Abstract
Several large studies in diabetes have shown that early initiation of intensive therapy is better for the prevention of long-term complications and suggest that patients with more advanced disease may be at increased risk of adverse cardiovascular events. Despite these findings, insulin initiation is often delayed in patients with type 2 diabetes, typically until A1C exceeds 8.5%. Barriers to the use of insulin are many, arising from both a patient and a physician perspective, and the decision to initiate insulin treatment can be influenced by cost, risk of hypoglycemia, convenience, and the potential for weight gain. Choosing when to initiate insulin and which insulin/treatment regimen to adopt in patients with type 2 diabetes is key, and the importance of tailoring treatment to the patient is widely acknowledged. However, there is currently no universal consensus on the optimal course of action. Once-daily basal insulin and twice-daily premix insulin are commonly used for insulin initiation. Relatively few studies have directly compared these starter treatment regimens, although general findings suggest that, although glycemic control appears to be similar with once-daily basal insulin and twice-daily premix, the lower hypoglycemia rates, lower weight gain, simplicity, and convenience associated with basal insulin support its first-line use as a starter insulin regimen in patients failing on oral antidiabetes agents. Variables such as age, body mass index, and bedtime or post-breakfast plasma glucose levels may alter the efficacy of the chosen treatment regimen, further supporting the need to tailor treatment to meet individual patient's requirements.
Introduction
Two commonly used insulin initiation regimens include the addition of once-daily basal insulin or twice-daily premix insulin. 4 –9 However, the benefits and drawbacks of each regimen are not clear. This article reviews the use of premix insulin versus basal insulin when starting patients with T2DM on insulin therapy. One key point to consider is that diabetes care must be individualized according to each patient's needs, without a particular rubber stamp made for all those with T2DM.
The Move Toward Primary Care: Assessing a Patient's Needs and Capabilities
It has been estimated that diabetes may affect as many as one in three Americans by the year 2050. 10 Fewer physicians are seeking training in endocrinology fellowships, resulting in a shift from specialized care to primary care. There are numerous and varied concerns with regard to this shift in practice. First, primary care physicians have less time to spend with their patients face-to-face; this is particularly problematic in view of the increasingly accepted need to tailor diabetes treatment to each individual patient. This process requires comprehensive knowledge of a patient's history and his or her adherence to therapy, as well as a full blood glucose profile. Once a patient's glycemic control is ascertained and the need for insulin therapy has been established, the patient's capabilities and needs must be assessed, as well as duration of the disease and any underlying complications. With all these factors assessed, the individual goals for the patient can be determined in accordance with existing recommendations (Table 1). 11
Adapted from Skyler et al. 11
It is widely understood that hypoglycemia is one of the greatest fears among physicians and patients alike and, together with a fear of weight gain, can adversely affect a patient's willingness to accept insulin therapy. Furthermore, patients may be unwilling and/or unable to inject insulin, often because of the perceived inconvenience and pain of injection. There is also a perception that starting insulin is complex and time consuming, and, for primary care physicians with limited time, this represents a challenge. There are many other factors that add to clinical inertia and as many processes and tools in place to reduce the impact of these factors (Table 2).
SMBG, self-monitoring of blood glucose.
When considering starting people with T2DM on insulin therapy, there are two primary questions to consider: (1) what is an appropriate time to initiate insulin and (2) what type of insulin(s) should be considered?
The Need for Tight Glycemic Control and the Importance of Early Intervention
The Diabetes Control and Complications Trial and the United Kingdom Prospective Diabetes Study (UKPDS) were landmark trials in demonstrating that intensive glycemic control could reduce the risk of microvascular complications compared with conventional therapy in patients with type 1 diabetes 12 and T2DM, 13,14 respectively. These studies are discussed in further detail by George Dailey in this supplement. 15 Follow-up data for both studies have demonstrated the persistence of microvascular benefits long after treatment cessation, a phenomenon known as the “legacy effect.”
