Abstract
Background:
We compared telecare and conventional self-monitored blood glucose (SMBG) programs for titrating the addition of one bolus injection of insulin glulisine in patients with type 2 diabetes uncontrolled on oral hypoglycemic agents for ≥3 months who were first titrated with basal insulin glargine.
Methods:
This randomized, multicenter, parallel-group study included 241 patients (mean screening glycosylated hemoglobin [HbA1c], 8.8% [73 mmol/mol]). In the run-in phase, any antidiabetes medication, except for metformin, was discontinued. Metformin was then up-titrated to 2 g/day (1 g twice daily) until study completion. Following run-in, all patients started glargine for 8–16 weeks, targeting fasting plasma glucose (FPG) ≤5.6 mmol/L using conventional SMBG. Patients with FPG ≤7 mmol/L added a glulisine dose at the meal with the highest postprandial plasma glucose excursion, titrated to target 2-h postprandial plasma glucose level <7.8 mmol/L using telecare or SMBG for 24 weeks. Patients with FPG >7 mmol/L at week 16 were withdrawn from the study.
Results:
After glargine titration, 224 patients achieved FPG ≤7 mmol/L, without any difference between telecare and SBMG groups (mean±SD, 6.2±0.8 vs. 6.0±0. 9 mmol/L, respectively). HbA1c levels were lower following titration and were similar for telecare and SMBG (7.9±0.9% vs. 7.8±0.9% [63 vs. 62 mmol/mol], respectively). Adding glulisine further reduced HbA1c in both groups (−0.7% vs. −0.7%); 45.2% and 54.8% (P=0.14), respectively, of patients achieved HbA1c ≤7.0% (≤53 mmol/mol). Weight change and hypoglycemia were similar between groups.
Conclusions:
Patients adding one dose of glulisine at the meal with the highest postprandial plasma glucose excursion to titrated basal glargine achieved comparable improvements in glycemic control irrespective of traditional or telecare blood glucose monitoring.
Introduction
The American Diabetes Association/European Association for the Study of Diabetes treatment algorithm recommends basal insulin for initiation of insulin treatment in patients with type 2 diabetes failing on oral hypoglycemic agents (OHAs). 5 The Treating to Target in Type 2 Diabetes study 6 has shown that adding basal or prandial insulin regimens to OHAs is effective in improving glycemic control, more so than biphasic insulin; several other studies have also demonstrated the possibility of achieving good glycemic control with basal insulin. 7,8 Although the basal–bolus strategy is considered to be the best approach to meet daily insulin requirement, experience has suggested that adding one bolus of fast-acting insulin at one meal (basal plus strategy) may offer a simpler and more effective evolution of basal insulin therapy than immediately starting a fully intensified basal–bolus insulin regimen or premix insulin. 9 –12
The Evaluation of Lantus Effect ON Optimization of use of single dose Rapid insulin (ELEONOR) study combines new technical and therapeutic strategies to compare telecare and conventional SMBG programs for titrating the addition of one bolus injection of insulin glulisine in type 2 diabetes patients with secondary failure to OHAs who were first titrated with basal insulin.
Subjects and Methods
Study population
Men and women, 35–70 years old, body mass index >25 kg/m2, with type 2 diabetes for at least 1 year, treated with OHAs or metformin at maximal doses for at least 3 months and glycosylated hemoglobin (HbA1c) 7.5–11.0% (58–97 mmol/mol) were eligible for this Italian, multicenter, parallel-group, randomized controlled study. Patients were excluded if they had a history of two or more severe hypoglycemic episodes within the past 3 months or history of hypoglycemia unawareness, active proliferative diabetic retinopathy, impaired renal or liver function, hypersensitivity to insulin, insulin analogs or excipients, or metformin, mental conditions rendering the subject unable to understand the nature, scope, or possible consequences of the study, or any clinically significant major organ system disease or were pregnant or lactating women.
The study was conducted according to good clinical practice and the Declaration of Helsinki and was approved by independent ethics committees or institutional review boards at each participating institution. All subjects provided written informed consent. Participating investigators and centers are listed in the Appendix.
Study design and treatment
The study design is shown in Figure 1. After screening, patients entered a 2–4-week run-in, during which any antidiabetes medication, with the exception of metformin, was discontinued. Metformin was then up-titrated in all patients to 2 g/day (1 g twice daily) until study completion. At the end of the run-in phase, glargine was started once daily at supper, and subjects were randomized into either the telecare or the conventional SMBG program but continued to use conventional SMBG during the titration phase.
