Abstract
Background:
A pooled analysis of randomized controlled trials of individuals with type 2 diabetes mellitus (T2DM) was conducted to compare dosing and impact of two basal insulin analogs, insulin glargine (glargine) and insulin detemir (detemir), on weight and hemoglobin A1c (A1c).
Methods:
Twenty-two studies of at least 20 weeks in duration in individuals with T2DM initiating glargine/detemir were included. Results were combined using a weighted-average method and a bivariate random effect model. Outcomes included changes in weight, A1c, and insulin dose from study start to end.
Results:
One study was head-to-head comparison of glargine and detemir. Detemir (four studies) was administered once or twice daily, with 50% starting on detemir once daily but needing therapy intensification. Glargine was used once daily in all 18 studies. The Egger test was borderline significant for change in weight over the course of the treatment for glargine (0.29; 90% confidence interval [CI] −0.01, 0.58), and heterogeneity was not observed for detemir (−0.18; 90% CI −0.59, 0.23). Heterogeneity was observed for change in A1c over the course of the treatment (glargine, −1.19, 90% CI −1.74, −0.63; detemir, −2.65, 90% CI −4.86, −0.45). Nonheterogeneity for change in A1c over the course of the treatment was achieved by excluding five studies for glargine and one study for detemir; however, all studies were included in subsequent analyses. In the unadjusted model, glargine and detemir showed similar results for mean A1c change (−1.4% vs. −1.4%), weight gain (2.5 vs. 1.7 kg), and weight/A1c (1.8 vs. 1.2 kg/%). A significantly higher detemir dose was needed to achieve the same A1c change (51.5 vs. 38.8 U/day).
Conclusions:
Although absolute weight gain was higher with glargine versus detemir, weight gain per A1c change was similar. A higher detemir dose was required to achieve a similar A1c reduction.
Introduction
The negative impact of weight gain on individuals with T2DM is both physiologically and psychologically. 4 –6 The fear of weight gain is recognized as a psychological barrier that can lead to a reluctance to initiate and titrate insulin appropriately, when oral antidiabetes agents (OADs) fail to maintain A1c targets below 7%. 7 –10 In addition, obesity predicts a poor response to any type of insulin therapy, especially if insufficient doses of insulin are used. 11,12 Managing weight gain is, therefore, an important consideration when initiating insulin therapy for the first time. Changes to lifestyle and diet can help to offset increases in weight due to treatment, and the concomitant use of a weight neutral treatment, such as metformin, can also be considered.
In the absence of an adequate and sustained response to lifestyle changes and OADs, basal insulin analogs, such as insulin glargine (glargine) and insulin detemir (detemir), offer a simple approach to the introduction of insulin. Once-daily dosing with glargine and once- or twice-daily dosing with detemir are associated with further improvement in glycemic control and a lower risk of hypoglycemia versus NPH insulin. 13,14 Some studies in type 1 diabetes mellitus and T2DM have suggested that treatment with detemir results in less weight gain compared with either glargine 2,15 or NPH. 14,16 –24 The differences, however, are relatively small in magnitude (<2 kg).
The aim of this review was to conduct a literature search to compare studies involving individuals with T2DM, with or without prior insulin use, and the impact of glargine and detemir treatment on weight gain in relationship to reductions in A1c. An analysis to determine the relationship between reductions in A1c and final insulin dose was also performed.
Subjects and Methods
Search terms
The aim of the literature search was to identify randomized controlled trials that evaluated glargine or detemir in persons with T2DM. A search of the MEDLINE database was conducted from January 1, 2000 to December 31, 2008 using the following terms: “glargine,” “insulin glargine,” “LANTUS®,” “HOE901,” “detemir,” “insulin detemir,” “levemir,” “NN304,” and “Type 2 diabetes.”
Study selection
Randomized, controlled trials were included if they had an active comparator, the treatment duration was at least 20 weeks, and reported clinical outcomes included A1c, change in weight or body mass index, insulin dose, and data on whether concomitant treatment with OADs was used. If numerous published articles existed on a trial, the article that included the most data was chosen; other related publications were omitted.
Outcomes
The outcomes analyzed included changes in weight, A1c, and insulin dose from start to end of study and relationship between reductions in A1c and changes in weight or insulin dose at the end of the study.
Statistical analysis
A statistical analysis was carried out involving all studies in order to assess the change in weight and final insulin dose, adjusted per 1% A1c reduction.
