Abstract
Background:
FlexTouch® (Novo Nordisk A/S, Bagsvaerd, Denmark) is a new prefilled insulin pen for people with diabetes, with a novel injection mechanism and no push-button extension at any dose setting. This study compared the injection force of FlexTouch with that of SoloStar® (sanofi-aventis, Paris, France) and KwikPen® (Eli Lilly & Co., Indianapolis, IN).
Methods:
Injection force was measured with the manufacturers' recommended needle attached to each pen (NovoFine® [Novo Nordisk] 32-gauge tip extra thin wall 6 mm needle for FlexTouch and BD [Franklin Lakes, NJ] MicroFine™ 31-gauge 5 mm needle for SoloStar and KwikPen) during injection of the maximum dose (60 IU for KwikPen and 80 IU for FlexTouch and SoloStar). Injection was performed at three different constant push-button speeds.
Results:
FlexTouch had a significantly (P<0.0001) lower injection force than SoloStar and KwikPen at all injection speeds. The mean±SD injection force of FlexTouch was 5.1±0.5 N. At 4, 6, and 8 mm/s push-button speeds, the injection force of SoloStar was 13.5±2.1, 19.1±1.9, and 26.9±2.4 N, respectively, and the injection force of KwikPen was 14.5±1.9, 20.9±1.4, and 28.2±1.4 N, respectively.
Conclusions:
The injection mechanism of FlexTouch means that insulin injection is driven by a torque spring and not the thumb pressure of the user. This results in a 62–82% lower injection force with FlexTouch than other prefilled insulin pens.
Introduction
The development of pens has addressed many of the issues with conventional vial and syringe, but the force required to press the push-button on pens and inject insulin (injection force or dose force) has an influence on the ease of use for some people with diabetes, particularly those with type 2 diabetes, who are often elderly, and those with co-morbidities affecting manual dexterity, such as arthritis. 13 –17 Modern prefilled pens such as Next Generation FlexPen® (Novo Nordisk A/S, Bagsvaerd, Denmark), KwikPen® (Eli Lilly & Co., Indianapolis, IN), and SoloStar® (sanofi-aventis, Paris, France) have helped to address the issue of injection force. 18 –22 One direct comparison of all three pens showed that Next Generation FlexPen had a significantly lower injection force than SoloStar and KwikPen when tested at a constant push-button speed (isokinetic). 18 Another study suggested lower injection force with SoloStar than with KwikPen and Next Generation FlexPen when tested at a constant volume flow rate (isoflow). 23 However, different needles were used on the pens in the study of van der Burg, 23 and the effect of the needle inner diameter was not taken into account. As the injection force is influenced by the inner diameter of the needle, rather than the outer diameter, as mentioned by van der Burg, 23 this may account for some of the differences. Also, the isoflow methodology used in that study may not replicate the normal use of pens as the user would have to constantly vary the rate of injection to achieve a uniform flow rate.
However, current prefilled insulin pens all function on the principle of dialing up a dose with a corresponding extension of the push-button as the dose increases and subsequent manual depression of the push-button to deliver the dose. FlexTouch® (Novo Nordisk A/S) is a new prefilled insulin pen that has no push-button extension at any dose, in which the injection is driven by a torque spring within the pen and not by the thumb pressure of the user. The torque spring is loaded during dose setting, and the spring energy is activated by pressing down the push-button. The activation force is determined by the push-button spring. With this unique spring-loaded mechanism, FlexTouch aims to eradicate the difficulties of injecting and, together with the absence of push-button extension, to improve the stability of holding the pen while injecting, to reduce bruising at the injection site, to make the administration of insulin smoother and more convenient, and to reduce the burden of injection. Here we present the results of a study to compare the injection force of FlexTouch with two other widely available prefilled pens, SoloStar and KwikPen.
