Abstract
Abstract:
Capsule Summary
Crisaborole is approved to treat mild-to-moderate atopic dermatitis (AD) in multiple countries. In the CORE studies, crisaborole was well tolerated, and a significantly greater percentage of crisaborole- versus vehicle-treated patients achieved improved global disease severity.
In this post hoc analysis, crisaborole treatment demonstrated greater efficacy versus vehicle and had no notable adverse events (AEs) in patients who were previously treated with systemic or topical corticosteroids or topical calcineurin inhibitors.
Crisaborole is effective and well tolerated in patients regardless of prior treatment status. This may affect therapy selection in AD, especially when attempting to avoid the occurrence of AEs.
INTRODUCTION
Atopic dermatitis (AD) is a complex chronic immune-mediated inflammatory skin disorder with a relapsing-remitting course.1–5 Both the innate and adaptive immunity are thought to contribute equally to the pathogenesis of AD, with various immune mediators (including Th-2 cytokines interleukin [IL]-4, IL-13, and IL-31) and regulators (including phosphodiesterase-4 [PDE4] and the Janus kinase-signal transducer and activator of transcription signaling pathway) playing a role in the induction of inflammation and pruritus.3,5–7 Characterized by eczematous lesions, oozing, crusting, lichenification, pruritus, and pain, AD has a significant impact on the quality of life (QoL) of patients.3,6,8–12
The goals of treatment in patients with AD are to minimize inflammation and pruritus, restore skin barrier function, and improve QoL. 13 Standard-of-care first-line treatment often involves the use of appropriate skin care and topical anti-inflammatory agents.6,8,14
Topical corticosteroids (TCSs) are traditionally considered the mainstay anti-inflammatory agents against which other agents are compared in AD. 8 However, long-term continuous TCS use is restricted to avoid local cutaneous atrophy and tachyphylaxis.8,14–16
Topical calcineurin inhibitors (TCIs) are approved for short-term and noncontinuous treatment in recalcitrant AD. 8 Because they do not cause cutaneous atrophy, TCIs are used when TCS use is problematic and when thinned-skin areas such as the face and intertriginous regions are affected. 14 Patient education is required due to a boxed warning for rare cases of malignancy (eg, lymphoma), although the risk is minimal with use in a circumscribed body surface area. 8
Emerging treatment options such as JAK inhibitors (including ruxolitinib) and biological drugs (including dupilumab [an IL-4 receptor alpha antagonist]) present alternative treatment options.7,17 Ruxolitinib has been shown to successfully reduce AD-related disease severity and pruritus. Treatment-emergent adverse events (TEAEs), including application site pain, were infrequent.18–20 However, long-term efficacy and safety studies with JAK inhibitors are limited. 7 Dupilumab has demonstrated improved AD signs and symptoms, with a favorable benefit–risk profile; however, this injectable treatment option may not be as convenient as oral or topical agents. 17
Crisaborole ointment, 2%, is a nonsteroidal PDE4 inhibitor approved for the treatment of mild-to-moderate AD in adult and pediatric patients ≥3 months of age in several countries, including Canada and the United States. In some countries, including Australia, crisaborole has only been approved for use in patients ≥2 years of age.21–23 Crisaborole has a favorable safety profile. It has no boxed warning and no limitations on duration of use; it does not cause skin atrophy and can be used on sensitive skin. 6
The efficacy and safety of crisaborole have been assessed in the CORE 1 (NCT02118766) and CORE 2 (NCT02118792) studies, which comprised patients aged ≥2 years with mild-to-moderate AD. In both studies, a significantly higher percentage of patients treated with crisaborole versus vehicle achieved Investigator's Static Global Assessment (ISGA) success (CORE 1, P = 0.038; CORE 2, P < 0.001). Crisaborole was well tolerated, and the incidence rate of treatment-related adverse events (TRAEs) was comparable with that of vehicle (pooled data from CORE 1 and CORE 2: 21.4% vs 15.8%). 1 The only TRAE that occurred in ≥1% of crisaborole-treated patients during both studies was application site pain.1,23
Crisaborole may be especially useful for situations in which AD is recalcitrant to or when patients are unable to use TCSs or TCIs. To minimize TCS-related adverse events (AEs), crisaborole may be used as monotherapy or as part of an alternating regimen with a TCS. In addition, current guidance documents and available data support crisaborole as a first-line agent in patients with mild-to-moderate AD.6,14
To evaluate the efficacy and safety of crisaborole when used as first-line treatment or as second-line treatment after prior treatment, we conducted a post hoc analysis of the key pivotal studies of crisaborole in patients with AD to assess its efficacy and safety in patients who had received prior treatment with (a) corticosteroids (systemic or topical) or TCIs or (b) TCSs or TCIs or (c) who were treatment-naive (TN). This analysis could assist in personalizing the sequencing of AD treatment options in the future.
