Abstract
In physiology, insulin is released continuously by the pancreas at a nearly constant rate between meals and in the fasting state (basal insulin secretion). The pivotal role of basal insulin is to restrain release of glucose from the liver and free fatty acids from adipose tissue, thus preventing hyperglycemia and ketosis. In type 1 diabetes mellitus (T1DM) (absolute insulin deficiency), the replacement of basal insulin is challenging because the currently available pharmacological preparations of long-acting insulin do not exactly reproduce the fine physiology of flat action profile of basal insulin of subjects without diabetes. NPH and NPH-based insulin mixtures no longer have a place in the treatment of T1DM because of their early peak effects and relatively short duration of action, which result into risk of nocturnal hypoglycemia and fasting hyperglycemia, respectively, after the evening injection. Only continuous subcutaneous (s.c.) insulin infusion (CSII) or long-acting analogs such as glargine (>24 h in duration, once a day) and detemir (<24 h in duration, once or more often twice a day) should be used as basal insulin in T1DM in combination with mealtime rapid-acting analogs. CSII and the long-acting analogs are nearly peakless and therefore reduce the risk for hypoglycemia (especially at night), blood glucose (BG) variability, and lower A1C with similar or less hypoglycemia. CSII is the “gold standard” of replacement of basal insulin because of better reproducibility of subcutaneous absorption of soluble insulin. Although CSII is not superior to multiple daily insulin injections in the general T1DM population, CSII might be indicated in subsets of T1DM (long-term T1DM with insulin “supersensitivity” and needs for low-dose insulin, some individuals with variable subcutaneous absorption of long-acting analogs) to minimize BG variability, reduce hypoglycemia, and benefit A1C.
Introduction
Physiology of Glucose Homeostasis: The Pivotal Role of Basal Insulin
