), efficacy (enhancement of effectiveness of insulin through dose adjustment), and flexibility for the patient (e.g., regarding dietary choices and physical activity). The guidelines generally recommend that SMBG be performed at least three times per day in patients with type 1 diabetes and that it should include both fasting and postprandial glucose measurements. 14 –18 Guidelines from the International Diabetes Federation also recommend that SMBG should be performed frequently during pregnancy in diabetes, and in those with preexisting diabetes this will relate to any previous pattern of testing and insulin regimen. 7 The guidelines also recommend that women with gestational diabetes are instructed in SMBG and perform testing four times daily (fasting and 1 h after each meal). 7
Clinical practice guidelines that incorporate recommendations on the use of SMBG in type 2 diabetes have been issued by several national and international healthcare bodies. 7,8,14 –16,18 –20 Key recommendations from the International Diabetes Federation, the American Diabetes Association, and the National Institute for Health and Care Excellence in the United Kingdom are summarized in Table 2. The available guidelines recommend the use of SMBG in insulin-treated type 2 diabetes and generally suggest that SMBG may be beneficial in non–insulin-treated type 2 diabetes, especially when incorporated into a comprehensive education and management program, and this is supported by recommendations from expert consensus groups. 10,21 –24 The International Diabetes Federation guidelines, in particular, recommend that SMBG protocols (intensity and frequency) should be individualized to address the individual needs of each patient, as well as the requirements of the healthcare provider. The guidelines also recommend that healthcare providers should discuss the purpose of SMBG with patients and get agreement on how it should be interpreted and that patient skills and the impact of SMBG should be monitored. However, the evidence-based nature of these guidelines remains questionable. A recent review of current guidelines on SMBG in non–insulin-treated type 2 diabetes concluded that clinical practice guidelines were generally more in favor of SMBG use than the systematic reviews that were cited. 25 The authors also concluded that the citation practice in the guidelines was nonsystematic.
HbA1c, glycated hemoglobin; MDI, multiple-dose insulin; SMBG, self-monitoring of blood glucose.
The evidence supporting SMBG use in type 1 and type 2 diabetes
Type 1 diabetes and insulin-treated type 2 diabetes
In spite of the strong rationale supporting the use of SMBG in conjunction with intensive insulin therapy in type 1 diabetes, the formal evidence base is relatively limited. 12 In the Diabetes Control and Complications Trial, intensive insulin therapy guided by frequent SMBG delayed the onset and slowed the progression of microvascular complications compared with conventional therapy in patients with type 1 diabetes. 26 However, rigorous studies comparing SMBG versus no SMBG are not available and would not be considered ethical in this patient group. 12 Nevertheless, a meta-analysis of four early studies comparing SMBG with urinary glucose testing found that SMBG was associated with 0.6% greater reduction in HbA1c. 27 Further support comes from observational analyses in type 1 diabetes, and several studies have found a strong association between higher SMBG frequency and lower HbA1c levels. 28 –31 Evidence also suggests that more structured, focused SMBG intervention in suboptimally controlled patients with type 1 diabetes can provide significant improvements in HbA1c compared with regular guideline-based care. 32
Like in type 1 diabetes, the rationale for the use of SMBG in insulin-treated type 2 diabetes is compelling and is supported by data from large prospective and observational trials. 30,33,34 Similar to the Diabetes Control and Complications Trial, the Kumamoto study showed that intensive insulin therapy guided by SMBG can reduce the risk of microvascular complications compared with conventional therapy in type 2 diabetes. 33 In the Diabetes Outcomes in Veterans Study (DOVES), intensified SMBG improved glycemic control (0.3% reduction in HbA1c over 1 year) in a large cohort of stable, insulin-treated veterans with type 2 diabetes. 34 Benefits were only seen in patients whose SMBG compliance exceeded 75% or those with baseline HbA1c >8% (64 mmol/mol).