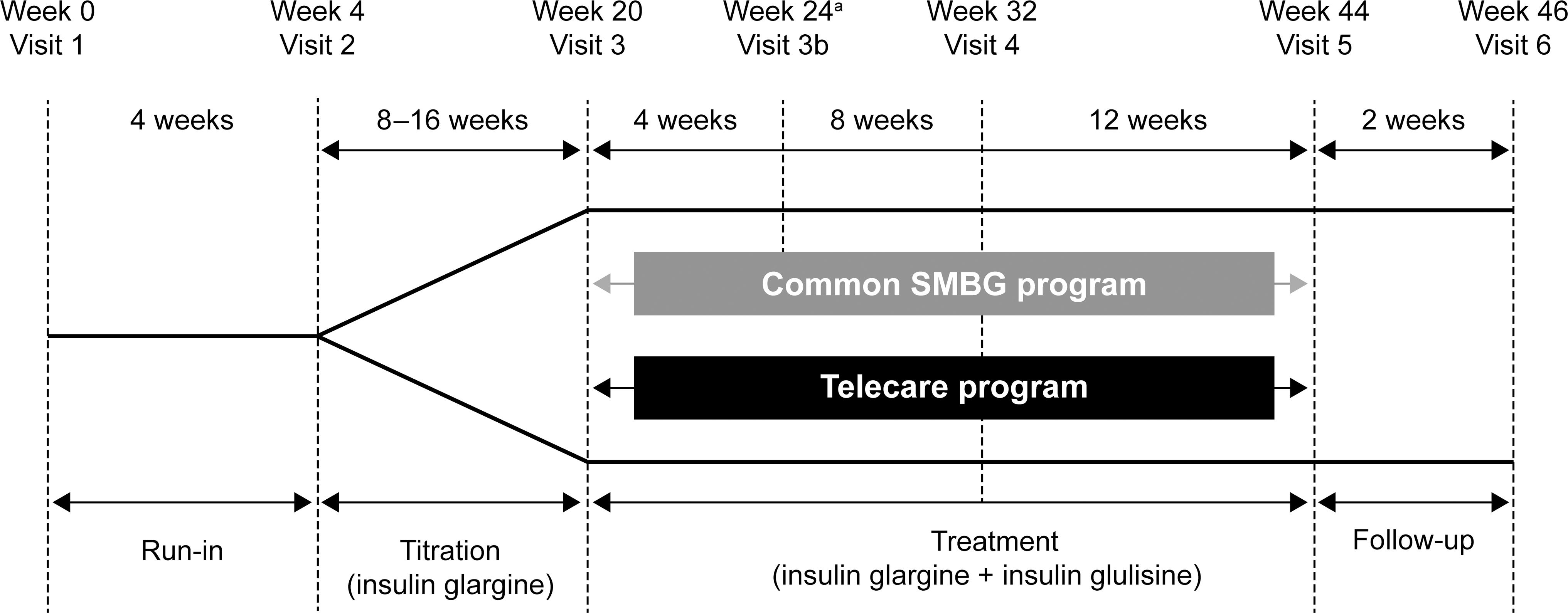
Study design. aDose adjustment (if required) was performed in the conventional self-monitored blood glucose (SMBG) group.
During the titration phase, glargine was adjusted to a fasting plasma glucose (FPG) target level of <5.6 mmol/L, starting with a dose of 10 U/day using a predefined titration algorithm (Table 1). During this phase, patients were required to perform a six-point blood glucose profile (pre- and 2-h post-breakfast, -lunch, and -supper) on two consecutive weekdays every week using standard glucometers (OneTouch® Ultra, Lifescan, High Wycombe, UK). The mean values of post-breakfast, -lunch, and -supper assessments were calculated to identify the meal with the highest postprandial glucose excursion.
Titration target was fasting plasma glucose (FPG) <5.6 mmol/L.
Titration target postprandial plasma glucose (PPPG)=7.8–5.6 mmol/L.
The treatment phase began when a patient achieved FPG ≤7 mmol/L, after 8, 12, or 16 weeks of the titration phase. Patients were withdrawn from the study if FPG remained >7 mmol/L after 16 weeks. Patients with FPG ≤7 mmol/L added glulisine (starting dose of 0.05 U/kg) before the meal identified as above. Patients optimized glulisine doses, with the goal of reducing the 2-h postprandial plasma glucose level to <7.8 mmol/L (Table 1), using either telecare or conventional SMBG. Each subject underwent an educational program designed to review either conventional capillary blood glucose reading by standard glucometer or features and mode of use of the Glucobeep® (METEDA S.r.l., San Benedetto Del Tronto, Italy) telecare system. The latter transforms glucose levels into tones that are transmitted by phone from the patient's home to a centralized server, from which the results are made available to the investigator's computer. The investigator can then transmit information (e.g., dose titration) to the centralized server, which is returned to the patient by phone. Patients in the conventional SMBG group used a standard glucometer and recorded values in a diary with dose adjustments discussed at each visit.