The hormone insulin is secreted by the beta cells of the islets of Langerhans in the pancreas and directly enters the portal bloodstream through the pancreatic vein. Insulin is secreted over the daily 24 h in the fasting state and after meals in response to rise in blood glucose (BG) as well as to ingestion of nutrients (incretin effect). 1 The close anatomy of the pancreas to the liver explains the primary physiological effect of insulin (i.e., restraining release of glucose from hepatocytes) (endogenous glucose output [EGO]) (Fig. 1). 2 The co-primary effect of insulin is suppression of lipolysis in adipose tissue, which is as insulin sensitive, or even more sensitive, than liver (Fig. 2). 3 In turn, lower free fatty acid levels contribute to restraining EGO. Finally, the secondary effect of insulin is targeting the muscle to promote entry of glucose as well as its intracellular metabolism, but this occurs intermittently at the elevated plasma concentrations of the postprandial state when meals are ingested. 2 Overall, the complex 24-h insulin secretion is finely tuned to prevent excessive increase in BG after meals (<140 mg/dL [8.0 mmol/L]) and to prevent hypoglycemia (plasma glucose <70 mg/dL [4.0 mmol/L]) between meals and during fasting (night) (Fig. 3). 4
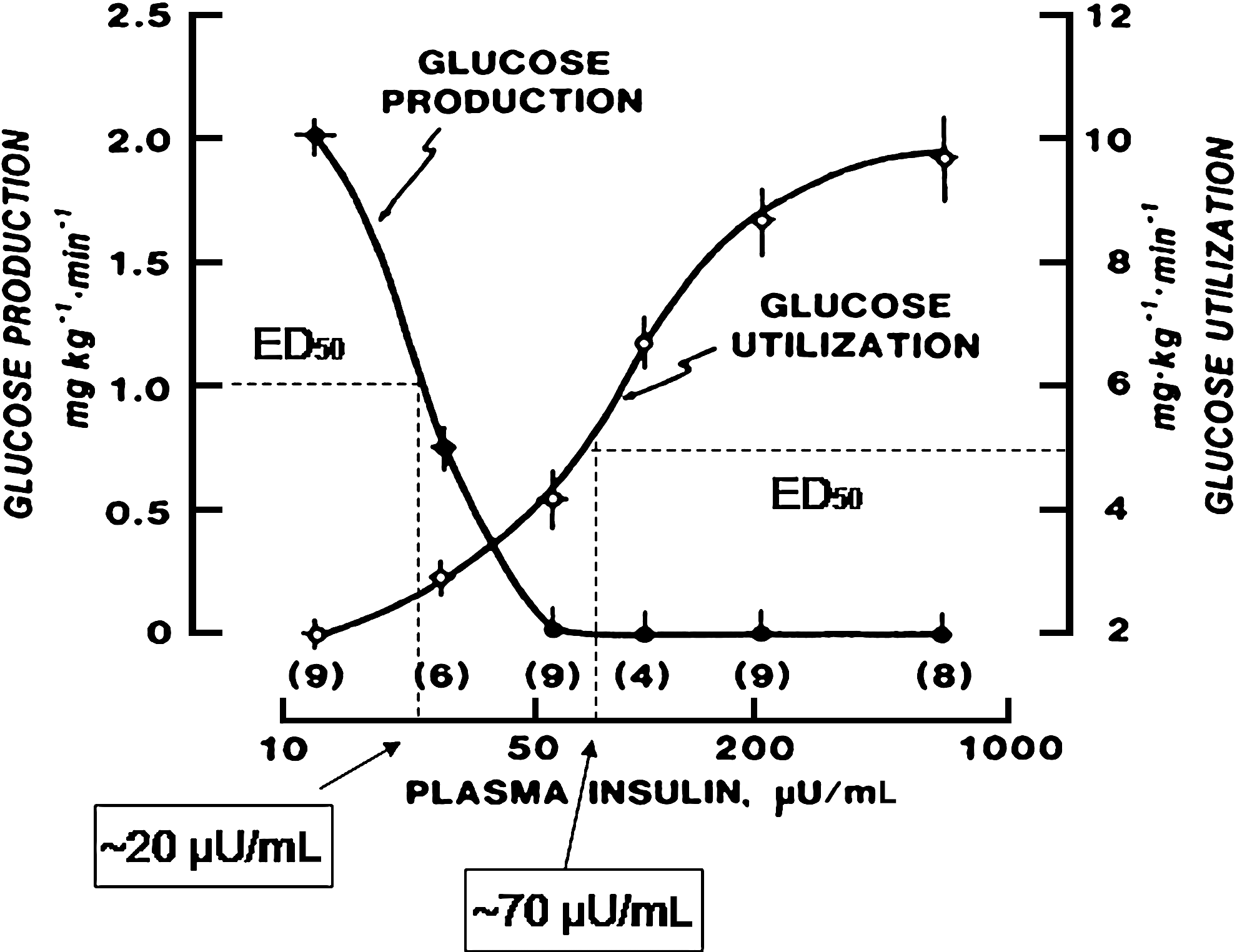
Dose–response curves showing (peripheral) plasma insulin concentration and insulin effect on (
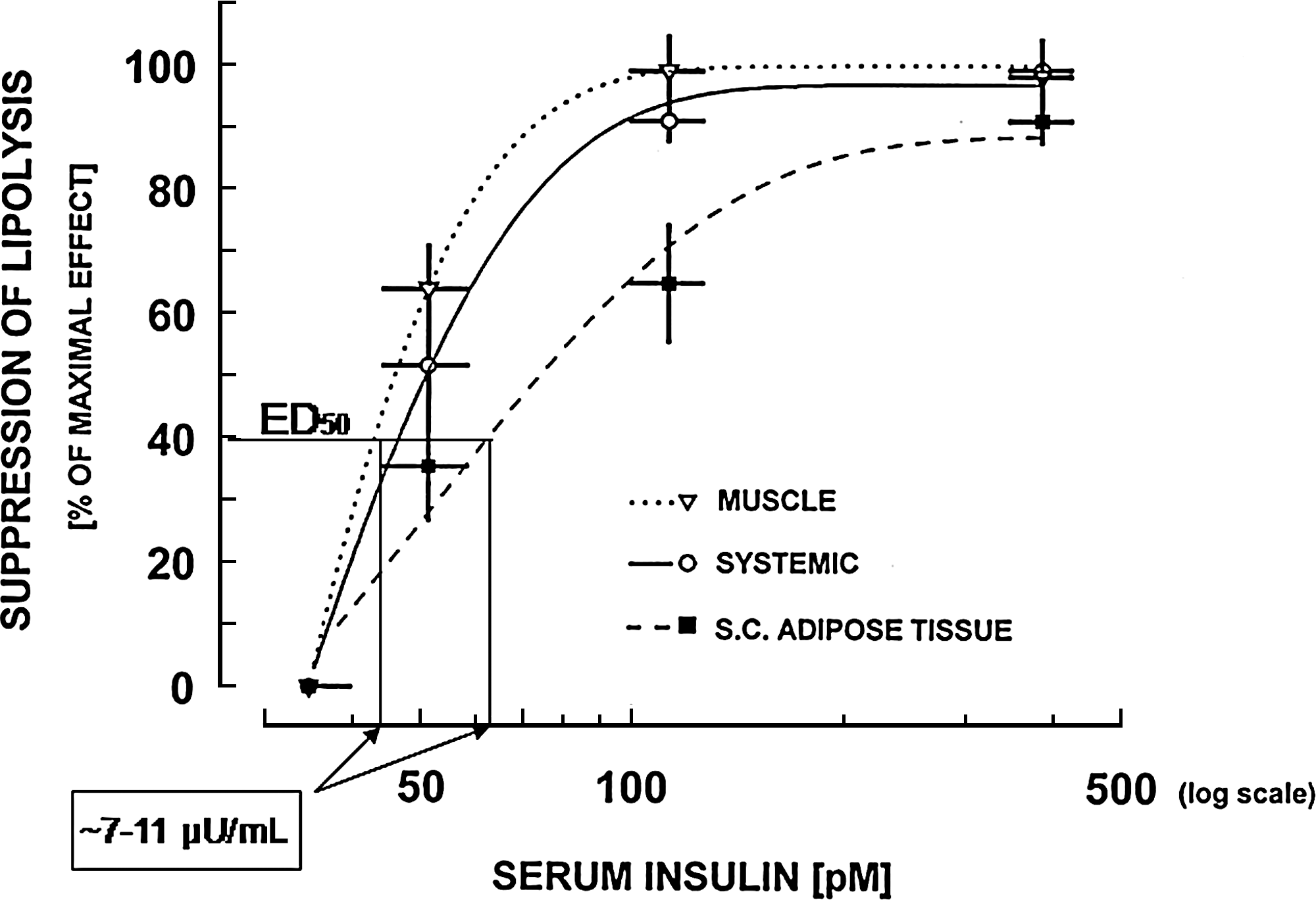
Dose–response curves showing plasma insulin concentration and insulin effect on suppression of lipolysis. The insulin sensitivity of adipose tissue is greater than that of liver and muscle as indicated by the lower estimated concentration of insulin at which the effect is 50% of the maximal (ED50) for suppression of lipolysis (∼7–11 μU/mL) compared with that of suppression of endogenous glucose output and muscle glucose utilization in Figure 1. S.C., subcutaneous. Reproduced with permission from Stumvoll et al. 3
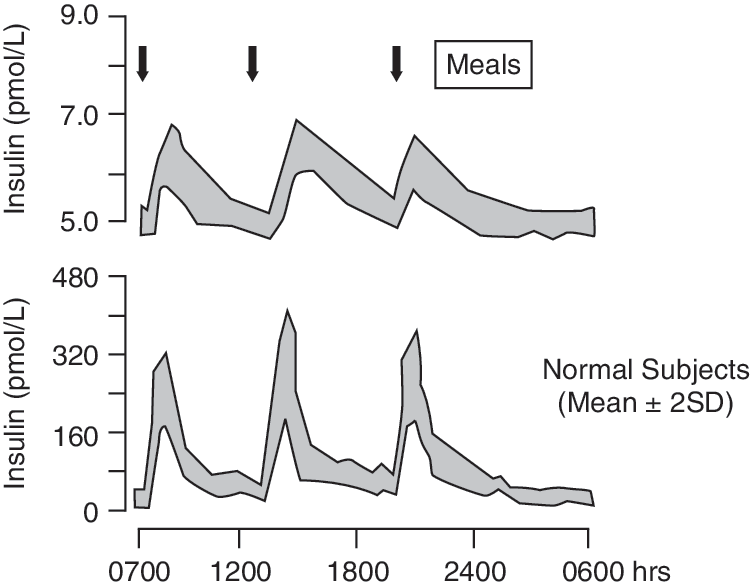
24-h plasma glucose and insulin profiles in individuals without diabetes. Reprinted from Rossetti et al., 33 with permission from Informa Healthcare.
