Abstract
Hypoglycemia has for the most part been studied inadequately for both of the commonly used long-acting insulin analogs in type 1 and type 2 diabetes. Almost all existing trials have been designed to investigate changes in glycemic control and not differences in hypoglycemia events. In this review, we present an overview of the hypoglycemic data available from the randomized controlled trials comparing insulin glargine and insulin detemir with NPH or continuous subcutaneous insulin infusion in type 1 and type 2 diabetes. The limited head-to-head glargine versus detemir data are also discussed with comments on early results relating to the newer insulin analog, degludec. Basal insulin analogs are associated with reduced nocturnal hypoglycemia in both type 1 and type 2 diabetes. Most studies have excluded participants with impaired awareness of hypoglycemia or previous severe events, however, and hypoglycemia reporting is variable and inconsistent. This limits interpretation for those with long-duration type 1 diabetes, and particularly impaired awareness of hypoglycemia, or long-duration more insulin-deficient type 2 diabetes. New optimally designed studies are required to elucidate the true impact of basal analogs on hypoglycemia burden in those living with long-term insulin therapy.
Introduction
However, hypoglycemia may itself be a risk factor for cardiovascular disease. Recently with regard to type 2 diabetes, it has been suggested that severe hypoglycemia is associated with significantly increased risk of macrovascular events, microvascular events, and all-cause death. 6 A recent review of the literature in this area describes how mechanisms involving counterregulatory responses, leukotriene secretion, and activation of platelets and neutrophils in response to hypoglycemia may increase cardiovascular risk. 7
As a 6,000-dalton peptide that naturally associates into 36,000-dalton hexamers if it is to maintain chemical and physical stability, unmodified insulin is quite unsuited to subcutaneous injection, rather than secretion into tributaries of the portal vein. 8 This problem was confounded by the necessary development of the extended-acting insulin preparations (based on protamine or zinc complexing) in the 1930s and 1940s, whereby the already problematic erratic absorption of dissolved insulin was coupled to erratic dissolution of the complex before absorption. 8 Lauritzen et al. 9 documented the problem with radioisotope studies in the 1970s.
Physiological basal insulin delivery at normoglycemia in normal-weight people is quite constant, even overnight. 3 In contrast, as reviewed by Porcellati et al. 10 in this supplement, the average profile of absorption of NPH insulin, first described in 1948, is entirely unsuited to reproducing that flat profile. The zinc insulin series (lente insulin) gave similar free insulin profiles to NPH insulin, and longer-acting preparations such as protamine zinc insulin and bovine ultralente insulin were associated with further problems, presumably because of degradation and thus poorer bioavailability with longer subcutaneous residence time.
Accordingly, the opportunity for chemical or biochemical modification of human insulin to produce new basal insulins was grasped in the 1990s, with the twin aims being to achieve a more physiological insulin profile (longer and flatter) and to reduce the within-patient between-day variation in insulin absorption. With these aims hypoglycemia should be reduced, allowing higher insulin dosage and thus lowering plasma glucose levels towards average.
Hypoglycemia has for the most part been inadequately studied for both of the commonly used basal insulin analogs, insulin glargine and insulin detemir. This is partly because clinicians, industry, and regulators (but not people with diabetes) see the need for insulin in terms of control of plasma glucose levels (as HbA1c), powering the pivotal studies accordingly.
Occurrence of hypoglycemia is more difficult to measure consistently and occurs with an unusual numerical distribution (with some participants having the majority of events in any one study), both of which lower statistical power. In people with type 2 diabetes, with a relatively low incidence of hypoglycemia, this is a particular problem.
Here we present an overview of the hypoglycemia data for insulin detemir and insulin glargine, with a comment on insulin degludec.
Type 1 Diabetes
Reporting of hypoglycemia in the randomized control trials (RCTs) comparing basal insulin analogs with conventional NPH insulin has not followed a consistent approach, with absolute values used to define low glucose ranging from <72 mg/dL (<4.0 mmol/L) to <36 mg/dL (2.0 mmol/L). Moreover, nearly all adequately powered trials to date have reported hypoglycemia only as a secondary end point and have not been designed appropriately to definitively compare rates. Type 1 diabetes basal insulin analog RCTs that have reported hypoglycemia outcome data are reported below and summarized in Table 1.
Unless otherwise stated data relating to basal analog are given before those relating to NPH comparator in the Results column.