Data from the randomized treatment period of the UKPDS showed that, in patients with newly diagnosed T2DM, intensive therapy with a regimen based on insulin or a sulfonylurea resulted in a 25% reduction in microvascular disease (P = 0.0099) compared with standard care. 14 The benefits of intensive therapy were even greater with a metformin-based regimen in overweight patients with newly diagnosed T2DM. 13,14 Following closure of the trial and the end of randomized treatment, the UKPDS population were followed up after 10 years. The groups that had originally received intensive treatment showed reductions in all diabetes-related end points, microvascular disease, myocardial infarction, and all-cause mortality compared with the conventional treatment group. These benefits were achieved despite the fact that the difference between the groups in A1C levels at the end of the UKPDS was similar for all of the follow-up period after 1 year. 16 These data suggest that early initiation of intensive therapy seems to confer a protective effect against micro- and macrovascular complications that persists even when tight glycemic control is subsequently lost.
More recently, the benefit of intensive glycemic control initiated later in the course of disease, and in higher-risk groups, has been evaluated in several large-scale, prospective trials. The Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial comprised over 10,000 patients with T2DM at high risk of cardiovascular (CV) disease (CVD) who received either intensive therapy aimed at achieving and maintaining A1C <6.0% or standard therapy targeting an A1C of 7.0–7.9%. 17 At the time of the interim analysis after a mean follow-up of 3.5 years, intensive therapy was not associated with a significant benefit over standard therapy in any efficacy end point, with the exception of a lower rate of nonfatal myocardial infarction (3.6% vs. 4.6%; P = 0.004). Surprisingly, significantly higher rates of death from any cause (5.0% vs. 4.0%; P < 0.05) and death from CVD (2.6% vs. 1.8%; P = 0.02) were observed in the intensive versus conventional treatment group. The Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) study likewise compared the impact of intensive glucose control with standard therapy in patients with relatively advanced disease and at high risk of CVD. 18 At 5 years of follow-up, the rate of new or worsening nephropathy was significantly lower with intensive versus standard therapy (4.1% vs. 5.2%; P = 0.006), but differences in the rates of all other single end points, including retinopathy, neuropathy, and macrovascular events, were not significantly different between the groups. In contrast to the ACCORD study, there was no increase in the rate of CV or all-cause mortality with intensive therapy in the ADVANCE study. One further study, the Veterans Affairs Diabetes Trial (VADT), randomized 1,791 military veterans with a mean duration of T2DM of 11.5 years to either an intensive or standard glycemic control regimen. 19 Despite the intervention arm achieving lower median A1C levels (6.9% vs. 8.5%), there were no significant differences between the two groups with regard to rates of macrovascular or microvascular events or of death from any cause.
Clearly, the benefits of glycemic control are less evident in trials of patients with more advanced disease versus early initiation in the UKPDS, although longer-term follow-up of the ACCORD, ADVANCE, and VADT studies may provide further information. The message to physicians and patients is that earlier initiation of intensive therapy is better for the prevention of long-term complications. Despite this evidence, however, initiation of insulin therapy is often delayed until the patient's A1C levels exceed 8.5%. 2 In view of the barriers to starting insulin therapy frequently encountered (Table 2), the appropriate choice of initial insulin regimen is imperative in achieving and maintaining optimal therapeutic success.
Starting Patients on Insulin Therapy: Should We Target Fasting or Postprandial Glucose Excursions?
There is no universal consensus for the optimal method of starting insulin therapy in patients with T2DM who are failing on OADs. Two commonly used insulin initiation regimens include the addition of once-daily basal or twice-daily premix insulin, containing both rapid-acting and basal components, to the OAD medications. 4 –9 The rationale for using premix insulin analogs, such as insulin lispro mix 75/25, in the management of T2DM is based on the need for both basal and postprandial glucose-lowering activity to mimic physiological insulin secretion. 20
In 2003, Monnier et al. 21 demonstrated that postprandial glycemic control accounts for approximately 70% of overall glycemic control in patients with A1C values <7.3% and for around 50% of overall glycemic control in patients with A1C values between 7.3 and 8.4%. However, in patients with A1C levels >10.2%, the situation is reversed such that fasting glucose levels account for the majority (∼70%) of overall glycemic control. It has, therefore, been argued that as A1C values approach target levels (6.5–7%), 22,23 it becomes increasingly important to address postprandial glucose control; conversely, in patients with poor glycemic control, fasting blood glucose should represent the primary target for therapy. There is also strong epidemiological evidence that postprandial plasma levels independently predict CV events, whereas evidence for the importance of fasting plasma glucose in this capacity is weaker. 24,25 It has, therefore, been argued that targeting postprandial glucose excursions is critical, not only to reduce microvascular complications, but also to limit the risk of CV complications and mortality in people with diabetes.