Endogenous insulin secretion can be divided essentially into two phases: the “basal” (or interprandial) component, when insulin is secreted continuously, and the stimulated or “bolus” component, when insulin is released in response to a nutrient or other challenge. In healthy, nonobese adults, the overall “basal” insulin is usually secreted at a rate of 0.5–1 U/h, 5 resulting in plasma insulin concentrations of 5–15 μU/mL (30–90 pmol/L), 6 which maintains the fasting plasma glucose concentrations in the range of 70–110 mg/dL (4.0–5.6 mmol/L) 5 (Fig. 3). In response to a meal, insulin secretion increases rapidly with plasma concentrations reaching a peak of 60–80 μU/mL (360–480 pmol/L) within 30–60 min and returning to basal levels approximately 2–4 h later. 4 In obese subjects without diabetes these figures are severalfold higher, related to the degree of obesity. Plasma glucose concentrations also vary in response to different meals; however, the postprandial BG level rarely exceeds 140 mg/dL (7.7 mmol/L). 7
The primary role of basal insulin secretion is to limit lipolysis and EGO in the fasting state, especially overnight, while ensuring sufficient glucose for cerebral function. Deficiency of basal insulin results in excess EGO (hence hyperglycemia) as well as excess lipolysis (increase in plasma free fatty acids, hepatic ketogenesis, and risk for ketoacidosis). In contrast, excess basal insulin results into inappropriate suppression of EGO (hypoglycemia). The latter condition occurs in insulinoma patients 8 and those insulin-treated subjects with diabetes mellitus given inappropriate insulin doses. 9 Physiology teaches and predicts how challenging it is to appropriately replace basal insulin in humans with diabetes mellitus. Because of the steep slope of the dose–response curve (Fig. 1), even a modest over- or under-replacement of (basal) insulin results in excess suppression or exaggerated stimulation of EGO with consequent hypo- or hyperglycemia. The presently still peripheral rather than portal administration of insulin (with consequent peripheral hyperinsulinemia and portal hypoinsulinemia) and the variability of its s.c. absorption are additional factors that make the replacement of basal insulin difficult in subjects with T1DM. Because endogenous insulin secretion “buffers” the effect of injected preparations of basal insulin, in type 2 diabetes mellitus (T2DM) the greater the residual insulin secretion, the less difficult and less challenging is replacement of basal insulin.
Supplementation of Basal Insulin Needs with Exogenous Insulin
The ideal approach to replace basal insulin in insulin-deficient subjects is continuous infusion (ideally through portal circulation) at a rate mimicking the endogenous insulin production by pancreas of individuals without diabetes. Because at present neither the portal nor the intravenous (i.v.) routes for insulin infusion are feasible, insulin is still administered in the peripheral s.c. tissues as it has always been ever since insulin has become available for treatment of diabetes in 1922.
Continuous infusion of insulin into the s.c. tissue (CSII) is the “gold standard” of basal insulin replacement in subjects with T1DM. The greater the deficiency of basal insulin in subjects with diabetes mellitus, the higher the value of such an approach. Thus, CSII is potentially indicated in all subjects with T1DM (who totally lack endogenous insulin), but only in the subjects with long-term T2DM approaching the end-stage phase of the disease when insulin secretion is eventually lost completely. Among the several reasons for superiority of CSII over s.c. administration (injection) of long-acting insulin preparations, the most important is the unique ability of such an approach to deliver in a continuous mode (like pancreas) amounts of insulin that may be quite precisely selected to prevent hyperglycemia and hypoglycemia. The rate of basal insulin by CSII can be regulated to deliver variable rates of insulin infusion during the 24-h period according to the subject's needs. Such an approach is successful because CSII infuses reliably the programmed rate of insulin, even at low rates of 0.1–0.05 U/h. This ensures the greatest flexibility of treatment for diabetes subjects with lowest risk for hyper- and/or hypoglycemia. This is also due to fact that CSII uses soluble insulin with better reproducibility of s.c. absorption, in contrast to administration of long-acting insulin preparations like NPH or lente. The latter are insoluble (suspensions) and therefore present large day-to-day variability of absorption 10 with important fluctuations of BG.
The Era of NPH and NPH-Based Insulin Mixtures
As mentioned by David Owens in his review earlier in this issue, 11 until the year 2000, the only insulin preparations with retarded activity were NPH and lente. The most popular NPH has been used for decades in subjects with T1DM or T2DM. However, as predicted by physiology, substitution of basal insulin is more challenging primarily in totally insulin-deficient subjects (Fig. 1). In T1DM, use of NPH has been responsible for most of the problems of glycemic control. NPH has caused either frequent and severe nocturnal hypoglycemia episodes, contributing to the syndrome of hypoglycemia unawareness, 12 and/or has resulted in overall hyperglycemia predisposing to the risk of development and progression of long-term vascular complications. These concepts are best illustrated in the Diabetes Control and Complication Trial 13 and Epidemiology of Diabetes Interventions and Complications studies, 14 where NPH was used as basal insulin generally as once a day administration. When NPH was used to intensify treatment, lowering A1C to ∼7.0%, the result was an elevated risk for severe hypoglycemia (up to 0.62 episodes/patient-year, half of which were nocturnal episodes). In contrast, when NPH was titrated to prevent hypoglycemia, A1C increased to ∼9.0%, and the poor glycemic control was followed over a few years by micro- (Diabetes Control and Complication Trial) 13 and later by macrovascular complications (Epidemiology of Diabetes Interventions and Complications). 14 These observations suggest that NPH is not a good candidate for replacement of basal insulin in T1DM aiming at prevention of hyper- and hypoglycemia. At the least, these studies 13,14 indicate that NPH should be used with a strategy different from that based on the once a day administration of the Diabetes Control and Complications Trial, as suggested by more recent studies where the NPH was administered three or four times daily, reconstituting a rather flat and nearly peakless basal insulin background. 15,16
How to Optimize the Use of NPH in T1DM
The well-recognized above limitations of NPH as basal insulin in T1DM along with advances in physiological insight into the mechanisms of glucose homeostasis 17 led investigators and clinicians at the end of the last century to propose new strategies to replace basal insulin. Yet, because at that time NPH was still the only available basal insulin, efforts were concentrated on optimization of its use.