Importantly, testing for heterogeneity of articles, i.e., that the results of the studies were similar enough that a combined estimate would be a meaningful description, was performed in relationship to A1c and weight using an Egger test and inverted funnel plots.
A weighted average method was used to combine the results of the studies using a bivariate random effects model. The PROC NLMIXED (SAS version 9.2, SAS Institute, Cary, NC) statistical procedure was used. A first model was built to estimate change in A1c and weight, and a second model was constructed with the ratio between the change in weight and A1c to estimate the final insulin dose with the A1c reduction and the ratio between the final dose and the A1c change. This method allows the retention of the maximum amount of data for the analysis, i.e., the change in A1c when the changes of weight over the course of treatment are missing.
When SDs were unknown, the maximum SDs observed in other studies were substituted. This is a conservative method, in that as a study with unknown variance and a small number of subjects will have a small weighting in the analysis, with a bigger weight given to studies with a large number of subjects. This again ensures the maximum amount of data can be utilized for the analysis. For the weight analysis, body mass index was used instead for the baseline value because it was more systematically reported in studies.
A secondary analysis of individuals without history of insulin use was performed using the above methodology.
Results
Summary of analyzed studies
A total of 22 studies were identified using our search criteria: 18 glargine 2,25 –41 and three detemir 14,42,43 trials, with one study 2 involving both glargine and detemir data. Glargine was prescribed once daily in all 18 studies. Detemir was given twice daily in one study 14 and once daily in one study, 42 and individuals were started on once-daily dosing and progressed to twice-daily dosing as required in two studies. 2,43
Study designs and baseline characteristics of the individuals enrolled in the glargine and detemir trials are presented in Table 1. In total, 4,091 and 1,086 glargine- and detemir-treated individuals, respectively, were included in the analysis. Individuals were non–insulin-treated subjects except in one study, 41 where participants who were receiving a rapid-acting insulin analog plus NPH either continued their treatment or had the NPH replaced with glargine. The treatment period was 24 weeks or more in all but one study for detemir, 42 where the study duration was 20 weeks. Basal insulin was given in combination with OADs in all but one study with glargine. 32 Table 2 summarizes the change in A1c, weight, and insulin dose at the end of the study period for each of the glargine and detemir trials.
Data are mean (±SD) values, unless otherwise stated.
Insulin glargine was administered once daily in all studies.
FPG measured (no FBG data).
Data provided by study sponsor.
A1c, hemoglobin A1c; B, bedtime administration; BID, twice daily; BMI, body mass index; DET, insulin detemir; FBG, fasting blood glucose; FPG, fasting plasma glucose; GLAR, insulin glargine; GLAR30, insulin glargine with 30 μg/mL zinc; GLAR80, insulin glargine with 80 μg/mL zinc; GLIM, glimepiride; Grp, group education program; Ind, individual education program; M, morning administration; MET, metformin; OAD, oral antidiabetes agent; OD, once daily; PG, plasma glucose; PIO, pioglitazone; ROSI, rosiglitazone; SU, sulfonylurea; TZD, thiazolidinedione.
Data are mean (with SD if available), unless otherwise stated.
Insulin glargine was administered once daily.
SD calculated from the maximum of SDs observed in the other studies.
No SD reported.
SD calculated from the SE.
For subjects on once-daily insulin detemir, escalation to BID was allowed.
A1c, hemoglobin A1c; B, bedtime administration; BID, twice daily; DET, insulin detemir; Ind, individual education program; GLAR, insulin glargine; GLAR30, insulin glargine with 30 μg/mL zinc; GLAR80, insulin glargine with 80 μg/mL zinc; Grp, group education program; M, morning administration; OD, once daily.
Glycemic control
Baseline A1c ranged from 8.1% to 9.8% and from 8.6% to 9.1% in the glargine and detemir studies, respectively (Table 1). Change in A1c from baseline to end point ranged from −0.30 to −2.36% for the glargine studies and from −0.8 to −1.8% for the detemir studies (mean change, 1.4% for both insulins) (Table 2).