Non–insulin-treated type 2 diabetes
Prior to 2001, data supporting the use of SMBG in non–insulin-treated type 2 diabetes were limited, in spite of increasing SMBG use, leading to repeated calls for more studies in this patient group. 35,36 However, until recently, the majority of randomized controlled trials (nine out of 14 performed up to the year 2010) failed to show any significant benefit on metabolic control 37 –39 (for reviews, see Davidson 11 and Kolb et al. 12 ). The inconsistent findings have been attributed to differences in study designs, populations, and the interventions, and the SMBG protocols and structures may not have allowed adequate SMBG-guided management. 7,12,13 However, several subsequent meta-analyses of SMBG studies in non–insulin-treated people with type 2 diabetes have suggested that SMBG is associated with modest, but nonetheless statistically significant, reductions in HbA1c of between 0.2% and 0.4%. 40 –47 It is notable that SMBG appeared to be useful only when SMBG results were used to adjust therapeutic regimens and was more effective in patients with higher baseline HbA1c levels. 42 However, no associations with general health-related quality of life, general well-being, or patient satisfaction were reported. 44,45,47
Individual studies also suggest that more intensive SMBG in non–insulin-treated type 2 diabetes patients may not necessarily lead to changes in self-reported health behaviors. 48 A recent cross-sectional study found that SMBG was not associated with improved metabolic control in type 2 diabetes. 49 Indeed, patients with worse glucose control tended to perform SMBG more often. However, only half of patients modified their behavior because of abnormally high results of SMBG, highlighting the need for better education to make effective use of SMBG.
In terms of other outcomes, a recent randomized controlled trial suggests that the use of SMBG for 6 months can provide significant improvement in calculated coronary heart disease risk scores. 50 The retrospective observational ROSSO study found that SMBG was associated with reduced incidence of micro- and macrovascular events and all-cause mortality, irrespective of insulin use. 51 –53 On the other hand, longitudinal observational data from the Fremantle Diabetes Study found that SMBG was not independently associated with improved survival, and there were inconsistent findings relating to the association of SMBG with cardiac death and retinopathy. 54 A recent cross-sectional study in Turkey suggested that regular use of SMBG was not superior to irregular/never use of SMBG on glycemic control, but it seemed to be good intervention for prevention of diabetic nephropathy. 55 It is notable that recent economic analyses from the United States and Europe suggest that the SMBG can be cost-effective in non–insulin-treated type 2 diabetes and that its costs can be partly offset by a reduction in complications. 56 –58
Structured SMBG in non–insulin-treated type 2 diabetes
Structured SMBG is an approach in which blood glucose data are gathered according to a defined regimen, interpreted, and then utilized to make appropriate pharmacologic and/or lifestyle adjustments. 13 An early prospective, randomized controlled study looking specifically at structured meal-related SMBG in non–insulin-treated type 2 diabetes found that SMBG provided a significant improvement in HbA1c over 6 months compared with non-SMBG control (−1.0% vs. −0.5%, respectively; P<0.01) 37 (Table 3). Recently, large numbers of studies (at least 10 since 2010) have specifically investigated the use of structured SMBG in non–insulin-treated type 2 diabetes, and most of these have not yet been included in meta-analyses (Table 3). 59 –67 Other studies on structured SMBG in this patient population are underway. 68 It is notable that all but one of these structured SMBG studies in non–insulin-treated type 2 diabetes found a significant benefit. 13,61 These results thus demonstrate a vast improvement in consistency.
BMI, body mass index; BP, blood pressure; CIMT, carotid intimal-medial thickness; HRQoL, health-related quality of life; LDL, low-density lipoprotein; MAGE, mean amplitude of glycemic excursions; OA, oral (glucose-lowering) agent; SF-36, 36-item Short Form; SMBG, self-monitoring of blood glucose; T1DM/T2DM, (type 1/type 2) diabetes mellitus.