To address this hypothesis, the Hyperglycemia and Its Effect After Acute Myocardial Infarction on Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus (HEART2D), a multinational, randomized trial, compared the effects of prandial versus fasting glycemic control on risk for CV outcomes in patients with T2DM after acute myocardial infarction. 26 It is interesting that the findings revealed only modest differences in fasting and postprandial blood glucose levels between the two insulin regimens, with comparable A1C levels. In addition, no difference in risk for future CV event rates was observed between the two regimens, which may reflect the somewhat similar glycemic control. 26 In reality, the failure to reach the predetermined difference in postprandial hyperglycemia between the groups in this study and the low overall number of CV events limit our ability to draw meaningful conclusions, and additional studies are required to provide further insight.
The Treat-to-Target trial, using a once-daily basal insulin algorithm based on target fasting plasma glucose levels, clearly demonstrated that patients can achieve the recommended American Diabetes Association A1C target ≤7% within 24 weeks. 5 According to the findings reported by Monnier et al., 21 the addition of a fast-acting insulin component to a basal regimen may enable more patients to achieve the recommended A1C targets by additional control of postprandial glucose levels. More recent findings presented at the American Diabetes Association's annual meeting in 2010 by Riddle et al., 27 however, contradict the earlier proposal of Monnier et al. 21 for the varying contributions of postprandial and fasting glucose to the overall glycemic profile at different A1C levels. Instead, Riddle et al. 27 reported that basal, rather than postprandial, glucose levels were dominant in the overall glycemic profile in a large study of patients with T2DM and that this association was unaffected by A1C level. Thus, A1C might be further improved with the addition of basal insulin, even in those whose A1C approaches 7%.
