Abstract
Type 2 diabetes is characterized by insulin resistance and progressive β-cell deterioration. As β-cell function declines, most patients with type 2 diabetes treated with oral agents, in monotherapy or combination, will require insulin therapy. Addition of basal insulin (glargine, detemir, or NPH/neutral protamine lispro insulin) to previous treatment is accepted as the simplest way to start insulin therapy in those patients. But even when basal insulin is adequately titrated, some patients will also need prandial insulin to achieve or maintain individual glycemic targets over time. Starting with premixed insulin is an effective option, but it is frequently associated with increased hypoglycemia risk, fixed meal schedules, and weight gain. As an alternative, a novel approached known as “basal plus strategy” has been developed. This approach considers the addition of increasing injections of prandial insulin, beginning with the meal that has the major impact on postprandial glucose values. Finally, if this is not enough intensification to basal–bolus will be necessary. In reducing hyperglycemia, this modality still remains the most effective option, even in people with type 2 diabetes. This article will review the currently evidence on the basal plus strategy and also its progression to basal–bolus therapy. In addition, practical recommendations to start and adjust basal plus therapy will be provided.
Introduction
When insulin should be added, addition of basal insulin (BI) (glargine, detemir, or NPH/neutral protamine lispro [NPL] insulin) to previous OAs is accepted as the simplest way to start insulin therapy in people with T2DM. 8 –10 To be effective, BI needs to be titrated based on fasting self-monitoring of blood glucose (SMBG) (100–110 mg/dL [5.5–6.1 mmol/L]) when administered once daily or before dinner/bedtime (“Treat to Target” concept). 9,11 If a second injection of BI is necessary (detemir or NPH/NPL insulin), morning insulin dose should be also adjusted based on SMBG before dinner. 12 This strategy has been successful bringing a significant proportion of patients (approximately 60–70%) to target, 9,10 at least during the first 6–12 months, or even up to 2–3 years. 13 However, a remaining important proportion of patients will need prandial insulin to achieve or maintain glycemic objectives. Alternatively, recent studies suggest that adding glucagon-like receptor-1 receptor agonists or dipetidyl peptidase-4 inhibitors to BI may be also effective and safe in this context, 14,15 considering that some patients may still retain enough pancreatic reserve.
Adding prandial insulin only (without BI) has been proposed as another alternative to start insulin when failing OAs in patients with T2DM. In terms of reduction of A1C, such a strategy is as effective 16 or even better 17 than starting insulin therapy with BI and achieves a superior postprandial control. 16,18 However, this approach does not seem to result into better cardiovascular outcomes in high cardiovascular risk T2DM patients. 18 Furthermore, the addition of prandial insulin results in a greater incidence of hypoglycemia, 16,17 even when BI is successfully titrated to obtain comparable A1C levels. 16 For these reasons, when insulin is needed, BI is considered the preferred starting option.
Therefore, progressing in insulin therapy needs a combination of basal and prandial insulin. Between the different therapeutic options, starting with premixed insulin twice or three times daily is one of the most widely used options. 19 Premixed insulin-based therapy, although effective in reducing hyperglycemia, is frequently associated with an increased risk of hypoglycemia, a need of fixed meal schedules, and weight gain. 20,21 As an alternative to premixed insulin, basal–bolus therapy (or multiple daily injections) still remains as the most effective insulin strategy, even in people with T2DM. 22,23 Because this option is rarely accepted by patients, at least initially, other options have been searched to facilitate a stepwise intensification of insulin therapy in people with T2DM when progressive deterioration of β-cell function is inevitable. Recently, a new therapeutic option, known as “basal plus strategy,” has been evaluated in these patients when BI in combination with OAs is not longer enough to achieve glycemic goals. 24
This article will review the currently evidence on the basal plus strategy and also its progression to basal–bolus therapy (Fig. 1). In addition, practical recommendations to start and adjust basal plus therapy will be provided.