In the St Carlos Study—a prospective, randomized clinic-based interventional study with parallel groups involving well controlled newly diagnosed type 2 diabetes patients—SMBG used as part of a structured educational program was associated with significantly greater reductions in median HbA1c (from 6.6% to 6.1%; P<0.05) and body mass index (from 29.6 kg/m2 to 27.9 kg/m2; P<0.001) over 1 year compared with a HbA1c-based control group. 60 Furthermore, the percentage of patients achieving a lifestyle score of >12 was significantly greater in the SMBG compared with the control group (38.4% vs. 9.7% respectively; P<0.001). In addition, pharmacological treatment changes occurred earlier and more frequently in the SMBG group.
One key recent trial was the Structured Testing Program (STeP) study—a cluster randomized trial in poorly controlled insulin-naive type 2 diabetes patients. 67 Primary care practices were randomized to the active control group or the structured testing group, the latter of which included quarterly review of structured SMBG results. The structured testing group patients used a paper tool that graphs seven-point glucose profiles over 3 consecutive days, and physicians received a treatment algorithm based on SMBG patterns. Among those who received at least one treatment modification recommendation, structured testing group patients demonstrated a greater reduction in HbA1c at 12 months compared with active control group patients (−1.2% vs. −0.8% [−13.1 vs. −8.7 mmol/mol]; P<0.03). The results suggest that collaborative use of structured SMBG data leads to earlier, more frequent, and more effective treatment modification recommendations for poorly controlled patients. Further analyses from this study found that structured SMBG led to significant increases in self-confidence and autonomous motivation associated with diabetes self-management. 69
The ROSSO-in-praxi study was an uncontrolled trial that evaluated the impact of a relatively inexpensive 12-week structured SMBG-based lifestyle intervention on metabolic and other outcomes. 62 Participants were advised to perform a seven-point SMBG diurnal profile every 4 weeks and were also urged to perform event-driven SMBG measurements. Participants significantly reduced weight, body mass index, waist circumference, blood glucose, blood pressure, low-density lipoprotein cholesterol, and HbA1c by 0.3% (3.3 mmol/mol) (all P<0.001), alongside increased physical and mental health and reduced depression measurements. After long-term follow-up of 2 years, the reduction in HbA1c was only maintained in those who continued with daily SMBG. 70 The Rosso-in-praxi-international study used the same intervention but randomly assigned patients to either SMBG or a non-SMBG control group. 70 After 12 weeks, the SMBG group had significantly improved HbA1c levels (from 7.4% to 6.9% [from 57 to 52 mmol/mol]; P<0.001) and weight (−0.9 to −1.9 kg; P<0.05), whereas changes were not significant in the control group. After 1.5 years of follow-up, HbA1c remained stable in the SMBG group but increased in the control group. It is notable that those who measured their blood glucose more than three times per week demonstrated an overall reduction in HbA1c of 1.0% (11 mmol/mol) (P=0.006 vs. three times or fewer per week) after 1.5 years. Another recent study also suggests that intensification of relatively simple structured SMBG policy (one profile every 2 weeks with pre- and postprandial values vs. one profile every month with fasting and postprandial values) can provide HbA1c improvements, as long as compliance is sufficient. 59
SMBG and hypoglycemia
Although the evidence for an association between SMBG and glycemic control is now extensive, the data remain more limited for hypoglycemia in non–insulin-treated type 2 diabetes. The few studies with hypoglycemia data have reported inconsistent findings, which likely reflect the inconsistent criteria used to define hypoglycemia, the low frequency of events in non–insulin-treated patients, and the relevance of the control groups when looking at this outcome. 38,72 –74 The study by O'Kane et al. 74 found no significant difference in hypoglycemia between SMBG and non-SMBG groups, whereas Farmer et al. 73 reported a statistically significant increase in the frequency of grade 2 hypoglycemic episodes with SMBG versus control. In the study by Guerci et al., 72 hypoglycemia (symptomatic or asymptomatic) was reported by 10.4% of subjects in the SMBG group and 5.2% in the non-SMBG group (P=0.003), but the difference was solely due to a significant difference in asymptomatic events. In the DINAMIC 1 study, symptoms suggestive of mild to moderate hypoglycemia were reported by 27 subjects (8.7%) in the SMBG group, which included 51 events in total (27 symptomatic, 11 asymptomatic, 11 SMBG-confirmed hypoglycemia, and two non-graded). 38 In the non-SMBG group, mild-to-moderate hypoglycemia was reported by 7.0% of subjects (66 events in total), with 64 symptomatic and two non-graded events. Thus, there was a decrease in symptomatic events with SMBG. This is to be expected, as patients in the SMBG groups are able to use their device to both detect asymptomatic episodes (that might otherwise go unreported) and also to confirm symptomatic episodes. 47 Evidence for severe hypoglycemic episodes is limited, but some evidence suggests that over half of severe hypoglycemic events in type 1 or type 2 diabetes can be predicted by specific glucose fluctuation patterns (based on at least three SMBG readings) in the 24 h before an episode. 75