In clinical practice, the choice of therapy will depend on the degree of failing glycemic control; as such, the patient's A1C value is a key criterion. If the patient is already on maximal oral medications but has an A1C of >8.5%, there is a lower probability of getting that patient back under good control without additional medication. Initial doses need to be calculated according to the degree of hyperglycemia, which will include fasting blood glucose, postprandial blood glucose, and A1C levels (Table 3). 28 –30 At this time, a very important discussion should ensue with the patient and caregivers (if available). The patient needs to be made aware of the different insulins available to him or her (Tables 4 and 5) 31 and their different action profiles (Fig. 1). 32 –36 Human insulin versus insulin analogs, cost implications, risk of hypoglycemia, pen availability, and potential weight changes must all be discussed with each individual patient.
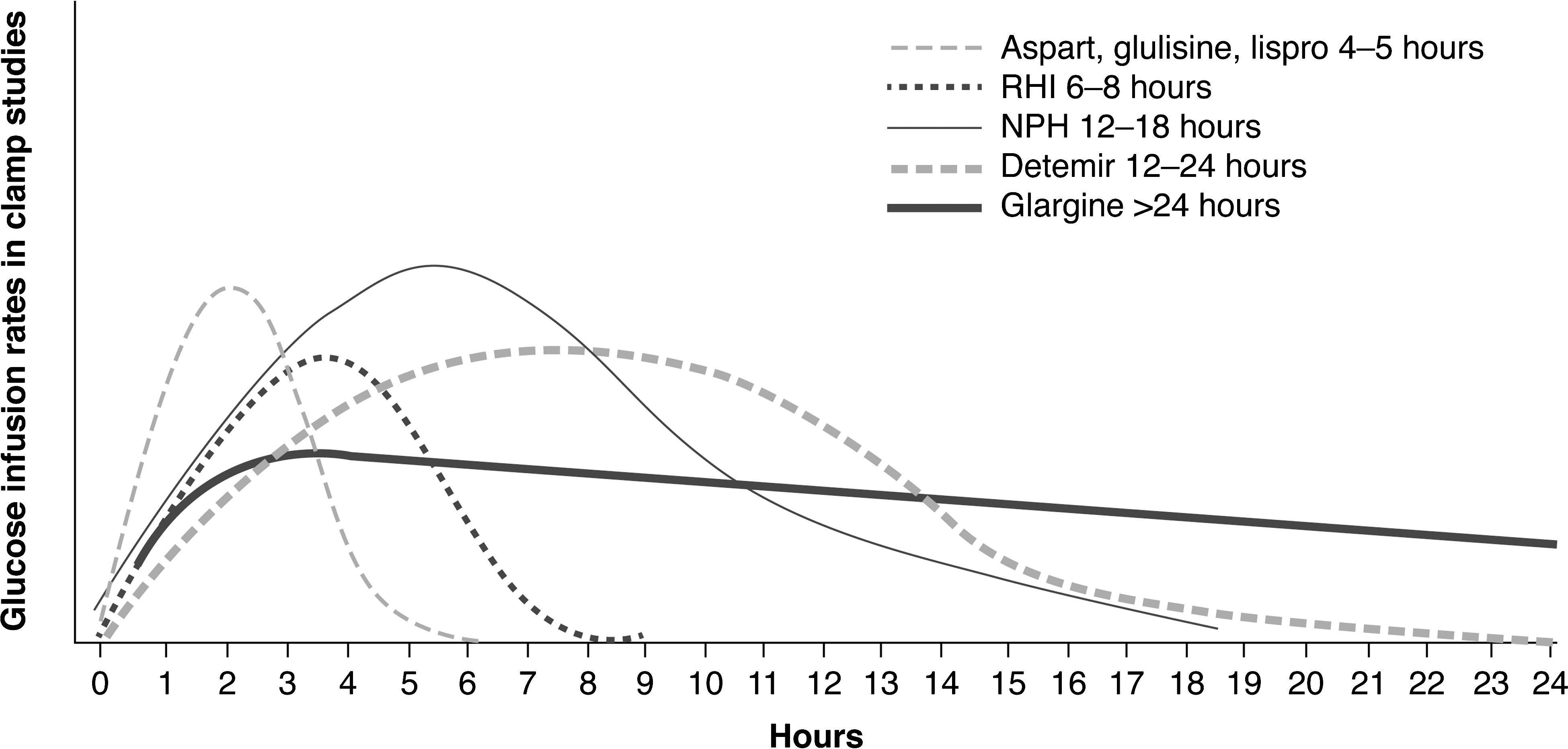
ADA, American Diabetes Association; ACE, American College of Endocrinology; IDF, International Diabetes Federation.
NPH, neutral protamine Hagedorn; RHI, regular human insulin.
Also known as bolus or premeal insulin; limits hyperglycemia after meals; administered one to three times per day; immediate rise and sharp peak at 1 h.
Also known as split-mixed or fixed-ratio regimen; contains both basal and rapid-acting insulin; administered twice daily; provides postprandial and fasting glucose control, but does not provide physiologic insulin replacement.
Clinical Evidence: Should We Start Our Patients on Premix Insulin or Basal Insulin?
There are few studies that have compared starter insulin regimens with OADs in individuals with T2DM. The outcomes of four relevant key clinical trials, either completed or ongoing, in insulin-naive patients with inadequate glycemic control despite OAD therapy are overviewed here.
