Abstract
Recent technological advancements in insulin administration and glucose monitoring have allowed patients with diabetes to become increasingly involved in their own care. Devices replacing the traditional vial and syringe, such as insulin pens, are gaining popularity and offer simple and convenient insulin administration. Pen devices are associated with improved dose accuracy, reducing the risk of hypo- or hyperglycemia, and are continually being updated with new safety features in order to optimize their performance. In patients for whom glucose variability remains a problem, continuous subcutaneous insulin infusion via an implanted canula or continuous intraperitoneal insulin infusion via an implanted pump is safe and effective when used correctly, although cost can be a limitation. More accurate retrospective and real-time continuous monitoring devices, which can better detect blood glucose excursions, have become standard components of modern-day diabetes management. The most recent devices have sensor-signaling capabilities with wireless data transmission, leading to reduced time delay and more accurate alerts. Ultimately, though, while self-management remains a critical factor in improving glycemic control at present, human error may undermine even the most accurate treatment interventions. A key long-term goal in diabetes management is, therefore, to develop an automated and accurate closed-loop system for blood glucose monitoring and insulin delivery to better reflect the physiological mechanisms of glucose homeostasis and remove the “human” element. This “artificial pancreas” would offer the most innovative intervention for diabetes management and has the potential to considerably reduce the patient's burden of self-care.
Introduction
A challenging component of insulin-dependent diabetes management is the need to tailor insulin regimens to adjust for differences between patients and, in an individual patient, to account for changes in insulin sensitivity and needs due to food consumed or physical activity undertaken. 2,4 At present, a patient's involvement in his or her own care is a critical factor in achieving optimal glycemic control. 4 Patients with advanced disease must subject themselves to a regimented daily routine that leaves little margin for “error” in terms of spontaneity and may impact their quality of life. Furthermore, the requirement for a daily regimen of insulin replacement combined with repeated glucose monitoring necessitates reliance on several devices.
This article reviews the history of technological developments underlying insulin delivery and glucose monitoring devices that have been designed to assist patients in achieving successful self-management of their condition. We also discuss recent progress in achieving the goal of closed-loop systems—the artificial pancreas—a means of automated insulin delivery and glucose monitoring that represents a step closer towards mirroring the physiological mechanism of glucose homeostasis and may reduce the burden of self-care for patients.
Insulin Delivery: A Brief Overview
Insulin delivery in patients with T1DM, and in some insulin-dependent T2DM patients, is achieved through multiple daily injections (MDIs) or continuous subcutaneous insulin infusion (CSII).
MDIs
An MDI treatment regimen is the most widely used by patients with diabetes and requires daily administration of both rapid- and long-acting insulin. Conventionally, an insulin vial and syringe was the most common method used to deliver MDI treatment, but the use of insulin pen devices is gaining in popularity. 5,6 Insulin pen devices have been designed to provide a convenient and simple means of insulin administration. These devices consist of an insulin cartridge or reservoir and a syringe and are available as either reusable/durable or disposable/prefilled types. Because the insulin is kept in the device, the patient only needs to screw on a small pen needle before administration. 5 The first insulin pen devices were introduced in the 1980s, and since then they have been continuously updated and improved in order to optimize their performance. 5
Insulin pens offer many advantages over the traditional vial and syringe method. These primarily relate to greater convenience, discretion, and ease of use and less pain associated with administration, which translate into greater patient preference, social acceptability, and treatment adherence. 5,7 –9 In addition, insulin pen devices are associated with increased dosing accuracy and safety features, such as audible clicks with each dose increase, 5 which reduce the risk of over- and under-dosing and consequent hypo- and hyperglycemia, respectively. Recent advances in insulin devices include a built-in recording function from which data can be downloaded. The HumaPen® Memoir™ (Eli Lilly, Indianapolis, IN) is the first reusable digital insulin device with a memory. The HumaPen records the dose, date, and time of the previous 16 injections, and this memory acts as a safety feature to prevent overdosing. Despite advances in MDI administration, variability in glucose levels remains a problem for some patients, and this can be partially addressed by CSII. 2
Despite the advantages of insulin devices over vial and syringe methods, the use of insulin pen devices varies widely between countries: around two-thirds of insulin prescriptions in Europe and approximately three-quarters in Japan are for pen devices; 10 in contrast, only 15% of patients are reported to use insulin pens in the United States. 11 It has been suggested that the higher prescription cost of insulin cartridges and prefilled insulin pens compared with insulin vials may account for the lower adoption rates in the United States. 6 However, it has been shown that despite the higher unit cost of insulin in pen devices versus vials, overall diabetes-related treatment costs are lower with pen devices than with vial and syringe, because of factors such as fewer hospital visits. 12,13
CSII
First introduced in 1976, CSII pumps mimic the pattern of insulin release from the pancreas, thus providing a more physiological regimen for insulin replacement. 14 Initially, CSII devices were equivalent in size to a large backpack, 15 and even though their size has subsequently been reduced, making them suitable for use in outpatients, the systems tend to be used primarily by patients experiencing severe hypoglycemia, hypoglycemia unawareness, or the dawn phenomenon in T1DM, 16 although restrictions on CSII pump use do vary by country. As well as providing a more physiological regimen, CSII offers increased lifestyle flexibility with the possibility of more convenient adjustment of insulin administration according to physical activity or food intake. 2,17
From a functional perspective, CSII involves the delivery of insulin via an infusion system that is attached through tubing to a canula that is subcutaneously implanted, usually into the abdomen. 15 The insulin pump consists of four main parts: a disposable plastic reservoir, filled with up to 300 units of insulin; a screw-drive mechanism that moves the insulin through the tubing at variable rates; a small computer that is programmed to dispense the insulin; and a battery as a power source. 15 With the application of the insulin pump system, continuous slow infusion of rapid-acting insulin analogs is used to reproduce basal secretion, which can be adjusted for each patient, while bolus injections are infused prior to meals and snacks.