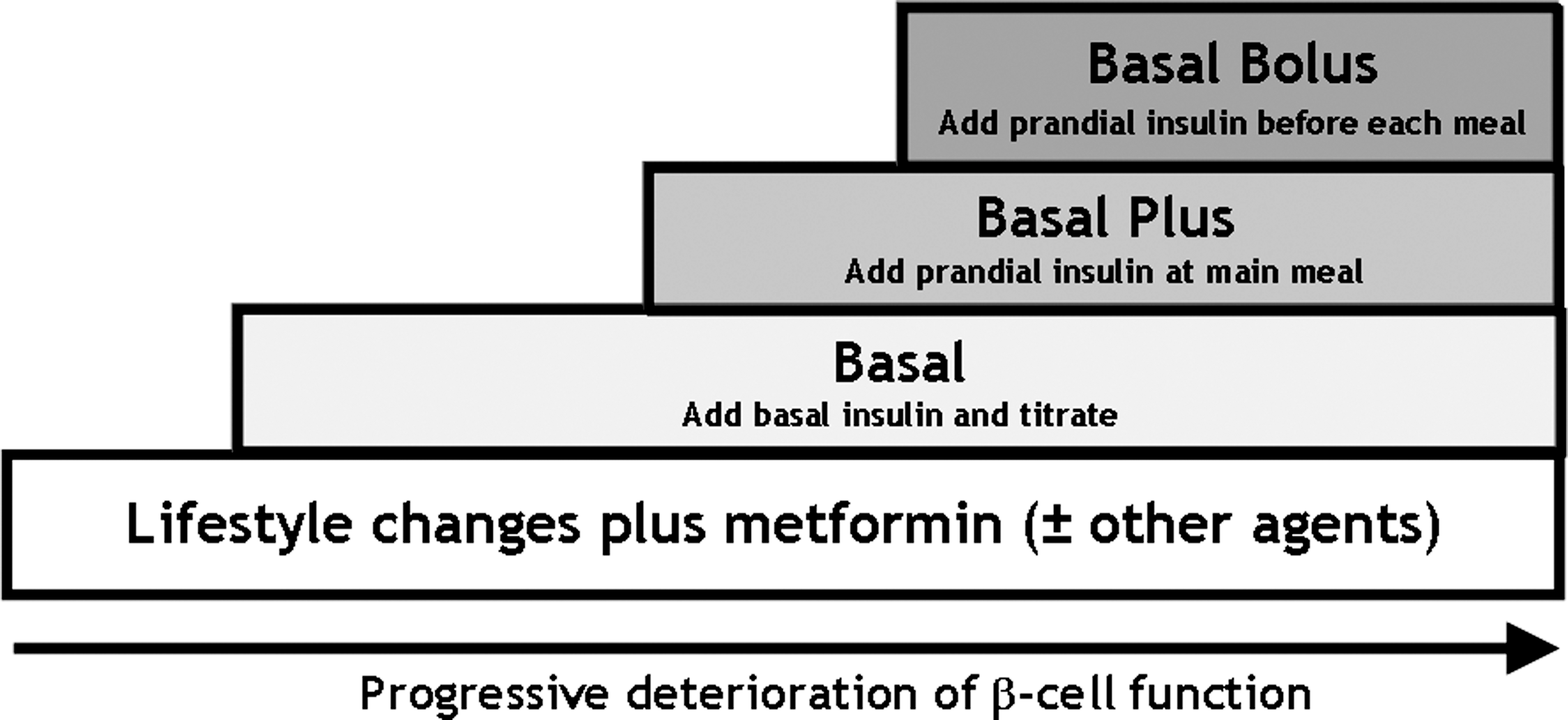
Matching treatment to disease progression in type 2 diabetes mellitus using a stepwise approach. Adapted from Raccah et al. 24 Type 2 diabetes is a progressive disease marked by insulin resistance and increasing failure of the pancreatic β-cell function. Therefore, as diabetes progresses, treatment will need to be intensified, including addition of insulin. Starting basal insulin in addition to previous oral agents is the simplest way to initiate insulin therapy in people with type 2 diabetes. When β-cell dysfunction progresses, prandial insulin is necessary and may be given using different strategies (see text). The progression from basal therapy to basal plus, and then to basal–bolus therapy, is shown schematically.