In weeks 9–12 and 21–24 of the treatment phase, all patients were asked to perform two eight-point (pre- and post-breakfast, -lunch, and -supper and at 11 p.m. and at 3 a.m.) glucose profiles on two consecutive weekdays. Eight-point glycemic profiles at Visit 2 and Visit 3 were calculated from available data from the patient's glucometer. Glycemic profiles were obtained from 126 patients (telecare, 52; SMBG, 74) who met the criteria of having ≥50% of planned assessments within the same day (three assessments for six-point glycemic profiles and four assessments for eight-point glycemic profiles) and with at least one glycemic profile (complete or partial) at all visits. Patients were required to test glucose whenever they had symptoms related to hypoglycemia and to record their blood glucose readings. At the end of the 24-week treatment phase, the subjects entered a 2-week follow-up phase.
The primary objective of this study was to compare the change in HbA1c from baseline (Visit 3) to the end of the treatment phase (Visit 5) between patients in the telecare and SMBG programs. Changes in SMBG six- or eight-point glycemic profiles, insulin dose, and body weight from baseline were also calculated.
All clinical chemistry/laboratory parameters were measured using standard techniques at the Exacta central laboratory, Verona, Italy.
Safety
Safety analyses included the frequency of hypoglycemia.
Statistical analysis
Statistical analyses were performed using the intention-to-treat (ITT) population (all randomized patients known to have taken at least one dose of study drug and providing enough data to assess the primary variable [i.e., having completed at least 12 weeks of the treatment phase]), the per-protocol (PP) population (all patients in the ITT population who completed the study protocol without a major protocol violation), or the safety population (all randomized patients known to have taken at least one dose of study medication). The primary end point and quantitative secondary end points were compared between groups using analysis of covariance with the baseline value as covariate. Categorical variables were compared using χ2 tests. Data are shown as mean±SD values unless otherwise specified.
Results
Patient disposition and characteristics