Optimal use of the insulin pump requires frequent decision-making, and, therefore, patients must have basic knowledge about insulin pharmacodynamics and carbohydrate counting and be able to assess situations, as well as have both the motivation and ability to operate a computerized pump. 15 The newer models have several advanced features, such as incorporation of bolus calculators and, when coupled with a continuous glucose monitoring (CGM) system, alarms. 17 Thirty years after its introduction, the use of CSII continues to increase, especially among children and adolescents, owing to its safety and efficacy when used correctly. 17
CSII versus MDIs
Evidence supports the effectiveness of CSII versus MDI in achieving glycemic control 18 –20 and improving treatment satisfaction and quality of life. 21 –24 Two meta-analyses comprising over 2,000 patients and a smaller pooled analysis in 139 patients demonstrated a decrease in glycosylated hemoglobin (HbA1c) of 0.4–0.5% and a decrease in insulin requirements with CSII versus MDI. 18 –20 In general, compared with MDI, CSII provides small, but clinically important, reductions in HbA1c, less blood glucose variability, decreased risk of severe hypoglycemia, and a more effective strategy for tackling the dawn phenomenon. 17,19,25 Although CSII offers several benefits over MDI, it may also be associated with a higher, albeit still relatively low, rate of diabetic ketoacidosis. A review of studies in children and adolescents suggested that in patients with good compliance and adequate family support, diabetic ketoacidosis rates were low or even nonexistent and that most episodes of diabetic ketoacidosis occurred early after pump start. This indicates a learning curve, which highlights the importance of patient education in CSII use. 26 Although the availability of insulin analogs has improved the treatment of diabetes, CSII remains the first option for patients experiencing severe hypoglycemic episodes, very poor glycemic control, and/or marked glucose variability despite treatment with optimized MDI (Table 1). However, despite the advantages of CSII, its use can be limited by cost implications as it requires a multidisciplinary team, intensive patient education, and continuous follow-up. Indeed, it has been suggested that CSII is only cost-effective for those patients who experience a substantial improvement in HbA1c or a decrease in severe hypoglycemic episodes. 17,27 Furthermore, a recent study conducted in patients with T1DM reported that CSII offered no benefits over insulin glargine-based MDI with regard to glycemic control and was associated with around fourfold higher costs. 28
Reproduced from Valla 15 under the Creative Commons Attribution License. Copyright © 2010 Vasiliki Valla. HbA1c, glycosylated hemoglobin; MDI, multiple daily injections; NMR, nuclear magnetic resonance.
Continuous intraperitoneal insulin infusion
An alternative to CSII is continuous intraperitoneal insulin infusion (CIPII), which is delivered through an implantable pump, as a treatment option for those patients with diabetes who fail to achieve satisfactory glycemic control with other intensive insulin treatment regimens. 27,29 The intraperitoneal administration of insulin allows blood glucose values to normalize more rapidly after a meal with more predictable insulin profiles than with subcutaneous (SC) insulin. 30 –32 Much of the intraperitoneal insulin is absorbed through the portal system, which closely mimics normal physiological action, resulting in improved hepatic uptake and lower peripheral plasma insulin levels. 33 Intraperitoneal insulin may also lead to improved glucagon secretion and hepatic glucose production in response to hypoglycemia. 34
CIPII has been reported to result in greater improvements in glycemic control compared with CSII, with a 0.8% decrease in HbA1c and an 11% increase in the time spent in euglycemia. 27 Others have reported a reduction in the incidence of severe hypoglycemia with CIPII (DiaPort™, Roche Diagnostics, Indianapolis), with improved quality of life and no weight gain. 35,36 A systematic review of the safety and efficacy of implantable insulin pumps over a 15-year period reported that, compared with CSII, use of implantable pumps significantly reduced blood glucose fluctuations and improved quality of life in both T1DM and T2DM patients, with significant weight reduction observed in those with T2DM. 29 However, it should be noted that CIPII may be associated with higher operating costs and local adverse events. 27,37,38
Blood Glucose Monitoring
Fingerstick capillary blood glucose monitoring, introduced in the 1970s, was the first step in achieving target glycemic control using intensive insulin therapy, leading to a substantial reduction in the risk of long-term complications from diabetes. 39 The correlation between sufficient glucose control and reduced microvascular complications was demonstrated in 1993 by the Diabetes Control and Complications Trial. 1 Frequent glucose monitoring was shown to play an important role in achieving glycemic control by providing guidance for insulin timing and accurate dosage. 1,15 Although self-monitoring of blood glucose (SMBG) is advocated in treatment guidelines for the management of insulin-treated patients with diabetes, 15,40 the technique is not without limitations. These primarily involve non-adherence, due to inconvenience and pain, and poor scheduling for measurements, which can hamper individualized treatment.