The INITiate Insulin by Aggressive Titration and Education (INITIATE) study compared the safety and efficacy of biphasic insulin aspart (BIAsp) 70/30, pre-breakfast and pre-supper with once-daily bedtime insulin glargine, with continued OADs in 209 individuals with T2DM who were inadequately controlled on OADs. 9 After 28 weeks, the mean A1C value was lower in the BIAsp 70/30 group versus the insulin glargine group (6.9% vs. 7.4%; P < 0.01). The A1C reduction was greater in the BIAsp 70/30 group than in the insulin glargine group (−2.8% vs. 2.4%; P < 0.01), particularly in those with baseline A1C >8.5% (−3.1% vs. −2.6; P < 0.05), and more BIAsp 70/30- versus insulin glargine-treated subjects achieved target A1C values (A1C ≤6.5%, 42% vs. 28%, P < 0.05; A1C <7.0%, 66% vs. 40%, P < 0.001). However, the superior glycemic control observed with the premix insulin was achieved at the expense of increased hypoglycemic episodes (minor hypoglycemic episodes/year: BIAsp 70/30 vs. insulin glargine, 3.4 vs. 0.7; P < 0.05), weight gain (BIAsp 70/30 vs. insulin glargine, 5.4 vs. 3.5 kg; P < 0.01), and higher daily insulin doses (BIAsp 70/30 vs. insulin glargine, 78.5 vs. 51.3 units/day). Despite these disadvantages, the authors concluded that, given its superior efficacy, BIAsp 70/30 is a viable choice for starting insulin therapy in this group of T2DM patients, particularly those with higher baseline A1C levels (>8.5%). 9,37
The findings of a study published by Janka et al. 6 in the same year (Lantus + Amaryl + metformin versus premix insulin Patients with Type 2 diabetes mellitus after failing Oral treatment Pathways [LAPTOP]) refute those reported by Raskin et al. 9 Janka et al. 6 studied the efficacy and safety of adding once-daily (morning) basal insulin (insulin glargine) plus OADs versus switching to twice-daily premix insulin (30% regular/70% human neutral protamine Hagedorn [NPH] insulin) in 371 insulin-naive T2DM patients insufficiently controlled by OADs in a multinational clinical trial. In contrast to the INITIATE findings, after 24 weeks, the reduction in mean A1C levels was significantly greater with the basal versus premix insulin (Fig. 2), and more basal- versus premix-treated patients reached A1C ≤7.0% without confirmed nocturnal hypoglycemia (45.5% vs. 28.6%; P = 0.0013). As was reported by Raskin et al., 9 basal insulin therapy was associated