In total, 352 patients were screened, of whom 291 were randomized (telecare, 142; SMBG, 149). The ITT population comprised 241 patients (telecare, 115; SMBG, 126); of these, 238 completed the study (telecare, 114; SMBG, 124) (Fig. 2). Overall, 14 patients in the telecare group and 13 patients in the conventional SMBG group were withdrawn from the study because of FPG >7 mmol/L at the end of the titration phase. Patient characteristics at screening were comparable in the telecare and conventional SMBG groups (Table 2). Patients who were withdrawn from the study during the titration phase had similar characteristics at screening as the ITT population. In the telecare group, 76 of 115 patients received full use of the telecare system, defined as patients transmitting data with telecare and receiving an answer from an investigator. Fourteen patients transmitted data to the investigator but did not receive answers; 25 patients never used the telecare system.
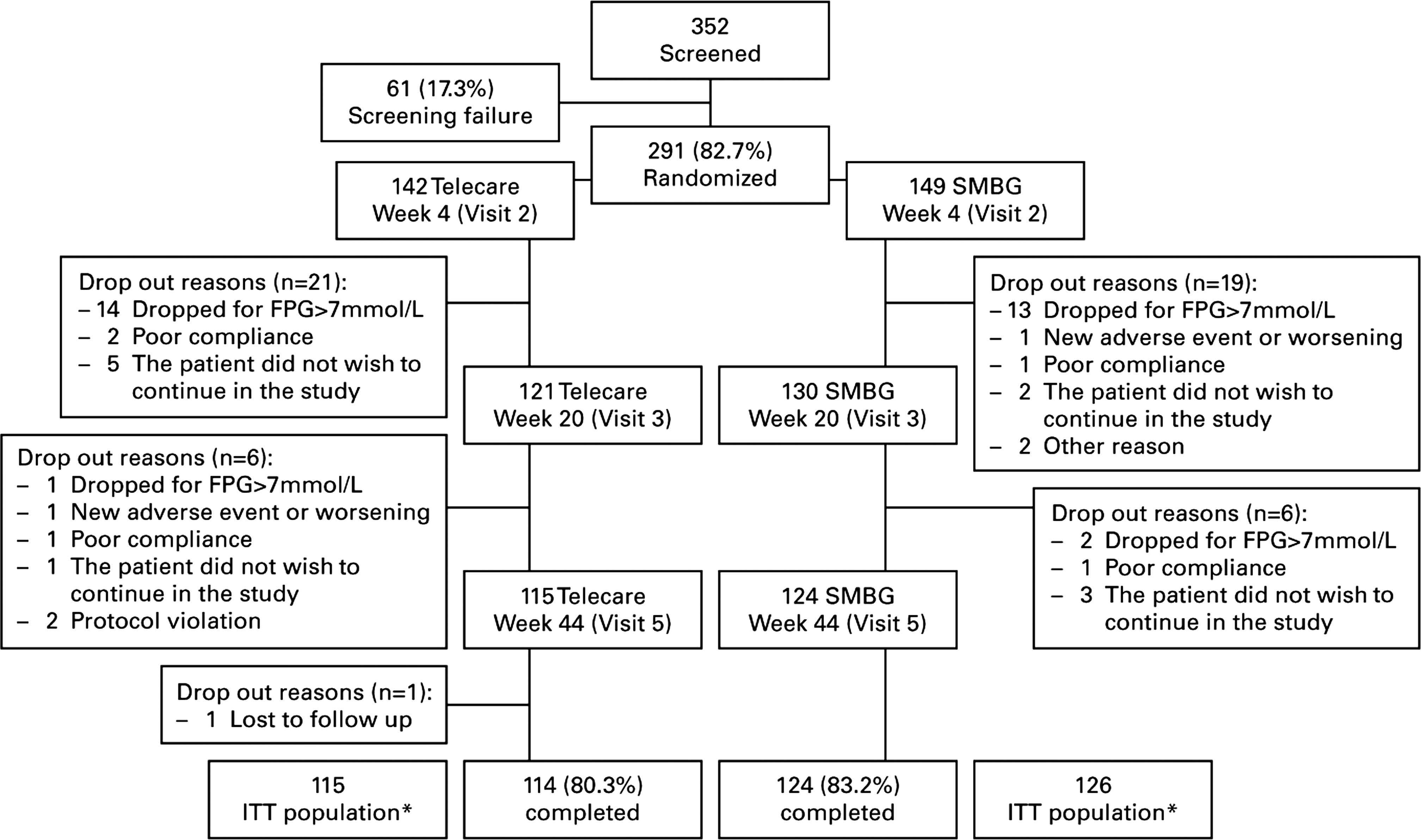
Patient disposition. *All randomized patients known to have taken at least one dose of study drugs and providing enough data to assess the primary variable (i.e., having completed at least 12 weeks of treatment phase). FPG, fasting plasma glucose; ITT, intention-to-treat population; SMBG, self-monitored blood glucose.
Data are mean±SD values or n (%).
A1c, glycosylated hemoglobin; BMI, body mass index; SMBG, self-monitored blood glucose.
Efficacy
The time course of HbA1c, FPG, and plasma insulin is shown in Figure 3. There was a marked reduction of FPG during the titration phase that was associated with a significant reduction in HbA1c, with no difference between telecare and SMBG patients. Fasting plasma insulin concentration increased in a similar manner in the two groups during this phase. Introduction of glulisine caused no further reduction in FPG, although HbA1c continued to decline, whereas fasting plasma insulin did not change in a significant manner. The change in HbA1c from baseline to the end of the treatment phase was significant for both telecare (adjusted mean±SE change from baseline, −0.7±0.06%; P<0.0001) and conventional SMBG patients (−0.7±0.06%; P<0.0001), with no difference between groups (point estimate, 0.07%; 95% confidence interval, −0.10, 0.25; P=0.40). Nearly identical results were obtained with the PP population. The proportion of patients achieving target HbA1c ≤7% (≤53 mmol/mol) was similar in the telecare (45.2%) and SMBG (54.8%) groups (P=0.14). At the end of the treatment phase, both the telecare and SMBG groups had received similar doses of glargine (28.6±17.8 vs. 27.8±16.0 U, respectively) and glulisine (9.5±8.3 vs. 9.5±6.8 U, respectively).
