Abstract
Background:
A1chieve® (Novo Nordisk A/S, Bagsværd, Denmark) was a prospective, multicenter, noninterventional study in 66,726 people with type 2 diabetes mellitus (T2DM) in 28 countries beginning biphasic insulin aspart 30 (aspart premix), insulin detemir, or insulin aspart in routine clinical care.
Subjects and Methods:
A subgroup of 27,594 insulin-naive people began therapy with aspart premix with or without oral agents. Safety and effectiveness data were taken from clinic records at baseline and after 24 weeks. Seven regional country groupings were prespecified.
Results:
Mean final insulin dose ranged from 0.68±0.26 U/kg/day (Middle East/Gulf) to 0.38±0.14 U/kg/day (South Asia). The baseline glycated hemoglobin (HbA1c) level varied from 10.5±2.0% (Latin America) to 9.2±1.3% (South Asia), with reductions from −2.9±2.1% (Latin America) to −1.9±1.3% (South Asia). The proportion of people reaching an HbA1c level of <7.0% was highest in China (56%) and lowest in North Africa (22%). Fasting plasma glucose level reductions were from −6.4±5.3 mmol/L (Latin America) to −3.6±2.6 mmol/L (South Asia). Most people began aspart premix twice daily, varying from 91% (North Africa) to 70% (Latin America). Improvement in HbA1c increased with baseline dose frequency (once daily, −1.5±1.4%; twice daily, −2.2±1.6%; three times daily, −2.9±2.2%).
Conclusions:
Insulin-naive people with T2DM beginning aspart premix insulin in routine clinical practice in non-western nations had clinically useful improvements in blood glucose control after 24 weeks in all seven regions. Improvements from baseline for glucose control variables were greater than cross-regional differences in those variables at 24 weeks.
Introduction
Observational studies can enroll a larger, more diverse group of people than RCTs and may better reflect typical clinical populations. A1chieve® (Novo Nordisk A/S, Bagsværd, Denmark) was a 24-week, prospective, noninterventional study conducted in 28 countries across Asia, Africa, Europe, and Latin America. 12,13 It was designed to evaluate the safety and clinical effectiveness of three insulin analogs (insulin detemir [Levemir®; Novo Nordisk A/S], insulin aspart [NovoRapid®; Novo Nordisk A/S], and biphasic insulin aspart 30 [NovoMix® 30/70 (aspart premix); Novo Nordisk A/S]) in people with T2DM who had recently begun insulin therapy.
This article reports safety and effectiveness findings of the insulin most commonly started in A1chieve (insulin aspart premix) and the cohort of most clinical interest: those beginning insulin from OGLDs.
Subjects and Methods
Study design and participants
The total A1chieve cohort involved 66,726 people (recruited January 2009–June 2010) at 3,166 sites across 28 countries. Details of the study design and methods and global primary data have been published elsewhere. 12,13 This study involved a subgroup who were insulin-naive at baseline and who started aspart premix. Some of these people may have had insulin earlier (maximal allowable duration was within the last 4 weeks before the start of the study) but were using only OGLDs when started on premix. Continuation and change in use of OGLDs were at the discretion of the investigator. All local requirements for health authorities or ethics committee approvals were obtained, according to country-specific regulations. This study was conducted in accordance with the Declaration of Helsinki and the Guidelines for Good Pharmacoepidemiology Practice.
Insulin therapy
Enrollment was within 4 weeks of starting insulin. The choice of aspart premix 30, dose optimization, and frequency of administration (once [OD], twice [BID], or three times [TID] daily) were determined by the person with diabetes and his or her physician.
Hypoglycemia, tolerability, and safety end points
Hypoglycemic events were collected for the 4 weeks before each study visit. Major hypoglycemic events were defined as events with severe central nervous system symptoms, accompanied by a plasma glucose level of <3.1 mmol/L (<56 mg/dL), or reversal of symptoms after either food intake or glucagon or intravenous glucose administration. Minor hypoglycemia was any event, with or without symptoms of hypoglycemia, with a plasma glucose reading of <3.1 mmol/L (<56 mg/dL) that the participant was able to self-treat. Nocturnal hypoglycemia was defined as a symptomatic event consistent with hypoglycemia that occurred during sleep between bedtime after the evening insulin injection and before getting up in the morning. Adverse events (AEs) were reportable whether or not considered related to the insulin. Serious AEs (SAEs) followed standard definitions. An adverse drug reaction (ADR) was an AE for which a causal relationship with insulin aspart premix was judged possible or probable by the investigator.
