Abstract
Abstract
Background:
Esophageal cancer (EC) is characterized by extreme genetic heterogeneity, advanced stage, high metastatic potential, and poor prognosis. High incidence and morbidity patterns of EC are seen globally even after the availability of new treatment options. The non-coding microRNAs (miRNAs) play a crucial role in EC carcinogenesis. The single nucleotide polymorphisms (SNPs) in the miRNA coding genes might impact the structure and expression of miRNAs, which imbalances cellular homeostasis and promotes tumor formation. In this study, we performed a case–control study to evaluate the correlation between two miRNA polymorphisms (miR-605 rs2043556A/G and miR-196a2 rs11614913C/T) and the risk of EC in the North-West Indians.
Methodology:
The miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms were screened in 320 EC patients and 356 controls using the polymerase chain reaction-restriction fragment length polymorphism method.
Results:
The GG genotype of miR-605 rs2043556A/G polymorphism was associated with a higher risk of EC in the total subjects as well as in female group, whereas no association of miR-196a2 rs11614913C/T polymorphism was observed with EC risk. The CT genotype of miR-196a2 rs11614913C/T polymorphism was associated with a reduced EC risk in patients ≤ 50 years. Genotype combinations analysis revealed a higher risk of EC with miR-605 rs2043556GG/miR-196a2 rs11614913 CT genotype combination.
Conclusion:
This study concluded that the miRNA polymorphisms were related to EC development in North-West Indians. The investigation of the miRNA polymorphisms that could influence EC susceptibility might help in designing better individualized treatment plans for EC treatment.
Introduction
Esophageal cancer (EC) is a highly metastatic, advanced, and aggressive gastrointestinal tract cancer that affects the health of millions of people worldwide. Globally, EC is positioned 11th in terms of most common cancers and is the seventh most common cause of cancer-related mortality with the majority of the EC cases and EC-related deaths reported from the Asian subcontinent.1 Different molecular subtypes of EC have been identified and are characterized by distinct etiologies.2 The development and progression of EC occurs through complex interactions between multiple genetic, epigenetic, and environmental factors.3,4 The major environmental risk factors for EC include alcohol consumption, smoking, consumption of hot drinks, Gastroesophageal Reflux Disease, Barett's esophagus, and obesity.5 Individuals exposed to the same environmental factors may not all develop EC, thus suggesting the role of genetic factors in determining inter-individual differences in EC susceptibility.
The regulation of the transcription and translation processes is necessary for the proper controlled expression of the protein-coding genes. Any dysregulation in these processes leads to abnormal protein function, ultimately leading to carcinogenesis. A class of non-coding RNAs has been identified that plays a major role in carcinogenesis.6 MicroRNAs (miRNAs) are 20–25 nucleotides long non-protein coding RNA molecules, which negatively regulate the post-transcriptional gene expression, either by degrading the target genes' mRNA or repressing the translation process.7–10 miRNAs are responsible for regulating the gene expression in one-third of all protein-coding genes.11 miRNAs play a critical role in regulating various normal biological activities and pathological conditions, including maintaining embryonic development, regulation of apoptosis, tissue repair, cell proliferation, and tumorigenesis.7,12,13
In cancer, the miRNAs regulate target gene expression, by acting as either oncogenes or tumor suppressor genes.14 The majority of the miRNA genes have been reported to be present in the cancer-susceptible region of the genome.15 Dysregulated miRNA expression significantly impacts the tumor progression, tumor characteristics, chemotherapeutic response, and prognosis in several cancers, including EC.16–21
It has been reported that genetic variants in a single miRNA can influence the expression of thousands of target genes.22 The genetic variants in the regulatory region of miRNA genes may alter the binding of miRNA to their target gene mRNAs or affect the maturation, expression, and function of the miRNAs,23–26 ultimately affecting carcinogenesis.25,27,28 Among the various miRNAs identified, miR-605 and miR-196a2 are the two important miRNAs involved in tumorigenesis.29 The miR-605 is localized on chromosome 10q21.1,30 whereas the miR-196a2 is positioned on chromosome 12q13.13.31 The genetic variants in the miR-605 and miR-196a2 might affect the miRNA expression as well as the binding of the mature miRNA to the target genes, leading to inter-individual variation in tumor susceptibility, development, and progression.32,33 Previous studies have reported that the CC genotype of miR-196a2 rs11614913C/T polymorphism is associated with higher levels of mature miR-196a expression in non-small cell lung34 and colorectal35 cancer. In contrast, the GG genotype of miR-605 rs2043556A/G polymorphism was associated with lower miR-605 levels in Li-Fraumeni Syndrome.36