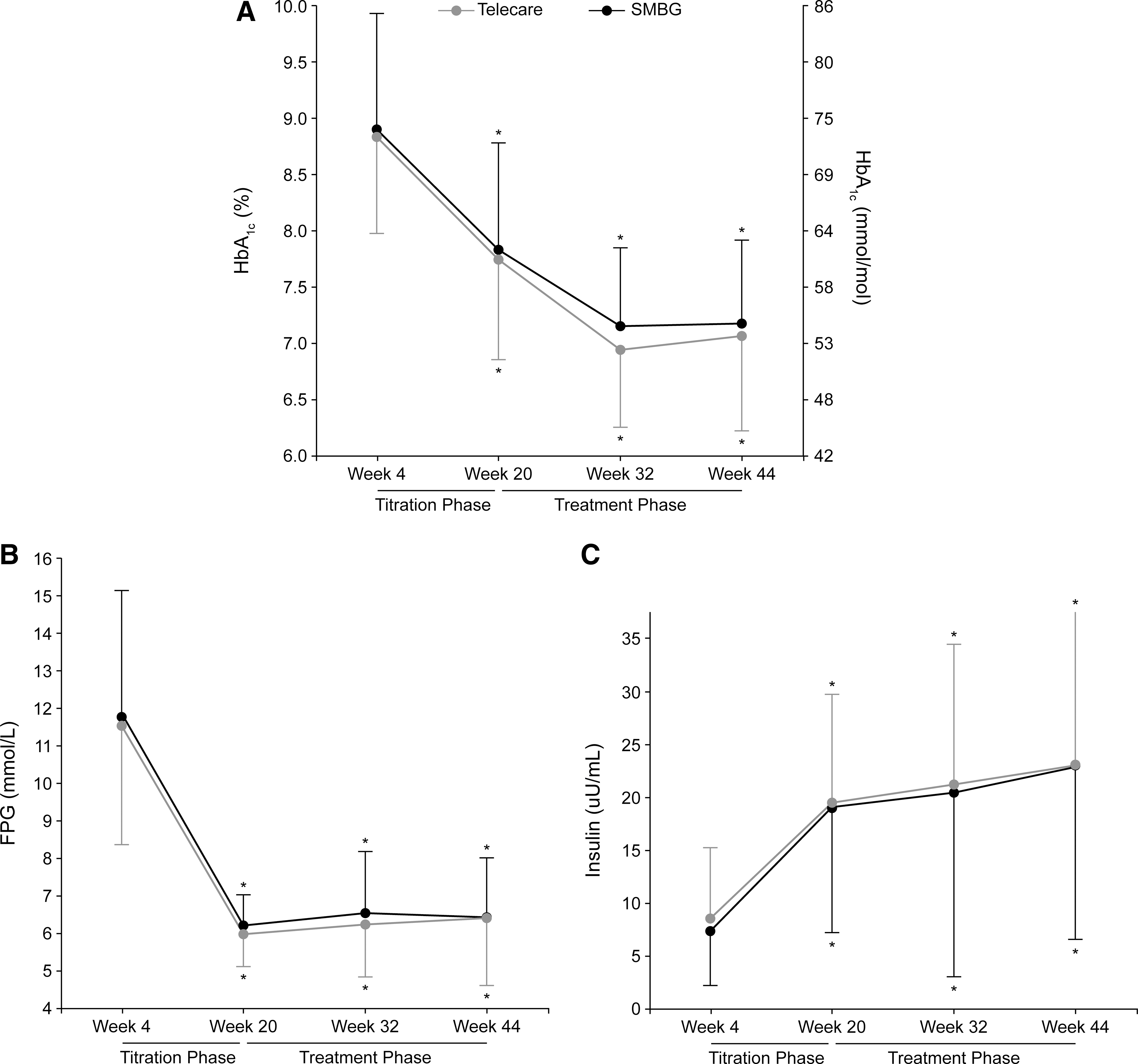
Time course of (
The eight-point glycemic profile at each visit is shown in Figure 4. Although glargine titration was very effective in reducing FPG, blood glucose levels increased progressively throughout the day to achieve the highest value at bedtime (Visit 3). Adding one injection of glulisine at the time of the meal with the largest glucose excursion resulted in the flattening of the blood glucose profile (Visits 4 and 5). Eight-point profiles demonstrated similar glycemic values for both treatment groups, regardless of whether the prandial injection was performed at breakfast, lunch, or supper (data not shown). More patients throughout the treatment phase injected glulisine at supper (telecare, 43%; SMBG, 45%) than at lunch (telecare, 36%; SMBG, 36%) or breakfast (telecare, 5%; SMBG, 5%). Approximately 15% of each group did not inject glulisine.