with significantly fewer hypoglycemic episodes than in the 70/30 premix insulin patients (mean, 4.07 vs. 9.87/patient-year; Fig. 2). Mean weight gain in patients treated with insulin glargine was numerically, but not statistically, lower versus the premix regimen (1.4 vs. 2.1 kg). As observed in the INITIATE study, daily insulin doses were substantially higher in the premix versus the basal treatment groups (64.5 vs. 28.2 IU). In accordance with these findings, Janka et al. 6 concluded that initiating insulin treatment by adding basal insulin glargine once daily to OADs was safer and more effective than beginning twice-daily injections of premix 70/30 insulin and discontinuing OADs in this population of patients.
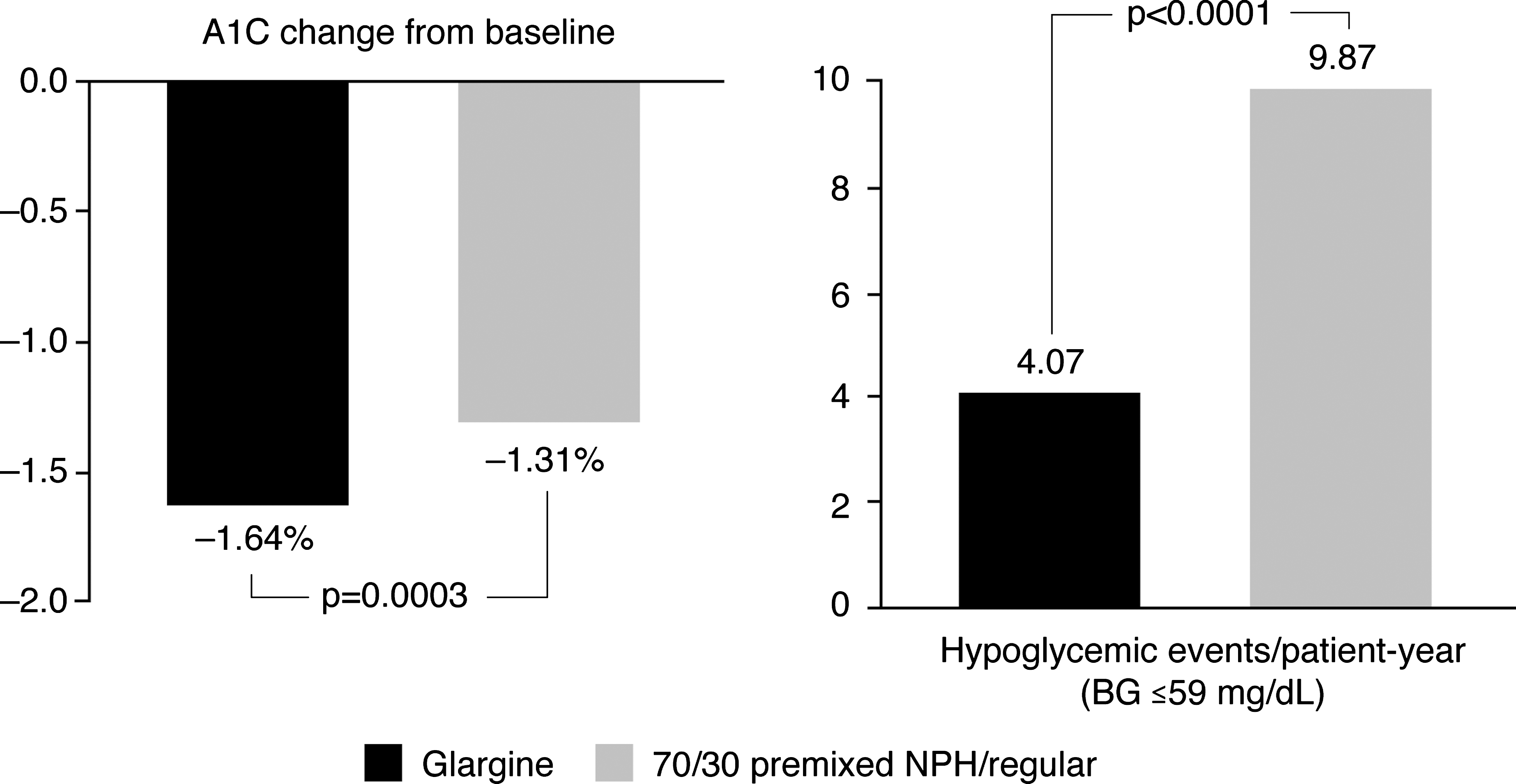
Combination therapy + glargine versus oral agent discontinuation + 70/30 premixed neutral protamine Hagedorn (NPH)/regular human insulin (RHI). 6
The Treating To Target in Type 2 diabetes (4-T) Study was an open-label, multicenter trial in over 700 patients with suboptimal A1C levels despite OAD therapy, which evaluated three different analog insulin regimens: BIAsp (premix) twice daily, prandial insulin aspart three times daily, or basal insulin detemir once daily (twice daily if required). 38 Although median A1C levels were similar for patients receiving premix (7.1%), prandial (6.8%), and basal (6.9%) insulin-based regimens (P = 0.28), fewer patients achieved A1C target ≤6.5% in the premix group (31.9%) than in the prandial group (44.7%, P = 0.006) or basal group (43.2%, P = 0.03). In addition, not only were median rates of hypoglycemia/patient-year lowest in the basal group (1.7) compared with the premix (3.0) and prandial (5.7) groups (P < 0.001 for overall comparison), mean weight gain was lowest in the basal (3.6 kg) versus the premix (5.7 kg) and prandial (6.4 kg) groups. Insulin doses were comparable between the basal and premix groups but higher in the prandial arm. The authors concluded that, on balance, patients who added a basal or prandial insulin-based regimen to oral therapy had better A1C control than patients who added a premix insulin-based regimen. 38 Because the basal insulin regimen was also associated with fewer hypoglycemic episodes and less weight gain, this treatment option may be the most appropriate choice in patients for whom risk of hypoglycemia or weight gain are a concern.