Several studies have investigated the role of miR-605 rs2043556A/G polymorphism in influencing cancer susceptibility in various cancers, such as breast,30,37–40 gastrointestinal,41 head and neck,42 lung,43,44 gastric,29 and cervical45 cancers (Supplementary Table 1). The miR-196a2 rs11614913C/T polymorphism has been extensively studied in several cancers, including hepatocellular,46 colorectal,47 non-small cell lung,34 breast,48 lung,32 gastric,49 and head and neck cancers.50
The involvement of miR-196a2 in the transformation of Barett's esophagus to highly aggressive esophageal adenocarcinoma has been reported in Caucasians.51 The miR-196a2 rs11614913C/T polymorphism has been reported as a potential candidate biomarker for EC susceptibility and prognosis in various studies (Supplementary Tables 2 and 3). It has been documented that miR-196a2 rs11614913 C/T polymorphism was associated with EC risk in Caucasians,52 Chinese,53–58 and North Indians.59 Wu etal. reported that the TT genotype of miR-196a2 rs11614913C/T polymorphism was significantly associated with worse survival in Chinese EC patients treated with platinum-based therapy.60 However, no role of miR-196a2 rs11614913 C/T polymorphism with clinical response has been reported in North Indian59 and Chinese61 EC patients.
To date, a single study has evaluated the role of miR-196a2 rs11614913C/T polymorphism with EC risk in the North Indians,59 whereas no study has reported the role of miR-605 rs2043556 A/G polymorphism in EC susceptibility. Considering the role of miR-196a2 rs11614913 C/T and miR-605 rs2043556 A/G variants in cancer development, we performed a case–control study to investigate the association of the miR-196a2 rs11614913 C/T and miR-605 rs2043556A/G variants in sporadic EC patients from Punjab, North-West, India.
Materials and Methods
Selection of the study participants and data collection
This study was approved by the institutional ethics committee of Guru Nanak Dev University, Amritsar, Punjab, India, and an informed written consent was obtained from all the study participants. In this study, 676 participants (320 sporadic EC patients and 356 healthy subjects) were included from Punjab state, North-West India. The patients were clinically confirmed at Sri Guru Ram Das Institute of Medical Sciences, Amritsar, whereas the controls were the cancer-free healthy subjects randomly recruited from the same geographical region as the EC patients. The EC patients on any treatment were excluded from the study. The controls were unrelated, healthy, age, and gender-matched subjects with no prior history of cancer or any other chronic disease. The demographic characteristics were collected on a pre-designed proforma by personally interviewing the study participants, whereas the clinical information was obtained from the clinical records of patients. Five milliliters of blood samples was collected from all the study subjects in a sterile ethylenediamine tetraacetic acid (EDTA)-coated vacutainer and stored at −20°C till further use.
Molecular genetic investigations
The genomic DNA was isolated from the stored or fresh blood samples using the standard phenol-chloroform method.62 Both the miRNA (miR-605 rs2043556A/G and miR-196a2 rs11614913C/T) polymorphisms were screened using the polymerase chain reaction-restriction fragment length polymorphism method. The targeted regions of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms were amplified using the specific set of primers.29 For miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms, the PCR reaction was carried out in a 15 µl reaction volume containing 50 ng of DNA, 1.5 µl of 10X Taq buffer with 15 mM MgCl2, 0.3 µl of dNTP's mix, 0.5 µl of forward and reverse primer (10 pmol/µl), and 0.6U of Taq polymerase. The amplification conditions used were initial denaturation at 95°C for 5 min, followed by 35 cycles with denaturation at 95°C for 45 s, annealing at 59°C for miR-605 rs2043556A/G and 62°C for mir196a2 rs11614913C/T polymorphism for 30 s, extension at 72°C for 45 s, and final extension at 72°C for 10 min. The amplified PCR products of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms were checked on 2% agarose gel and then overnight digested with HinfI and MspI restriction enzymes, respectively, at the recommended temperature as per the manufacturer's instructions. The restriction-digested PCR products were analyzed on 2.4% agarose gel (Fig.1). The genotyping was performed based on the restriction digestion pattern as detailed in Table1. The results were validated by genotyping 10% randomly selected samples with the Sanger sequencing, and 100% concordance in the results was observed (Fig.2).