Outcome assessment and reporting
Full details of the methodology of reporting, collecting, and analyzing of data have been published previously. 12 There were no defined study-related procedures; physicians evaluated safety and effectiveness of therapy at usual clinic visits, defined as baseline (visit 1), at approximately 12 weeks (visit 2; not reported here), and 24 weeks (visit 3). Data were obtained from medical records, participant recall, and self-monitoring diary/meter at each visit, as available. The primary safety end point was the number of serious ADRs (SADRs) from baseline to final visit, including all major hypoglycemic episodes. Effectiveness and related measures included change from baseline in glycated hemoglobin (HbA1c) and blood lipids using local assays, fasting plasma glucose (FPG), postprandial plasma glucose (PPG) following breakfast, lunch, and dinner, body weight, and insulin dose. Heath-related QoL (HRQoL) was measured using the EQ-5D questionnaire, 14 which describes HRQoL across five dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) and includes a rating for each person's current health state measured by visual analog scale (from 0=worst imaginable health to 100=best imaginable health). The health dimensions can be converted to a single utility value (QoL UK, where 1.00=perfect health and 0.00=dead).
Statistical analysis
For analysis purposes, countries were grouped a priori into seven regions. 12 All analyses were performed on the full analysis set (FAS), defined as all participants with a baseline visit and who used study insulin at least once at baseline, week 12, or week 24. Change from baseline in HbA1c, FPG, PPG, blood lipids, body weight, and HRQoL were analyzed using Student's paired t test. Changes in frequency of overall, minor, major, and nocturnal hypoglycemia were analyzed using Fisher's exact test. For this publication, only data for the subgroup beginning therapy with insulin aspart premix and insulin-naive at baseline were used. The intention to explore data according to frequency of injection at baseline was also made a priori.
Results
Participant characteristics
There were 27,594 people in the insulin-naive cohort at baseline and who started insulin aspart premix. There were marked variations in baseline participant characteristics among regions (Table 1). Sex ratio varied from 62% male in South Asia to 26% male in Russia. Body mass index (BMI) was relatively low in most regions but was highest in Russia (mean±SD, 30.9±5.1 kg/m2) and lowest in East Asia (24.5±4.4 kg/m2). Duration of diabetes from diagnosis when starting insulin was highest in Latin America (10.4±7.3 years) and lowest in China (5.0±5.4 years) (overall mean, 6.2±5.2 years).
Data are mean (SD) values unless otherwise stated.
BMI, body mass index; OGLD, oral glucose-lowering drug.
The majority of patients (23,342; 85%) were on OGLDs pre-study, whereas 4,252 (15%) had no diabetes treatment. Of those on OGLDs, 5,572 (23.9%) were using one OGLD, 12,893 (55.2%) were using two, and 4,877 (20.9%) were using more than two. Most patients were using metformin (n=19,130; 82.0%) and/or sulfonylureas (n=17,146; 73.5%). At 24 weeks, 9,855 (57.1%) of 17,261 patients were using one OGLD, 6,548 (37.9%) were using two, and 858 (5.0%) were using more than two. The number of patients using metformin was 14,008 (81.2%); 6,892 (39.9%) were using sulfonylureas.
Insulin dose and regimens
At 24 weeks across all regions, insulin dose was 32.6±16.1 U/day, ranging from 54.6±21.3 U/day (Middle East/Gulf) to 25.9±10.1 U/day (South Asia) (Table 2). The dose difference was less extreme when adjusted for body weight, ranging from 0.68±0.26 U/kg/day (Middle East/Gulf) to 0.38±0.14 U/kg/day (South Asia).
Data are mean (SD) unless otherwise stated.
Data are presented and statistical testing was only done where n≥100.
P<0.001.
FPG, fasting plasma glucose; HbA1c, glycated hemoglobin.
Across all regions, most people began insulin aspart premix BID, ranging from 70% (Latin America) to 91% (North Africa). TID use was most common in China (18%), Latin America (17%), and Russia (11%) but much less frequent in other regions. At 24 weeks, BID use ranged from 61% (Russia) to 89% (South Asia). A TID regimen was most frequently observed in Russia (35%) and ranged between 20% (Latin America) and 1.4% (South Asia).
At the end of the study, 26,702 people (96.8%) were still using insulin aspart premix insulin. Other study treatments recorded for this cohort were insulin detemir (1.7%), insulin aspart (0.5%), basal insulin + insulin aspart (0.2%), and other (0.8%).