Materials and Methods
Materials
Pens tested were FlexTouch (filled with Levemir® [batch YP51685] or NovoRapid® [batch XL70014], both from Novo Nordisk), SoloStar (filled with Lantus® [batch 40C531] from sanofi-aventis), and KwikPen (Humalog® [batch A638534F] from Eli Lilly). The manufacturers' recommended needles were used: NovoFine® (Novo Nordisk) 32-gauge tip extra thin wall (etw) 6 mm needle for FlexTouch and BD (Franklin Lakes, NJ) MicroFine™ 31-gauge 5 mm needle for SoloStar and KwikPen. Only one needle of each type was used for all injection force tests to avoid variation in measured injection force caused by the flow stress of different needles.
Equipment
All testing was carried out in a climate-controlled laboratory at 20°C (±2°C) and 45% relative humidity (±7.5%). All pens were acclimatized for at least 4 h before testing. The tensile tester (Lloyd LF Plus, Lloyd Instruments Ltd., Humlebaek, Denmark) was set to compression mode, and a calibrated transducer (measuring cell) with a maximum of 50 N was used.
SoloStar and KwikPen injection force testing
The injection force of SoloStar and KwikPen was measured as an isokinetic comparison (injection force at a constant push-button speed, corresponding to the speed a user would depress the push-button when injecting) while delivering maximum doses (60 IU for KwikPen and 80 IU for SoloStar) at three constant push-button speeds (4 mm/s, 6 mm/s, and 8 mm/s).
The manufacturers' recommended needle was attached, and air was expelled by performing air shots until liquid appeared at the tip of the needle (i.e., the pen was primed). The pen was set to the maximum dose, and the first dose was discarded. The pen was then set to the maximum dose and placed in a fixture in the tensile tester with the push-button pointing upward, allowing free flow from the needle. A dose was delivered by depressing the push-button at a speed of 4 mm/s; this was repeated at a speed of 6 mm/s and then 8 mm/s (maximum extension of push-button was 32 mm for SoloStar and 29 mm for KwikPen). The whole procedure was repeated until 25 measurements were obtained at each push-button speed for each pen type.
FlexTouch injection force testing
Because FlexTouch has the spring-loaded mechanism for delivery of the dose, the push-button speed has no influence on the rate of insulin delivery, and therefore FlexTouch was not tested at three speeds as above. Instead, the injection force of FlexTouch was measured as activation force at the maximum dose (80 IU).
The manufacturers' recommended needle was attached to the pen, and air was expelled by performing air shots until liquid appeared at the tip of the needle (i.e., the pen was primed). The pen was then set to 80 IU and placed in a fixture in the tensile tester with the push-button pointing upward, allowing free flow from the needle. The lowest speed for pushing down the injection button was set (0.2 mm/s), which enabled a precise and highly sensitive increase in the pressure on the push-button of the pen. This pressure was increased until the “ignition” point was reached, and the torque spring was activated. The force registered at the point of “ignition” was recorded as the activation force for FlexTouch. This procedure was repeated for 25 pens.
Needle inner diameter testing
To determine if the different needles influenced the injection force measured in this study, the inner diameter of the NovoFine 32-gauge tip etw 6 mm needle and BD MicroFine 31-gauge 5 mm needle was measured. The inner diameter was calculated by measuring the flow resistance and total length of the needle. Each needle's inner diameter was measured five times, and the mean was calculated.
Results
The mean injection force was significantly lower for FlexTouch with NovoFine 32-gauge tip etw 6 mm needles compared with SoloStar with BD MicroFine 31-gauge 5 mm needles and KwikPen with BD MicroFine 31-gauge 5 mm needles at all injection speeds (P<0.0001) (Fig. 1 and Table 1). The injection force of FlexTouch was 65%, 76%, and 82% lower than KwikPen at 4 mm/s, 6 mm/s, and 8 mm/s, respectively (P<0.0001), and 62%, 73%, and 81% lower than SoloStar at 4 mm/s, 6 mm/s, and 8 mm/s, respectively (P<0.0001).