“Basal Plus” Therapy
The concept of basal plus considers the addition of increasing injections of prandial insulin, beginning with the meal that has the major impact on postprandial glucose values, and maintaining previous treatment with BI and OAs. 25 As explained below, this represents an effective strategy for a “soft” transition to the more physiologic (and effective) replacement of insulin by means of classical basal–bolus therapy. 24
When to move from basal to basal plus
Patients eligible to start basal plus therapy are those treated with basal therapy with or without OAs who do not reach A1C goals (<7.0% for most patients), even after adequate titration of BI to target (100–110 mg/dL [5.5–6.1 mmol/L]). 25 In addition, it has been suggested that also patients using high doses of BI without success (>0.7 U./kg) or who had limitations on increasing BI doses because of high risk of nocturnal hypoglycemia are good candidates. 26 Although evidence is scarce, other patients may potentially profit of basal plus therapy, as suggested by a recent experts' document, summarized in Table 1. 26
Modified from Merchante et al. 26
Most patients will have a slightly elevated A1C (between 7.0% and 8.0%). Indeed, Monnier et al. 27 have suggested that excessive postprandial glucose excursions are the most important component of hyperglycemia for patients with less elevated A1C levels (∼7.3%). In addition, using continuous glucose monitoring it has been demonstrated that high postprandial glucose levels are the first recognized glucose abnormality, especially after breakfast, even before fasting glucose values deteriorates. 28 Recently, it has been reported that the relative contribution of postprandial hyperglycemia increases to approximately 60% for an A1C below 8%, independent of the A1C ranges, but only when BI is titrated appropriately. 29 In any case, it seems reasonable to suggest that when patients are closed to the A1C target the addition of a fitting dose of prandial insulin administered at the main meal might be effective.
Review of the current evidence
Because the basal plus strategy is a relatively new concept coming out in the last years, only a few trials have been published until now. In the “Proof Of Concept” (POC) trial, 106 patients with T2DM treated with insulin glargine in combination with OAs and A1C > 7.0% (60% women; age [mean ± SD], 60 ± 8 years; body mass index, 33.3 ± 4.8 kg/m2; A1C, 7.9 ± 0.6%; fasting plasma glucose, 111 ± 22 mg/dL) were randomized to continue with the same treatment (control group) or to the addition of insulin glulisine before the main meal (intervention group). 30 After 3 months, patients assigned to the glulisine group (0.14 ± 0.07 U/kg/day) achieved a greater reduction of A1C (−0.36 vs. −0.13% in the control group, P = 0.029), without a significant increase of hypoglycemic episodes. BI increased in the control group from 0.59 ± 0.26 to 0.65 ± 0.32 U/kg/day (no statistical value reported), but not in the glulisine group (from 0.57 ± 0.31 to 0.59 ± 0.35 U/kg/day). In addition, in the intervention group a higher proportion of patients reached an A1C < 7.0% (22% vs. 9% in the control group). This POC study confirmed that the addition of only one injection of prandial insulin may reduce further A1C by approximately 0.3–0.4% without increasing the risk of hypoglycemia.
In the “Oral Plus Apidra and Lantus” (OPAL) trial, 393 patients with T2DM receiving treatment with insulin glargine and OAs and insufficient glycemic control (mean basal A1C was ∼7.4%) were randomized to the addition of insulin glulisine before breakfast or before the main meal. 31 The main meal was defined as the meal associated with the highest postprandial glycemic excursion based on several daily glycemic profiles. The initial dose of insulin glulisine (∼5 U) was individually increased in both groups, at the discretion of the investigators, using a similar target of 2-h postprandial glucose values for titration (≤135 mg/dL [≤5.5 mmol/L]). After 24 weeks, A1C was reduced similarly ∼0.4% in both groups using similar doses of insulin glulisine (∼11 U), although a greater proportion of patients reached the goal of A1C < 7.0% in the main meal group (52% vs. 37% in the breakfast group). No differences in the frequency of hypoglycemias were found between the groups (∼2.52 hypoglycemic events per patient per year). Body weight increased similarly about ∼1 kg in both groups.
In the “Optimisation of Insulin Treatment of Type 2 Diabetes Mellitus by Telecare Assistance for Self Monitoring of Blood Glucose” (ELEONOR trial), 200 patients failing on one or more OAs (54% men; age, 58.9 ± 8.2 years; body mass index, 29.9 ± 4.3 kg/m2; duration of diabetes, 10.9 ± 6.9 years; basal A1C, 8.9 ± 0.9%) were randomized to insulin dose adjustment by either electronic transfer of capillary blood glucose readings (Telecare program) or standard SMBG (control group). 32 After an initial phase of 8–16 weeks, in which insulin glargine was optimized to achieve fasting blood glucose ≤100 mg/dL (5.5 mmol/L), patients were randomized to glulisine once daily, at the main meal, for an additional 24 weeks. Insulin glulisine adjustments in the follow-up were based on postprandial glucose measurements (target, 100–140 mg/dL) using also Telecare or SMBG. Mean A1C decreased similarly from baseline to end point (8.8% to 7.1% for Telecare; 8.9% to 7.0% for SMBG). The single injection of glulisine was responsible for the last 0.7–0.8% decrement (both P < 0.0001 vs. with glargine alone). The proportion with A1C ≤ 7.0% at end point did not differ significantly between the Telecare and SMBG groups (50.6% vs. 54.6%). There was no difference in weight changes (Telecare vs. SMBG, +0.4 ± 3.0 vs. +0.1 ± 5.0 kg), glargine final dose (29 ± 16 vs. 28 ± 17 U/day), or glulisine final dose (8.3 ± 7.1 vs. 8.1 ± 8.1 U/day). Severe hypoglycemia episodes were experienced by three subjects in the Telecare group and one patient in the SMBG group. In this study, the efficacy of basal plus strategy was unaffected by the insulin dose adjustment method used.