CSII, continuous subcutaneous insulin infusion; NS, not significant; pt-mo−1, per patient-month; pt-yr−1, per patient-year.
Insulin glargine
Two early short-duration studies comparing insulin glargine and NPH insulin, in which all participants used unmodified human insulin prandially, 11,12 reported contrasting hypoglycemia results, although both achieved significantly lower fasting plasma glucose (FPG) in the glargine group. In the first, 11 with 333 participants, glargine (before bed) was associated with a reduced number of participants with at least one episode of symptomatic nocturnal hypoglycemia compared with NPH insulin (36% vs. 56%, P = 0.004). However, this reduction was significant only when glargine was compared with NPH taken once daily (also before bed), not when compared with NPH insulin taken twice daily (morning and bedtime). In the study by Rosenstock et al., 12 including 256 participants, incidence of hypoglycemic episodes was lower in the NPH insulin group (P = 0.03). The authors stated that this may have been due to the study design whereby the initial glargine dose was derived from the total prestudy dose of NPH insulin, reflecting the observation that some of the NPH dose would be for prandial blood glucose control, and this should not be substituted when changing to a true basal insulin.
Two 28-week comparative studies with 534 13 and 394 14 participants also using unmodified insulin prandially in both groups reported significant reductions in hypoglycemia and FPG in favor of glargine. When findings were confirmed with a measured blood glucose of <36 mg/dL (<2.0 mmol/L), Ratner et al. 13 found significant reductions in the percentage of patients experiencing at least one episode of any hypoglycemic episode (glargine before bed 40% vs. NPH once or twice daily 49%), nocturnal hypoglycemic episodes (18% vs. 27%), and severe hypoglycemic episodes requiring third-party intervention (1.9% vs. 5.6%). In the study of Hershon et al., 14 the percentage of participants reporting at least one episode of symptomatic hypoglycemia with a confirmed blood glucose of <50 mg/dL (<2.8 mmol/L) and <36 mg/dL (<2.0 mmol/L) was significantly reduced to 73% versus 82% and 37% versus 46%, respectively. No significant differences in the occurrence of any category of hypoglycemia were detected in a large multicenter 28-week study of glargine versus NPH (n = 585) when using unmodified human insulin at mealtimes. 15
Trials comparing insulin glargine and NPH insulin using prandial rapid-acting insulin analogs have also reported variable findings with respect to hypoglycemia. A 16-week trial 16 with 310 participants and a recent 24-week multicenter RCT of 175 participants 19 reported no significant differences in overall hypoglycemia between glargine and NPH groups. The latter study did, however, report significantly reduced serious nocturnal hypoglycemia as confirmed by a blood glucose of <42 mg/dL (2.3 mmol/L) with significantly reduced FPG in the glargine group from baseline to end point.
A 52-week single center RCT 17 including 121 participants reported a significant reduction in both daytime and nocturnal hypoglycemia with a confirmatory blood glucose of <72 mg/dL (<4.0 mmol/L) in the glargine group compared with NPH. A 30-week RCT by Fulcher et al. 18 recruited 125 participants all with initial HbA1c >8% and randomized to either once-daily glargine or NPH at bedtime with both groups receiving rapid-acting insulin analogs at mealtimes. The study reports a significantly lower study event rate for severe nocturnal (requiring assistance of a third party) and moderate (<50 mg/dL [<2.8 mmol/L]) nocturnal hypoglycemic events in the glargine group. However, there were comparable rates of overall hypoglycemia, and there was an increased study rate of minor daytime hypoglycemia (50–65 mg/dL [2.8–3.6 mmol/L]) in the glargine group compared with NPH.
A single-center clinical trial 20 with 51 participants comparing NPH insulin given four times daily and glargine given at dinnertime or bedtime over 3 months was published in 2003. All participants used rapid-acting insulin analogs prandially. This showed a significantly reduced frequency of monthly nocturnal hypoglycemia episodes in both the glargine at dinnertime (1.7 ± 0.2 episodes/patient-month) and glargine at bedtime (2.0 ± 0.19 episodes/patient-month) groups compared with the NPH group (3.6 ± 0.4 episodes/patient-month; P < 0.05). There was no difference reported in hypoglycemia rates between glargine groups.