CGM
When CGM was initially introduced in the 1960s, glucose levels were obtained every 1 s to 15 min via intravascular access, a method that was associated with a significant risk of infection and thrombosis. 2 Progressive improvements over the last 20 years have led to the development of devices that measure glucose levels in the interstitial fluid (ISF) and are, therefore, less invasive. 2,40,41 For example, an enzyme-based electrode inserted directly into the SC tissue detects current generated from the oxidation of glucose and oxygen to hydrogen peroxide via glucose oxidase. 42 Oxidation of glucose by the enzyme-coated sensor removes an electron from each glucose molecule in the ISF, resulting in an electrical current, which the sensor converts into an interstitial glucose reading. The accuracy of SC CGM readings is dependent on the equilibrium of glucose levels between ISF and plasma and whole blood, while accounting for a time delay of glucose diffusion between plasma and ISF. 43
Retrospective CGM
Retrospective continuous glucose monitors, introduced in 1999, were the first CGM devices to become commercially available. Manufactured by Medtronic (Northridge, CA), the CGMS® provided a 3-day glycemia history upon measurement of ISF glucose levels. 41 The latest advance in retrospective CGM technology is the iPro™ 2 Professional CGM, recently launched by Medtronic. The device consists of a small glucose sensor that is inserted subcutaneously and records glucose level readings every 5 min for up to 7 days; after the 168-h monitoring period, the readings are provided for physician review. The system is intended to guide future management of a patient based on response to trends noticed, such as when best to take fingerstick glucose measurements to enable better management and outcomes.
Real-time CGM
Real-time CGM devices were the successors of retrospective glucose monitors. The first such device, Glucowatch (originally manufactured by Cygnus, Inc., Redwood City, CA), was beset by problems when initially introduced more than 10 years ago. 41 Advances in technology have, however, enabled the establishment of real-time CGM devices as a standard component of diabetes management.
Currently approved real-time CGM models include the DexCom™ (San Diego, CA) SEVEN®, the Abbott (Alameda, CA) FreeStyle Navigator®, and the Menarini (Florence, Italy) GlucoDay®, which, with the exception of GlucoDay, all use a glucose oxidase-based sensor. The DexCom STS and the FreeStyle Navigator also use an SC enzyme-based electrode, and both are wire-free devices with recording times of 7 and 5 days, respectively. 40,44,45 In a recent comparator analysis conducted in patients with T1DM during hypoglycemic clamp studies, numerical accuracy was similar between the FreeStyle Navigator and GlucoDay devices, and both had high clinical accuracy during hypoglycemia. 46 Real-time CGM studies can be used in conjunction with MDI- or CSII-administered treatment. In a recent, prospective, real-life study that compared use of CGM (DexCom Seven PLUS) in patients with T1DM on MDI therapy versus CSII therapy, 6-month use of real-time CGM was shown to provide similar benefits in usability, persistence, and glycemic control when used with either regimen. 47
Real-time CGM serves as a means not only to facilitate self-management of glucose levels, but also to enable patients to better understand the concept of dose adjustments and treatment effects. 15 For example, some patients are instructed that upon an increase or a decrease in glucose level of 1–2 mg/dL · min, they should adjust (increase or decrease according to the direction of the trend) their insulin dose by 10% from the dose they would have injected for their meal if they had been unaware of the change rate of blood glucose. 41
Sensor-augmented insulin pump therapy
In 2006, the sensor-augmented insulin pump, Medtronic's Paradigm® REAL-Time system, was launched, introducing the first device that had all of the required components communicating together that were necessary for an ambulatory closed-loop artificial pancreas product. 48 Continuous glucose sensing-augmented insulin pump therapy is a further technological advance, whereby sensor-augmented insulin pump therapy integrates real-time CGM with CSII, a technique that has demonstrated improvements in glycemic control in clinical trials and real-world settings. 49 –51
SMBG versus retrospective CGM
Although the CGMS detected significantly more incidences of hypoglycemia and postprandial hyperglycemia compared with standard blood glucose testing, the day-to-day variability was similar between the two groups, and, with a sensor failure rate of 28%, 52 the effectiveness of this method of CGM is debatable.