A gel photograph showing the restriction digestion pattern of (A) miR-196a2 rs11614913C/T and (B) miR-605 rs2043556A/G polymorphism.
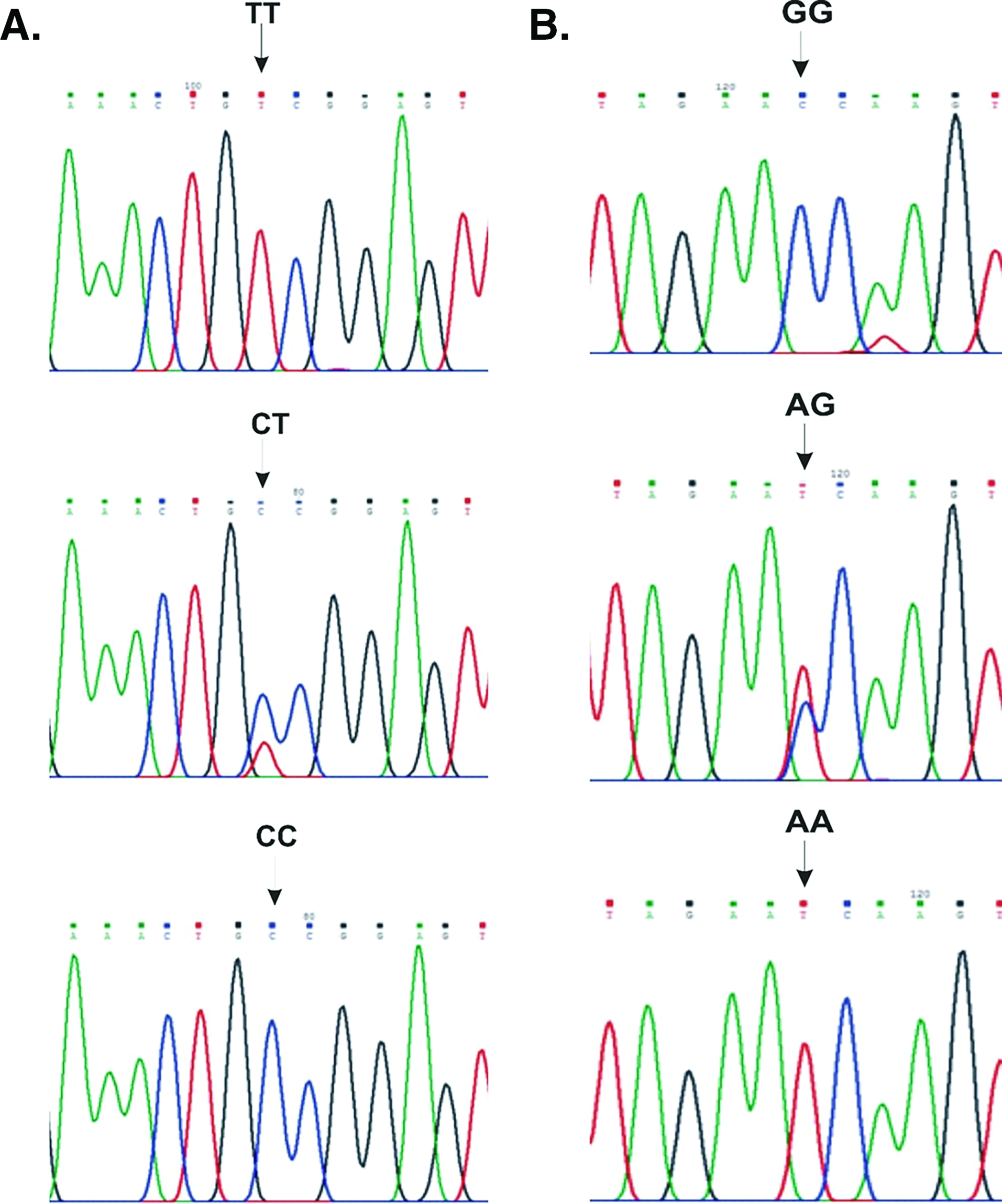
(A) A part of electropherograms representing the TT, CT, and CC genotypes of mir-196a2 rs11614913C/T polymorphism (forward strand). (B) A part of electropherograms representing the GG, AG, and AA genotypes of mir-605 rs2043556A/G polymorphism (reverse strand).