SMBG and current controversies
Recently, two major debates around SMBG are thriving. One concerns accuracy of SMBG devices, as glucose meters are increasingly expected to deliver most accurate results. Independently conducted studies show that over 80% of marketed devices in Europe meet the high system accuracy requirements of DIN EN ISO 15197:2003, and all of them show acceptable measurement reproducibility. 76
A second debate, even more heated, evolves around cost-effectiveness of SMBG, particularly in patients not treated with insulin. However, more and more studies show that having considered long-term impact of increased risk of developing vascular complications SMBG is cost-effective across the whole spectrum of diabetes patients. 57,77,78
Conclusion on SMBG evidence
In spite of the recent increase in the number of studies consistently showing a benefit of structured SMBG in non–insulin-treated type 2 diabetes, the relative merits of improved glycemic control versus expense and inconvenience continue to be debated. 79 –81 The data suggest that SMBG may benefit both well-controlled and poorly controlled patients not on insulin therapy. Recent consensus statements have acknowledged the increase in high-quality efficacy data, but also highlight the need for further well-defined studies with end points beyond HbA1c. 21,24 Guidelines of SMBG may require updating in light of the large number of recent studies showing benefits on metabolic and other outcomes. Data on the impact of SMBG on hypoglycemia remain scant, and this is an area that warrants further study. However, the available evidence suggests that SMBG can help patients to detect asymptomatic episodes and also to confirm symptoms suggestive of hypoglycemia, which may be particularly appropriate in those with impaired hypoglycemia awareness. Indeed, hypoglycemia unawareness is one of the utmost important indications for frequent SMBG. The importance of hypoglycemia avoidance is exemplified by growing evidence for an association between a history of severe hypoglycemic episodes and serious adverse outcomes over the long term, including death, major vascular events, and dementia. 82,82
It has been suggested that the utility of SMBG can be improved through attention to several key factors, including (1) sufficient SMBG frequency and appropriate timing (and the use of a more structured approach), (2) education and skills training for patients, (3) education and skills training for physicians and nurses or educators, and (4) the use of easy-to-read ways of displaying SMBG profile data that make sense to patients, physicians and nurses, or educators. 81 Such strategies should aid motivation and adherence to SMBG and remove barriers to optimal SMBG implementation. 84 Furthermore, appropriate education and regular patient monitoring may also help to avoid inappropriate overuse of SMBG, which has cost implications and may adversely affect patient quality of life. 85 Other factors, such as use of the palm rather than finger testing, may be beneficial in some patients. 86 It is interesting that some evidence suggests that SMBG values appear to predict HbA1c better in patients on oral therapy compared with patients on insulin therapy. 87
Practice nurses and other nurses working in the community are well situated to support people with diabetes to make effective use of SMBG. 88 They can play a key role first in identifying those patients who are most likely to benefit from SMBG and second in providing suitable education and problem-solving skills. 88 The latter role should involve education on the purpose of SMBG and the techniques required to use specific glucose meters and strips effectively. 88 Furthermore, patients should be taught how to interpret and use SMBG test results to adjust treatment, diet, and other lifestyle factors, thus empowering them to manage their diabetes more optimally and, it is hoped, avoiding hypoglycemic episodes and improving glycemic control. 88 Actually, having practice nurses—or even better, diabetes nurse specialists or educators—is a prerequisite of successful diabetes care as they are the best suited to provide essential and advanced diabetes education. At present, with doctors often unable to devote sufficient time for individual visits, a patient may obtain the necessary knowledge of how to manage her or his disease either from a professionally trained diabetes nurse or from no one. It is regrettable that in all countries from Central and Eastern Europe, nursing in diabetes is still in its infancy, with educational infrastructure and human resources being slowly built.