METHODS
Study Design and Patient Population
This is a pooled post hoc analysis of 2 identically designed multicenter randomized double-blind pivotal phase 3 trials (CORE 1 and CORE 2) in patients aged ≥2 years at time of informed consent/assent who had a clinical diagnosis of AD per Hanifin and Rajka criteria. 24 Details of the study design have been previously published. 1 In brief, patients who were included had mild-to-moderate AD defined as a baseline ISGA score of 2 (mild) or 3 (moderate) and involving a percentage of treatable body surface area (%BSA; excluding the scalp) of ≥5 and were randomly assigned to receive crisaborole or vehicle (2:1, respectively) twice daily (BID) for 28 days. 1
In this analysis, patients in CORE 1 and CORE 2 were divided into 3 subgroups: treatment-experienced (TE) patients who had received prior treatment with corticosteroids (systemic or TCSs; TE-CS) or TCIs (TE-CS/TCI), TE patients who had received prior treatment with TCSs or TCIs (TE-TCS/TCI), and TN patients who had received no prior treatments within the 90 days before screening. Patients treated with other types of dermatological medications, such as biologic therapy, were excluded from this analysis.
In CORE 1 and CORE 2, patients were instructed to apply a layer of the study drug ointment (crisaborole or vehicle) to cover each lesion BID during the 28-day study. This included all treatable areas affected by AD (excluding the scalp) at baseline/day 1 and newly identified AD lesions that appeared after day 1. In both studies, the patients were allowed to use bland emollients in areas around (but not overlapping) treatable AD lesions. 1
Assessments
Efficacy assessments
ISGA was used to assess global disease severity during study visits at baseline and on days 8, 15, 22, and 29. The primary efficacy endpoint was ISGA success, defined as an ISGA score of 0 (clear) or 1 (almost clear) with a ≥ 2-grade improvement from baseline.1,25
Severity of pruritus was measured BID through an electronic diary with the Severity of Pruritus Scale (SPS), a 4-point rating scale, which measures the severity of pruritus for a 24-hour period and in which a score of 0 indicates none (no itching) and 3 indicates severe (bothersome itching/scratching that disturbs sleep). 26 The severity of pruritus at baseline was calculated as the average of ≥2 assessments on day 1. Weekly SPS scores for each patient were calculated as the average of all post-baseline SPS scores during the respective week. The additional efficacy endpoint used was improvement in pruritus, defined as achieving a weekly average SPS score of ≤1 with a ≥ 1-grade improvement from baseline at week 4. 1
Post hoc efficacy assessment
The Atopic Dermatitis Severity Index (ADSI) total score was used as a post hoc efficacy endpoint to assess disease severity. The ADSI score is the sum of 5 component subscores of erythema, excoriation, exudation, lichenification, and pruritus. 27
QoL measures
Patient QoL was measured at baseline and day 29 using the Dermatology Life Quality Index (DLQI) in patients aged ≥16 years or the Children's Dermatology Life Quality Index (CDLQI) in patients aged 2–15 years. The least squares mean (LSM) change from baseline in DLQI or CDLQI score at day 29 was calculated to assess the improvement in patient QoL.
The Dermatitis Family Impact (DFI) questionnaire was completed at baseline and at day 29. The LSM change from baseline in DFI score at day 29 was calculated to assess the improvement in QoL of the parent/guardians of a child with AD. 28
Safety assessments
Frequencies of AEs were evaluated at baseline, during scheduled and unscheduled investigator visits, and at study conclusion for the pooled CORE 1 and CORE 2 studies. TEAEs were defined as AEs with an onset on or after the day of the first dose of study drug. TEAEs were classified as treatment related if they were determined by the study investigator to be definitely, probably, or possibly related to treatment with crisaborole.
Statistical Analyses
Efficacy analyses were performed on the intention-to-treat population, which included all patients who were dispensed the study drug or vehicle, regardless of discontinuation. Statistical significance was a nominal 0.05 level. 1 The percentages of patients achieving success in ISGA at day 29 were compared between the crisaborole-treated arm and the vehicle-treated arm for all subgroups. The differences were tested based on normal approximation to response rates. The percentages of patients achieving improvement in pruritus at week 4 (weekly average) were compared between the crisaborole and the vehicle arm for all subgroups. The differences were tested based on normal approximation to response rates.
Changes from baseline DLQI, CDLQI, and DFI scores at day 29 were analyzed using a mixed-effects model with fixed effects for the treatment and baseline values. Change from baseline in ADSI score at each timepoint was analyzed using a mixed-effects model with fixed effects of treatment, visit, treatment-by-visit interaction, and baseline value.
