Abstract
Background:
Lack of self-monitoring of blood glucose (SMBG) records in actual practice settings continues to create therapeutic challenges for clinicians, especially in adjusting insulin therapy. In order to overcome this clinical obstacle, a “Symptom-based Insulin adjustment for Glucose Normalization” (SIGN) algorithm was developed to guide clinicians in caring for patients with uncontrolled type 2 diabetes who have few to no SMBG records. This study examined the clinical outcome and safety of the SIGN algorithm.
Subjects and Methods:
Glycated hemoglobin
Results:
Mean HbA1c improved by 0.29% at 3 months (P=0.015) and 0.41% at 6 months (P=0.006) after algorithm implementation. A slight increase in HbA1c was observed when the algorithm was not implemented. There were no major hypoglycemic episodes. The number of minor hypoglycemic episodes was minimal with the majority of the cases due to irregular meal habits.
Conclusions:
The SIGN algorithm appeared to offer a viable and safe approach when managing uncontrolled patients with type 2 diabetes who have few to no SMBG records.
Introduction
In order to overcome this challenge without compromising the care of patients who do not provide SMBG records, a “Symptom-based Insulin adjustment for Glucose Normalization” (SIGN) algorithm was developed and implemented in an outpatient diabetes clinic where more than half of the patients refuse to perform SMBG. 7 In this pilot study, we report the clinical outcome and safety in the use of our SIGN algorithm to manage patients with uncontrolled type 2 diabetes.
Research Design and Methods
Study design and setting
This was a prospective study conducted in a diabetes specialty clinic located within a government-owned primary care facility that provides subsidized outpatient medical care to patients with chronic diseases. The diabetes specialty clinic is managed by a multidisciplinary care team and receives referral from general clinics for the care of patients with uncontrolled diabetes defined as a glycated hemoglobin (HbA1c) level of ≥8%.
Development of SIGN algorithm
The SIGN algorithm was developed and implemented in 2008 by a clinical pharmacist with the endorsement from a family physician for the use of regular insulin, neutral protamine Hagedorn (NPH), and biphasic insulin isophane 70/30 only. 7 The algorithm was subsequently revised and updated in 2010, incorporating all rapid-, short-, and long-acting insulin products as well as the premixed insulin products (Fig. 1). The algorithm is designed for the use of patients with type 2 diabetes with HbA1c ≥9% or HbA1c ≥8% with maximized metformin or sulfonylurea unless otherwise contraindicated. The algorithm focuses on the severity and frequency of hypoglycemic episodes reported by patients, and it is only to be used upon assurance of patients' adherence to all antidiabetes agents.
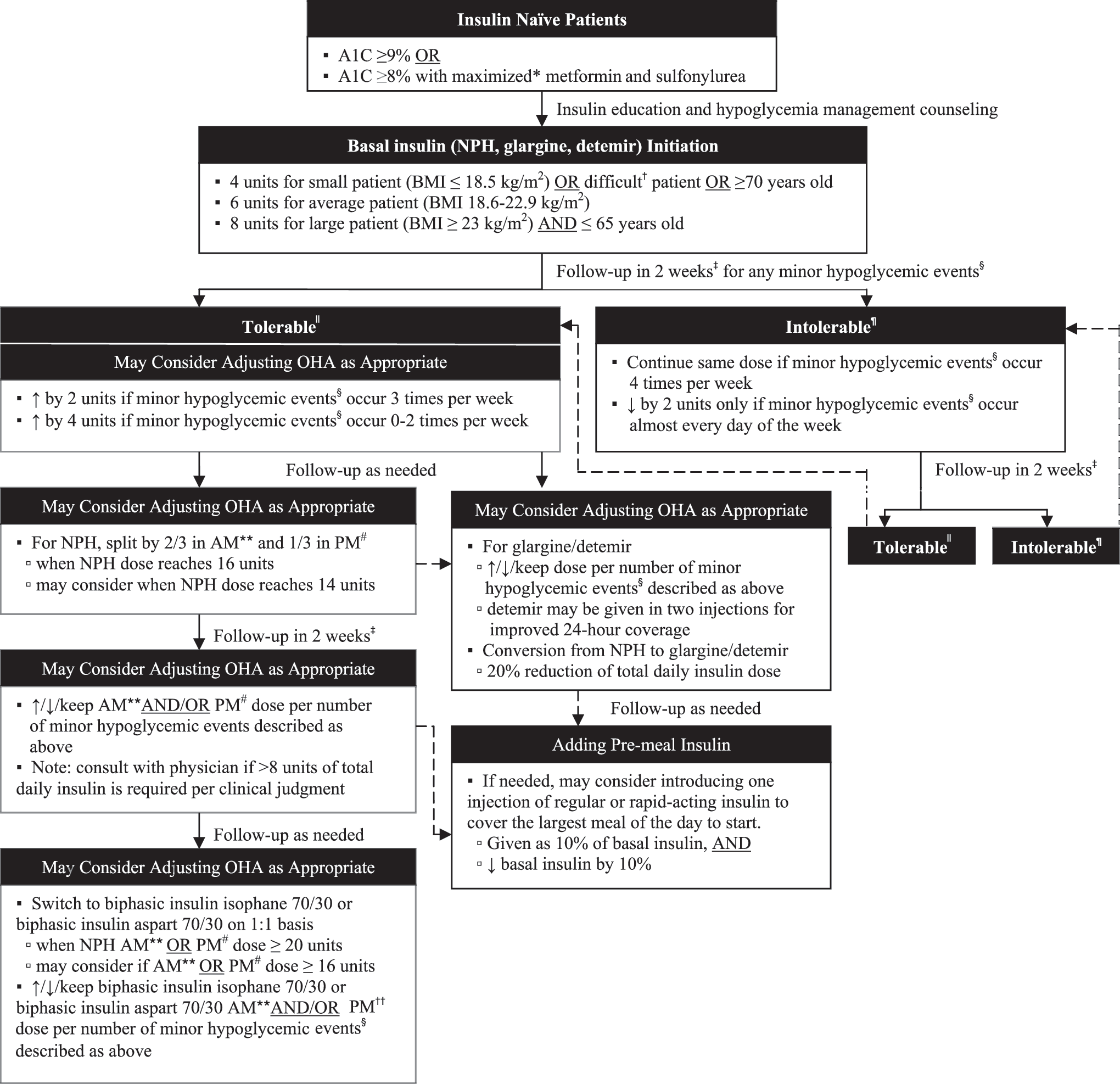