Clinical experience as well as controlled studies indicated the important limitations of morning injection of NPH because of the high risk for hypoglycemia before lunch 18,19 (Fig. 4). At the same time, the high risk of nocturnal hypoglycemia when NPH was injected at dinnertime was established. 20 These studies were the basis for developing new strategies of use of NPH. 15,16
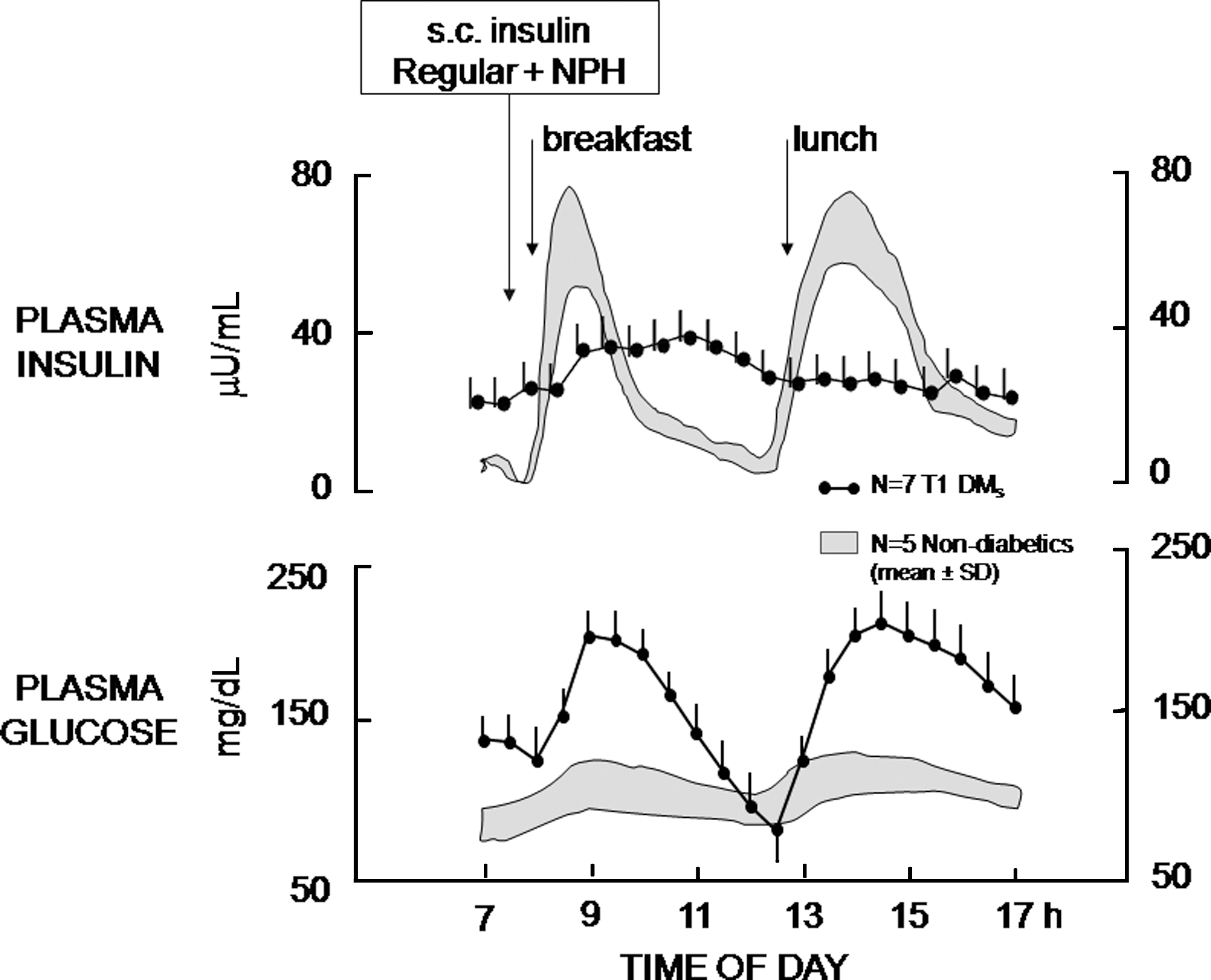
Administration of a mixture of regular + NPH insulin in the morning before breakfast in subjects with type 1 diabetes mellitus (T1 DM) induces hypoglycemia at noon because plasma insulin remains inappropriately elevated compared with normal subjects without diabetes. s.c., subcutaneous. Reproduced with permission from Dimitriadis and Gerich. 18
The first step to optimize the use of NPH was to postpone the evening injection from dinner to bedtime (“the later the better”) to minimize risk for nocturnal hypoglycemia and target near-normoglycemia in the fasting state. 20,21
The second step was to add a second daily NPH injection (in the majority of, although not all, subjects with T1DM) to ensure 24-h coverage. This second NPH was administered ∼12 h after the bedtime injection (i.e., at lunchtime, with a dose of ∼30–40% that of bedtime). The twice-daily NPH regimen was popular and successful as basal insulin replacement in the era of human regular insulin (HRI) as mealtime insulin preparation. 16
The third step was introduced more recently, when rapid-acting insulin analogs became popular in place of HRI as mealtime treatment, 17 and consisted of additional intensification of NPH administration up to three or four daily injections. 15 –17 Because the duration of action of rapid-acting insulin analogs is shorter than that of HRI, 17 mealtime administration of the former in place of the latter unmasks the deficiency of basal insulin prior to the next meal when NPH is given once a day. 22 This observation led to the understanding that until 1996, when HRI was “the” mealtime treatment for T1DM, the long “tail” of action contributed to replace interprandial (basal), in addition to prandial, insulin. The “basal” effect of HRI was no longer seen when rapid-acting analogs were introduced for mealtime treatment. 15 As predicted, rapid-acting insulin analogs at mealtime resulted in greater hyperglycemia prior to the next meal and at night, 15,22 thus offsetting the potential of lowering the percentage of A1C as a consequence of their ability to lower early postprandial BG. 16 Therefore, the advantages of rapid-acting insulin analogs over HRI as mealtime treatment (lower postprandial BG, less risk for late postprandial hypoglycemia, convenience as administration immediately at mealtime or during/after the meal) 17 called for optimization of replacement of basal insulin. This was easily achieved with CSII. 23 With multiple daily insulin injections (MDII) in the NPH era, the idea was to add a few units of NPH to each mealtime administration of rapid-acting analog to build up a nearly flat and long-lasting basal insulin replacement to better mimic basal insulin in CSII (Fig. 5). This regimen of combination of rapid-acting analogs at mealtime and optimized three or four times a day NPH successfully demonstrated the superiority (lower A1C and less risk for hypoglycemia, hypoglycemia unawareness, and impaired counterregulation) compared with HRI at mealtime and NPH. 15,16 This was similar to CSII using rapid-acting insulin analog compared with HRI. 23
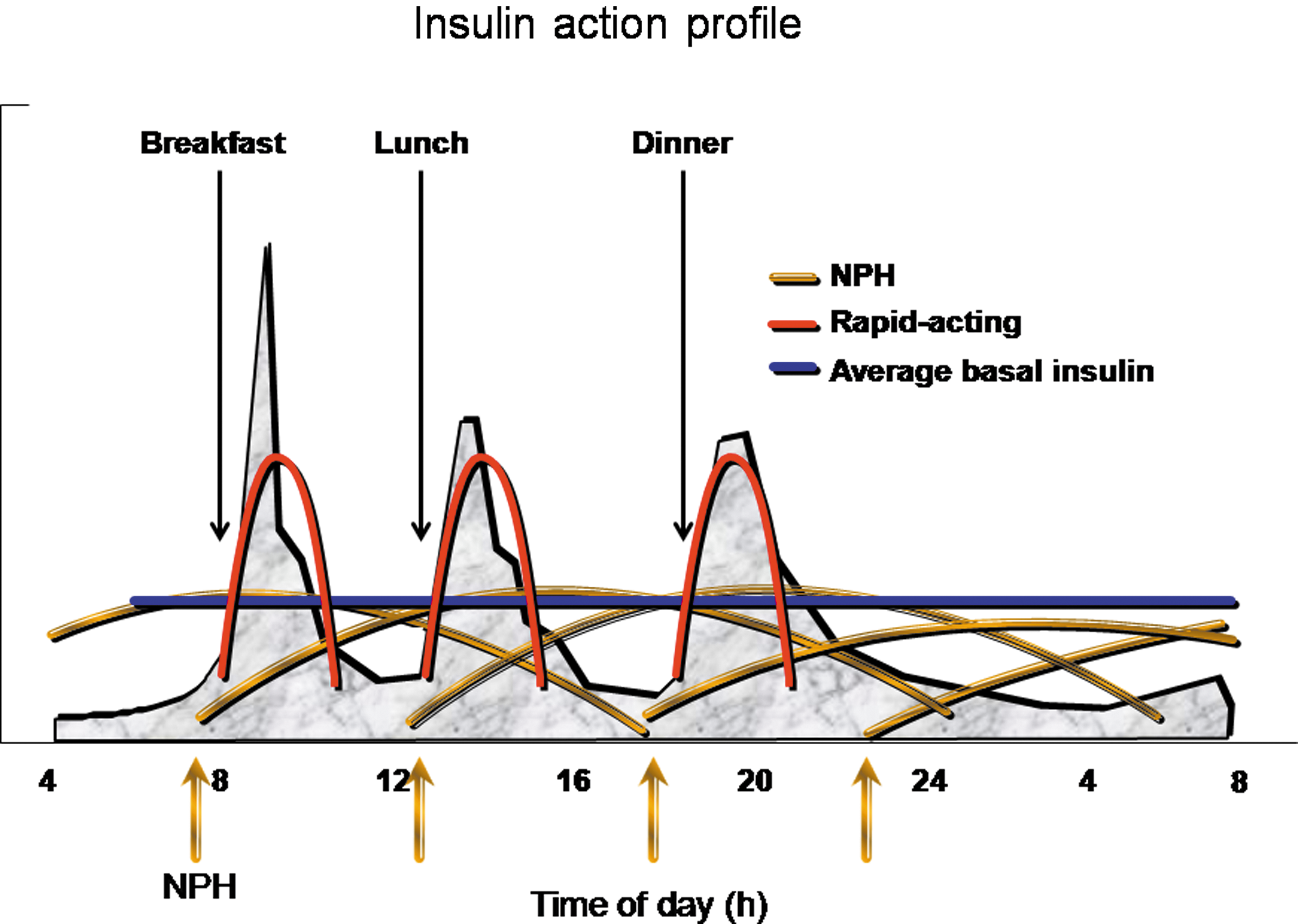