For all studies, the safety populations contained all randomly assigned patients who received ≥1 dose of study drug and had ≥1 post-baseline assessment. Frequencies of patients with TEAEs, serious TEAEs, and TRAEs were summarized and tabulated (Table 1).
Summary of Treatment-Emergent Adverse Events by System Organ Class and Preferred Term (All Causalities)
AE, adverse event; BID, twice daily; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid; TN, treatment-naive.
Ethical Approval
This was a post hoc analysis and was exempt from institutional review board approval. All patients or parent(s)/guardian(s) provided written informed consent for participation in the studies. The studies were approved by the Quorum Review Institutional Review Board and were conducted in accordance with the ethical principles originating in the Declaration of Helsinki.
RESULTS
Patient Population at Baseline
The intention-to-treat population of the CORE 1 and CORE 2 studies consisted of 1522 patients, of whom 1016 patients were randomly assigned to receive crisaborole and 506 patients were randomly assigned to receive vehicle. 1 Baseline and demographic characteristics of the 3 subgroups are shown in Table 2.
Key Demographic and Baseline Characteristics for Core 1 and Core 2 (Pooled) by Previous Treatment
%BSA, percentage of treatable body surface area; BID, twice daily; ISGA, Investigator's Static Global Assessment; max, maximum; min, minimum; SD, standard deviation; SPS, Severity of Pruritus Scale; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid; TN, treatment-naive.
The mean age of patients was similar between treatment groups across all subgroups, ranging from 10.1 to 13.8 years. Most patients (61.9%) were aged 2–11 years (Table 2).
The percentage of patients with mild or moderate AD was similar at baseline across the 3 subgroups, ranging between 31.2% and 43.8% for mild AD (ISGA score of 2) and between 56.2% and 68.8% for moderate AD (ISGA score of 3). The percentage of patients who had mild AD in the TN subgroup (43.3%–43.8%) was greater than the percentage of patients with mild AD in the TE subgroups (31.2%–32.1%). Inversely, the percentage of patients with moderate AD in the TE subgroups (67.9%–68.8%) was greater than the percentage with moderate AD in the TN subgroup (56.2%–56.7%) (Table 2).
The SPS scoring across the subgroups was similar at baseline. The majority (61.9%) of patients had a baseline SPS score of 2 (moderate) to 3 (severe). The percentage of patients who had an SPS score of 1 (mild) in the TN subgroup was greater than that in the TE subgroups. Inversely, the percentage of patients who had an SPS score of 2 (moderate) in the TE subgroups was slightly higher in the TN subgroup (Table 2).
Efficacy Endpoints
ISGA success at day 29
A significantly higher percentage of crisaborole-treated patients versus vehicle-treated patients achieved ISGA success in the TE-CS/TCI subgroup (27.9% vs 15.9%; P = 0.001), the TE-TCS/TCI subgroup (27.4% vs 14.7%; P = 0.0008), and the TN subgroup (35.0% vs 26.8%; P = 0.0166) at day 29
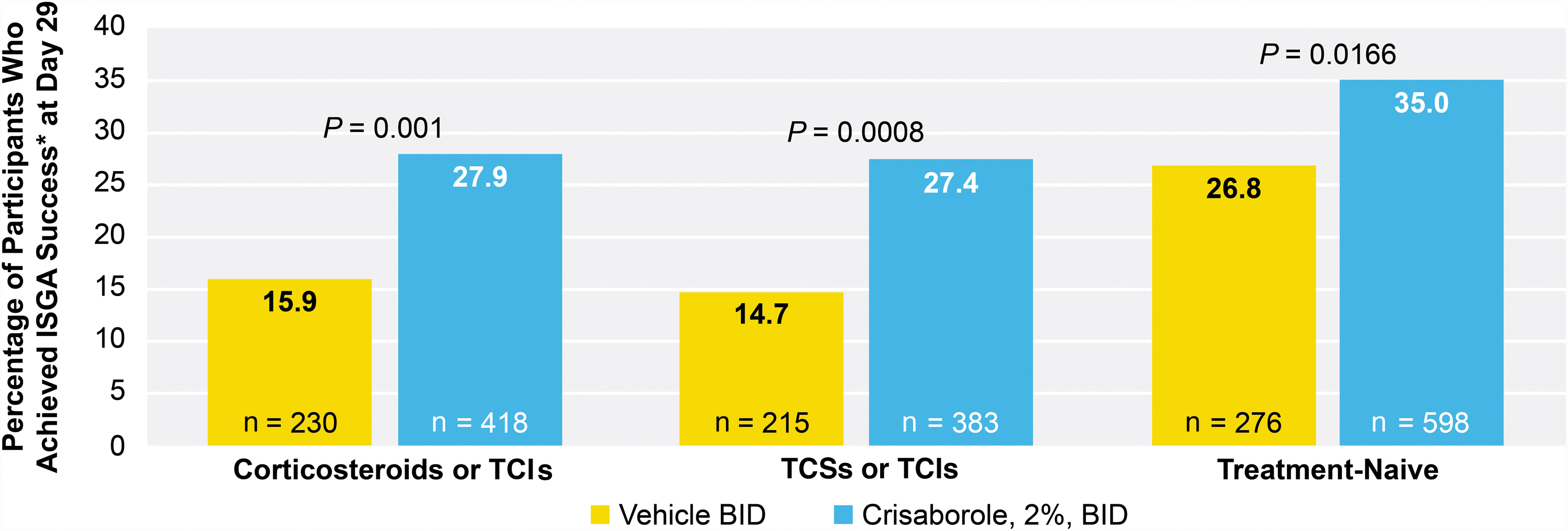
ISGA success at day 29. *ISGA success was defined as an ISGA score of 0 or 1 with a ≥ 2-grade improvement from baseline. BID, twice daily; ISGA, Investigator's Static Global Assessment; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid.