“Symptom-based Insulin adjustment for Glucose Normalization” (SIGN) algorithm. *Defined as 50% to 66% of the Food and Drug Administration upper limit for recommended dosage. †Refers to any patient with strong or uncompromisable psychological insulin resistance. ‡Follow-up in 2 weeks only if there are any major changes made in oral medications or insulin (e.g., initiate, add, discontinue, hold, or switch), nonadherence, retrial of medications, or other unusual circumstances per clinical judgment. §Defined as common signs and symptoms due to known causes such as irregular eating habits, increased daily activity, stress, or other causes that are modifiable. ||Tolerable if minor hypoglycemic events occurred three or fewer times per week and quickly recovered with rest or 15 g of fast-acting glucose. ¶Intolerable if minor hypoglycemic events occurred more than three times per week despite quickly recovered with rest or 15 g of fast-acting glucose. **Before breakfast. #At bedtime. ††Before dinner. A1C, glycated hemoglobin; BMI, body mass index; NPH, neutral protamine Hagedorn; OHA, oral hypoglycemic agent(s).
Inclusion and exclusion criteria
All insulin-treated patients with uncontrolled type 2 diabetes who refused to use SMBG or performed SMBG once a day for less than three times per week were included in this study. Patients with type 1 diabetes, patients with documented nonadherence to diabetes medications, and patients who were not on insulin therapy any time during the study period were excluded from this study. In addition, patients with known history of hypoglycemia unawareness, cognitive impairment, or trouble communicating were also excluded from this study.
Measures
The primary outcomes were mean reduction in HbA1c and incidence of insulin-related adverse events, which included mean change in body weight and number of major and minor hypoglycemic episodes. All parameters were collected 3 months prior to the implementation of the algorithm and 3 and 6 months after the implementation of the algorithm.
Major hypoglycemic episodes were defined as any hypoglycemic symptom that required help from another person. 8 Minor hypoglycemic episodes were defined as signs and symptoms with known precipitating causes such as irregular eating habits, increased daily activity, or other attributes that can be modified; minor hypoglycemic events should allow the patient to recover quickly with or without the ingestion of fast-acting glucose. 9
Statistical analysis
Statistical Package for Social Sciences version 17.0 software (SPSS, Inc., Chicago, IL) was used for analysis. Paired-samples t test was performed to determine significance between the baseline and follow-up HbA1c values and incidence of insulin-related adverse events. A two-tailed value of P<0.05 was considered statistically significant.
Results
Patient demographics
Of the 366 patients recruited, 114 (31.1%) met the inclusion criteria. The mean age was 57.4±9.0 years with equal number of males and females. Hypertension (n=90 [78.9%]) and dyslipidemia (n=110 [96.5%]) were the most common co-morbidities. A majority of patients (n=105 [92.1%]) were on a combination of insulin with at least one oral diabetes medication, and nine (7.9%) were on insulin therapy only. Cost of the strips and inconvenience were the main reasons for not having SMBG records.