In a multicenter randomized 32-week crossover trial 21 with 56 participants, NPH insulin and unmodified human insulin were compared with glargine and insulin lispro. This study showed no difference in the monthly rate of all symptomatic hypoglycemia between groups. However, it did demonstrate a significant reduction in the monthly rate of nocturnal hypoglycemia (0.66 ± 0.02 vs. 1.18 ± 0.02 episodes/month, P < 0.001) in favor of the combined analog group. The reduction in nocturnal hypoglycemia in this group was accompanied by a significant reduction in HbA1c and FPG. A study 22 with a similar design in 28 adolescents with type 1 diabetes assessed nocturnal hypoglycemia by undertaking overnight metabolic profiling. Nocturnal hypoglycemia was confirmed by the presence of two consecutive blood glucose values of <63 mg/dL (<3.5 mmol/L) on a venous blood sample taken between 22:30 and 08:00 h. Although only two episodes of nocturnal hypoglycemia were symptomatic, statistical analysis of total nocturnal hypoglycemic episodes revealed a significant reduction in favor of the combined analog treatment group (P < 0.05).
In a randomized 32-week (2 × 16-week treatment period) crossover study with 60 participants by Chatterjee et al. 23 comparing insulin aspart in combination with NPH at breakfast and dinnertime or glargine at bedtime, continuous subcutaneous glucose monitoring profiles were collected in addition to hypoglycemic rates as measured by a blood glucose of <50 mg/dL (2.8 mmol/L). No difference in hypoglycemia rates was found between the two basal insulin groups. From the 27 participants who had continuous glucose monitoring data no significant difference was found between either overall or nocturnal hypoglycemia. In this study, however, fasting glucose was >3.0 mmol/L lower in the glargine group in addition to significantly lower HbA1c.
An RCT with 378 participants 24 compared metabolic effects of giving insulin glargine at different times of the day (before breakfast, before the evening meal, and before bed). Total symptomatic hypoglycemia and severe hypoglycemia did not differ significantly between treatment groups. However, incidence of nocturnal symptomatic hypoglycemia was reported to be significantly less in the breakfast group (60% of participants) compared with both the dinnertime (72%, P = 0.029) and bedtime (78%, P = 0.0013) groups. This was despite occurrence of the lowest mean nocturnal blood glucose value in the breakfast group, suggesting this group did not have higher overall blood glucose levels. Ashwell et al. 25 also undertook a study comparing blood glucose control when glargine was given once daily at lunchtime, dinnertime, or bedtime, including 24-h in-patient metabolic profile. There was no difference in overall frequency of hypoglycemia, but timings of hypoglycemia varied between groups. Highest incidence occurred between 9.5 and 15.5 h after bedtime glargine without a nocturnal peak, whereas glargine before the evening meal was associated with peak incidence between 10 p.m. and 10 a.m.
Glargine has been compared when given once and twice daily. An 8-week, two-way, crossover study including 20 participants without history of recurrent severe hypoglycemia 26 compared these two regimens. There was no difference in hypoglycemia rates between participants given either once- or twice-daily glargine.
Hypoglycemia data from RCTs comparing continuous subcutaneous insulin infusion (CSII) pumps versus basal insulin analogs are extremely limited. Indeed, a recent meta-analysis 45 comparing severe hypoglycemia with CSII and multiple daily insulin injections (MDII) did not include any basal insulin analog studies as all were either of too short duration or had too low an initial frequency of severe hypoglycemia.
Two small RCTs comparing glargine with CSII in participants with type 1 diabetes with 27 and without 28 previous recurrent severe hypoglycemia showed no significant differences in frequency of hypoglycemia between the two interventions. Similarly, hypoglycemic rates were not different between CSII- and MDII-treated patients in a small 8-month randomized crossover study with 39 participants. 46
A short-duration 10-week multicenter randomized, crossover study 29 suggested a lower occurrence of nocturnal minor hypoglycemic episodes for subjects treated with CSII than for subjects treated with MDII therapy including insulin glargine. However, this study also suggested a higher rate of daytime minor hypoglycemic episodes for subjects treated with CSII than for subjects treated with MDII therapy.
Insulin detemir
When twice-daily detemir combined with regular human insulin was compared with NPH insulin also with regular human insulin by Standl et al., 30 only a trend toward reduced nocturnal hypoglycemia with detemir was demonstrated. This study also found no significant difference in overall glycemic outcomes.