Improvement in pruritus at week 4
A significantly higher percentage of crisaborole-treated patients versus vehicle-treated patients achieved improvement in pruritus in the TE-CS/TCI subgroup (35.1% vs 14.9%; P < 0.0001), the TE-TCS/TCI subgroup (34.5% vs 13.5%; P < 0.0001), and the TN subgroup (36.3% vs 26.0%; P = 0.0099) at week 4
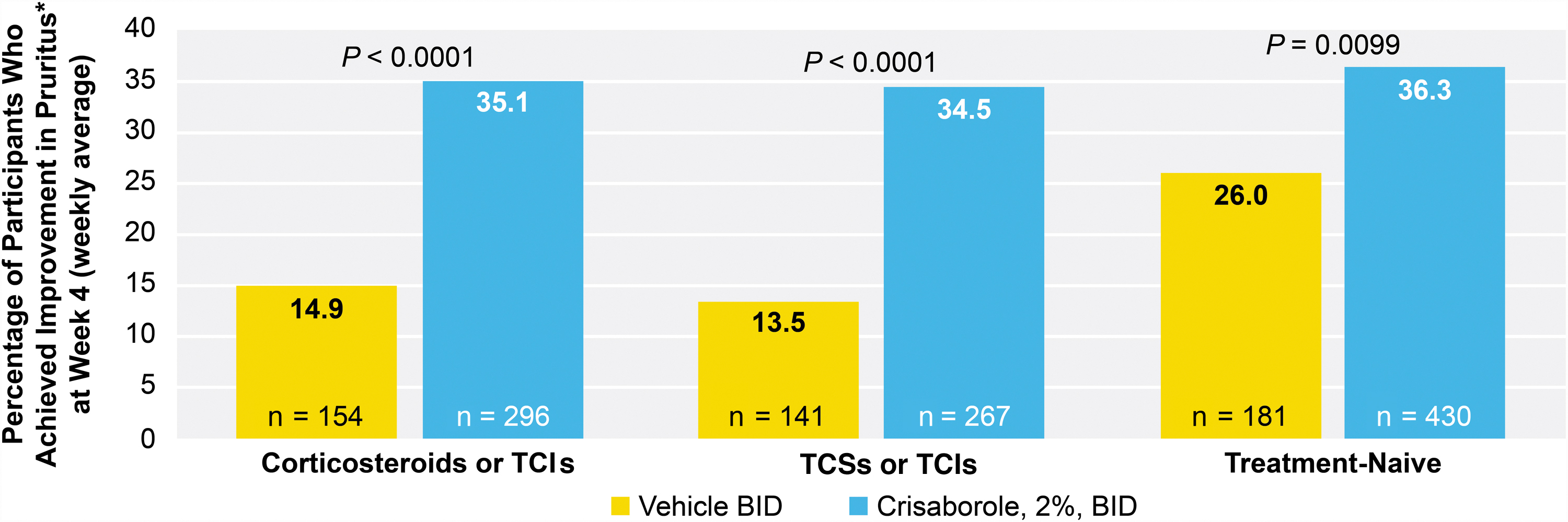
Improvement in pruritus at week 4 (weekly average). *Improvement in pruritus was defined as achieving a weekly average SPS score of ≤1 with a ≥ 1-grade improvement from baseline. BID, twice daily; SPS, Severity of Pruritus Scale; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid.