Strategy of intensive insulin treatment of type 1 diabetes mellitus with mealtime rapid-acting insulin analog added with NPH at each meal, in addition to bedtime NPH administration. The multiple daily NPH insulin administrations build up a relatively flat basal insulin profile over the 24-h period. This optimized NPH strategy of basal insulin supplementation has been used in previous studies. 15,16
A few years elapsed between the conception of this unconventional and to some extent complex, but efficacious and safe, NPH regimen and the availability of the more convenient once-a-day long-acting insulin analog glargine. 24 Clearly, the latter gained immediate popularity over the multiple daily NPH administrations as the basal regimen to be associated with rapid-acting analogs at mealtime. 17
At present, NPH is no longer used in T1DM because of the superiority of long-acting analogs glargine and detemir. 17 However, still in some countries worldwide, long-acting analogs are not accessible to a large number of subjects with T1DM because they are more expensive than NPH. It is hoped that insulin analogs, both rapid- and long-acting, become in the near future easily available to the universal community of subjects with T1DM all over the world with no discrimination between diabetes subjects living in rich compared with poor countries. However, it is important for those subjects with T1DM who are still restricted nowadays to the use of NPH to take advantage of the above-described strategy of optimization of NPH in combination with either HRI or rapid-acting insulin analogs.
Year 2000 and the Post-NPH Era: The Long-Acting Insulin Analogs Glargine and Detemir
The limitations of NPH as basal insulin have led toward the end of the last century to intense research for new long-acting insulin preparations that possibly would better mimic the basal insulin of physiology. Earlier in this issue, Porcellati et al. 25 have reviewed the superior pharmacokinetics (PK) and pharmacodynamics (PD) of glargine and detemir compared with NPH, as well as the differences between these two long-acting insulin analogs.
Studies on glargine and detemir insulins are good examples of how careful experimental evaluation of PK and PD in clamp studies appropriately designed to mimic real life of subjects with T1DM, 26 –28 and most recently T2DM, 29 has predicted the later clinical results after long-term treatment. In clinical studies, glargine has proven less BG variability, lower fasting BG, and lower frequency of nocturnal hypoglycemia compared with NPH. 30
Clinical experience with glargine has started in the summer of the year 2000. Initially, the relatively flat action profile of glargine was confirmed from the similar BG control with no increase in nocturnal hypoglycemia when glargine once a day was given at dinner compared with bedtime. 31 This study established the relative independence of the time of day of administration of glargine and is the basis for the present preference of its dinnertime compared with bedtime administration. This is different from NPH, which should be given at bedtime to minimize nocturnal hypoglycemia. 20 The superiority of glargine once a day over NPH in intensive treatment of T1DM has then been demonstrated in a long-term study where glargine has lowered A1C, reduced hypoglycemia (primarily nocturnal), and improved awareness of and counterregulation to hypoglycemia. 32 It is notable that in these studies 31,32 the comparator treatment (NPH) was the above-described optimized regimen of three or four daily administrations of NPH. This suggests that the benefits of glargine might be even greater in subjects with T1DM given NPH only one or two times a day.