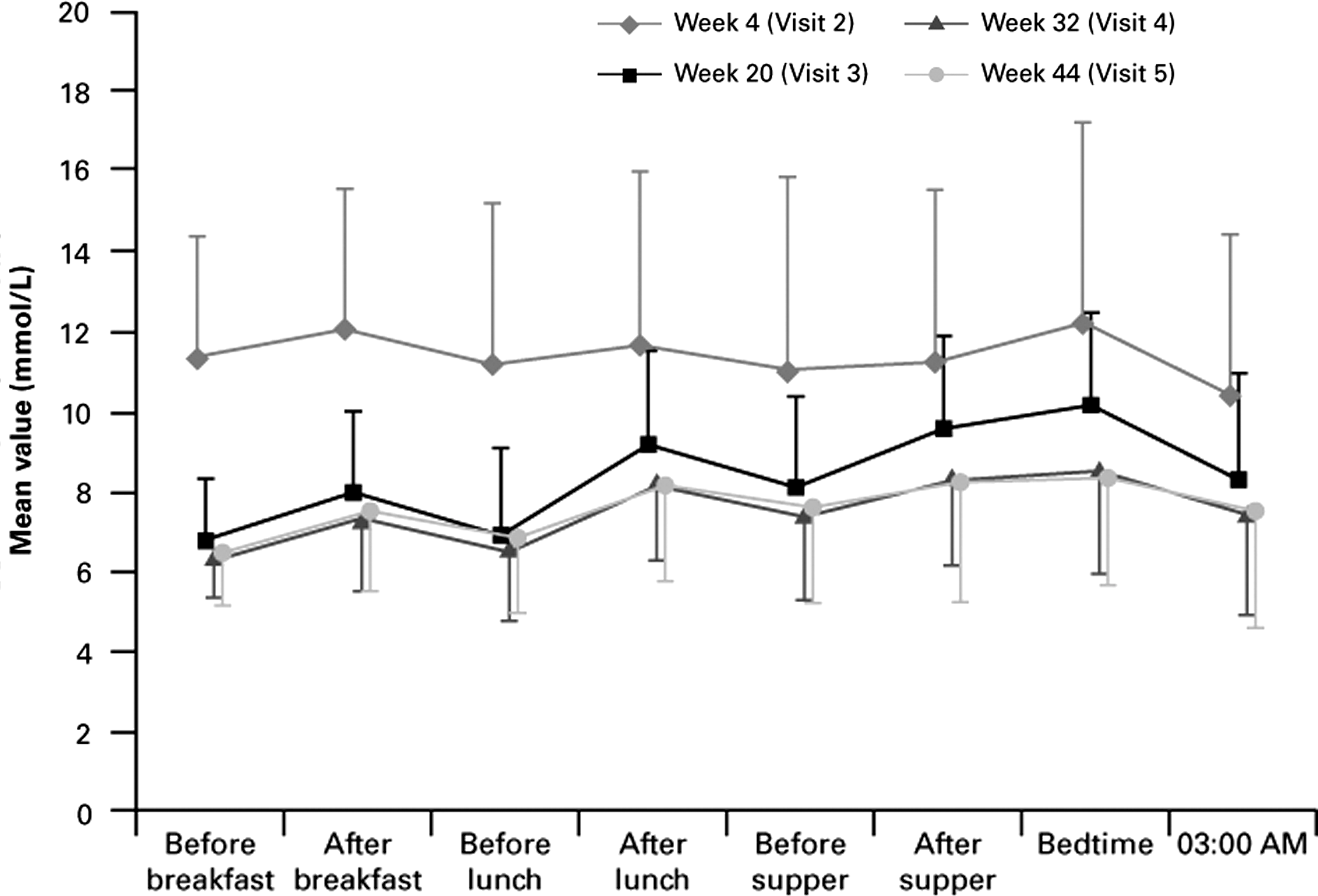
Eight-point glycemic profile at each visit for the intention-to-treat population.
There was no change in body weight from baseline to end point (telecare, 0.4±3.4 kg; SMBG, 0.4±5.1 kg), with no difference between treatment groups.
Hypoglycemia
The incidence (events per patient-year) of total symptomatic hypoglycemia (telecare, 1.89; SMBG, 1.76), severe hypoglycemia (telecare, 0.04; SMBG, 0.02), and severe nocturnal hypoglycemia (telecare, 0.02; SMBG, 0.01) was low and comparable between the telecare and conventional SMBG groups.
Discussion
This study was designed to determine whether the use of a telecare system was superior to conventional SMBG in terms of change in HbA1c in type 2 diabetes patients with poor glycemic control with OHAs initiated on insulin glargine plus insulin glulisine. Our results show that both monitoring systems provided comparable and significant improvements in HbA1c on our chosen insulin treatment strategy. This switch to, and titration of, glargine followed by the addition and titration of a single dose of glulisine at the meal with the highest postprandial glucose excursion was associated with significant improvements in glycemic control, regardless of the blood glucose monitoring intervention.
In prior studies evaluating telecare programs, telemedicine was associated with some advantages in type 1 diabetes patients in terms of glycemic control. 1 –3 However, information in type 2 diabetes patients is much scarcer. A significant reduction in HbA1c has been reported by Kim and Kim, 13 but that study had no control group. Similarly, Cho et al. 14 showed an improvement in glycemic control using an internet system or mobile phone assistance. A more direct comparison between telecare and standard care was performed by Rodriguez-Idigoras et al. 15 in a 1-year study, but the authors failed to find a significant difference in HbA1c between the two groups.