The 1-2-3 trial was designed to evaluate the effectiveness of the increasing addition of prandial insulin injections in patients with T2DM previously treated during a 14-week run-in period with BI and OAs and having bad metabolic control. 33 Thereafter, 343 patients (age, 53 years; body mass index, 37 kg/m2; A1C, 7.9%; fasting plasma glucose, 120 mg/dL; duration of diabetes, 10 years) were randomized to three groups depending of the number of injections of insulin glulisine administered before the main meal. After 24 weeks, A1C values were reduced similarly in all groups (−0.46% vs. −0.46% vs. −0.58% in the group with one, two, or three additional injections of insulin glulisine, respectively). However, the proportion of patients achieving an A1C ≤ 7.0% was higher with increasing number of injections (30% vs. 33% vs. 45% with one, two, or three injections of insulin glulisine, respectively). In addition, the hypoglycemia rate increased with the rising number of glulisine injections (0.10 vs. 0.30 vs. 0.26 events/patient/year with one, two, or three injections, respectively), although only significantly higher when comparing the group with one versus two shots of insulin glulisine (P = 0.043).
Recently, two additional trials have been reported evaluating the efficacy and safety of the stepwise intensification of prandial insulin. In one trial, 296 patients with T2DM and not controlled with insulin detemir and OAs (mean values: age, 58.3 years; A1C, 8.8%; duration of diabetes, 12.3 years) were randomized to stepwise addition of insulin aspart in the largest perceived meal (n = 150) or in the meal with the largest postprandial glucose increment (n = 146). 34 After a run-in period, in which basal doses were optimized, patients received increasing doses of insulin aspart after 12-week periods if A1C was ≥7% according to the different protocols. Sulfonylureas were discontinued before randomization. Both strategies were equally effective in reducing A1C by ∼1.2% (by ∼0.5% in period 1, by a further ∼0.5% in period 2, and by ∼0.2% in period 3 in both groups). The overall rate of hypoglycemia was low, and weight gain was comparable, between 2.0 and 2.7 kg, at the end of the trial.
In the other trial, stepwise addition of prandial insulin with or without discontinuation of sulfonylureas was compared with the basal–bolus therapy in patients with type 2 diabetes. 35 After a 6-month period in which insulin glargine was optimized (36 U/day; mean A1C, 8.3%), patients with A1C > 7% and fasting plasma glucose <6.7 mmol/L (<120 mg/dL) (n = 476) were then randomized to the three different strategies. All patients received insulin glulisine as prandial insulin, which was titrated according to postprandial values. Increasing doses of insulin glulisine were added at 4-month intervals upon the next meal with the highest postprandial glucose value. After 1 year, full basal–bolus therapy was not superior to stepwise intensification of prandial insulin, without or with sulfonylureas (change of A1C, −0.72% vs. −0.47% vs. −0.40%, respectively; prespecified margin of non-inferiority, ≤0.4%). Mean glulisine dose at end point was 29, 20, and 17 U/day, respectively, with 33% and 40% of patients remaining on one glulisine injection in the group without or with sulfonylureas. The incidence of symptomatic hypoglycemia was highest with sulfonyluresa and lowest without (between-group differences were not significant). Weight gain was also significantly lower in the group with stepwise addition of prandial insulin without sulfonylureas compared with the basal–bolus group (+1.30 vs. +2.03 kg, P < 0.05).