Change in weight
The baseline body mass index ranged from 24.8 to 34.6 kg/m2 in the glargine studies and 28.9 to 30.6 kg/m2 in the detemir studies (Table 1). The initiation of glargine or detemir was associated in all studies with increases in weight (Table 2). In individuals taking glargine, weight gain from baseline to the end of the study ranged from +1.4 to +3.9 kg compared with +0.7 to +3.0 kg in the detemir studies. Of note is that no difference in weight gain was observed between morning and evening glargine administration. 27,37 Weight gain was higher in individuals receiving detemir twice daily compared with those receiving once-daily detemir. In the single detemir study providing results stratified by both once- or twice-daily dosing, the insulin dose in the twice-daily group was almost double that in the once-daily group, with 55% of participants requiring twice-daily dosing. 2 However, the change in A1c in the twice-daily group was just 0.04% more than that in the once-daily group (A1c change, −1.58% vs. −1.52%), whereas weight gain was 61% higher in the twice-daily group (3.7 vs. 2.3 kg; Table 2). 2
Change in insulin dose
Insulin dose at end point ranged from 23 to 68 U in the glargine studies and from 37 to 71 U in the detemir studies (see Supplementary Appendix 1 at
Model Results: Weight Gain and Insulin Dose Adjusted for A1c Reduction
Of note is that 13, 2,14,25,28,29,31,33,35,36,38 –41 eight, 14,26,28,33,37,41,42 and five 2,29,31,32,43 studies had missing SDs for A1c, weight, and insulin dose, respectively, and therefore the maximum SDs from other equivalent studies included in the pooled analyses were used instead.
Results of the Egger test were borderline significant in terms of the change in weight over the course of the treatment for glargine (0.29; 90% confidence interval [CI] 0.01, 0.58). If five studies were excluded, 32,39 –41,44 the Egger test was no longer significant (0.03; 90% CI −0.34, 0.39). The Egger test was not significant in terms of the change in weight over the course of the treatment for detemir (−0.18; 90% CI −0.59, 0.23).
However, heterogeneity was observed for change in A1c over the course of the study for glargine (−1.19; 90% CI −1.74, −0.63) with the exclusion of two studies 32,41 and detemir (−2.65; 90% CI −4.86, −0.45). For the change in A1c analysis, involving the glargine studies, the absence of heterogeneity was obtained after excluding five studies 32,34,39 –41 (−0.18; 90% CI −0.85, 0.49). For the detemir studies, if the study by Holman et al. 43 was excluded, the Egger test was no longer significant (−0.68; 90% CI −5.86, 4.50).
For the purpose of this pooled analysis, as the Egger test demonstrated limited heterogeneity in weight gain among the studies, the absence of excessive variation between all identified studies validates the pooling of the data. This ensured that studies with a large number of patients would not be excluded and that the impact of covariates could be studied.
Change in weight per 1% A1c reduction
For the unadjusted model (inclusion of all studies and without adjustment for baseline covariates), the changes in A1c and weight were similar for glargine and detemir: −1.4 versus −1.4% (Fig. 1A) and +2.5 versus + 1.7 kg (Fig. 1B), respectively. There were no important differences seen when comparing these variables between the two treatment groups (see Supplementary Appendix 2). Similarly, there was no relevant difference in the A1c/weight ratio for the glargine versus detemir groups at 1.8 kg/% versus 1.2 kg/% (Fig. 1C), respectively.