Unfortunately, it appears that no general agreement exists at present between healthcare professionals regarding the advice on various aspects concerning SMBG (e.g., on frequency, timing, and practical considerations) highlighting the need for education and more uniform guidelines. 89,90 A recent European expert panel has provided recommendations regarding frequency and timing of SMBG also for various clinical scenarios and suggested a less intensive and an intensive scheme for SMBG across the type 2 diabetes continuum, depending on clinical circumstances and the quality of glycemic control. 21 The expert panel also recommended further evaluation of various schemes for SMBG in type 2 diabetes in clinical studies. 21 However, as noted by the International Diabetes Federation, SMBG should only be used when individuals with diabetes and/or their healthcare providers have the knowledge, skills, and willingness to incorporate SMBG monitoring and therapy adjustment into their diabetes care plan. 7,8 The key word that should always accompany “SMBG” in non–insulin-treated diabetes patients is “structured,” and as long as patients and their care providers do not learn to use these two together, SMBG will never develop to its full potential in diabetes control. There is a sufficient body of evidence to show that structured SMBG is beneficial for all type 2 diabetes patients, regardless of the therapy they are using.
SMBG in Central and Eastern Europe—The Reality
The Central and Eastern European countries represented in this statement make up a group with relatively limited financial resources by the standards of many Western European nations (Table 4). In terms of gross domestic product (purchasing power parity) per capita, they have a relatively broad range, from $7,374 (Ukraine) to $28,195 (Slovenia). 91 Thus, they overlap with the poorest Western European nations (approximately $23,000–27,000 for Portugal, Greece, Malta, and Cyprus) but are all well below the levels of the major Western European economies (approximately $35,000–40,000 for France, the United Kingdom, and Germany) and are all below the European Union average ($32,021). In spite of this financial disadvantage, the available evidence suggests that, in general, patients with diabetes in Central and Eastern Europe receive a quality of care (e.g., in terms of control of both glycemic levels and cardiovascular risk factors) comparable to patients in Western Europe, although there are many potential areas for development. 92 Any disparities with Western European countries are related mainly to limited economic resources. 93 Cost-effective improvements in care are therefore highly relevant to the countries represented in this statement, some of which (e.g., Poland and Slovenia) have a particularly high burden of diabetes, in terms of both prevalence and healthcare spending, and many people with diabetes remain undiagnosed (Table 4). As in Western Europe, both glycemic control and cardiovascular risk factor control remain unsatisfactory in many patients, and there is a continuing need for improvements in education and management guidelines, as well as access to cost-effective drug therapies and devices, such as those required for SMBG. 92,93
Adult data are for the 20–79 year age range.
Source for total population and GDP data was the IMF World Economic and Financial Surveys. 91 Source for adult population data was the IDF Diabetes Atlas, 5th ed. 99
Gross domestic product (GDP) based on purchasing-power-parity (PPP) per capita GDP (PPP takes into account differences in the cost of living between countries).
Source for IDF estimates was IDF Diabetes Atlas, 5th ed. 99
National prevalence provides a measure of actual DM burden with a country. Comparative prevalence adjusts for age and allows between country comparisons independently of population age distribution.