SMBG versus real-time CGM
The advent of CGM offers the opportunity to increase the duration of time spent within glycemic thresholds for adults and children, 53 –55 thereby improving overall diabetes management. Unlike SMBG, CGM provides detailed information on glucose levels at time points throughout the day, enabling identification of postprandial glucose excursions and hyperglycemic or hypoglycemic episodes. 15,56
Several pivotal studies have demonstrated the ability of CGM to reduce HbA1c levels and glycemic variability, while increasing hypoglycemia awareness, compared with SMBG. 57,58 In the largest study of CGM, sponsored by the Juvenile Diabetes Research Foundation and comprising 322 patients, CGM achieved greater reductions in HbA1c (mean difference, −0.5%) versus SMBG in patients ≥25 years old. 58 The European GuardControl Trial assessed the efficacy of CGM in 156 patients wearing the Guardian® RT monitor (Medtronic) and, likewise, reported that individuals with CGM experienced better improvements in HbA1c levels (0.6% improvement difference) than those using traditional SMBG. 57 Furthermore, the 6-month RealTrend study compared glycemic control in 132 adults and children with uncontrolled T1DM (HbA1c ≥ 8%) using MDI who transitioned to insulin pump therapy using either CGM or SMBG; 59 one group was fitted with the Medtronic MiniMed Paradigm REAL-Time system, an insulin pump with integrated CGM capability, whereas CSII was initiated in the other group. In patients who were fully protocol-compliant (including wearing the CGM sensor ≥70% of the time), HbA1c improvement was significantly greater in the Paradigm REAL-Time group versus the CSII group (−0.96 vs. −0.55%; difference, P = 0.004), with no impact on the rate of hypoglycemia.
Indeed, current guidelines recommend the use of CGM in conjunction with intensive insulin regimens as a tool to help lower HbA1c in adults with T1DM, and this tool may prove useful in younger patients, although adherence is a possible obstacle in these younger individuals. CGM may also be a supplemental tool to SMBG in patients with hypoglycemia unawareness or those prone to frequent hypoglycemic episodes. 60
Sensor-augmented insulin pump therapy versus MDIs and SMBG
The largest study investigating this new technology is the randomized controlled study Sensor-Augmented Pump Therapy for A1c Reduction (STAR), which compares sensor-augmented pump therapy with MDI. The STAR 3 Study compared sensor-augmented pump therapy versus MDI in adult and pediatric patients with inadequately controlled T1DM and found that, over 12 months, the reduction in HbA1c was significantly greater with the integrated sensor and pump therapy (−0.8 vs. −0.2%; P < 0.001). 50 The difference was also significant in pediatric patients (pump vs. injection group, −0.4 vs. +0.2%; P < 0.001). Furthermore, the improvement in glycemic control observed with the integrated sensor and pump therapy was not associated with an increased rate of severe hypoglycemia. 50 It should be noted, however, that the design of this study has been criticized owing to the lack of a third arm comprising patients on MDI also using CGM. This omission limits the interpretation of the study findings because the effects reported in the STAR3 study may be entirely due to the use of CGM in the integrated pump and CGM group. 47
New Advances in Real-Time CGM Systems
New technology is aimed at improving the use of CGM devices, such as facilitating insertion and calibration of the sensor. The next generation of CGM devices should be smaller, simpler devices with increased reliability and accuracy. 39 Although the majority of CGM devices provide threshold and predictive alarms to warn the user of impending or occurring hypo- or hyperglycemic episodes, widespread adoption of these features is often hampered by low accuracy and false alerts. As such, there has been a drive toward the development of a device with increased accuracy, particularly at potentially dangerous low blood glucose levels.
A recent study has evaluated a new glucose-sensor calibration algorithm that is used in the Paradigm Veo™ (Medtronic) insulin pump recently launched in Europe. 56 The Paradigm Veo calibration algorithm demonstrated performance improvement over the existing Paradigm REAL-Time system (using Guardian) in terms of accuracy. Hypoglycemia sensitivity was increased by more than 50%, with 90.5% and 94% of all hypoglycemic events detected for prediction horizons of 15 and 30 min, respectively, and the sensor signal was more reactive to fast glucose excursions. Furthermore, sensor signal-processing delays were substantially reduced in the Veo algorithm. This reduction in time delay provides earlier and more accurate alerts, whereby hypoglycemia can be detected and predicted sooner, thereby enhancing patient safety and affording more time to self-treat potential reactions. 56 Wireless transmission to the Paradigm Veo has been made possible by the introduction of Bayer's CONTOUR® LINK meter (Bayer, Leverkusen, Germany). CONTOUR LINK is also compatible with Medtronic's MiniMed Paradigm insulin pumps and the Guardian REAL-Time system. The system is designed to simplify diabetes control and enhance convenience by (1) simplifying the steps needed to calculate bolus dosing (using the Bolus Wizard® calculator), (2) simplifying calibration of the CGM system, and (3) eliminating the risk of data-entry mistakes. The completely integrated wireless insulin pump system connects with the insulin pump, blood glucose meter, software, and the continuous glucose monitor to enable the storage of information on one device.