Detemir, the second long-acting basal insulin to reach the market after glargine, is also superior to NPH in subjects with T1DM. Because its action profile is nearly flat compared with NPH (Fig. 6), 33 detemir reduces the risk for nocturnal hypoglycemia similar to glargine. 30 Because detemir insulin is reversibly bound to interstitial and circulating albumin, its effect is less variable than that of NPH. 30 However, detemir differs from glargine not only in terms of primary structure, mechanism of action, and metabolism, but also for PK and PD. 28 In contrast to glargine, detemir at therapeutic doses of 0.35 IU/kg has a duration of action of <24 h in the majority of subjects, and its effects on glucose and lipid metabolism are weaker than those of glargine. 28 This explains the observation in clinical studies that similarly successful long-term intensive treatment of T1DM is achieved with either basal analog, but detemir has to be administered at a higher dose than glargine and twice daily in the majority of subjects compared with once-daily glargine. 34
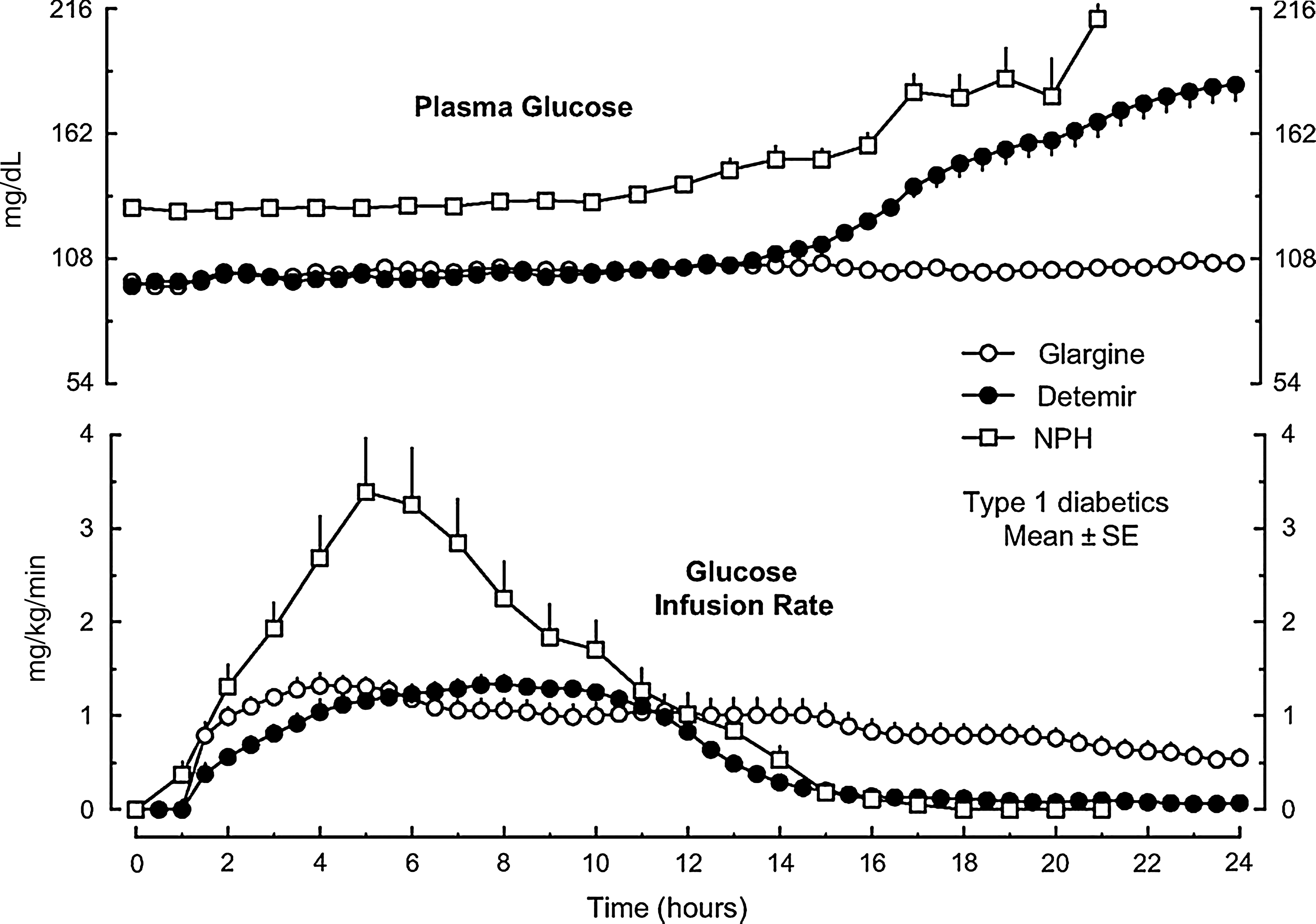
Pharmacodynamics of long-acting insulin analogs glargine and detemir compared with NPH injected in the evening in type 1 diabetes mellitus examined by the glucose infusion rate of the hyperglycemic clamp (NPH) or euglycemic clamp (glargine and detemir). NPH results in peak activity after midnight with a risk for hypoglycemia after midnight. In contrast, the activities of glargine and detemir are nearly flat overnight. Insulin activity of detemir wanes earlier than that of glargine. Reprinted with permission from Rossetti et al. 33
Neutral Protaminated Lispro Insulin
Recently, a retarded, protaminated preparation of the insulin analog lispro (neutral protaminated lispro [NPL]) has reached the market in some countries. At present, there is little information on NPL's PK/PD. Radziuk et al. 35 reported no differences compared with NPH, but the study was in normal subjects without diabetes, which does not predict PD in T1DM. A recent comparison between NPL and detemir in T1DM subjects 36 has reported a peak action profile for NPL similar to that of NPH. 26
Modern Insulin Regimens of Intensive Treatment of T1DM
Insulin can be delivered in T1DM by means of CSII or MDII (Fig. 7). CSII is the gold standard of basal insulin replacement because the insulin effect is more reproducible compared with that observed with MDII. This was easily evident in the NPH era, 37 as indicated by Hoogma et al. 38 In the post-NPH era (i.e., the glargine era), it has been difficult to prove superiority of CSII versus MDII. 39,40 In an unselected population of subjects with T1DM, either CSII or glargine-based MDII achieved similar results (mean BG, A1C, risk for hypoglycemia, BG variability). 39 This indicates that a basal insulin like glargine, better than NPH, makes MDII a more competitive treatment closely approaching the effects of CSII in the general T1DM population. However, such a study has not examined subgroups of T1DM with special needs (excessive variability in s.c. insulin absorption, low insulin requirements in long-term T1DM) in whom CSII might be preferable to glargine-based MDII.