BG control
Mean HbA1c decreased significantly from baseline (P<0.001) (Fig. 1) in all regions by 24 weeks (overall by −2.2±1.7% to 7.3±1.0%). Reductions varied (Table 3), by −2.9±2.1% achieving 7.6±1.2% at 24 weeks in Latin America to −1.9±1.3% achieving 7.4±0.8% in South Asia. The size of change was not just a function of baseline HbA1c, as the China cohort fell −2.7±2.2% from an average baseline of 9.6±2.2%. The proportion of people reaching HbA1c<7.0% was 56% in China, 32% across all regions, but only 22% in North Africa (Table 3).
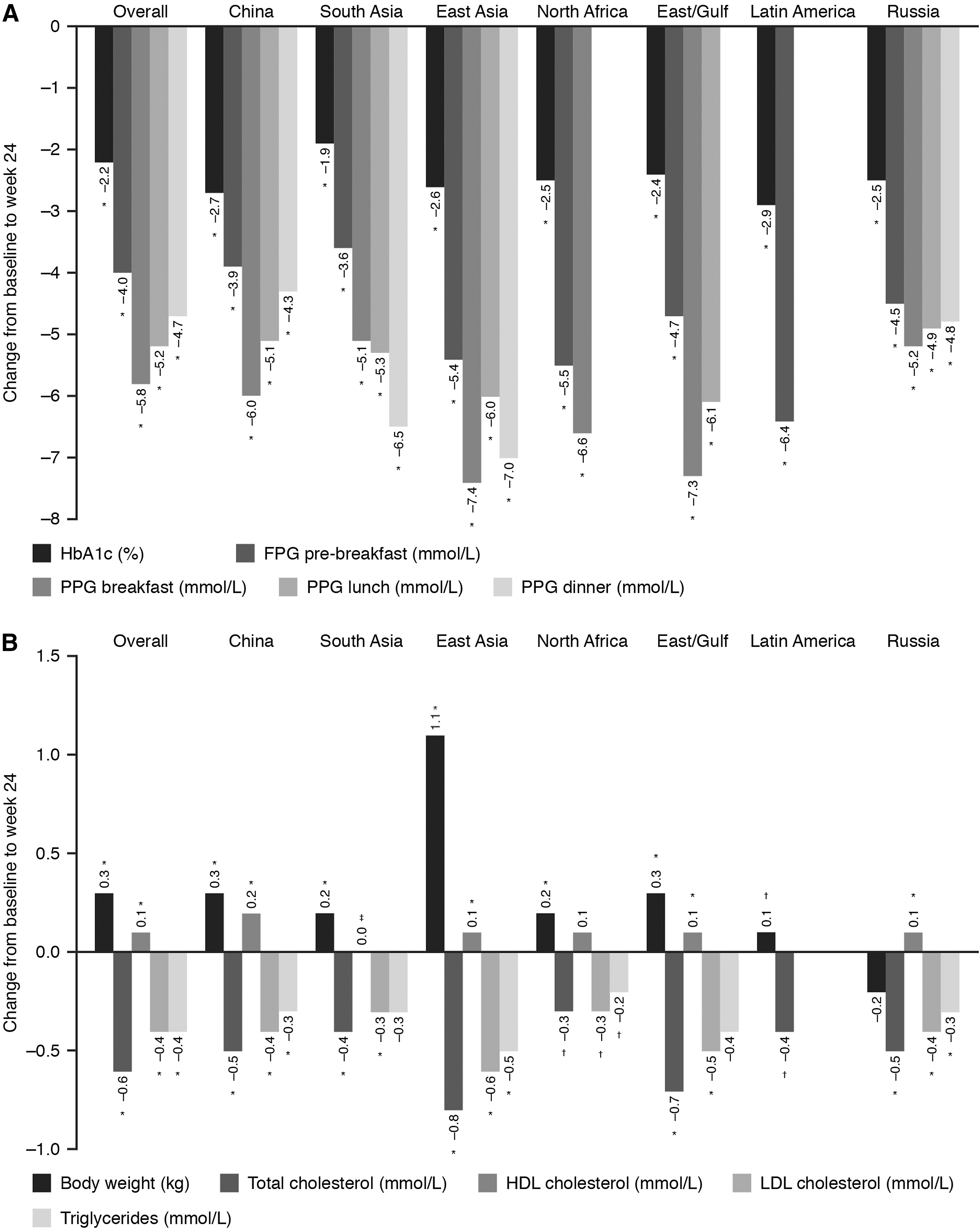
Change from baseline to week 24 in
Data are mean (SD) unless otherwise stated. Numbers for patients reaching glycated hemoglobin (HbA1c) target are larger than numbers for change in HbA1c because the latter requires data from both baseline and 24 weeks.