Despite the clear utility of CGM, the element of human error that can be introduced by the failure of a patient to respond to the information provided by the device signals a need for further automation of glucose monitoring.
Closing the Loop: The “Artificial Pancreas”
The concept of the artificial closed-loop system was pioneered in the late 1950s, but success was limited by practical constraints. 61 –64 Research efforts in this area have recently been reinvigorated by the introduction of continuous glucose sensors. 65 For ultimate success, the closed-loop system needs to reflect the physiological state in terms of the timing of the insulin response to changing glucose levels with the aim of restoring stable near-normal glycemia in insulin-deficient patients with diabetes. 66 To achieve optimal replacement therapy, the closed-loop system, as shown in Figure 1, should contain three crucial elements: a sensing arm (performs continuous or repeated measuring of glucose; continuous glucose monitor); a computer (controls insulin delivery timing; control algorithm); and an insulin delivery arm (to inject insulin on demand; insulin infusion pump). 2,15,65,66,68 –70 The glucose sensor is used to generate an almost continuous signal corresponding to the blood glucose level. This signal serves as input for the algorithm of the control system that computes the amount of insulin to be delivered in order to maintain glucose levels within a narrow, normoglycemic range. 66 Historically, this combination was made available by the development of the bedside external artificial pancreas in the 1970s: the Biostator® (Miles Laboratories, Elkhart, IN). 71 In this model, an intravenous (IV) infusion of insulin from a peristaltic pump is modulated by a continuous blood glucose assessment using glucose oxidase, with algorithms that define insulin delivery according to glucose variations. 69 However, the whole system is cumbersome and requires almost constant human assistance. Thus, although it is still used for physiological investigations, this system cannot serve as a replacement for insulin secretion in the everyday life of a person with diabetes. 69
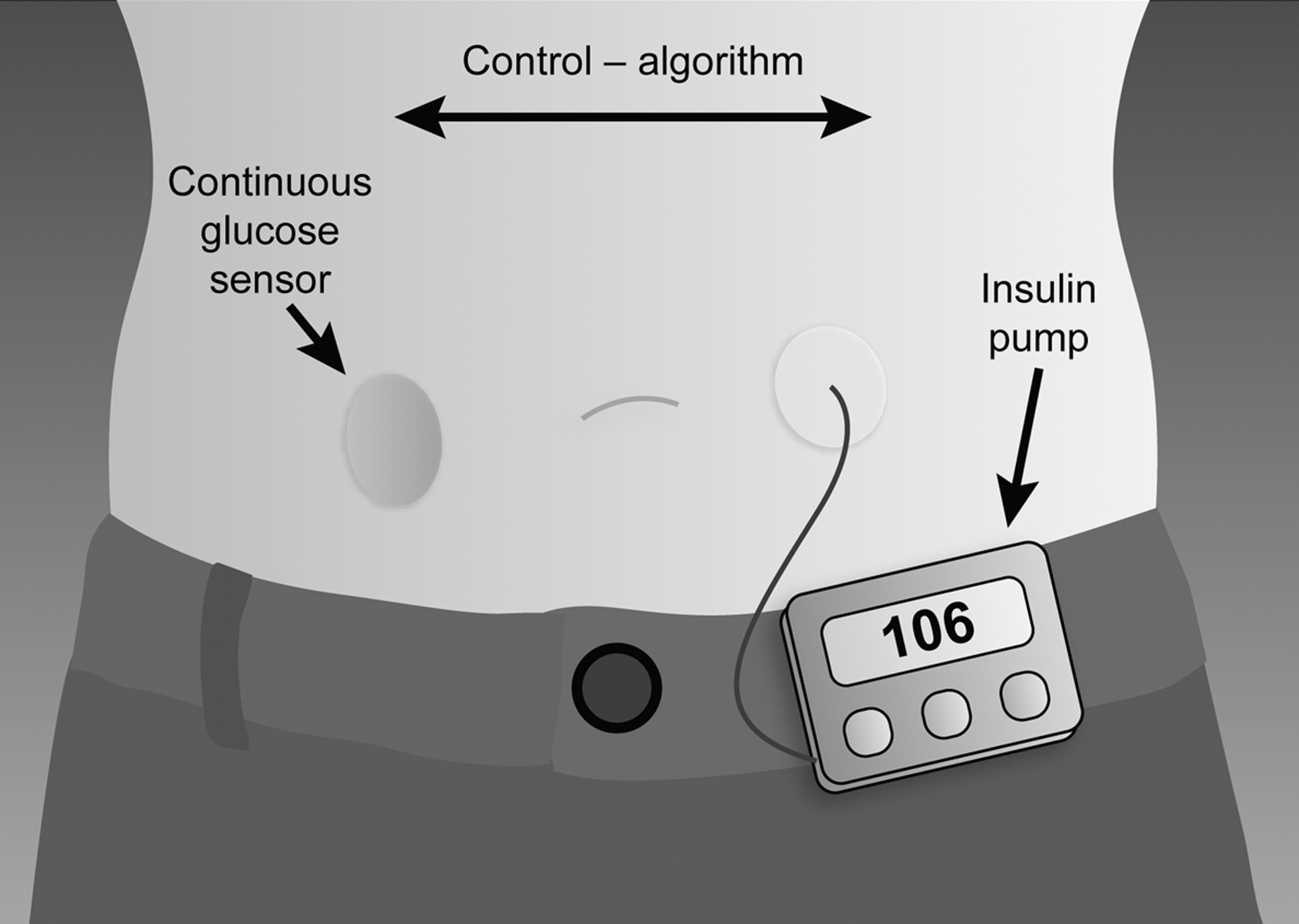
Components of the closed-loop insulin delivery system. 67
Over the past few years, more advanced insulin delivery systems that better mimic physiology, with more accurate glucose sensing devices, have been developed that have affirmed the feasibility of closed-loop insulin delivery. 48 The optimal process should operate continuously without errors so as to avoid mistakes leading to severe hypo- or hyperglycemia. This autonomous function would resemble glucose-responsive insulin secretion from the pancreas via a fully closed-loop approach, 72 monitoring glucose levels and supplying insulin accordingly. 73 Thus, by definition, the artificial pancreas will, theoretically, free patients with diabetes from the responsibility of self-management of their condition.