Change from baseline in ADSI at day 29
A significantly greater reduction in the change from baseline in ADSI was noted in crisaborole-treated patients versus vehicle-treated patients in the TE-CS/TCI subgroup (−3.41 vs −2.18; P < 0.0001), the TE-TCS/TCI subgroup (−3.35 vs −2.09; P < 0.0001), and TN subgroup (−3.37 vs −2.50; P < 0.0001) at day 29 (Fig. 3).
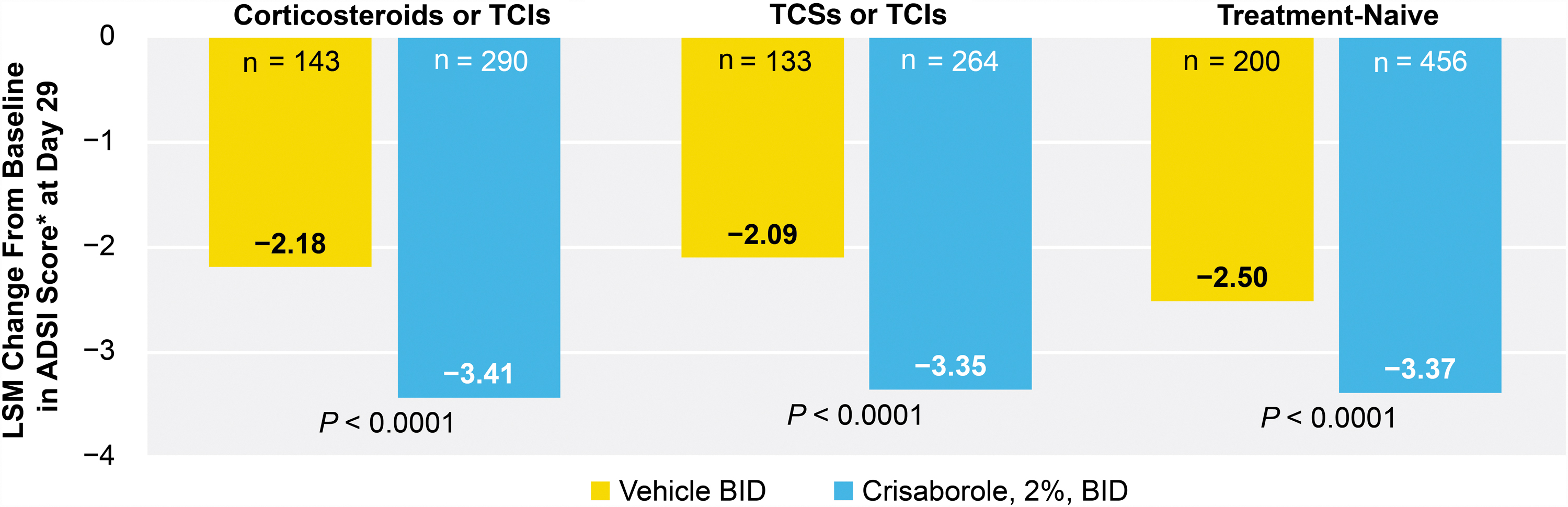
LSM of change from baseline in ADSI score at day 29. *The ADSI score is the sum of 5 component subscores of erythema, excoriation, exudation, lichenification, and pruritus. ADSI, Atopic Dermatitis Severity Index; BID, twice daily; LSM, least squares mean; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid.
Change from baseline in DLQI at day 29
A significantly greater reduction in the change from baseline in DLQI was observed in crisaborole-treated patients versus vehicle-treated patients in the TE-CS/TCI subgroup (−5.81 vs −2.71; P = 0.0006) and the TE-TCS/TCI subgroup (−5.91 vs −2.80; P = 0.0011) at day 29. The change from baseline in DLQI was not significantly reduced in crisaborole-treated patients versus vehicle-treated patients in the TN subgroup (−4.87 vs −4.01; P = 0.1872) at day 29 (Fig. 4).