Data are presented and statistical testing was only done where n≥100.
P<0.001, c P<0.01, d P<0.05.
FPG, fasting plasma glucose; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PPG, postprandial plasma glucose.
Improvement in BG control, as measured by a reduction in FPG values, ranged between −6.4±5.3 mmol/L (Latin America) to −3.6±2.6 mmol/L (South Asia). PPG control improved to a greater degree, and in all regions, post-breakfast from −7.4±5.2 mmol/L in East Asia to −5.1±3.7 mmol/L in South Asia, although not proportionately greater than basal levels, given baseline levels were 2–5 mmol/L higher for PPG than FPG (Fig. 1 and Table 3). Post-lunch and post-dinner glucose values also decreased in all regions where numbers were high enough to assess (P<0.001), with the degree of reduction being similar to that observed for post-breakfast levels (Table 2).
Overall, the magnitude of improvement in HbA1c increased with greater dosing frequency (OD, −1.5±1.4%; BID, −2.2±1.6%; TID, −2.9±2.2%; all P<0.001). Similarly, decreases in FPG (pre-breakfast) were greater with increasing frequency of injection overall (OD, −3.3±3.4 mmol/L; BID, −4.0±3.2 mmol/L; TID, −4.6±3.9 mmol/L; all P<0.001). However, it was difficult to discern any obvious association between improved control and injection frequency when regions were examined individually (Table 2). PPG decreased significantly (P<0.001) at all dosing frequencies and after breakfast, lunch, and dinner in all regions where numbers were high enough to assess (Fig. 1).
Hypoglycemia
Overall, the reported rate of all hypoglycemic events showed no change with treatment: from 0.95 to 1.04 events/person-year (not statistically significant) for the 4 weeks before baseline and 24-week visits.
The reported proportion of people experiencing one or more minor hypoglycemic events varied considerably among regions, with the largest proportion in North Africa (baseline, 15%; 24 weeks, 22%) and the smallest in Latin America (baseline, 0.5%; 24 weeks, 5%) (Table 4).
P<0.001, b P<0.01, c P<0.05.
Nocturnal hypoglycemic events were reported at baseline by <1–2% of people in all regions except North Africa (11%) (Table 4). At week 24, changes in nocturnal events varied by region, with statistically significant increases in the Middle East/Gulf (P<0.0001) and Russia (P=0.01) but a decrease in South Asia (P<0.001), with no statistically significant change reported in other regions.
Major hypoglycemic events were reported in the 4 weeks to baseline by <1% of people in all regions except North Africa (7%) (Table 4). At 24 weeks, major hypoglycemic events were further reduced in most regions, except Latin America and Russia, where the reported event rate was already negligible, and the Middle East/Gulf (no statistically significant change).
Other cardiovascular risk markers
Overall, body weight increased after 24 weeks by a clinically small amount (0.3±3.5 kg; P<0.001). This varied by region, from 2.2±4.6 kg in North Africa to −0.2±3.4 kg in Russia (Table 3). There were modest but statistically significant improvements in blood lipid profile by 24 weeks in all regions (Table 3). Overall, the mean total serum cholesterol level fell − 0.6±1.2 mmol/L, and the low-density lipoprotein cholesterol level fell −0.4±1.0 mmol/L, whereas the high-density lipoprotein cholesterol level rose 0.1±0.4 mmol/L (all P<0.001). The mean triglycerides level also fell (− 0.4±1.0 mmol/L). Overall, the mean systolic blood pressure (SBP) decreased significantly (−7.1±17.0 mm Hg; P<0.001) to 126.9±12.2 mm Hg at 24 weeks. The largest decrease was in Russia (−10.1±14.6 mm Hg; P<0.001), and the least decrease in North Africa (−2.8±18.1 mm Hg; P=0.003). Complete data were not available concerning use of antihypertensive or lipid-lowering medications.