Detail of the miRNA polymorphisms and genotyping methodology
Statistical analysis
The Chi-square test was applied to calculate the deviation from the Hardy–Weinberg Equilibrium (p-value > 0.05) in the control subjects. The odds ratios (ORs) and the 95% CI were calculated to determine the association between the miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphism with the EC risk using theMedCalc software. The genotype data were also analyzed based on different genetic models (codominant, dominant, recessive, overdominant, and log-additive model) using the online SNPstats software.63 The genotype combinations of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms were prepared, and the association of miRNA polymorphisms genotype combinations with the EC risk was evaluated using theMedCalc software. A probability value of <0.05 was considered statistically significant for the statistical analyses.
Results
Characteristics of the study participants
The present study included 320 EC patients (138 males and 182 females) and 356 healthy subjects (163 males and 193 females). The mean ages of the EC patients and controls were 56.42 ± 12.66 years and 54.12 ± 12.56 years, respectively. In 61.3% of the EC patients, the age of diagnosis was more than 50 years. The majority of the study participants lived in rural regions and were exposed to toxic agricultural chemicals, fertilizers, and pesticides. The frequency of smokers (14.1%) was higher in the EC patients group compared to the control group (3.1%). Most of the study subjects were non-alcoholic and vegetarian. Esophageal squamous cell carcinoma was the main histological subtype in the EC patients, with the majority of the patients suffering from Stage II and Stage III cancer. The demographic and clinical data of the study participants have been reported in Table2.
Demographic and clinical features of the EC patients
Association of miR-605 rs2043556A/G polymorphism with EC risk
The genotype distribution for miR-605 rs2043556 A/G polymorphism was in complete Hardy–Weinberg equilibrium in healthy controls (p = 0.08). The genotype distribution of miR-605 rs2043556A/G polymorphism of both EC patients and controls has been mentioned in Table3. In total subjects, individuals with the GG genotype had a 2.58-fold higher risk of EC (OR = 2.58, 95% CI = 1.23–5.42, p = 0.01) (Table3). The miR-605 rs2043556A/G polymorphism was associated with an increased risk of EC under the codominant model (p = 0.01) and recessive model (p = 0.009) in the total subjects (Table4).
Association of miR-605 rs2043556 A/G and miR-196a2 rs11614913C/T polymorphisms with EC risk in total, female, and male subjects
Statistically significant p values displayed in bold.
Association of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms with esophageal cancer risk in total, female, and male subjects under different genetic models
Statistically significant p values displayed in bold.
The data were further stratified based on gender, and it was observed that in female subjects, the risk of EC was significantly higher with the GG genotype (OR = 3.83, 95% CI = 1.36–10.75, p = 0.011) (Table3). Genetic model analysis revealed a significant association of miR-605 rs2043556A/G polymorphism with EC risk in female subjects under codominant (p = 0.01) and under the recessive model (p = 0.004) (Table4). However, no association of miR-605 rs2043556A/G polymorphism with EC risk was observed in the male group (p > 0.05) (Tables3 and 4). The genotype data were stratified based on the age at diagnosis of the patients, and no association was observed (Table5).