In summary, although the current evidence is still scarce and the designs of the different trials were heterogeneous, the addition of increasing number of prandial insulin injections seems to be an effective and safe treatment modality. Therefore, the basal plus strategy should be view as an alternative for those patients treated with BI and OAs who are not able to achieve or maintain their individual glycemic goals. 36 Recently, this concept has been also incorporated in the treatment algorithm supported by a group of experts from the European Association for the Study of Diabetes and the American Diabetes Association. 11 However, the currently algorithm is sparse regarding how to initiate, titrate, and intensify prandial insulin doses.
Practical issues regarding basal plus strategy
How to initiate and titrate prandial insulin
In the basal plus strategy, the initial dose of prandial insulin can be calculated using different methods, although all of them offer similar results (Table 2). To start, you can use a fixed dose or find an individual dose depending on weight, previous BI dose, or currently postprandial glucose values. Initiating with a very small fixed dose is the simplest way to add prandial insulin and avoid hypoglycemia, although this dose may be very low depending on the patient. In any case, each healthcare provider should try one of the offered possibilities or search for an alternative.
The frequency of dose adjustments should be individualized, but changes can be done every 3 days or weekly. Patients should be trained in self-dose adjustments.
Finding the most appropriate dose of prandial insulin safely requires using an adequate titration algorithm and defining the most valuable glucose target. Insulin doses may be adjusted based on either preprandial or postprandial glucose values. Some guidelines recommend achieving a 2-h postprandial glucose value lower than 140 mg/dL (7.8 mmol/L), 37,38 which in many cases means limiting the glucose excursion to less than 40–50 mg/dL (2.2–2.8 mmol/L). 39 In the case of using rapid-acting insulin analogs (RAIAs), adjustments based on postprandial glucose values seem to be more worthwhile.
Increasing the dose of prandial insulin can be performed with fixed dose increments (i.e., 1 or more units each time) or depending of the previous prandial insulin dose or level of postprandial glucose values (see Table 2). Titration should be performed regularly based on SMBG. Patients should be instructed to be able to implement titration algorithms at home. Changing insulin doses when necessary every 3 days or weekly seems to be reasonable.
Selection of the prandial insulin to be used will depend on the patient's characteristics, availability of insulin, and costs. RAIAs (lispro, aspart, glulisine) have to be administered with the meals and try to reproduce the rapid, potent, and short-acting insulin-secretory response that takes place with endogenous insulin after intake in individuals without diabetes. Compared with regular insulin, RAIAs reduce postprandial glycemic excursions more effectively, as well as the incidence of late hypoglycemia (before the next intake). 40 Moreover, because of their rapid action, RAIAs may be administered just before the meal or even after, which is particularly appreciated by some patients, in particular, by elderly persons with unpredictable intake. In contrast, regular insulin requires administration 30–45 min before the meals, which in fact rarely is accomplished by patients on a daily basis.
Finally, when basal plus therapy is implemented no changes in previous BI dose are necessary. However, it seems logical to avoid nocturnal hypoglycemias, reducing the dose of BI by ∼10% if the selected main meal to start with prandial insulin is the dinner. 26
Recommendations about the use of OAs in the basal plus concept
In general, OAs used in combination with BI should be maintained also in the basal plus strategy. Metformin should be continued and is beneficial in association with insulin, irrespective of the insulin regimen. In relation to glitazones (only currently pioglitazone is available in Europe), although these agents may have potential benefits in some patients, it seems prudent to avoid their combination with insulin because of the potential for increased fluid retention and weight gain. Other OAs such as sulfonylureas, glinides, or dipeptidyl peptidase-4 inhibitors (sitagliptin) may be maintained during basal plus. A recent study compared continuation versus discontinuation of insulin secretagogues (sulfonylureas and glinides) in addition to metformin when initiating BI in patients with T2DM over 24 weeks. 41 A1C reduction was similar in both groups (continuing vs. stopping secretagogues, −1.59% vs. −1.30%, P = 0.382). However, in patients continuing insulin secretagogues, although lower BI doses were necessary, a higher hypoglycemia rate (40.0% vs. 24.5% with at least one symptomatic hypoglycemic event, P < 0.001) and an increased weight gain (+1.44 vs. +0.43 kg, P < 0.001) were found. Conversely, stopping these agents at initiation of basal plus therapy may result in a significant increase of insulin doses. 26 In any case, when increasing prandial insulin injections are needed, which may suggest a more important deterioration of β-cell function, it will be advisable to suspend treatment with sulfonylureas or glinides, and probably also with dipeptidyl peptidase-4 inhibitors. 42
Other aspects
In relation to how many and how often patients should perform SMBG when transferring to basal plus therapy, there are no well-established recommendations. At the start, to identify the main meal, it will be necessary to recollect at least two or three daily glycemic profiles, including preprandial and 2-h postprandial glucose values after breakfast, lunch, and dinner. These procedures should be repeated when increasing prandial insulin injections are needed in order to identify the next main meal.