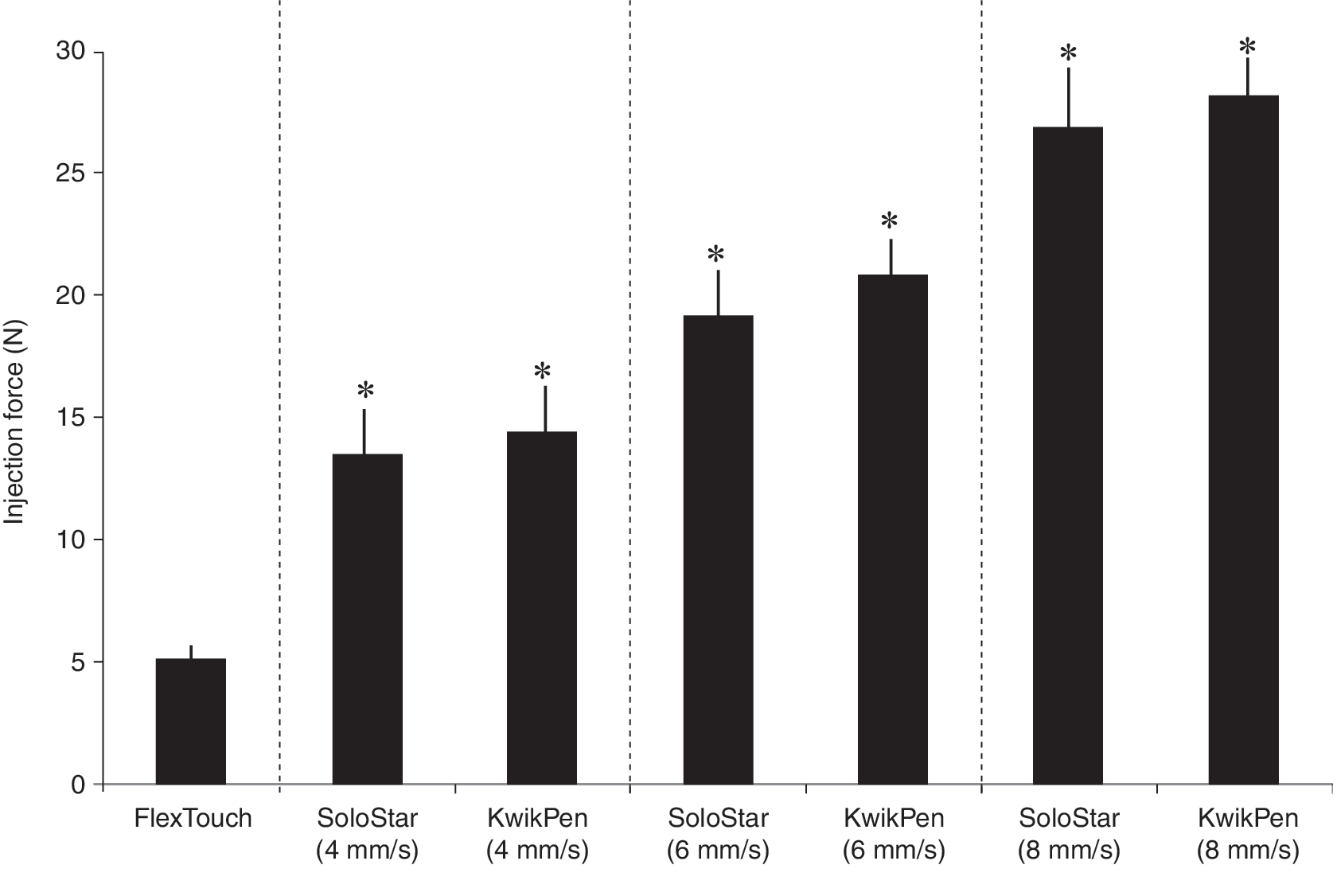
Injection force for FlexTouch, SoloStar, and KwikPen with manufacturers' recommended needle attached. Data are mean values from 25 measurements for each pen type at each push–button speed. Columns represent means, and lines represent +SD. *P<0.0001 versus FlexTouch.
Injection force is in newtons.