Association of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms with esophageal cancer risk in patients stratified by age
Statistically significant p values displayed in bold.
Association of miR-196a2 rs11614913C/T polymorphism with EC risk
The genotype distribution of miR-196a2 rs11614913C/T polymorphism was in Hardy–Weinberg Equilibrium in the controls (p = 0.14). The genotype distribution of miR-196a2 rs11614913C/T polymorphism of both EC patients and controls has been mentioned in Table3. The genotype and allele frequency of the miR-196a2 rs11614913C/T polymorphism did not show any significant differences in the total subjects as well as in males and female groups (p > 0.05), indicating that miR-196a2 rs11614913C/T polymorphism was not associated with EC risk (Table3). Genetic model analysis revealed no significant association of miR-196a2 rs11614913C/T polymorphism with EC risk in any genetic model in total subjects, female subjects, and male subjects (p > 0.05) (Table4).
Age-wise stratification of the data revealed a decreased risk of EC in patients having age ≤ 50 years with the CT genotype (OR = 0.52, 95% CI = 0.32–0.85, p-value = 0.008) and T allele (OR = 0.67, 95% CI = 0.48–0.94, p-value = 0.02) (Table5).
Association of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T genotype combinations with EC risk
Various genotype combinations of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms were prepared to evaluate their association with EC risk. It was observed that the individuals having the miR-605 rs2043556GG/miR196a2 rs11614913 CT genotype combinations had a higher risk of EC (OR = 2.87, 95% CI = 1.05–7.80, p-value = 0.04) (Table6).
Genotype combinations of miR-605 rs2043556A/G and miR-196a2 rs11614913C/T polymorphisms and esophageal cancer risk
Statistically significant p values displayed in bold.
Not calculated.
Discussion
The genetic variants in the miRNA act as the potential biomarkers of tumor susceptibility, metastasis, progression, and prognosis. In this study, we examined the relationship between the miRNA genetic variants (miR-605 rs2043556A/G and miR-196a2 rs11614913C/T) with the susceptibility of EC in patients from Punjab, North-West India. The miR605 rs2043556A/G polymorphism might influence cancer susceptibility by affecting the miRNA structure and expression. One of the main targets of the miR605 is MDM2. The miR-605 inhibits the interaction between the p53 and MDM2 during cellular stress. The miR-605 rs 2043556 A/G polymorphism can impair the functioning of miR-605 in regulating the p53-MDM2 signaling pathways, ultimately leading to tumor development.64 In the present study, we observed that the GG genotype of the miR-605 rs2043556A/G polymorphism was associated with higher EC susceptibility. Similarly, the GG genotype of miR-605 rs2043556A/G polymorphism was associated with higher lung squamous cell carcinoma risk in Chinese nonsmoker females.44 The miR-605 rs2043556AG and GG genotypes were associated with increased susceptibility of gastric cancer in Brazilians29 and breast cancer in Iranians.30 In Chinese population, the AG/GG genotype of miR-605 rs2043556A/G polymorphism was associated with reduced risk of colorectal cancer41 and oral cancer.42 In South Americans, the miR-605 rs2043556 G allele was associated with a reduced risk of early-onset non-familial breast cancer.40 No association of miR-605 rs2043556A/G polymorphism was observed with lung43 and cervical cancer risk in the Chinese population45 and risk of breast cancer in the Chinese37 and Iranian population.39
The major targets of miR-196a2 rs11614913C/T polymorphism are the HOX gene cluster and ANXA1.65 ANXA1 is mainly involved in promoting cell proliferation and suppressing apoptosis.54 The miR-196a2 rs11614913C/T polymorphism might affect the expression of mir196a and further the expression of ANXA1, affecting cancer development.66 Individuals with the CC genotype of the mir196a2 rs11614913C/T polymorphism might have altered HOX expression, further affecting their risk of cancer development and progression.67 In the present study, we did not observe any relationship between the miR-196a2 rs11614913C/T polymorphism with the EC risk. A previous study from Jammu and Kashmir, North India, also reported no association of the miR-196a2 rs11614913C/T polymorphism with the EC risk.59 The association of CC genotype of mir196a2 rs11614913C/T polymorphism with higher EC susceptibility has been reported in the Chinese population.53,57 The association of miR-196a2 rs11614913C/T polymorphism with reduced EC risk has been reported in Chinese subjects under the recessive genetic model,54 whereas higher EC susceptibility was observed in the Caucasians.52 Another study conducted on Chinese subjects reported a reduced risk of EC in individuals carrying the TT genotype of miR-196a2 rs11614913C/T polymorphism.55 The inconsistency in the findings of the studies could be due to variations in sample sizes, different ethnicity, and exposure to different lifestyle factors.