The lack of any difference in our study may have several explanations. First, the telecare system used in our study had the most current technology available at the time. However, it was not able to support dose decision-making in real-time, allowing communication from patient to physician but not direct feedback from the physician. This limitation may have contributed to the suboptimal use of the system, so that of 114 patients, only 76 (67%) used the system fully. It is possible that the more sophisticated interactive systems available today may have produced different results. However, it is worth considering that the introduction of glulisine resulted in a smoother glucose profile (i.e., reduced postprandial glucose excursions) with a very low rate of hypoglycemic events; therefore, the more stable glycemic control may have limited the need for more frequent insulin adjustments. In agreement with this interpretation are the consistency of both insulin doses and the distribution of glulisine doses for the three main meals for the duration of the study.
An alternative or additional explanation may be that the basal plus approach was so simple that there were no differences between groups. While the algorithm used in this study for glargine titration (Treat-to-Target) has already been validated in previous studies, 8,16 our algorithm for prandial insulin titration provides information not only on the optimal dosage, but also the optimal timing of insulin injection. This user-friendly, conceptually simple approach is expected to provide good glycemic control, even in the SMBG patient group, thus reducing the need for telecare programs. Indeed, in both treatment groups, good glycemic control was achieved, as indicated by the HbA1c level at the end of the study and the percentage of patients with HbA1c ≤7.0%. This interpretation is further supported by a positive effect on quality of life assessment. 17
The magnitude of improvement in glycemic control in both groups was consistent with those reported in earlier studies of basal–bolus regimens combining long-acting (i.e., glargine, detemir) plus short-acting insulins (i.e., glulisine, aspart). 9,18 –21 In our study, the physiological rationale for the intensification of the insulin regimen in patients with type 2 diabetes was (1) to manage FPG and (2) to identify the mealtime with the greatest daily postprandial glucose excursion. Consistent with dietary habits in Italy, only a small percentage of patients injected glulisine at breakfast with an almost equal distribution of patients receiving glulisine at lunch and supper. Nevertheless, in both groups, a single dose of glulisine was sufficient to maintain glycemic control during the study.
It is interesting that there was no significant change in body weight in spite of significant changes in insulin dose and fasting plasma insulin levels. The reasons are unclear and need confirmation, but it may be that the low rate of hypoglycemia contributed to reduced defensive eating. 22
In terms of study limitations, we must acknowledge the lack of demographic data for patients at treatment initiation (Visit 3) and the potential for the treatment groups to have become unbalanced as a result of patient drop-out during the titration phase, primarily for failing to reach FPG ≤7 mmol/L. However, as demographic data for the drop-out patients were similar at Visit 1 to data from the ITT population across both treatment groups, we do not expect any significant or inconsistent changes to have occurred to the overall group demographics during the titration phase. It should also be noted that patients who entered the treatment phase had achieved target FPG levels during the titration phase and, thus, may have been more compliant both to treatment and to monitoring. Although in other studies telecare has shown a cost benefit, 1 –3 the cost benefit ratio was not estimated in this study.
In conclusion, the telecare system did not provide an advantage in glycemic control over conventional monitoring in this study population. Both patient groups did, however, still achieve a significant reduction of HbA1c with our treatment regimen based on basal insulin plus one injection of prandial insulin given at the time of the meal with the most evident glucose excursion. This improved glycemic control was achieved by a marked improvement in daily plasma glucose profile while maintaining low FPG levels, a low risk of hypoglycemia, and a neutral effect on body weight, using simple algorithms for insulin dose adjustments. Further investigation will be required to explore how long the improvement in glycemic control can be maintained before introducing a second injection of insulin and to what extent this regimen can be applied in patients with long-standing basal insulin therapy.