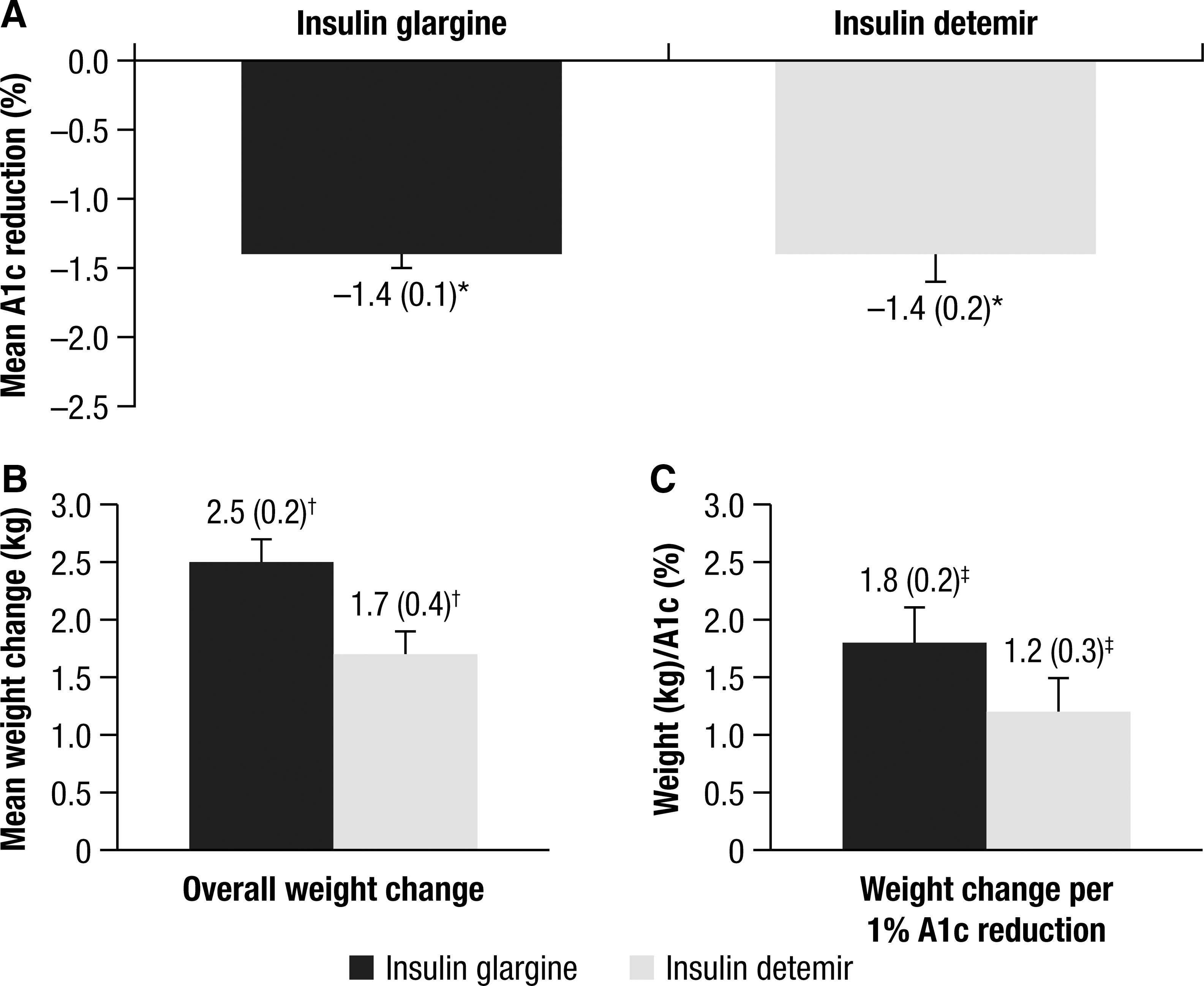
Changes in
Change in insulin dose per 1% A1c reduction
Using the unadjusted results, individuals required a higher detemir dose (36.1 U/day) compared with those receiving glargine (27.2 U/day) in order to decrease A1c by 1% (difference, −8.84; 95% CI, −17.7, 0.03 U/day [see Supplementary Appendix 2]).
In our pooled analysis, the final insulin doses were lower with glargine than with detemir (38.8 vs. 51.5 U/day; difference, −12.75; 95% CI, −25.72, −0.21 U/day). The final mean (±SE) insulin dose per kg for glargine and detemir was 0.45 (0.02) and 0.59 (0.07) U/kg, respectively.
Discussion
In the 22 studies identified for this review, the introduction of glargine or detemir to individuals with T2DM inadequately controlled by OADs was associated with a similar reduction in A1c levels of 1.4%. Both treatments were also associated with a modest increase in weight that was within the expected range (increase of 2 kg per 1% reduction in A1c). 45 Furthermore, our analysis shows that weight change with glargine therapy is borderline statistically significantly greater than that with detemir (P = 0.049). There was no difference between the treatments per 1% reduction in A1c levels. A previous meta-analysis 15 has reported significantly less weight gain with detemir than glargine, where the analysis was restricted to only one study of detemir and four glargine trials, all of which are included in the present analysis. 15
Our analysis demonstrates that individuals treated with detemir require a higher dose of insulin compared with individuals treated with glargine in order to achieve the same improvement in glycemic control. In the study by Rosenstock et al., 2 55% of the individuals required twice-daily dosing with detemir by the end of the 52-week treatment period. Twice-daily dosing was associated with a higher overall daily insulin dose and greater weight gain compared with once-daily dosing. Johnson and Shimshi, 46 in their recent meta-analysis of seven randomized clinical trials comparing detemir with other basal insulins, demonstrated that the mean total daily insulin dose was greater for detemir than other basal insulins.