A significant relative risk reduction in nocturnal hypoglycemia with insulin detemir was seen in a large study including 747 participants in which insulin detemir was compared with NPH insulin, both taken once daily. 39 Human insulin was used prandially by all participants.
In studies 31,32 comparing twice-daily insulin detemir and NPH insulin, where both groups used rapid-acting insulin analog prandially, significant reductions in hypoglycemia have been attained. In the first study 31 a significantly lower monthly rate of nocturnal hypoglycemia was seen in the detemir group compared with the NPH group (1,378 episodes in 180 patients vs. 926 episodes in 87 patients, P = 0.016) during the maintenance phase. There were no differences in overall monthly hypoglycemia rates or glycemic control assessed by HbA1c and FPG. In the latter study 32 including 447 participants, there was a reduction in both overall and nocturnal hypoglycemic events per subject-month with insulin detemir compared with NPH insulin (5.2 vs. 6.7 [P = 0.03] and 0.64 vs. 0.96 [P < 0.005], respectively). Again, HbA1c and FPG were not significantly different between groups, but there was a significant reduction in intra-person variation in self-measured fasting plasma glucose with detemir in comparison with NPH (SD, 3.37 vs. 3.78 mmol/L, P < 0.001).
A 24-month RCT was published in 2008 comparing insulin detemir and NPH insulin. 33 In this longer-term study that included 495 patients, detemir was found to reduce the risk of both severe (requiring assistance of a third party) and nocturnal hypoglycemic episodes per patient-year (0.2 vs. 0.8 [P < 0.001] and 3.4 vs. 6.4 [P < 0.001], respectively) although not overall hypoglycemia rates. In this study all participants used insulin aspart with meals, and all started on a once-daily basal regimen, although a second basal dose could be added if necessary. There was also significantly reduced HbA1c, FPG, and intra-patient variation in FPG in the detemir group.
Another study comparing insulin detemir and NPH insulin with both groups using insulin aspart prandially was designed and adequately powered to investigate whether there was reduced incidence of hypoglycemia in the combined analog group. 34 This crossover trial with two 16-week treatment periods included 130 individuals but actively excluded those with a history of impaired awareness of hypoglycemia and recurrent major hypoglycemia. The primary end point was defined as the total self-recorded hypoglycemic episodes with detemir relative to NPH during the last 10 weeks of each treatment period. This study found significant reductions in total and nocturnal episodes of hypoglycemia per patient per year (53.3 vs. 64.7 [P = 0.001] and 6.0 vs. 12.0 [P < 0.0001], respectively) in favor of detemir. This low-intensity study demonstrated a reduction in hypoglycemia in the detemir group without deterioration in HbA1c. Intra-individual variability in both mean and fasting self-monitored plasma glucose was significantly reduced in the detemir group.
Studies have been undertaken comparing NPH insulin with two different detemir regimens. Both demonstrated reduced intra-person variability in self-monitored FPG with insulin detemir, but they reported contrasting hypoglycemia results. 35,36 In the 16-week study of Pieber et al. 35 no differences were found in either overall or nocturnal hypoglycemia rates between NPH insulin and insulin detemir given either morning and before dinner or morning and bedtime. Home et al. 36 compared twice-daily NPH insulin with both detemir given in the morning and 12 h later and detemir given before breakfast and at bedtime. This 16-week study including 408 participants reported significantly reduced risk of nocturnal hypoglycemia in the detemir morning and bedtime group compared with the NPH insulin group (P < 0.001). There were significantly lower FPG levels in both detemir groups compared with the NPH group in addition to again significantly lower within-person between-day variation in self-monitored FPG.
Once- and twice-daily insulin detemir regimens have been compared. 37 In this study, 520 participants all took insulin aspart prandially. No significant difference was found either in the rate of hypoglycemia between the two groups or in overall glycemic outcome measures.
An 18-week RCT 38 including 595 patients compared the combined analog regimen of twice-daily detemir and prandial aspart with twice-daily NPH insulin and prandial regular human insulin. As well as a significantly reduced HbA1c in the analog group (7.9% vs 8.1%, P < 0.001), there were fewer overall and nocturnal hypoglycemic episodes per person-year (37.1 vs. 48.2 [P = 0.036] and 4.0 vs. 9.2 [P < 0.001], respectively). Within-person day-to-day variation in plasma glucose as measured by all self-monitored glucose values during the last week of the study was lower with insulin detemir/insulin aspart than with NPH insulin/regular human insulin (SD, 2.88 vs. 3.12 mmol/L; P < 0.001).