HRQoL
QoL scores improved significantly in all regions by 24 weeks (P<0.001) (Table 5); however, the magnitude of improvement varied. The overall increase in EQ-5D visual analog scale was 15.0±16.9%, ranging from 20.4±16.3% (South Asia) to 6.1±14.4% (China). Similarly, the mean improvement in QoL UK score was 0.19±0.29, again highest in South Asia (0.31±0.30) and lowest in China (0.01±0.22).
All changes P<0.001.
QoL, quality of life; VAS, visual analog scale.
Overall, across all regions, there were significant improvements at 24 weeks (P<0.001 for all changes at 24 weeks vs. baseline, respectively) in the proportion of patients who were not anxious or depressed (77.2% vs. 49.1%), had no problems with self-care (80.1% vs. 59.9%), had no problems performing usual activities (78.4% vs. 53.5%), had no pain or discomfort (74.4% vs. 45.9%), and had no problems walking about (86.4% vs. 57.0%).
Safety
Only 49 SAEs were reported by 40 of >27,000 patients in 28 countries during this study. Four events were judged mild, 14 moderate, and 31 severe. Six SAEs were assessed by investigators as probably (n=4) or possibly (n=2) related to the insulin aspart premix treatment, being hypoglycemic events. Two of these led to withdrawal from the study.
Discussion
These results (previously reported in abstract form 15 ) are comparable to three other groups of data: the whole A1chieve study including other insulin analogs, RCTs with analog premixes, and other observational studies. These results in insulin-naive people beginning insulin aspart premix are similar to those reported for all insulin analogs in the whole A1chieve insulin-naive population and show the same clinically encouraging profile of marked improvements in BG control without any suggestion of a resulting problem from increased hypoglycemia (reportedly reduced for the most part) or of body weight gain. 12 Although, at first sight, this combination is surprising, the results suggest that, in people with poor glucose control, beginning an insulin analog is usually an opportunity for enhanced patient education and self-care. The improvements in average BG control as measured by HbA1c are in the order of a 22% reduction in average glucose concentration or a 60% reduction in hyperglycemia exposure above the upper limit of normal of 6.0% units. This is explained by the changes in basal glucose levels (measured here as FPG) and proportionate (i.e., greater) reductions in postprandial levels, the latter in the current substudy appearing to be equivalent throughout the day.
RCTs tend to come from very different populations in western economies. Although Raskin et al. 16 achieved a somewhat greater reduction in HbA1c in North America with insulin aspart premix (mean±SE, −2.8±0.11%) from a baseline not dissimilar to that in the present study, insulin doses used were notably higher with the benefit of trial dose titration (mean±SD, 78.5±39.5 U/day), perhaps explained by the much higher baseline BMI in the RCT than in any of our global populations. In Europe, and from a somewhat lower baseline HbA1c and with similar insulin doses to those in A1chieve, Kann et al. 17 reported a somewhat lower improvement in glucose control (HbA1c of −1.6%, from 9.2±1.4% at baseline), consistent with the 4T study (multicenter, United Kingdom only) where the reduction in HbA1c at 1 year (−1.3±1.1%) was also from a much lower baseline of 8.6±0.8%. 18
In all these studies, only a minority achieved target HbA1c<7.0%, despite the relatively low incidences of reported hypoglycemia. In China, a bare majority achieved the target (56%), notably in the population that had the shortest duration of diabetes and the lowest baseline BMI. It is unclear to what extent dose titration was actively pursued in A1chieve participants, but the small increase in dosage between beginning insulin and 24 weeks in the overall cohort suggests it was not usual. 12
Results from A1chieve with premix insulin aspart complement data from another large noninterventional observational trial, IMPROVE, which included participants from developed economies. 19 Nevertheless, the overall reduction in measures of glucose control was nearly identical in the two studies, 19 as were findings for hypoglycemia, and results from China.
Adverse outcomes, notably potential for body weight gain and expected increase in hypoglycemia, are a significant barrier to beginning insulin therapy. 2,3 Hypoglycemia increases in the RCTs, albeit to a low incidence rate, something not found in the observational studies including both the overall and aspart premix cohorts of A1chieve (Table 4). 12,16 –19 It is possible that lower hypoglycemia rates could reflect improved self-care behaviors, and the failure of body weight gain despite presumed amelioration of urinary glucose excretion, plus the fall in SBP, does suggest improved attention to dietary measures. Also, the falling usage of sulfonylureas, which are associated with an elevated risk of hypoglycemia, 20 during the 24-week period from 73.5% to 39.9% may play a role. However, as there is an absence of meaningful data on hypoglycemic episodes, it is not possible to make any definitive conclusions.