Sources were Diabetes. The Policy Puzzle: Is Europe Making Progress?, 3rd ed. 100 and IDF Diabetes Atlas, 5th ed. 99
DM, diabetes mellitus; IDF, International Diabetes Federation; OA, oral (glucose-lowering) agent; SMBG, self-monitoring of blood glucose; T1DM/T2DM, type 1/type 2 diabetes mellitus.
In most countries in the region, diabetes care is provided by both general practitioners and diabetologists/endocrinologists, with patients typically managed by general practitioners, in the first instance, with subsequent referral to specialists if insulin treatment is required or where there is deterioration of glycemic control. However, in some countries (the Czech Republic, Romania, Slovakia), the majority of patients are managed by specialists. Diabetes education is typically provided by doctors, nurses, or dieticians, although access to structured education may be limited in some countries.
Reimbursement for glucose meters and testing strips varies widely across the region (Table 4). Depending on the country, glucose meters may be fully reimbursed (typically for children/adolescents, patients on insulin, and during pregnancy), partially reimbursed, or not reimbursed at all. However, glucose meters may also be distributed free of charge by the manufacturers in some instances. Similarly, there is wide variation in the levels of reimbursement for glucose test strips, in terms of both patient eligibility and the number of strips provided. In most countries, strips are reimbursed for patients on insulin therapy, but the number of strips may depend on the number of daily injections and other factors such as age (see Table 4). Some countries in the region still have no specific clinical guidelines on the use of SMBG.
Recommendations
SMBG is an essential tool that should, as a minimum, be accessible to all patients with diabetes, including those with non–insulin-treated type 2 diabetes. Like other treatments, SMBG needs to be individualized by the physician in partnership with the patient, and guidelines can play an important role when deciding on an appropriate level of intervention. The ultimate goal of any SMBG strategy is to maximize the efficacy and safety of glucose-lowering therapies and to empower the patients to play a more active role in the management of their diabetes. Unfortunately, access to SMBG can be limited in many countries in our region of Central and Eastern Europe. To some extent, this may reflect the lack of local SMBG guidelines, which is in stark contrast to the extensive guidelines available for glucose-lowering drug therapy. As we come from similar historical, political, and social environments, it seems appropriate for us to make some recommendations for SMBG in diabetes that would be