We are not aware of any published RCTs comparing detemir and CSII.
Glargine versus detemir studies
In 2007, a 26-week RCT 40 including 320 patients comparing once-daily glargine with twice-daily detemir using premeal aspart achieved similar glycemic control and overall hypoglycemia rates. FPG was significantly lower in the glargine group, associated with higher bedtime insulin dose and overall bolus dose. This was associated with an increase in nocturnal and severe hypoglycemia. A multinational 52-week RCT comparing the two basal insulin analogs 41 including 443 participants showed no significant difference in either hypoglycemia rates or glycemic outcome measures between the two groups.
Hypoglycemia risk with detemir and glargine in addition to NPH has also been studied specifically in relation to risk after exercise. 42 This showed an increase in post-exercise hypoglycemia 5 h after a morning glargine injection in comparison with either detemir or NPH. However, it is notable that use of twice-daily NPH and detemir but once-daily glargine led to almost twice the morning dose of basal insulin in the glargine group.
Insulin degludec
Data for this novel basal insulin analog remain limited to date particularly regarding hypoglycemia. The once-daily formulation has been associated with reduced numbers of hypoglycemic episodes compared with insulin glargine. A phase 2 16-week RCT 43 with 178 participants with type 1 diabetes comparing two different formulations of insulin degludec with insulin glargine was published in March 2011. This study suggested a reduction in all confirmed episodes of hypoglycemia (blood glucose <56 mg/dL) including nocturnal hypoglycemia with insulin degludec compared with glargine. Overall control as assessed by end-of-study HbA1c was similar in all treatment groups. A Novo Nordisk press release to investors in December 2010 reported that a 52-week phase 3a degludec study in 629 people with type 1 diabetes using mealtime insulin aspart showed 24% lower nocturnal hypoglycemia compared with insulin glargine despite similar changes in HbA1c. 44
Type 2 Diabetes
Insulin glargine
As noted above, the relatively low incidence of hypoglycemia in people with type 2 diabetes on insulin poses power problems for anything other than larger studies. Furthermore, registration studies of insulin glargine were performed against NPH insulin at a time when treat-to-target protocols were not properly deployed, with people having high HbA1c at randomization remaining in poor even if improved control at the end of the studies. In the subpopulation who were previously insulin naive in one such study, while overall plasma glucose control remained poor (mean HbA1c, >8.0%), numbers of both all and nocturnal hypoglycemia episodes were significantly reduced on insulin glargine compared with NPH insulin by 59% (10% vs. 24%) over 52 weeks. 47 This was not confirmed for the entire population, including those previously insulin-treated. 48 In the other registration study of only 24 weeks in duration with marginally better overall blood glucose control and in people already using insulin but not in combination with oral agents, nocturnal hypoglycemia was also reduced in the glargine group (study prevalence from 36% to 27%, P = 0.014), as possibly was severe hypoglycemia (10% to 7%, P = 0.055). 49
Concepts of insulin management in type 2 diabetes were, however, changed by the treat-to-target study of insulin glargine versus NPH insulin once daily (insulin-naive population, continued oral agents) in which more respectable overall glucose control was achieved (7.0% on both insulins), but with a reduction of prevalence of nocturnal hypoglycemia over 24 weeks from 6.9% to 4.0% (P < 0.001), with this driving a similar absolute difference in overall symptomatic hypoglycemia. 50 Analysis of rate of hypoglycemia by time of day showed advantage for insulin glargine for every hour between 01:00 and 07:00 h, the times of peak frequency of hypoglycemia on NPH insulin.