Regarding the titration process, prandial insulin doses should be adjusted taking into consideration postprandial (preferable) and preprandial (before the next meal) glucose values. Therefore, in addition to monitor fasting blood glucose to adjust prospectively BI doses, daily monitoring of postprandial glucose values at least after the main meal is important initially. If RAIAs are used, this practice is even more important, and postprandial values remain the best in the dosing process. When glucose values are stable, how many times and how often SMBG should be performed by patients should be established based on an individual basis.
Instructing patients on carbohydrate counting may be advisable in some patients and can be very effective when used in the context of a structured insulin dosing algorithm, 22 but in general this is not necessary and should not be implemented in patients with T2DM transferred to the basal plus strategy.
Transition to Basal–Bolus Approach
As commented on before, in clinical practice, the basal plus strategy is acceptable for many patients. When β-cell dysfunction deteriorates further and more complicated insulin regimens are needed, this therapeutic alternative may facilitate moving from the previous strategy of BI combined with OAs, which is easier, to the basal–bolus therapy. 43 Although addition of a BI to OAs allows approximately 60–70% of insulin-naive patients to achieve the goal of A1C < 7%, 9,10,12 data from the United Kingdom Prospective Diabetes Study and Kumamoto studies clearly demonstrated that for most patients attainment of glycemic goals requires early intensification of insulin therapy with the addition of prandial insulin to basal. 4,5,44,45 This concept has been recently confirmed in the 3-year follow-up of the 4-T trial. 46 However, the choice between a basal–prandial and a premixed “biphasic” insulin regimen, as well as the use of analogs instead of human insulins, is a matter of debate.
Basal–bolus versus premixed insulins
When the intensification of hypoglycemic treatment is needed, the use of prandial premixed insulin is an effective option with a mean reduction in A1C of about 1.2–1.5% 23,47 and 45–50% of patients achieving an A1C < 7%. 48 In comparison, the use of a basal–bolus strategy (four or five injections per day) is associated with a slightly greater reduction in A1C (about 1.5–1.8%) and a greater proportion of patients achieving A1C < 7% (50–55%). 22,23,49,50 The high efficacy of basal–bolus therapy in T2DM has been proven in the study from Bergenstal et al. 22 Here, a population of insulin-treated (36% premixed, 37% glargine plus one or more shots of rapid-acting insulin, 27% various other regimens) obese T2DM patients was switched to basal–bolus and randomized to two different algorithms for prandial insulin titration. Independent of the mealtime insulin dose adjustment strategy, 70–75% of subjects reached the goal of A1C < 7%, with a mean reduction from baseline of about 1.5% in both groups. In the very few direct comparisons between the premixed and basal–bolus regimens, 23,50,51 the latter resulted in a greater proportion of patients in target (mean difference, 10–15%) and better postprandial blood glucose. 50,51 Also in the 4T trial, although not specifically designed to compare a flexible basal–bolus with premixed regimen over 3 years of follow-up, the superiority of basal–bolus was confirmed. 46 In contrast, a recently published substudy of the DURABLE trial 52 suggests no difference between the two insulin regimens. In this trial two different premixed regimens (Lispro Mix 50/50 three times a day or Lispro Mix 75/25 twice a day) achieved similar results as basal–bolus therapy. 52 However, glycemic control was poor, with <20% of patients reaching the goal of A1C < 7% regardless of treatment group. Therefore, non-inferiority of premixed insulin was probably the result of suboptimal treatment intensification.