Only a small proportion of participants starting insulin aspart premix used it OD. In Asia, they tended to be people with lower baseline HbA1c (Table 2). This might partly account for the smaller reduction in HbA1c with fewer injections, but equally more injections enabled higher overall insulin dosage at 24 weeks (Table 2), which might account for some of the better glucose control.
In A1chieve as a whole, as in this subgroup, the incidence of AEs was low, and no problems of note were identified. It is not possible to know if the low rates of reporting of AEs and SAEs reflect the selection of participants or underascertainment in the context of this being an observational study with no monitoring of investigator reporting. However, that is suggested by the considerable variation in event reporting between regions.
Regional differences
There was considerable consistency in the magnitude of improvement of glucose-lowering effectiveness measures across all regions, with the exception of the proportion of patients reaching HbA1c<7.0%, where the artifact of the forced categorical cutoff, together with somewhat better average findings, results in China having more than twice the achievement of North Africa. Although weight gain in most regions was clinically unimportant, North Africa reported a much greater gain (2.2±4.6 kg) after 24 weeks versus other regions, as well as the highest reported rates of all forms of hypoglycemia. This suggests that either educational initiatives when starting the analog were less effective in North Africa or that, having already been as effective as possible, this cohort could not gain from further education. As noted above, we cannot be certain that hypoglycemia reporting was complete, and variations in consistency might account for regional differences.
Some of the regional differences are likely to contribute to differences in outcome, as mentioned above, notably the short duration of diabetes and lowest BMI in China, against the longer diabetes duration and much higher BMI in the Middle East/Gulf, Latin America, and Russia.
Limitations of this study
The standard limitations of observational studies apply here. In particular, changes in all measures cannot be causally attributed to the insulin itself, as other confounding changes and interventions may have been made at the same time, including study effects, and be influencing the findings. Indeed, we suggest above that this is the case regarding diabetes education. A further possibility is of some degree of regression to the mean if, for example, those studied came to the notice of physicians because of an acute event (e.g., hospitalization for infection) that disturbed BG control and started insulin in that context. HbA1c, however, being retrospective by 2–3 months provides some protection against that. In the absence of control interventions, the observed improvements can then only be judged against values at baseline. Additionally, there is a problem of possible changes in concomitant medications, although in this A1chieve subgroup this was mainly a reduction in sulfonylurea use, which would have been expected to impact negatively on BG control. Lastly, the short duration of the study (24 weeks) may be a limitation when predicting long-term outcomes.
Conclusions
Insulin-naive people with T2DM in predominantly non-western nations who began therapy with insulin aspart premix in routine clinical practice achieved clinically important HbA1c reductions with a good safety profile. In terms of effectiveness, the results reflect those found in RCTs. However, with regard to safety, observational data do appear to show more favorable frequency of hypoglycemia when insulin aspart premix is initiated. Insulin-naive patients starting insulin aspart premix, as well as gaining from the insulin treatment itself, may benefit from a simultaneous opportunity to enhance lifestyle behaviors. Despite significant improvements in FPG and PPG, only one-third of the patients reached target glycemic control (below HbA1c of <7.0%) in this study. However, improved FPG and PPG were associated with a marked improvement in QoL scores.
Footnotes
Acknowledgments
The authors would like to thank all of the participants who provided data and all of the investigators involved in the A1chieve study. The A1chieve study was sponsored by Novo Nordisk A/S. Watermeadow Medical provided writing assistance, funded by Novo Nordisk.
Author Disclosure Statement
S.S. or institutions with which he is connected receive funding from all major manufacturers of glucose-lowering products, including from Novo Nordisk, for his educational, research, and advisory activities. W.Y. has no conflicts of interest to disclose. M.I.H. is a member of the A1chieve advisory board. R.M. has participated in several advisory boards for Novo Nordisk, has participated in studies sponsored by Novo Nordisk, and has also participated in symposia run by Novo Nordisk, Sanofi, Eli Lilly, Merck, and Pfizer. O.M.B. is an employee of and owns shares in Novo Nordisk. P.H. or institutions with which he is connected receive funding from all major manufacturers of glucose-lowering products, including from Novo Nordisk, for his educational, research, and advisory activities. These include the AstraZeneca/Bristol-Myers Squibb alliance, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Merck (MSD), Merck Serono, Novo Nordisk, Roche Pharma, Sanofi, SkyePharma, and Takeda.