The ongoing DURABLE (assessing DURAbility of Basal versus Lispro mix 75/25 insulin Efficacy) study compares the efficacy, safety, and durability of two starter insulin regimens: twice-daily insulin lispro mixture 75/25 (LM75/25) (Humalog® [Eli Lilly, Indianapolis, IN] Mix 75/25: 75% insulin) with once-daily insulin glargine, with continued OADs, in a large, ethnically diverse population with T2DM. 20 At 24 weeks, compared with the basal insulin regimen, premix insulin therapy resulted in significantly lower A1C levels (7.2% vs. 7.3%; P = 0.005), greater reductions in A1C (−1.8% vs. −1.7%; P = 0.005), and a higher percentage of patients reaching A1C target <7.0% (47.5% vs. 40.3%; P < 0.001). However, as observed with the INITIATE study, the premix regimen was also associated with a higher insulin dose (0.47 vs. 0.40 units/kg/day; P < 0.001) and more weight gain (3.6 vs. 2.5 kg; P < 0.0001). Although the premix regimen was associated with a higher overall hypoglycemia rate versus the basal group (28.0 vs. 23.1 episodes/patient-year; P = 0.007), the rate of nocturnal hypoglycemia was lower in those receiving premix versus basal insulin (8.9 vs. 11.4 episodes/patient-year; P = 0.009), with low rates of severe hypoglycemia rates with both regimens. The authors concluded that both regimens led to clinically meaningful reductions in A1C and that physicians should interpret the findings within the context of their own treatment practices and preferences. 20 Again, the study findings highlight the need to balance the advantages and limitations of each regimen and choose the most appropriate starter insulin regimen according to the patient's needs.
A summary of the key findings of all four studies with regard to the main outcomes discussed here is provided in Table 6. A direct comparison between these four studies is beyond the scope of this review, and interpretation is limited by several factors, the most important of which is differences in study design. The regimens in each study were not identical: indeed, the study performed by Janka et al. 6 has been criticized because OADs were discontinued in the premix arm, which may have favored the basal insulin arm. Overall, the two regimens appear to have comparable efficacy, as acknowledged by others. 38,39 Furthermore, the results of a recent study suggest that in insulin-naive individuals with T2DM, who are starting insulin therapy, premix analogs may be a better choice to target A1C values in older individuals and those with higher bedtime plasma glucose. Basal insulin (insulin glargine, insulin detemir, or NPH insulin) may be more appropriate to target fasting plasma glucose in patients with lower body mass index and higher post-breakfast plasma glucose levels. 40 This finding further supports the need for tailoring therapy according to individual needs of the patients. In a substudy of AT.LANTUS (A Trial comparing Lantus Algorithms to achieve Normal blood glucose Targets in patients with Uncontrolled blood Sugar), patients who switched from premix insulin with or without OADs to once-daily insulin glargine experienced a significant improvement in A1C levels with a low incidence of severe hypoglycemia, suggesting that a basal insulin regimen may represent a more appropriate treatment option in those failing on premix insulin regimens. 41
A checkmark denotes the favored insulin regimen, and an equals sign denotes equivalence between regimens.
HbA1c, hemoglobin A1c.
If we accept that initiating insulin-naive patients on premix versus basal analog regimens has the same effect in terms of glycemic control, then the other two key factors to consider are the risk of hypoglycemia and weight gain. The fact that a basal insulin regimen is associated with fewer hypoglycemic episodes and less weight gain than the premix insulins (Table 6) is widely acknowledged. Other important factors that influence treatment management also need to be considered—namely, insulin dose, number of daily injections, complexity when monitoring blood glucose, incidence of hypoglycemia, weight gain, and quality of life. Whereas insulin glargine is injected only once daily, premix insulin requires twice-daily administration and blood glucose monitoring two to four times daily, a likely barrier to achieving treatment success, particularly in clinical practice with insulin-naive patients being initiated to insulin therapy. Aside from the debilitating effect of hypoglycemia on the patient and carers, hypoglycemia has important health economic implications, and cost-effectiveness plays an integral part of the decision-making process.
Conclusions
The similarities in glycemic control, lower hypoglycemia rates, less weight gain, simplicity and convenience observed with basal insulin versus premix insulin may support the use of basal insulin as first-line add-on therapy in those patients failing on OADs. Whichever regimen is chosen, an individually tailored regimen with minimal disruption to patient lifestyle will facilitate the transition from oral to insulin therapy.
Footnotes
Acknowledgments
F.L. is the Founder and Director of the North Broward Diabetes Center. The author would like to acknowledge Huw Jones, Ph.D., of Medicus International for his editorial support. The funding of editorial assistance was provided by sanofi-aventis.
Author Disclosure Statement
The author serves on the speaker's bureau for Abbott Pharmaceuticals.