Indeed, the basal–bolus approach is best suited to the “treat to target strategy,” allowing for independent titration of both basal and prandial insulin to address both pre- and postprandial hyperglycemia and more flexibility. In contrast, premixed insulins (which contain fixed proportions of rapid- and intermediate/long-acting insulin) are used preferentially for targeting preprandial hyperglycemia. 53 Of note is that when an intensive approach is used, better glycemic control is obtained with basal–bolus with a similar (or even lower) incidence of hypoglycemia in comparison to premixed regimens. 23,50,51 For these reasons, premixed insulins should be restricted only as nonintensive therapy with special caution in patients with long-standing T2DM, who are likely to be more insulin deficient and prone to hypoglycemia. 54 –56
Human insulins versus insulin analogs
Recently, it has been suggested that insulin analogs should not be used in place of human insulin because they do not improve glycemic control, providing only “minor” benefits in terms of reduction of hypoglycemia. 57 –59 This recommendation is based on the results from meta-analysis and systematic reviews showing no benefit of insulin analogs over human insulin, based on A1C reduction. However, measuring metabolic control with only A1C may be misleading. In fact, RAIAs have more favorable pharmacokinetics/pharmacodynamics profile resulting in appreciable advantages compared with regular human insulin, reflected by a higher reduction of postprandial hyperglycemia and of postabsorptive hypoglycemia. 40 In addition, long-acting insulin analogs show less variability and reduction of nocturnal hypoglycemia compared with NPH insulin. 60 Consequently, improvement in postprandial glycemic control along with the reduction of the incidence of hypoglycemia is likely to result in lower glycemic variability compared with human insulins. All these advantages might have significant impact on diabetes-related morbidity and mortality. Indeed, both acute and postprandial hyperglycemia 61 –65 and glucose variability 65 might theoretically contribute to the development/progression of micro- and macroangiopathic complications and incidence of cardiovascular events. In particular, a plethora of data from basic science 65 have shown that fluctuating glucose activates the pathways involved in the pathogenesis of diabetes complications. On the other hand, retrospective analysis of data from large clinical trials seems to rule out an independent role of glycemic variability 66 in clinical outcomes of people with diabetes. Unfortunately, this apparent contrast between basic science and clinical data will remain unsolved until intervention trials aimed at reducing glucose variability demonstrate (or exclude) a reduction in the risk of diabetes complications. However, while waiting for an answer that may never come, should we take care of glycemic variability? It is our opinion that yes, we should. Indeed, addressing variability probably allows for the reduction of the incidence of hypoglycemia, 67 the main complication of diabetes treatment.
Results from the ACCORD and VADT studies have recently emphasized the relationship between adverse outcomes in T2DM and hypoglycemia, especially severe hypoglycemia. 68,69 Although this issue is neglected by some authors, 70 others underlined how relevant this issue is in T2DM patients, 71 increasing in frequency with progression of the β-cell defect and impairment of hypoglycemia counterregulation. 72 In this context, the irrational use of insulin may lead to unnecessary increment in the rate of hypoglycemia with potentially fatal adverse events, especially in the frail elderly, who are rarely included in clinical trials. 55 Therefore, because of its better performance and safety profile compared with human insulin, the combined use of BI and RAIAs may contribute to improvement in the care of diabetes patients. However, long-term trials are needed to find out if the above-mentioned improvements in glucose control lead to improved clinical outcomes.
Conclusions
Progressive deterioration of β-cell function in people with T2DM is responsible for the initiation of insulin therapy when OAs are not enough to achieve or maintain individual glycemic goals. Addition of BI to previous OAs is the simplest way to start insulin therapy in these patients. If this approach is insufficient, adding prandial insulin is necessary over time. Recently, the basal plus therapy (i.e., a stepwise introduction of prandial insulin beginning with the main meal) has been accepted as an effective and safe alternative to premixed insulin. Additionally, this strategy will help patients if necessary in the transition to basal–bolus therapy, which remain the most effective and flexible insulin therapy even for people with T2DM.
Footnotes
Author Disclosure Statement
F.J.A.-B. has received honoraria as speaker and/or consultant from Abbott, AstraZeneca, Bristol-Myers Squibb, Glaxo-SmithKline, LifeScan, Lilly, Madaus, MannKind Corp., Medtronic, Menarini, Merch Farma y Química, SA, MSD, Novartis, Novo Nordisk, Pfizer, Roche, sanofi-aventis, Schering-Plough, and Solvay. In addition, F.J.A.-B. has participated in clinical trials supported totally or partially by AstraZeneca, Glaxo-SmithKline, LifeScan, Lilly, MSD, Novo Nordisk, Pfizer, sanofi-aventis, and Servier. P.R. has no potential conflicts of interest to declare. J.F.A. has received honoraria as speaker and/or consultant form AstraZeneca, Ferrer, Glaxo-SmithKline, Laboratorios Dr. Esteve, Lilly, MSD, and Solvay.