Effects on HbA1c
The mean HbA1c at baseline was 9.34±1.35%. The mean HbA1c levels 3 months before and 3 and 6 months after the algorithm implementation were 9.30±1.33%, 9.05±1.36%, and 8.93±1.44%, respectively. Mean HbA1c was slightly increased by 0.04% when the algorithm was not implemented (P=0.57). Mean HbA1c was reduced by 0.29% (P=0.015) at 3 months and 0.41% (P=0.006) at 6 months after the implementation of the algorithm (Table 1).
HbA1c, glycated hemoglobin.
Insulin-related adverse events
Overall, an average titration of 8.6 (range, 6.0–16.0) units of basal insulin per patient was observed over the 6-month period using the algorithm. Mean body weight increased from 77.1 kg at baseline to 77.6 kg 6 months after the algorithm implementation (P=0.008). There were no major hypoglycemic events. Total numbers of minor hypoglycemic episodes 3 months before and 3 and 6 months after the algorithm implementation were six, four, and two, respectively (Table 1). All minor hypoglycemic episodes reported during the algorithm implementation were due to irregular meal habits.
Discussion
This pilot study illustrated the clinical and safety data of the first symptom-based insulin adjustment algorithm known in the literature. Although it is generally acknowledged that when clinicians care for patients with few to no SMBG records, the titration of insulin is largely dependent on their clinical judgment. However, depending on the practice experience, clinicians' confidence in carrying out an effective insulin titration without SMBG is often compromised by fear of hypoglycemia. 10 The proposed algorithm implemented in this study served as a guide to assist clinicians in adjusting the insulin dose with a focus on the severity and frequency of hypoglycemic episodes reported by patients.
Our study showed that use of the SIGN algorithm resulted in significant and persistent reductions in HbA1c at 3 and 6 months in patients with uncontrolled type 2 diabetes who have few to no records of SMBG. Although the reduction of HbA1c at 6 months was moderate at 0.41%, the decrease was continuous from the HbA1c level at 3 months, and this trend of improvement was not observed before the algorithm implementation. Furthermore, the reduction in HbA1c level in our study was comparable to that in a prospective study where the mean change in HbA1c level from the baseline of 9.7% in insulin-treated patients with type 2 diabetes who performed SMBG for at least twice a day every day was 0.5% at 12 months. 11 In another prospective study where SMBG was performed four times per day every day of the week, the HbA1c level of insulin-treated patients with type 2 diabetes was reduced by 0.31% from the baseline of >8% at Week 52. 12 Therefore, our algorithm appeared to be at least as effective as in situations where SMBG records were performed regularly two to four times per day, every day.
Adverse events related to the use of the algorithm appeared to be minimal. In this study, a weight gain of 0.50 kg was observed. Based on the average insulin-related weight gain of 1.5–3.2 kg over a 1-year period, 13 the average weight gained by the subjects in this study was less than what has been reported. Furthermore, the overall minor hypoglycemic events were reduced after the algorithm implementation, and the incidence was less compared with the reported range of eight to 16.5 hypoglycemic episodes per patient per year. 14,15 Although the incidence of hypoglycemia in this study was minimal, it is nevertheless a major concern in diabetes management. Therefore, appropriate education regarding hypoglycemia and its triggers must be carried out before the algorithm implementation and to be reinforced regularly.
There were several limitations. First, the algorithm was only designed for patients with an HbA1c level of ≥8%; hence extra caution must be practiced when applying this algorithm to patients with an HbA1c level of <8%. Second, this pilot study was a single-group design with patients serving as their own controls before and after the implementation of the algorithm. Although incorporation of a control group may have strengthened the study design, the nonstandardized approach in caring for patients with few to no SMBG records makes it difficult to have a control group. As such, the single-group design was considered as a better approach to capture the effectiveness of the algorithm within a group of subjects who received the same interventions. Lastly, the duration of this study was only for a period of 6 months. Future studies should focus on the long-term effects of the algorithm in insulin-treated patients with type 2 diabetes and their sustainability in the improvement of HbA1c.
In conclusion, in patients with uncontrolled type 2 diabetes who do not perform SMBG, the use of the SIGN algorithm in our pilot study appeared to be an effective and safe approach in adjusting for insulin requirement. Although the algorithm should not replace SMBG as the use of a blood glucose meter is still the gold standard in diabetes care, clinicians may consider utilizing this algorithm in situations where patients are present with few to no SMBG records to ensure timely management of blood glucose control.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