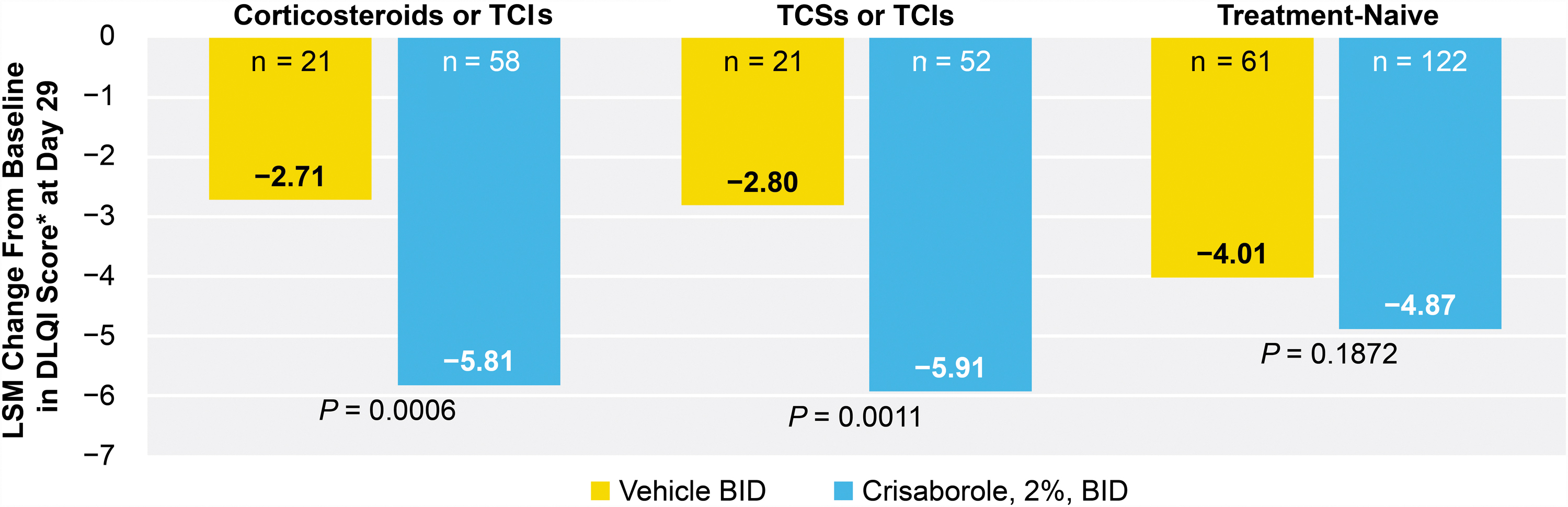
LSM of change from baseline in DLQI score at day 29. *Patients were ≥16 years of age at baseline. BID, twice daily; DLQI, Dermatology Life Quality Index; LSM, least squares mean; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid.
Change from baseline in CDLQI at day 29
A significantly greater reduction in the change from baseline in CDLQI was demonstrated in crisaborole-treated patients versus vehicle-treated patients in the TE-CS/TCI subgroup (−4.31 vs −2.45; P = 0.0001), the TE-TCS/TCI subgroup (−4.35 vs −2.63; P = 0.0001), and the TN subgroup (−4.66 vs −3.84; P = 0.0118) at day 29 (Fig. 5).
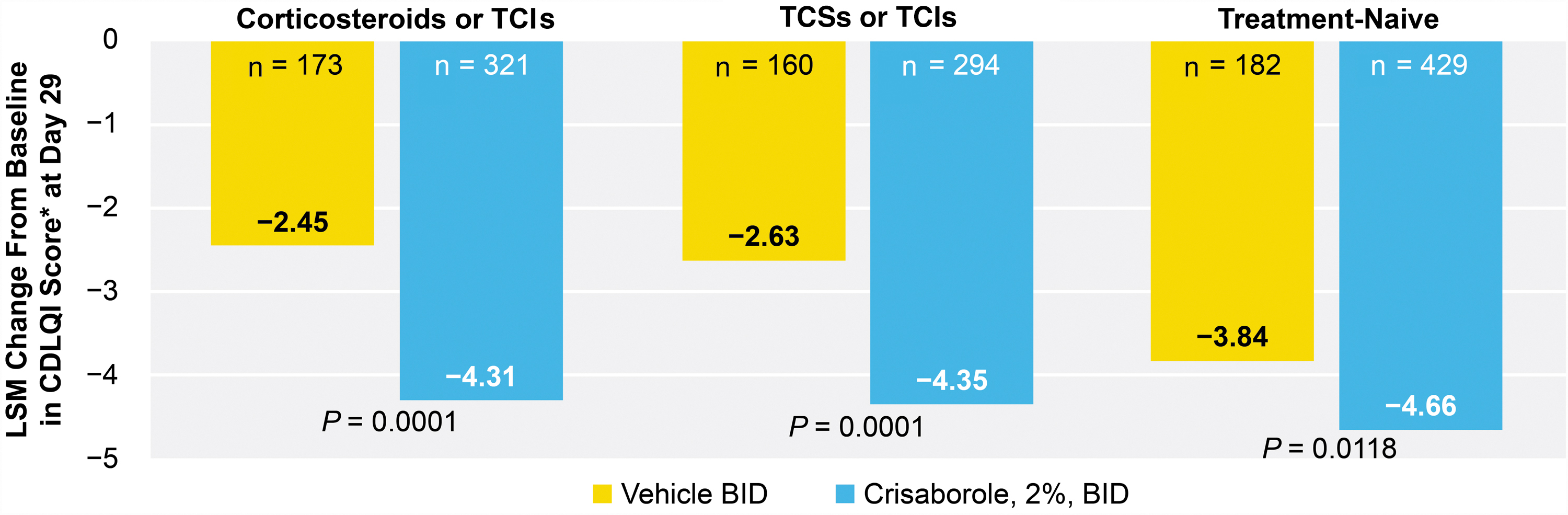
LSM of change from baseline in CDLQI score at day 29. *Patients were 2–15 years of age at baseline. BID, twice daily; CDLQI, Children's Dermatology Life Quality Index; LSM, least squares mean; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid.