This study used evening administration of basal insulins, once daily, and several other studies have reported similar data also using a treat-to-target approach in insulin-naive people and from diverse populations. A recent meta-analysis combined the data from five studies (evening glargine administration) for which patient level data were available, in an attempt to more precisely estimate the reduction in hypoglycemia for insulin glargine compared with NPH insulin. 51 For nocturnal hypoglycemia the odds ratios clustered around 0.50, being 0.44–0.52 whatever definition (including severe hypoglycemia) was used, with all estimates statistically significant. Daytime hypoglycemia was not, however, improved or deteriorated, so that for overall (anytime) hypoglycemia only the more frequent symptomatic definitions had the power to reach statistical significance. Not included in this analysis was the similar study from Finland for which equivalent data were not available, but where results were consistent with the meta-analysis. 52 For morning glargine injection compared with evening NPH insulin, a single study was found, with this tending also to show reductions in nocturnal hypoglycemia, statistically significant only for the most frequent definition of confirmed glucose <3.9 mmol/L. 53
Such data can be used to calculate the number of patients needed to treat to avoid one episode of hypoglycemia if glargine is used rather than NPH over the average time period of the studies (25 weeks). For nocturnal hypoglycemia this was eight for evening glargine injection and five for morning injection. 51
Poisson regression analyses describing the relationship between hypoglycemia and HbA1c for glargine and NPH insulin have been published for the Treat-to-Target study and as meta-regressions of multiple studies (Fig. 1) 54,57,58 (see separate discussions of Riddle et al. 54 and Yki-Järvinen et al. 57 in Rosenstock and Owens, 55 pp. 121–122 and 119–120). The validity of meta-regression analysis in this situation can be challenged as simply pooling data can give misleading results if the shape of the curve differs between studies. Nevertheless, the overall message of these analyses is the same, namely, that the difference in hypoglycemia rates between insulins is largest at lower HbA1c levels and is much less at levels above 10%.
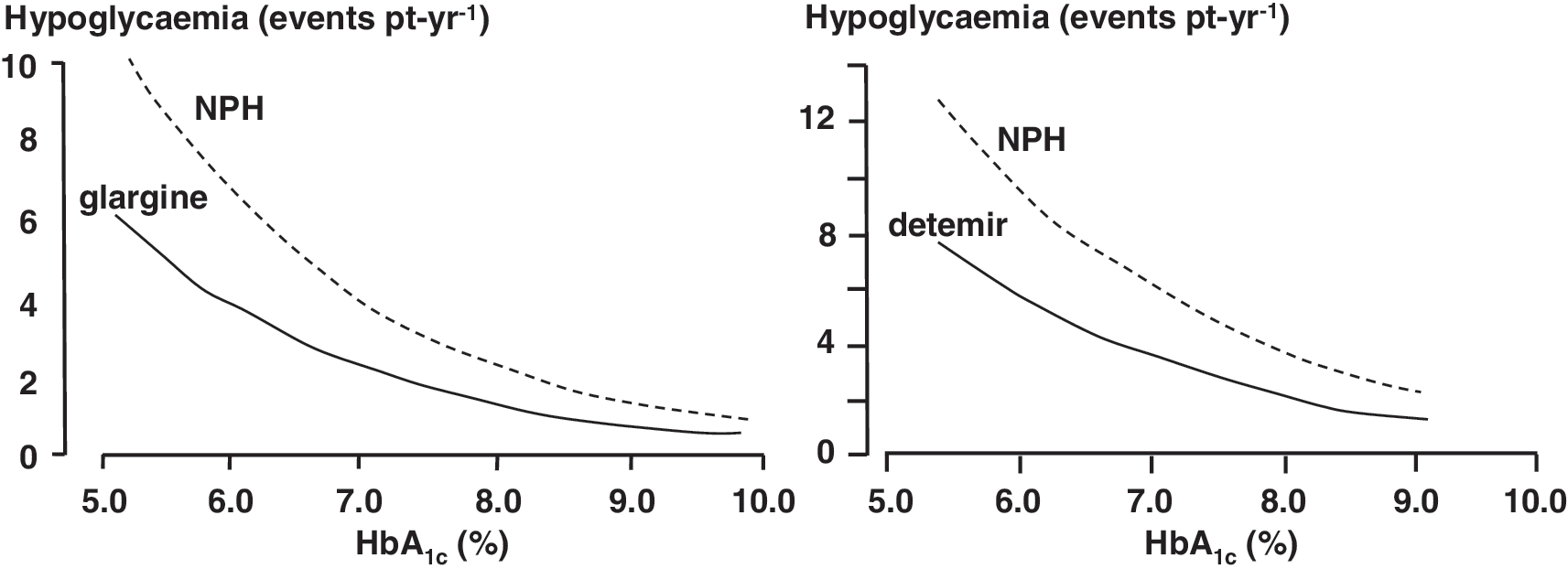
Lastly, hypoglycemia data are available for comparisons between insulin glargine and glucagon-like peptide-1 mimetics. 59,60 In these studies the rates did not differ, but were low, because of undertitration of the insulin doses.