The needle diameter for both the NovoFine 32-gauge tip etw 6 mm needle and the BD MicroFine 31-gauge 5 mm needle was measured five times for each needle. The mean (±SD) inner diameter was 0.153±0.00000 mm for NovoFine and 0.154±0.00055 mm for BD MicroFine. The two needles had almost equivalent inner diameters. Therefore, injection force results for each pen were not biased by the choice of needle used in this study.
Discussion
The aim of this study was to determine whether the injection force of FlexTouch is significantly lower than that of currently available prefilled insulin pens—specifically, SoloStar and KwikPen. The injection force of FlexTouch was significantly lower than that for SoloStar and KwikPen at all injection speeds—with the injection force of FlexTouch being 62–82% lower than the two other widely available pens tested.
Each pen was tested with the manufacturer's recommended needle with FlexTouch being recommended with NovoFine needles and SoloStar and KwikPen being recommended with BD MicroFine needles. Because the injection force of FlexTouch is driven by a torque spring, its low injection force is unlikely to be affected by needle diameter. However, for most prefilled insulin pens, the needle inner diameter influences the injection force needed to deliver a dose, with a larger diameter usually associated with reduced force. 24 Thinner needles are generally preferred by patients as they are associated with less pain, bleeding, and bruising than thicker needles. 25 –27 To overcome these two conflicting issues, therefore, needles such as NovoFine etw have thin-wall technology that allows a thin outer diameter while maintaining the inner diameter. Such thin-wall needles are preferred by patients. 28 In this study, there was no significant difference in inner diameter between the needles, meaning that the injection force measurement was unrelated to the needles used and solely a result of the pens' mechanisms. Some recent reports of injection force of insulin pens have failed to assess the possible influence of the needle used on the injection force measured. 22,29
The injection force of FlexTouch is driven by a spring-loaded mechanism within the pen and not by the thumb pressure of the user, which means, as shown by the results of this study, that a substantially and significantly lower injection force is required to deliver the insulin dose. This may represent an advantage for patients with diabetes. In particular, this may have benefits for people with diabetes who have less hand strength, smaller hands, or impaired manual dexterity because the lower force required for injection contributes to the ease with which the insulin dose is injected. The lack of push-button extension on FlexTouch (alongside the very low injection force) may help patients keep the pen stable and more easily and smoothly inject when delivering their insulin—again, this may be especially useful in patients with impaired manual dexterity and during the injection of high doses.
The design features of FlexTouch were made with the needs of patients in mind, with the aim of making the administration of insulin easier and more convenient than with currently available pens. Improved convenience and ease of use of insulin pens may improve adherence to the patient's insulin regimen. However, this report is a technical report of injection force, and therefore the possible effects of the reduced injection force of FlexTouch on patient perception, preference, and adherence has yet to be determined.
Further studies will be needed to assess the impact of the novel spring-loaded mechanism on the usability of FlexTouch and on other functional properties of this pen. In a complementary study comparing dose accuracy of FlexTouch, SoloStar, and KwikPen, accuracy of FlexTouch was demonstrated, and FlexTouch was accurate and consistent for delivering low (1 IU), medium (40 IU), and high (80 IU) doses. 30 Furthermore, a similar mechanism has been used for a pen to deliver growth hormone therapy, and usability studies among children suggest that the pen is well accepted, easy to use, intuitive, easy to learn to use, and reliable. 31,32
FlexTouch is a new prefilled insulin pen with a unique spring-loaded mechanism that significantly reduces injection force compared with other prefilled pens. The simplification to insulin injection conferred by this pen could benefit all patient populations by reducing the burden of insulin treatment. Furthermore, it could potentially increase uptake of such devices in countries where vials and syringes are still widely used. The unique features of FlexTouch are a clear advance in insulin pen technology compared with all other currently available insulin pens.
Footnotes
Acknowledgments
The study was funded by Novo Nordisk A/S, Bagsvaerd, Denmark. Editorial assistance was provided by Martin Gilmour at ESP Bioscience (Sandhurst, UK).
Author Disclosure Statement
H.H., M.N., M.R.H., D.B., and N.B.T. are employees of Novo Nordisk A/S.