Change from baseline in DFI at day 29
There was a significantly greater reduction from baseline in DFI in crisaborole-treated patients versus vehicle-treated patients in the TE-CS/TCI subgroup (−3.69 vs −1.83; P < 0.0001) and the TE-TCS/TCI subgroup (−3.74 vs −2.04; P = 0.0002) at day 29. The change from baseline on the DFI was not significantly reduced in crisaborole-treated patients versus vehicle-treated patients in the TN subgroup (−3.64 vs −3.85; P = 0.4952) at day 29 (Fig. 6).
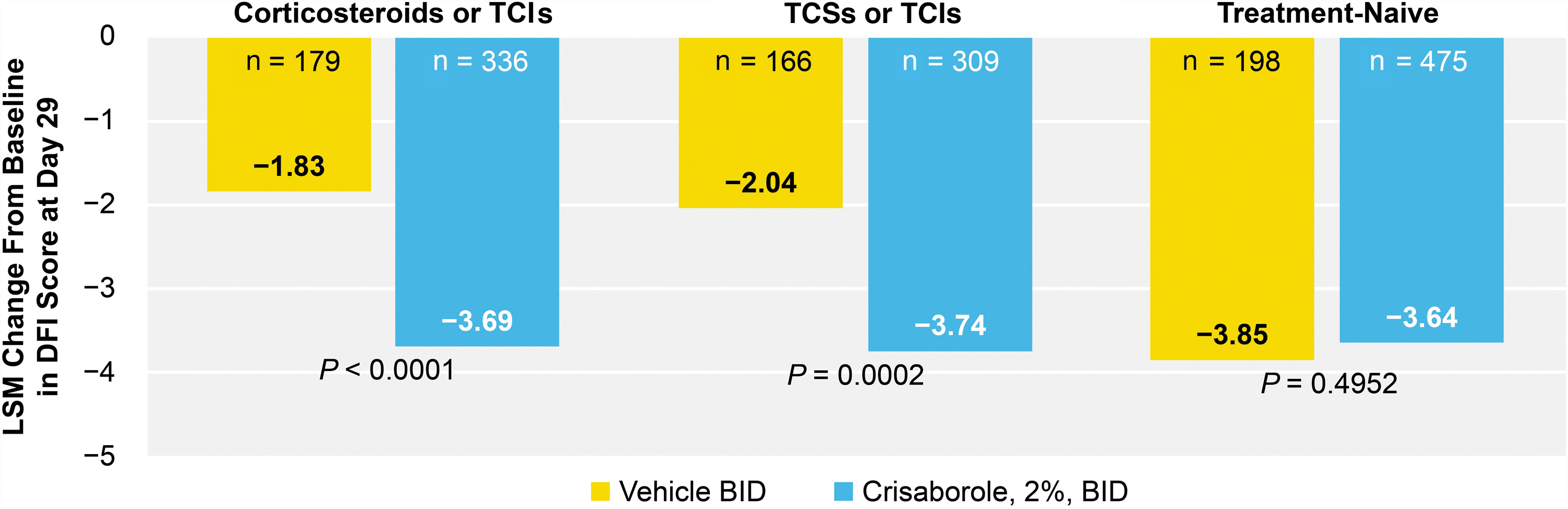
LSM of change from baseline in DFI questionnaire score at day 29. BID, twice daily; DFI, Dermatitis Family Impact; LSM, least squares mean; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid.
Safety Endpoints
Crisaborole was well tolerated and effective for the treatment of mild-to-moderate AD in all 3 subgroups. In the TE-CS/TCI subgroup, 33.2% and 32.9% of patients receiving crisaborole and vehicle, respectively, experienced all-causality TEAEs. All-causality TEAEs occurred in 33.6% and 33.8% of crisaborole-treated and vehicle-treated patients, respectively, in the TE-TCS/TCI subgroup. In the TN subgroup, 26.5% and 19.9% of patients receiving crisaborole and vehicle, respectively, experienced all-causality TEAEs (Table 1).