Similar to CSII, patients likely to derive the greatest benefit from the closed-loop system are those who are highly motivated and capable of managing the technology. In particular, children and adolescents 51,74 and other age groups in which poor adherence may be a determining factor represent ideal target groups for this system, as well as patient groups, such as pregnant women, in whom rapid achievement of tight glucose control in a safe manner is essential. 2,75 The closed-loop system may be particularly useful for children and adolescents for whom other potentially “curative” therapeutic options, such as islet cell transplantation, are not suitable. 2
The development of closed-loop prototypes has, however, been hindered because of suboptimal accuracy and reliability of monitoring devices, slow absorption of subcutaneously administered rapid-acting insulin analogs, and insufficient control algorithms. 76 Because of these limitations—although closed-loop systems could, theoretically, transform management of insulin-dependent diabetes—their implementation is likely to be gradual, starting with straightforward applications such as shutting off the pump at low glucose concentrations 77 or overnight closed-loop delivery, and proceeding to more complex applications providing 24-h control. 76 Overnight closed-loop delivery is particularly sought after because it addresses the problem of nocturnal hypoglycemia. A recent study by Hovorka et al. 76 demonstrated that closed-loop insulin delivery enables safe and tight overnight glucose control in children and adolescents with diabetes. In their closed-loop system, continuous information provided by a subcutaneously inserted needle-type glucose sensor was used to modulate SC insulin delivery from a portable pump according to a model predictive controller. The results showed that although mean blood glucose concentrations were comparable to those achieved without the controller, controller use was associated with several benefits, including reduction of blood glucose excursions outside the near-normal range and, importantly, reduced time spent in nocturnal hypoglycemia. 76 By demonstrating nocturnal glycemic control, this study is the logical first step toward progressive implementation of automated insulin delivery at home. The next challenge is to address glycemic control at mealtimes and during various activities as part of an individual's daily life.
Advancements in glucose sensing technologies could further improve performance of closed-loop systems. Fully automated closed-loop delivery will need wireless data transmission to replace manual control of the pump by nurses. 76 Ultimately, the aim is to develop a wearable artificial pancreas, so that the patient is not confined to bedside systems. A newly designed wearable closed-loop system is shown in Figure 2.