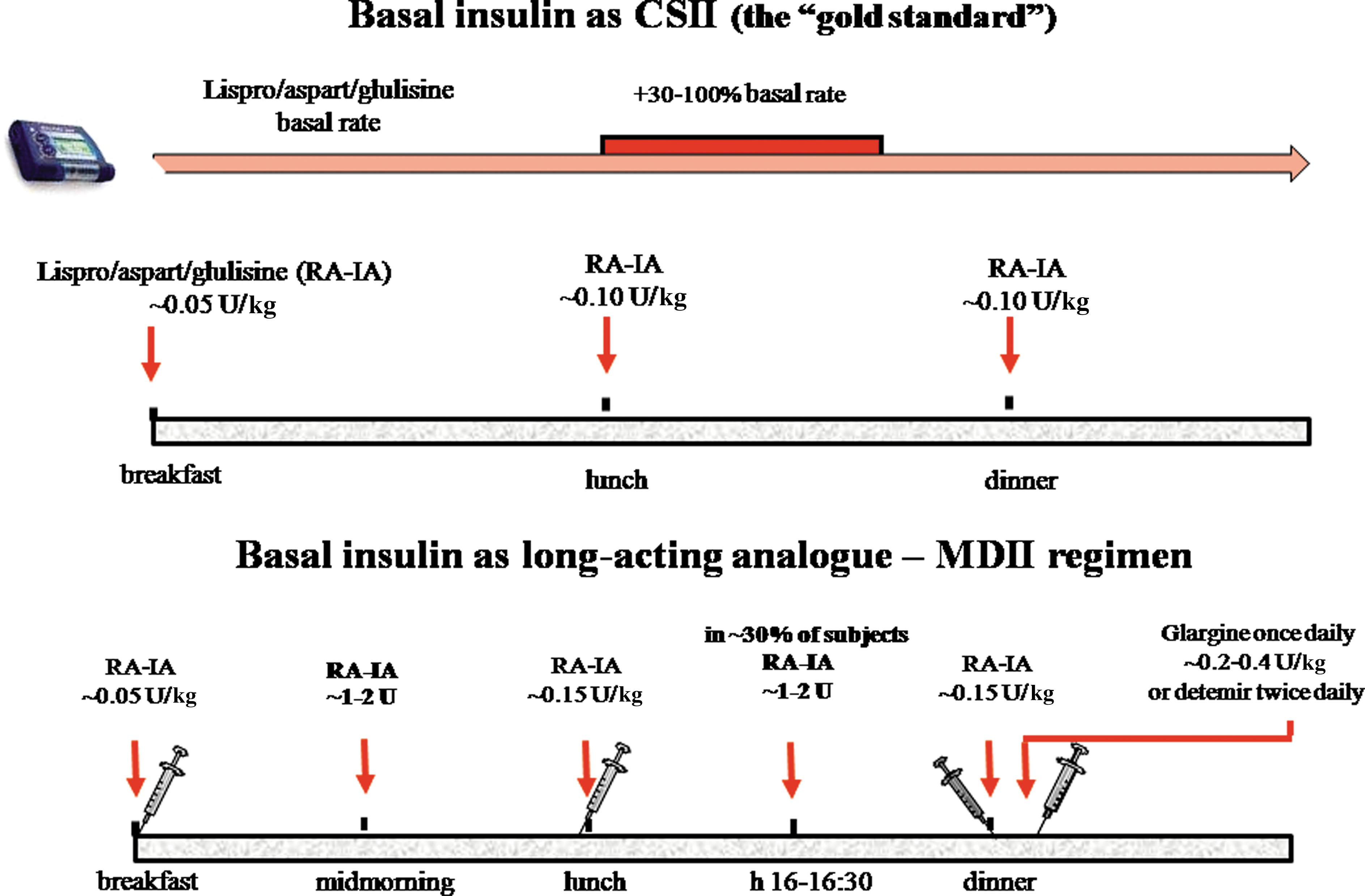
Modern strategies of replacement of insulin in type 1 diabetes mellitus. (
The CSII regimen is easily understood. The basal rate may be varied during the 24-h period, but experience indicates that two to four basal rates are usually sufficient in the large majority of subjects, primarily to prevent nocturnal hypoglycemia and late afternoon hyperglycemia. Too numerous changes in the everyday basal rate are not necessary.
With MDI, the same principles of CSII should be followed. Glargine (or detemir) should be titrated to target fasting euglycemia while avoiding nocturnal hypoglycemia. The afternoon hyperglycemia requires an additional bolus of rapid-acting insulin analog (1 or 2 U) 3–4 h after lunch to mimic the increase in basal rate of CSII (Fig. 8).
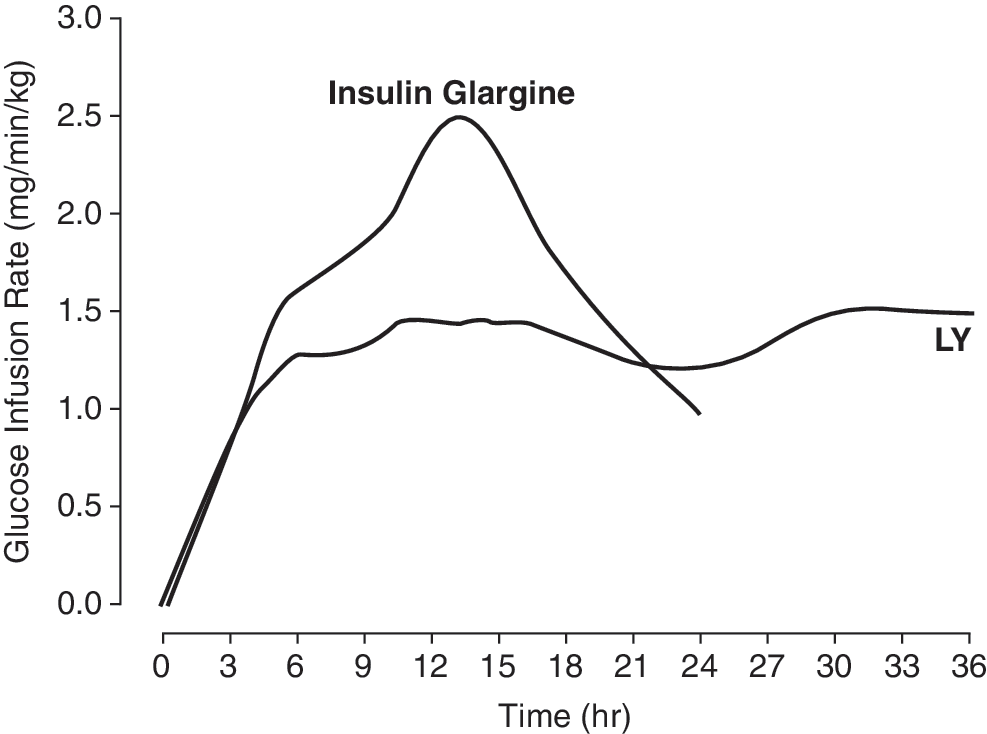
Pharmacodynamics of poly(ethylene glycol)ylated insulin lispro [poly(ethyleneglycol) 20 kDa-B28 (≥∼95%)/A1 (≤∼5%)–insulin lispro (LY) (0.225 mg/kg)] versus insulin glargine (0.5 U/kg) in normal subjects without diabetes. Reproduced from
To be successful, both CSII and MDII require education by doctors and patients to life-style, nutrition, and BG monitoring. Only under this circumstance do CSII and MDII result into optimal BG regulation.