applicable to the region as a whole (summarized in Figure 1). It should, however, also be stated that it would take major changes in several countries (most notably Bulgaria, Romania, and Ukraine) to introduce the following recommendations into routine practice. Nevertheless, we are convinced that these changes are necessary for the patients' well-being and that it is only a matter of time until clinical practice in our region changes in the directions outlined below.
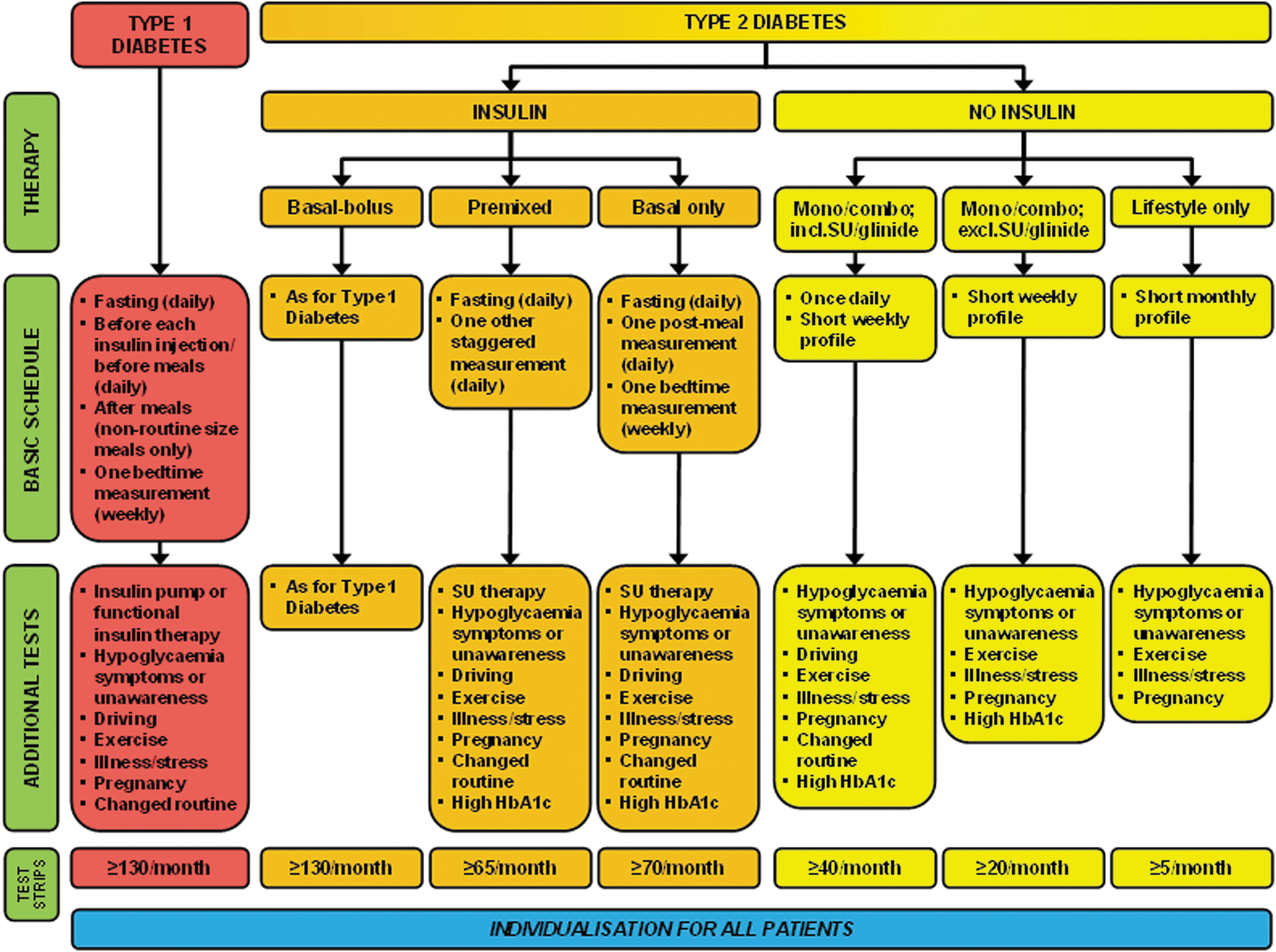
Consensus recommendations for self-monitoring of blood glucose in diabetes. HbA1c, glycated hemoglobin; SU, sulfonylurea.
The grading of the recommendations has been conducted according to the adopted system proposed by the U.S. Agency for Health Care Policy and Research as shown in Table 5. 94
Type 1 diabetes
It is impossible to treat type 1 diabetes safely without SMBG. Patients with type 1 diabetes are at risk of both severe hypoglycemic episodes and acute hyperglycemic crises, as well as the long-term microvascular risk associated with poor overall glycemic control. As such, SMBG is an integral part of type 1 diabetes management and crucial for optimizing the safety and efficacy of the complex insulin regimens required in these patients (Grade A). The minimum requirements for SMBG in patients with type 1 diabetes are (1) daily testing of fasting glucose, (2) premeal testing before every insulin injection, (3) postmeal testing after non–routine-size meals, and (4) at least one bedtime measurement per week (Grade C). This should be sufficient to obtain a full SMBG profile under normal conditions and to allow patients to identify periods of hyper- or hypoglycemia and to adjust insulin doses and diet accordingly. The bedtime measurement should help patients make adjustments to avoid nocturnal hypoglycemia. Postmeal testing addresses the potential risk of vascular complications, which is well documented in type 2 diabetes, but obviously cannot be excluded in patients with type 1 diabetes either. 95 However, additional testing should be implemented as often as required in patients experiencing symptoms of hypoglycemia or those with impaired hypoglycemia awareness, and therefore no limit should be imposed, in justifiable cases, on the number of test strips that a patient can access. Patients using insulin pump therapy and more intensive “functional” insulin regimens also usually require more frequent testing. Furthermore, additional testing should also be performed before driving, during pregnancy, during periods of illness, stress, or disruption of normal routine, and before, during, and after exercise, as well as more generally in any patient deemed to be at increased risk of hypoglycemia (Grade A).