The genetic differences between the individuals are responsible for different chemotherapeutic responses to cancer treatment and the prognosis of the patients. Identification of the cancer-related miRNA polymorphisms helps in the designing of better personalized treatment plans for EC patients. The therapeutic response of miR-196a2 rs11614913C/T polymorphism in EC patients has been reported in several studies (Supplementary Table 3). It was observed that in EC patients treated with platinum-based chemotherapy, the TT genotype of miR-196a2 rs11614913C/T polymorphism was associated with worse overall survival,60 whereas no relationship between the miR-196a2 rs11614913C/T polymorphism and therapeutic response was observed in the North Indian chemotherapy-treated EC patients59 and Chinese patients treated with different chemotherapy regimens.61
It has been reported that certain miRNAs are expressed differently in males and females.68 We observed a gender-specified association in our study with female subjects carrying the GG genotype of miR-605 rs2043556A/G polymorphism having a higher risk of EC. No association of the mir196a2 rs11614913C/T polymorphism with EC risk was observed on sex-based stratification analysis. The G allele of miR-605 rs2043556A/G polymorphism was associated with decreased colorectal cancer risk in Chinese male subjects,41 whereas the miR-196a2 rs11614913C/T polymorphism was associated with a decreased risk in Chinese female EC patients.54,58
In our study, there was a higher frequency of smokers in the EC patients compared to the healthy subjects. Smoking is known to influence the expression of various miRNAs and miRNA-target genes.69,70 The combined role of lifestyle factors such as smoking and miRNA polymorphisms in influencing cancer risk has been previously studied. It was reported that the miR-196a2 rs11614913 CC genotype was associated with a higher EC risk in Chinese smokers individuals.53 Another study from China reported a reduced EC risk with CC genotype in nonsmokers and nondrinkers.54 Shen etal. reported a higher EC risk in nonsmokers under the recessive model of miR-196a2 rs 11614913 polymorphism.57 The miR-605 rs2043556 AG/GG genotype was associated with higher gastric cancer risk in smokers41 and higher lung cancer risk in females exposed to cooking oil fumes.43
The miRNA expression profiles have been reported to be different in older and younger subjects.71 In our study, we observed a reduced risk of EC in the younger subjects carrying the CT genotype and the T allele of miR-196a2 rs11614913 polymorphism. A previous study by Wang etal. reported a higher EC risk in patients less than 60 years of age carrying the T allele of miR-196a2 rs11614913 polymorphism,56 whereas a decreased risk was observed in EC patients >63 years of age under the additive model.58
In the present study, the majority of the patients lived in rural regions of Punjab state and were exposed to highly toxic insecticides and pesticides. Higher utilization of insecticides and pesticide products in farming practices is responsible for the higher EC prevalence in Punjab.72,73 It has been reported that pesticide exposure leads to aberrant miRNA expression,74 ultimately affecting cancer development.75
There are some limitations in this study. First, the small sample size limits the statistical power of the study. We could not perform functional analysis to determine the impact of miRNA polymorphisms on the expression levels of miRNAs and their target genes.
Conclusion
In this study, we concluded that miR-605 rs2043556 polymorphism was significantly associated with increased EC risk in total patients as well as in the female group, whereas miR-196a2 rs11614913 polymorphism was associated with reduced risk of EC only in the younger subjects. Further larger case–control studies on different ethnicities as well as functional analysis are needed in future to validate our findings.
Footnotes
Acknowledgements
We would like to thank all the study participants.