In Vivo Metabolism of Glargine
Recently, important advances have been made in the understanding of metabolism of insulin glargine after s.c. injection in subjects with T1DM and T2DM. After s.c. injection, glargine undergoes a cleavage of the C-terminal amino acids of the B chain. 41 Initially, the two arginines at position 30 are removed, and A21-Gly-insulin (metabolite 1 [M1]) is formed. Then the threonine at position 30 is lost with formation of A21-Gly-des-30B-Thr (metabolite 2 [M2]). Initial studies in normal individuals without diabetes 41 and diabetes subjects 42 provided evidence for such an enzymatic biotransformation by carboxypeptidase(s) in vivo at the injection site as well as in serum. The interest for glargine metabolism derives from the observation that in vitro, at supraphysiological concentrations, glargine has greater affinity for the insulin-like growth factor-1 receptor (IGF-1R) and the ability to promote DNA synthesis (mitogenic activity) in some cell lines in vitro. 43,44 In contrast to glargine, M1 and M2 do not differ from human insulin in terms of IGF-1R binding and mitogenic characteristics while retaining full metabolic activity. 45
Glargine metabolism has recently been evaluated in subjects with T1DM 46 and T2DM 47 using a novel, specific, and sensitive method (liquid chromatography/mass spectrometry) that detects glargine, M1, and M2 without cross-reactivity with human insulin. Within 1–2 h from s.c. injection of glargine, intact glargine is detectable in serum in only a minority of subjects and only for a few hours. When glargine is detected, its concentration lies just above the detection limit (∼30 pmol/L). In contrast, M1 is detected in all subjects, and its level increases progressively in serum over time and correlates with insulin activity. M2 is not detected in circulation at any time. These data suggest glargine is a pro-drug, which exerts its metabolic effects primarily via the deaminated M1. Although additional studies on larger patient groups are needed, it appears that after glargine injection, there is virtually no glargine circulating in plasma, but only M1, which does not differ from human insulin in terms of IGF-1R binding and mitogenesis. These observations emphasize the limitations of studies in vitro in cell line models and reinforce the caution required in translating the result in vitro into the in vivo situation. These new data suggest that the hypothesis that treatment with insulin glargine might induce cancer or accelerate growth of preexisting malignant cells to clinically overt cancer in humans with diabetes mellitus 48 does not appear to have a rationale.
New Candidates for Basal Insulin in T1DM
At present, the majority of T1DM subjects may achieve and maintain long-term optimal BG control with the long-acting analogs glargine and detemir. Still, questions and unmet needs about replacement of basal insulin remain even in the glargine and detemir era.
Compared with the physiology of basal insulin in subjects without diabetes, the present long-acting insulin analogs still present limitations, primarily variability of effects on BG control. Although the variability of s.c. absorption of soluble long-acting analogs glargine and detemir is lower compared with NPH, 30 glargine and detemir still present variable day-to-day effects on BG control. Part of the variability is due to the s.c. tissue where basal insulin is injected. This component of variability, intrinsic to the characteristics of the tissue, cannot be improved, unless in the future alternative sites of insulin administration will be used. Innovative formulations of basal insulin might reduce the absorption variability characteristic of individual insulin formulations.
Degludec is a new long-acting acylated insulin developed by NovoNordisk (Bagsvaerd, Denmark). 49 Its ultra-long effect is primarily a result of the slow release of insulin degludec monomers from soluble multihexamers that form after s.c. injection, resulting in a long half-life and a smooth and stable pharmacokinetic profile at steady state. 49 The preliminary data indicate longer duration of action compared with detemir, similar to glargine. 49 Also, intra-subject variability of the insulin effect has been reported to be lower with degludec compared with glargine. 50 Non-inferiority results with degludec compared with glargine have been reported in a short-term study in T1DM 51 and T2DM 52 as well. In the latter study the idea of basal insulin administration every 2 days compared with every day should not be translated into a treatment option because it would be highly diseducational for patients. The promising PK/PD characteristics of the new basal insulin degludec await confirmatory observations in longer and larger clinical studies.
An alternative approach to retard insulin absorption is poly(ethylene glycol) (PEG)ylation. 53 When electrophilically activated derivatives of low-molecular-weight monomethoxyPEG (mPEG) are chemically coupled to insulin via its amino groups at positions phenylalanine-B1 or lysine-B29, an amide bond is formed between the polymer and protein. 53 The site-specific attachment of mPEG to insulin does not substantially alter the insulin's secondary/tertiary structure, self-association behavior, or potency in vivo. However, mPEG attachment does significantly enhance insulin's resistance to aggregation. 53 A novel controlled release formulation has been developed with PEGylated human insulin encapsulated in microspheres that produces multi-day release in vivo. In animal studies, PEG–insulin microspheres administered subcutaneously as a single injection produced <1% release of insulin in the first day but then lowered the serum glucose levels of rats with diabetes to values <200 mg/dL for approximately 9 days; when doses were given at 7-day intervals, steady-state drug levels were achieved after only two doses. 54 Eli Lilly (Indianapolis, IN) has described a long-acting lispro insulin formulation that is PEGylated with high-molecular-weight PEG, is highly soluble at physiological pH, has an extended duration of action, and is characterized by PK, PD, and/or activity peak-trough ratios of less than 2. 55 Initial PD data in normal humans without diabetes indicate that PEGylated lispro has a flatter action profile compared with insulin glargine along with a prolonged activity (Fig. 8). 55 However, as clearly stated by Porcellati et al. 25 earlier in this issue, meaningful PD data are generated only in subjects with T1DM, not subjects without diabetes.
When these new long-acting insulin formulations will eventually reach the market, it will be important to compare them with the gold standard, basal insulin infusion by CSII, as was done for glargine, 26 and with glargine and detemir as well in subjects with T1DM. For now, it is important that subjects with T1DM learn how to optimally use glargine and detemir for good BG control.
Conclusions
The NPH era was associated with description of syndrome of “brittle” diabetes, 56 and with the definition of insulin treatment being “elusive.” 57 In reality, it was brittle insulin (NPH) that caused brittle diabetes, and this was the reason why insulin treatment was defined elusive. The modern physiological basal insulin replacement, along with mealtime insulin, BG self-monitoring, and education of doctors and patients, makes the goals of A1C <7.0% with minimal hypoglycemia realistic targets.
Footnotes
Author Disclosure Statement
G.B.B. has received honoraria for lecturing and scientific advising from Sanofi-Aventis, Eli Lilly & Co, NovoNordisk, and MannKind. No competing financial interests exist for A.M.A. and P.L.