Insulin detemir
Not unlike the insulin glargine studies, the early insulin detemir studies in type 2 diabetes did not attain good blood glucose control, and hypoglycemia was of relatively low prevalence. In previously insulin-treated people, when transferred to a mealtime + basal insulin regimen incorporating either insulin detemir (and insulin aspart) or NPH insulin (with human short-acting insulin), although nocturnal hypoglycemia was reduced by 38%, this was not statistically significant. 61 Similarly, where detemir and NPH were compared again in people already using insulin but using insulin aspart only at mealtimes, the low prevalence (detemir, 16%; NPH, 24%) of hypoglycemia did not differ significantly between groups, even though between-day variability in pre-breakfast glucose concentration was reduced with detemir. 62
A treat-to-target study of insulin detemir compared with NPH insulin in insulin-naive people with diabetes subsequently achieved mean HbA1c levels of 6.8% and 6.6%, respectively. 56 In this situation there was a highly statistically significant reduction in nocturnal hypoglycemia (55%, P < 0.001) in the detemir group driving a reduction in any hypoglycemia of 47% (P < 0.001). Within the article the relationship of hypoglycemia and HbA1c was explored by negative-binomial regression, again showing that the greatest numerical reduction in hypoglycemia is at lower HbA1c, although relative risk reduction is similar throughout the range (Fig. 1).
A Cochrane review by Horvath et al. 63 published in 2007 assessed studies comparing both insulin glargine and detemir to NPH insulin. This review reports significantly reduced rates of symptomatic, overall, and nocturnal hypoglycemia in patients treated with either insulin glargine or detemir.
Basal insulin analogs in combination with glucagon-like peptide-1 mimetics
Hypoglycemia has been investigated in patients treated with insulin glargine in combination with the glucagon-like peptide-1 mimetic exenatide. A 30-week RCT with 261 participants used glargine together with placebo or twice-daily exenatide. 64 This study showed no significant difference in hypoglycemia rates between groups while demonstrating a significant reduction in HbA1c (−1.7% vs. −1.0%) in the glargine plus exenatide group.
Basal insulin analogs and continuation with sulfonylurea therapy
In a non-randomized comparison of people continuing or stopping insulin secretagogues when starting a basal insulin, 65 there was more hypoglycemia in the secretagogue group, despite lower insulin doses; however, the change in HbA1c in the two groups was not similar, further limiting the conclusions that can be drawn.
Glargine versus detemir studies
Insulin detemir and insulin glargine have been compared in people with type 2 diabetes. 66 The study was limited by licensed indications resulting in an asymmetric protocol for number of injections allowed. However, overall glucose control was not different, within-participant variability for self-monitored FPG and pre-dinner plasma glucose did not differ by insulin treatment, and relative risk of overall and nocturnal hypoglycemia did not differ. A further study comparing the two basal analog insulins published in 2010 67 also demonstrated no difference in rates of overall hypoglycemia between treatment groups. This study suggested lower FPG with glargine than with detemir but lower daytime plasma glucose with detemir. The mean reduction in HbA1c was similar between groups; however, fewer patients treated with detemir reached an HbA1c of <6.5%.
Insulin degludec
At the time of writing only data from a small phase 2 study were available. While confirmed hypoglycemia prevalence on once-daily degludec (8%) was lower than on once-daily glargine (23%), the total number of events was too small for any conclusions to be reached. 68
Conclusions
Administering NPH insulin at bedtime compared with administration at dinnertime was shown in the 1980s to allow better pre-breakfast plasma glucose control without worsening the dip in the nighttime glucose profile. 69 However, if the administered dose is unchanged as a result of absorption peaking at around 5 h, at least with the more physiological doses used in type 1 diabetes, a major contribution to the risk of nocturnal hypoglycemia is inevitable. In keeping with their flatter pharmacokinetic and pharmacodynamic profiles (described earlier in this supplement), 10 lower rates of overnight hypoglycemia were to be expected with the long-acting analogs, and the evidence reviewed above confirms that for the most part this promise is delivered (Table 1). To some extent this is surprising, as the studies were mostly not powered for hypoglycemia, an outcome that is highly variable in people with diabetes.