Appendix
Participating principal investigators are given with their institutional affiliation(s): Stefano Del Prato, M.D., Department of Endocrinology and Metabolism, University of Pisa, Pisa, Italy; Giacomo Vespasiani, M.D., Presidio Ospedaliero Madonna del Soccorso, San Benedetto del Tronto, Italy; Renato Lauro, M.D., Azienda Ospedaliera Universitaria Policlinico Tor Vergata, Rome, Italy; Francesco Dotta, M.D., Azienda Ospedaliera Universitaria Senese–Policlinico Le Scotte, Siena, Italy; Andrea Corsi, M.D., Ospedale La Colletta, Arenzano, Italy; Giuseppe Rosti, M.D., Azienda Ospedaliera Nazionale Santi Antonio e Biagio e Cesare Arrigo, Alessandria, Italy; Franco Gregorio, M.D., Ospedale E. Profili, Fabriano, Italy; Francesco Fallucca, M.S., Azienda Ospedaliera Sant'Andrea, Rome, Italy; Rossella Iannarelli, Nuovo Ospedale San Salvatore, L'Aquila, Loc. Coppito, Italy; Maurizio Di Mauro, M.D., Presidio Ospedaliero Garibaldi Ambulatorio Diab e medicina interna, Catania, Italy; Emanuele Bosi, M.D., Ospedale San Raffaele di Milano, Milan, Italy; Francesco Giorgino, M.D., Azienda Ospedaliera Policlinico Consorziale, Bari, Italy; Roberto Torella, M.D., Azienda Universitaria Policlinico della Seconda Università degli Studi di Napoli, Naples, Italy; Anna Vittoria Ciardullo, M.D., Ospedale Civile Bernardino Ramazzini, Carpi, Italy; Pasqualino Calatola, ASL SA/2, Ex Ospedale Vernieri, Salerno, Italy; Paolo Di Bartolo, M.D., Ospedale Santa Maria delle Croci, Ravenna, Italy; Massimo Boemi, M.D., INRCA, Ancona, Italy; Maurizio Carlini, M.D., Ospedale Maria Vittoria, Torino, Italy; Paolo Foglini, M.D., Ospedale Civile AZ. USL 11, Fermo, Italy; Pietro Pata, M.D., Azienda Ospedaliera Piemonte, Messina, Italy; C. Augusto Lovagnini-Scher, M.D., Centro di attenzione al diabetico, Presidio territoriale di Cusano Milanino, Azienda Ospedaliera San Gerardo di Monza, Milan, Italy; Sergio Leotta, M.D., Ospedale “Sandro Pertini,” Rome; Fiorella Massimiani, M.D., and Rita Amoretti, M.D., P.O. San Maria, Complesso Ospedaliero San Giovanni Addolorata, Rome, Italy; Carlo Bruno Giorda, M.D., Ospedale Maggiore, Chieri, Italy; Adolfo Arcangeli, M.D., Azienda USL 4 di Prato ospedale misericordia e dolce, Prato, Italy; Nazario Melchionda, M.D., Policlinico San Orsola Malpighi, Bologna, Italy; Geremia B. Bolli, M.D., Policlinico Monteluce, Perugia, Italy; Mauro Cignarelli, M.D., Azienda Ospedaliera Ospedali Riuniti di Foggia, Foggia, Italy; Aldo Galluzzo, M.D., Azienda Ospedaliera Universitaria Policlinico “Paolo Giaccone,” Palermo, Italy; Emanuela Orsi, Fondazione Ospedale Maggiore IRCCS, Policlinico Mangiagalli e Regina Elena, Milan, Italy; Cecilia Invitti, M.D., Centro Auxologico, P.O. San Michele, Milan, Italy; Angelo Venezia, M.D., Ospedale Madonna delle Grazie, Matera, Italy; Brunella Capaldo, M.D., Dipartimento di Medicina Clinica e Sperimentale, Università degli Studi di Napoli Federico II, Naples; Celestino Giovannini, M.D., Poliambulatorio, ASL 11, Reggio Calabria, Italy; and Gabriele Maolo, M.D., Azienda Sanitaria Unica Regionale Marche, Zona territoriale 9, Ospedale Generale Provinciale, Macerata, Italy.
Footnotes
Acknowledgments
This study was supported by sanofi-aventis. Editorial support was provided by Anisha Mehra, Ph.D., of Medicus International and funded by sanofi-aventis.
Author Disclosure Statement
S.D.P., A.N., and G.V. were involved in the conception of the study and its design. A.C.L.-S., S.T., and S.L. were involved in carrying out the study and collecting the data. All authors contributed to the writing of this manuscript, including critical review and editing of each draft, and approval of the submitted version. The authors have no competitive financial interests to disclose. S.D.P. has served on advisory panels for Novartis Pharmaceuticals, Merck & Co., Roche Pharmaceuticals, Roche Diagnostics Corporation, Pfizer Inc., Eli Lilly and Co., Amylin Pharmaceuticals Inc., Mannkind Corp., Boehringer Ingelheim, Bristol-Myers Squibb, Astra Zeneca, GlaxoSmithKline, sanofi-aventis, and Takeda Pharmaceuticals, has received research support from Merck & Co. and Takeda Pharmaceuticals, and has served as a member of a speakers bureau for GlaxoSmithKline. A.N. has received research support from sanofi-aventis, Novo Nordisk, Bayer, Novartis, and Bristol-Myers Squibb and has served as a member of a speakers bureau for Eli Lilly and Bristol-Myers Squibb. G.V. has served on advisory panels for Eli Lilly, Novo Nordisk, Roche Diagnostics, GlaxoSmithKline, Meteda, and sanofi-aventis and has received research support from Lifescan. A.C.L.-S., S.T., and S.L. have no conflicts of interest to disclose.