When the analysis was conducted using only those studies that included individuals without a history of previous insulin use (the majority of the studies), the results were very similar, except that the limit of the 95% CI of weight gain difference was borderline in favor of detemir. The final insulin dose for detemir was higher (i.e., twice the dose for glargine) in order to achieve the same levels of glycemic control with glargine.
Caution should be taken, however, when interpreting results from the current analysis, as direct comparison is limited owing to the small number of detemir studies. However, the results are in agreement with other data indicating that weight change with detemir is dose-dependent, 2 as it is for other insulins. Moreover, in order to help reach glycemic targets, many individuals need to inject detemir twice daily, thus necessitating an increase in the frequency of administration and dosage. 7,8
The direct comparison of glargine and detemir data also supposed that the estimators obtained for each product are close to the data that would be obtained in real life conditions. It was not possible to adjust on a potential effect that could interfere with this difference estimation, as only a meta-analysis of only randomized head-to-head studies could address this issue.
The low numbers of detemir studies compared with the glargine studies did not allow testing for covariates such as hypoglycemia and fasting blood glucose. There is also only one study directly comparing detemir and glargine 2 and no studies comparing once- versus twice-daily regimens of either insulin. However, the results from our analyses are reassuring in that the weight gain was within the expected range per 1% reduction in A1c for both insulin preparations. Our pooled analysis indicates that weight changes, when adjusted for 1% reduction in A1c, are consistent with the 4T Trial; in this study, basal insulin (detemir), not sufficient for all individuals in terms of A1c change, was associated with significantly less weight gain than the other two regimens that also contained rapid-acting insulin. 43
This analysis did not adjust for baseline covariates, as this was only possible for the glargine studies, with the small number of studies with detemir precluding adjustment. Therefore, to provide a similar assessment, we have only presented unadjusted data here. However, even with adjustment for baseline covariates for the glargine studies, the pattern of results did not differ. In addition, patient-level data were not available for the studies used in the analysis, and therefore the analysis was carried out with the data as reported in the published studies.
Hypoglycemia is often a limiting factor of insulin therapy and may limit the magnitude of A1c change 13 ; similarly, the presence of other adverse events may result in inadequate dose titration and limit A1c change. However, in this analysis, we did not adjust for the effect of rates of hypoglycemia on an individual patient basis (only proportion of patients experiencing hypoglycemia was included in the model) or examine the association between hypoglycemia and change in weight or A1c over the course of the treatment. Although these were considered, the methods of reporting the incidence of hypoglycemia on an individual basis varied greatly between trials, and a meaningful analysis of the effect of hypoglycemia on the change in weight over the course of the treatment was not possible.
Nevertheless, concerns regarding changes in weight are still a major barrier for both individuals with diabetes starting insulin therapy and their caregivers, leading to reduced adherence to insulin regimens and inadequate titration. 7,9 Implementation of effective education programs, including diet and exercise management and self-monitoring of blood glucose, in addition to pharmacotherapy for individuals with T2DM is a vital element of addressing this barrier.
Conclusions
The evidence from the individual studies included in this review and the pooled analysis presented confirm that the use of glargine once daily or detemir once or twice daily as required was associated with relatively low weight gain. Our pooled analysis suggests that although the absolute weight gain was numerically higher with glargine versus detemir, the weight gain with glargine does not differ from detemir when adjusted for the reduction in A1c. Individuals treated with detemir, however, required a higher daily dose of insulin compared with the individuals treated with glargine in order to achieve the same A1c reduction. Therefore, at equipotent dose, the weight gain was not significantly different between the two basal insulin preparations glargine and detemir.
Footnotes
Acknowledgments
Editorial support for this review was provided by the Global Publications Group of sanofi-aventis.
Author Disclosure Statement
G.D. has received research grants and speaker's honoraria and served as an occasional consultant for sanofi-aventis. K.A. is an employee of sanofi-aventis. F.M. has received honoraria for statistical analyses conducted for sanofi-aventis. D.O. has received research support and honoraria for consultancy and lecture fees from and has served on the clinical advisory board and as a consultant for sanofi-aventis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