Lower rates of overnight hypoglycemia have also been demonstrated in type 2 diabetes with both insulin glargine and detemir across the full range of HbA1c (Fig. 1). The differences between NPH insulin suspensions and the basal insulin analog solutions serve as a reminder that hypoglycemia is a consequence of variability of insulin absorption from day to day as much as inappropriate mean pharmacokinetic/pharmacodynamic profiles. Variability of absorption will not be better in type 2 diabetes, but thanks to residual endogenous insulin secretion background glucose levels are more consistent. The significance of variability may account for why insulin glargine, although of longer duration of action in physiological doses, does not seem to perform differently from insulin detemir, with the latter insulin showing evidence of lesser variability of pre-breakfast plasma glucose levels than NPH insulin 36 and perhaps better performance after exercise. 42 This would be consistent with the large plasma pool of albumin-bound detemir, able to buffer any acute changes in absorption. It should be noted, however, that relatively few glargine studies have specifically reported variability in fasting glucose following bedtime basal insulin administration.
As alluded to above, the potential benefit is greatest in those with absolute β-cell deficiency without insulin resistance who are at highest risk of hypoglycemia per se and progression to impaired awareness and severe events. The majority of trials to date have actively excluded those with previous recurrent severe hypoglycemia 18,29 –32,34 –36,39,45,46 or impaired awareness of hypoglycemia. 8,33,35,39,45 Trial participants with type 1 diabetes have thus tended to be relatively young (mean age usually between 35 and 45 years old with mean duration of diabetes of usually less than 20 years), whereas those with type 2 diabetes have had a relatively short duration (or none) of preceding insulin therapy. This, in addition to inconsistencies and inadequacies in collecting/reporting hypoglycemia, means that only limited interpretations of the data can be made for those with long-duration type 1 diabetes, and in particular impaired awareness of hypoglycemia, or long-duration more insulin-deficient type 2 diabetes. Further research in this area is ongoing.
The clinical trial focus on mean glucose levels is also unfortunate in trying to make more profound judgments about how difference between the analogs might influence diabetes control and quality of life. In the real clinical situation people with diabetes and physicians are much more cognizant of the risks and inconveniences of hypoglycemia and will sacrifice attainment of acutely better glucose levels for current quality of life. This does not seem to happen in the quality-assured atmosphere of the modern treat-to-target clinical trial and may be handicapping the ability of the long-acting analogs to achieve better blood glucose control at the same rate of hypoglycemia as NPH insulin. This is of some importance as the measurable decrement of quality of life due to acute hypoglycemia is quite small when averaged over the year, such that healthcare funders have been reluctant to pay a premium price for it. 70 Meanwhile, it might suggest that a better measure of advantage (or otherwise) of different analogs might be a composite of HbA1c and hypoglycemia.
Although glargine is commonly used twice daily, comparative studies have almost exclusively been confined to once-daily injection in accordance with its license, in contrast to detemir, which has been used both once and twice daily. This asymmetry again limits interpretation of comparative hypoglycemia risk. Minimization of overnight hypoglycemia can be achieved by avoiding an evening basal insulin injection, but this is often associated with suboptimal control through the night. Bedtime administration of both detemir and glargine is associated with lower nocturnal hypoglycemia risk than injection before the evening meal. 25,36 In those with type 1 diabetes, including glargine users, who cannot attain glucose target consistently before the evening meal, a second morning injection may be helpful in avoiding both excessive prandial insulin doses and a high basal insulin dose before bed associated with falling glucose throughout the night and thus increased risk of nocturnal hypoglycemia.
In order to properly determine the relative merits of different basal insulins and CSII with respect to hypoglycemia, novel studies are required specifically including participants with impaired awareness and previous problematic hypoglycemia. Newer studies should be adequately designed to optimally capture hypoglycemia incidence/severity, including silent nocturnal events, perhaps by more accurate continuous glucose monitoring devices, and powered to detect hypoglycemia-specific differences.
Footnotes
Author Disclosure Statement
S.L. has received reimbursement of travel costs to attend international diabetes meetings from Novo Nordisk. J.S. has received research funding and reimbursement of travel costs to attend international diabetes meetings from Novo Nordisk, sanofi-aventis, and Medtronic. P.H. or the institutions with which he is associated receive funding from all major insulin manufacturers for his research, advisory, or educational activities.