Insulin-treated type 2 diabetes
Patients with type 2 diabetes on insulin therapy represent a heterogeneous group, as insulin regimens can vary widely from a single daily injection of long-acting insulin (typically added to existing oral glucose-lowering therapy) to the complex basal-bolus regimens used in type 1 diabetes. Furthermore, the progressive nature of type 2 diabetes and the frequent need for intensification of therapy mean that SMBG requirements may evolve over time. As well as an increase in the need for insulin therapy with more advanced disease, patients may become more prone to hypoglycemia due to impaired glucose counterregulation (associated with declining insulin production, increasing glucagon release, and attenuated increase in adrenaline) and the development of hypoglycemia unawareness (the result of attenuated increase in sympathoadrenal activity). 96 As such, it is difficult to make general SMBG recommendations for this patient group, and the pattern and frequency will depend on the particular insulin regimen and use of other specific glucose-lowering drugs, as well as the intrinsic risk of hypoglycemia. For patients on a single injection of long-acting insulin, the minimum SMBG requirement should be two measurements per day (a fasting measurement and one postmeal measurement), plus one bedtime measurement per week (Grade C). Morning fasting measurements are important for adjusting insulin doses to achieve target fasting blood glucose, and a single postmeal measurement should help to determine if postmeal glucose control is adequate (Grade C). For patients on premixed insulins, the minimum should be a fasting measurement plus on postmeal measure each day (using a staggered pattern by testing after different meals on different days). Patients on insulin who are also receiving sulfonylurea or glinide oral agent therapy will require additional testing owing to the increased risk of hypoglycemia with this combination. For patients on basal-bolus insulin regimens, the recommendations should mirror those for type 1 diabetes. Everyday circumstances and other factors contributing to an increased risk of hypoglycemia (e.g., illness, exercise, etc.) should also be taken into consideration. Furthermore, if the HbA1c level remains elevated in spite of good fasting glucose control, more postmeal SMBG measurements may be required to determine the extent of postmeal glucose control.
Non–insulin-treated type 2 diabetes
For patients receiving glucose-lowering drugs other than insulin, the pattern of SMBG testing should be dictated by the risk of hypoglycemia associated with specific agents, as well as the other factors contributing to hypoglycemia risk. For patients receiving agents known to have a low propensity to cause hypoglycemia (e.g., metformin, thiazolidinediones, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide-1 receptor agonists), either as monotherapy or in combination with each other, a short weekly SMBG profile (consisting of four or five measurements) is usually adequate (Grade C). However, if therapy includes higher-risk agents (i.e., sulfonylureas or glinides), an additional single daily test is recommended in order to detect episodes of hypoglycemia (Grade C). For patients being treated with lifestyle intervention alone (i.e., not receiving any glucose-lowering drug therapy), a short SMBG profile performed once per month is usually sufficient (Grade D). In all patients with non–insulin-treated type 2 diabetes, additional testing may be required during illness or other unusual circumstances or if glycemic control is poor (Grade B).
Footnotes
Acknowledgments
The authors would like to acknowledge educational support from LifeScan, part of the Johnson & Johnson Diabetes Solutions Companies, as well as thank Ms. Lynne Trenery for editorial and organizational help.
Author Disclosure Statement
No competing financial interests exist.