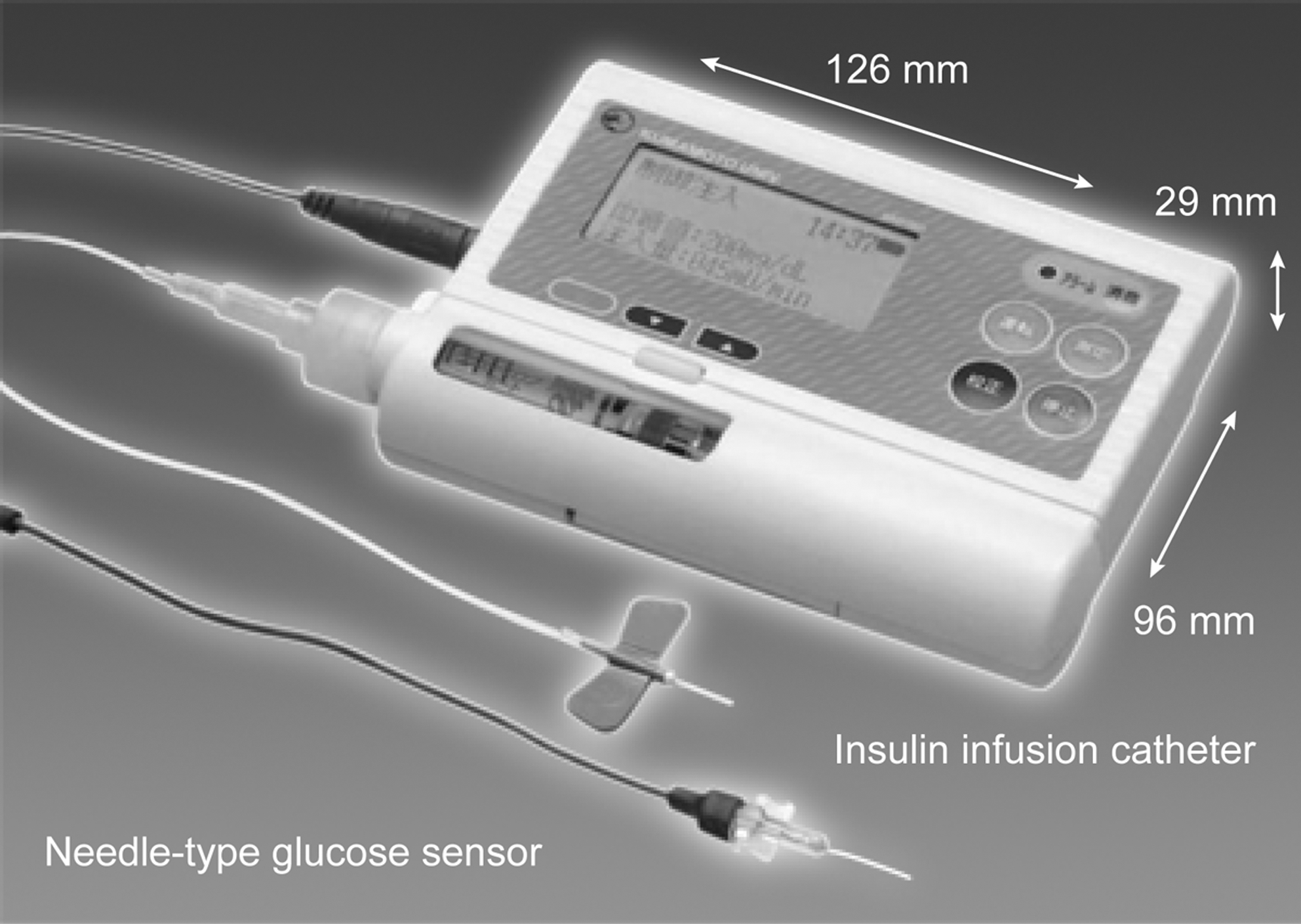
Newly designed wearable artificial endocrine pancreas. 61 Reproduced under the Creative Commons Attribution Non-commercial License, © 1995–2011 Baishideng Publishing Group Co., Ltd.
Inside the closed-loop system
Closed-loop systems are categorized according to meal-related insulin delivery and body interface types. Depending on the SC or IV body interface, three major types of closed-loop systems are in development: SC sensing and SC delivery system; IV sensing and intraperitoneal delivery system; and IV glucose sensing and IV insulin delivery system. Algorithms follow two main models: one option aims to reproduce the physiological characteristics of insulin secretion, including proportional, integral and derivative components, whereas the other option is a “predictive-control” model based on an observed relationship between blood glucose and plasma insulin variations. 69
Differences between systems mainly lie in the control algorithms used and, to some extent, the method and site of glucose measurement and insulin delivery. 65 The first two systems are the most promising and are described in further detail here. The first is an extracorporeal SC–SC approach that utilizes SC glucose monitoring combined with subcutaneously delivered insulin. 78 Although the minimally invasive nature of this system offers a strong advantage, a significant drawback lies in the delayed time between glucose measurement and insulin delivery, which may be up to 40 min. 15 In the implantable IV–intraperitoneal system, 69 glucose monitoring occurs intravenously, and insulin delivery is performed intraperitoneally, which represents a more physiological delivery and absorption. Although the lags in the system are shorter than with the SC–SC method, physiological delays still occur. 15,69 Of the closed-loop systems in development, it is likely that the SC–SC system will be introduced first because of its practical advantages. Indeed, a preliminary study supported the feasibility of this type of system for improving glycemic control. 17 However, the SC–SC system is currently associated with several methodological limitations, including those related to delivery, sensor, and infusion site problems. 79 Intraperitoneal insulin delivery, although still marginally used in diabetes care, may offer an alternative because it more closely mimics physiological plasma insulin profiles and provides sustained stability and reliability of insulin delivery. 66 Compared with the SC–SC system, both the IV-sensing/intraperitoneal-delivery systems and the IV-glucose sensing/IV-insulin delivery systems will allow more rapid insulin delivery, but have the disadvantage that they require surgery. 66
Expert opinion predicts a gradual introduction of closed-loop systems, with initial emphasis on hypoglycemia prevention or nocturnal glucose control, as supported by a recent analysis, 80 followed by a system that moves further toward removing the element of human error. 76,79
Continuous Innovation in Diabetes Management
Ongoing development continues to improve insulin replacement therapy, such as the introduction of devices with incorporated insulin-dose calculators. One such concept includes the OmniPod® system (Insulet, Bedford, MA), which features the Pod, a lightweight, watertight, self-enclosed insulin pump with automated cannula insertion. 81 The Pod delivers insulin according to preprogrammed instructions transmitted wirelessly from the personal diabetes manager, a hand-held device that programs the pump with customized insulin instructions, monitors the operation of the pump, performs suggested bolus calculations, and contains an integrated blood glucose meter. “Patch pumps” such as the OmniPod system offer greater convenience and comfort for patients while lowering HbA1c levels. 66,81 Other patch pumps in development include V-Go™ (Valeritas, Parsippany, NJ), Picosulin (Picosulin, Geneva, Switzerland), Cellnovo (Cellnovo, Swansea, Wales), and Solo™ (Medingo, Yoqneam Illit, Israel), which was approved by the Food and Drug Administration in 2009.