Application site pain and AD were the most frequently reported TEAEs in crisaborole-treated and vehicle-treated patients, respectively; most AEs were mild. The incidence of application site pain in the 3 subgroups ranged from 2.7% to 7.1% in crisaborole-treated patients and from 1.1% to 1.4% in vehicle-treated patients. The TN subgroup had the lowest incidence of application site pain. The incidence of AD was consistent (0.7%–1.8%) among both crisaborole-treated and vehicle-treated patients in all 3 subgroups, with vehicle-treated patients in each subgroup having a higher incidence versus crisaborole-treated patients (Table 1).
No treatment-related serious adverse events (SAEs) were reported for crisaborole-treated patients. Only 1 SAE was reported by a vehicle-treated patient in the TE-CS/TCI subgroup, and 1 SAE was reported by a vehicle-treated patient in the TN subgroup.
In the TE-CS/TCI subgroup, 2 crisaborole-treated and 4 vehicle-treated patients discontinued from the study due to an AE (specifically, AD) that was considered treatment related. In the TE-TCS/TCI subgroup, 2 crisaborole-treated and 4 vehicle-treated patients discontinued from the study due to a TRAE. Both crisaborole-treated patients discontinued the study due to application site burning, and the 4 vehicle-treated patients discontinued the study due to different AEs (application site blister, acute urticaria, Henoch–Schonlein purpura, and facial swelling). In the TN subgroup, 5 crisaborole-treated patients and 1 vehicle-treated patient discontinued from the study due to an AE of AD that was considered treatment related.
DISCUSSION
In the CORE 1 and CORE 2 studies, crisaborole-treated patients were found to have had improved therapeutic outcomes versus vehicle-treated patients. 1 This analysis enabled us to extrapolate these data to further assess and compare the efficacy of crisaborole in TE and TN patients. Crisaborole was effective and well tolerated in TE and TN patients with mild-to-moderate AD with no new safety signals identified.
Current guidance documents suggest the use of crisaborole as another potential first-line treatment option for both acute and maintenance treatment of mild-to-moderate AD. 6 In this post hoc analysis, the percentage of AEs (all causalities) in crisaborole-treated and vehicle-treated patients in the TE subgroups were similar. Crisaborole was well tolerated, and no new safety signals were identified in any of the 3 subgroups. The overall results from this post hoc analysis demonstrated favorable safety and efficacy of crisaborole in TE and TN patients, and may be used as a basis for clinical application and future studies.
Although emerging drug treatment options such as crisaborole (a PDE4 inhibitor) have proven efficacy and favorable safety profiles when used to treat AD-related signs and symptoms, their use is often limited by cost, comfort of physicians and patients with traditional treatment options, and insurance requirements.6,29
As has been observed previously, 1 vehicle-treated patients also had improved primary and key secondary endpoints at the end of the treatment period versus baseline. The main excipient and base of crisaborole ointment, 2%, is white petrolatum, which has been shown to upregulate antimicrobial peptides, induce expression of barrier differentiation markers, increase the thickness of the stratum corneum, and decrease T-cell infiltrates.30–33 White petrolatum, therefore, assists in repairing the skin barrier and often enhances the skin's innate immune defenses, preventing the development of AD. 30
Notably, the CORE 1 and CORE 2 studies were not designed to assess the effects of crisaborole on patients who had been very recently treated with corticosteroids (topical or systemic) or TCIs. In these pivotal studies, exclusion criteria prohibited previous use of systemic corticosteroids within 28 days and TCS or TCI use within 14 days of randomization.
One of the weaknesses of this analysis was that prior treatment status in this post hoc analysis was only established within 90 days before screening. In the crisaborole trials, only 43% of patients had received prior therapy (TCSs or TCIs) within 90 days before screening; however, it is uncertain whether this percentage represents real-world clinical practice. A further limitation of this analysis of the CORE 1 and CORE 2 studies was that it was not long enough to assess the long-term efficacy and safety of crisaborole in patients who received prior treatments. Lastly, the adult population aged ≥18 years in this analysis was small (12.59%). Future studies including a greater proportion of adult patients may be useful.
CONCLUSIONS
Crisaborole is an effective and well-tolerated treatment option in TE and TN patients. Data from this post hoc analysis extrapolate on prior work and expands on the research done to evaluate the efficacy and safety of different therapeutic alternatives used to manage AD. Although crisaborole is a favorable treatment option, more extensive head-to-head comparative trials comparing crisaborole with other AD treatment options available are needed to expand its current role in treatment guidelines.
Without head-to-head trials, only indirect comparisons to other treatment options (not definitive conclusions regarding the comparative efficacy and safety to other topical treatment options) can be made. In the future, data from this analysis could assist in personalizing the sequencing of therapy, especially when attempting to avoid certain AEs associated with conventional therapies.