Other developments have been aimed at improving monitoring and communication. The Diabeo system comprises software, uploaded onto smartphones with an internet connection that provides the following to the patient: (1) bolus calculators using validated algorithms, which account for carbohydrate intake, premeal blood glucose, and anticipated physical activity, as reported by the patient; (2) plasma glucose targets; (3) automatic algorithms for the adjustment of carbohydrate ratio and basal insulin or pump basal rates when the postprandial or fasting plasma glucose levels are off target; and (4) data transmission to medical staff computers, through radio services and secured web sites, to allow easy telemonitoring and teleconsultations. 82 Several studies have now demonstrated the feasibility, safety, and accuracy of Diabeo in improving glycemic control in people with T1DM. 82,83 Diabeo significantly improved HbA1c control (−0.9%) using a telemedicine system in poorly controlled T1DM, without increasing medical time or expenses, and without an increase in hypoglycemic episodes. 82
Another open-label randomized study assessed the impact of the Accu-Chek® (Roche) insulin dosing guidance software (Advisor) on glucose control and hypoglycemia in patients with T1DM. 84 The Advisor software is similar to bolus calculators currently used on many insulin pumps. Garg et al. 84 were the first to report improved glucose control over 1 year with no weight gain or change in insulin dose with use of insulin guidance software. However, it should be noted that, contrary to the above study with Diabeo, 82 the use of Advisor was associated with an increased rate of severe hypoglycemia. 84
With regard to one of the most readily accessible technologies in diabetes management, the mobile phone, a recent study has shown mobile communication technologies to have promise within a web-based collaborative care program for T2DM, 85 while a 3-month pilot study in adolescents with T1DM demonstrated that a novel mobile phone glucose monitoring system had significant potential for improving glucose management. 86 Future developments in this arena should focus on combining simple applications with familiar mobile phone technologies, while still permitting sufficient interaction with care providers. 85
Conclusions
As advancing technologies continue to improve the treatment of diabetes, the ultimate goal is to allow more patients to reach glycemic targets without increasing, or even potentially decreasing, the risk of hypoglycemia. However, despite technological progress in glucose monitoring and insulin delivery, successful glycemic control is hampered by adverse effects that are the likely consequence of a failure to achieve responses that reflect the physiological nature of insulin replacement. Against this background, the closed-loop system or artificial pancreas arguably offers the most revolutionary intervention for diabetes management next to achieving an actual cure. Such a device would liberate patients from the need for relentless, several-times-daily, monitoring of blood glucose levels and injections of insulin. The ultimate goal is a fully automated artificial pancreas, and, with each technological advance, we are one step closer to achieving this goal and freeing our patients from the burden of their diabetes care.
Footnotes
Acknowledgments
The authors would like to acknowledge Huw Jones, Ph.D., of Medicus International for his editorial support. The funding of editorial assistance was provided by sanofi-aventis.
Author Disclosure Statement
Prof. Penfornis is a member of the boards of AstraZeneca, Bristol-Myers Squibb, Lilly France, Novartis, Novo Nordisk, and Sanofi Aventis. He has received payment for the development of educational presentations including speakers' office services from Abbott, Eli Lilly, Medtronic, Merck-Serono, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, Sanofi Aventis, and Takeda. He had travel and accommodation expenses covered or reimbursed by Abbott, AstraZeneca, Boehringer-Ingelheim Pharmaceutical, Eli Lilly, GlaxoSmith-Kline, Medtronic, Merck-Serono, Merck Sharp & Dohme, Novartis, Novo Nordisk, Sanofi-Aventis, and Servier.
