Abstract
Abstract
Background:
Cytomegalovirus (CMV) disease affects mainly immunosuppressed individuals such as stem cell and solid organ transplant recipients as well as those patients infected with the human immunodeficiency virus (HIV). Cytomegalovirus colitis has been diagnosed increasingly in normal hosts.
Case Presentation:
A 65-year-old male with a history of alcohol abuse had been hospitalized for severe bloody diarrhea for three months in an outside hospital; he tested negative for clostridial toxin. He was transferred to our intensive care unit in septic shock and multi-organ failure with bloody diarrhea and a megacolon on computed tomography (CT) scan. Emergency sigmoid colectomy was performed, however, the patient continued to have bloody diarrhea; subsequently, completion sub-total colectomy with ileostomy was performed. On immunohistochemistry a diagnosis of CMV colitis was made; blood CMV polymerase chain reaction (PCR) was positive, the patient tested positive for anti-CMV immunoglobulin (Ig) G but non-IgM antibodies. Ganciclovir was started and the patient's CMV PCR became negative within one week. The patient developed multiple other complications including Aspergillus tracheobronchitis, Pseudomonas aeruginosa sepsis, and Candida krusei pneumonia, but ultimately recovered from the multiple infections. A low natural killer (NK) cell count (<1%) was found on immunologic workup; the patient tested negative for HIV.
Conclusion:
Cytomegalovirus colitis is a rare disease in the non-immunocompromised host but should be considered in patients with pseudomembranous colitis testing negative for clostridial toxin.
Cytomegalovirus (CMV) has been associated with a specific fetopathy, pneumonitis in the newborn, and diseases in the immunocompromised host. Before the development of highly active anti-retroviral therapy (HAART), CMV disease was a common complication in human immunodeficiency virus (HIV)-infected individuals. Cytomegalovirus is the most important systemic virus complicating solid organ transplantation. Cytomegalovirus disease manifests most commonly as a mononucleosis-like syndrome, however, the virus may also cause interstitial pneumonitis, retinitis, and meningoencephalitis as well as diseases of the gastrointestinal tract including esophagitis, gastritis, gastric ulcers, enteritis, and colitis. Although CMV colitis is typically observed in severely immunosuppressed patients, increasingly cases of life-threatening CMV colitis have been diagnosed in the normal host [1–12].
Cytomegalovirus plays a pivotal role in inflammatory bowel disease (IBD) by potentially triggering IBD and/or emerging after onset of immunosuppressive therapy including disease modifiers [13–15]. The clinical presentation of CMV colitis ranges from mild diarrhea with minimal mucosal changes to toxic megacolon with pseudomembranes similar to Clostridium difficile-associated colitis (CDAC). Co-infection of CMV together with C. difficile has been reported [16]. Once diagnosed, CMV colitis responds to antiviral therapy with ganciclovir [4,7], however, in the non-immunocompromised host diagnosis is made commonly on colectomy specimens because normal patients with colitis are not tested routinely for CMV. Cytomegalovirus polymerase chain reaction (PCR) is not available universally in many hospitals or not performed routinely on a daily basis but rather once or twice weekly for pooled monitoring of high-risk patients [17]. Antibody testing is usually not conclusive because the seroprevalence of CMV exceeds 60% and only in rare cases new infection with this virus as determined by presence of CMV-specific IgM antibodies occurs in adults. In the majority of cases, the virus re-emerges from its latent state in endothelial, epithelial, and polymorph nuclear cells after a change in the immune status of patients. This change in the immune competence against CMV may be subtle and difficult to diagnose. Because the most important immune response to the virus is a specific T-cell response, alterations in the T-cell immunity are associated most commonly with CMV re-activation although also critically ill individuals are at risk for this opportunistic infection [18]. We herein report on a middle-aged male who developed pseudomembranous colitis caused by CMV.
Case Presentation
A 65-year-old male had been treated for several weeks at an outside facility before transfer to our hospital. He was a smoker and had been drinking substantial amounts of alcohol in the past. He had a three-month history of watery and bloody diarrhea, weight loss, malaise, and chronic fatigue. At the outside hospital he received several courses of antibiotics including ciprofloxacin and metronidazole for presumed bacterial diarrhea. He had tested negative for Salmonella, Shigella, Yersinia, Escherichia coli as well as C. difficile. No work up for immunologic disorders or for viral pathogens or parasites had been performed. He was malnourished and had lost more than 15 kg over the past three months; his creatinine was elevated to 1.7 mg/dL and his bilirubin was 2.5 mg/dL. The patient had been started on coumadin for deep venous thrombosis at the outside hospital and developed supratherapeutic international normalized ration (INR >5). Because of respiratory failure he was intubated and transferred to our hospital, where he was admitted to the intensive care unit and fluid resuscitation was begun.
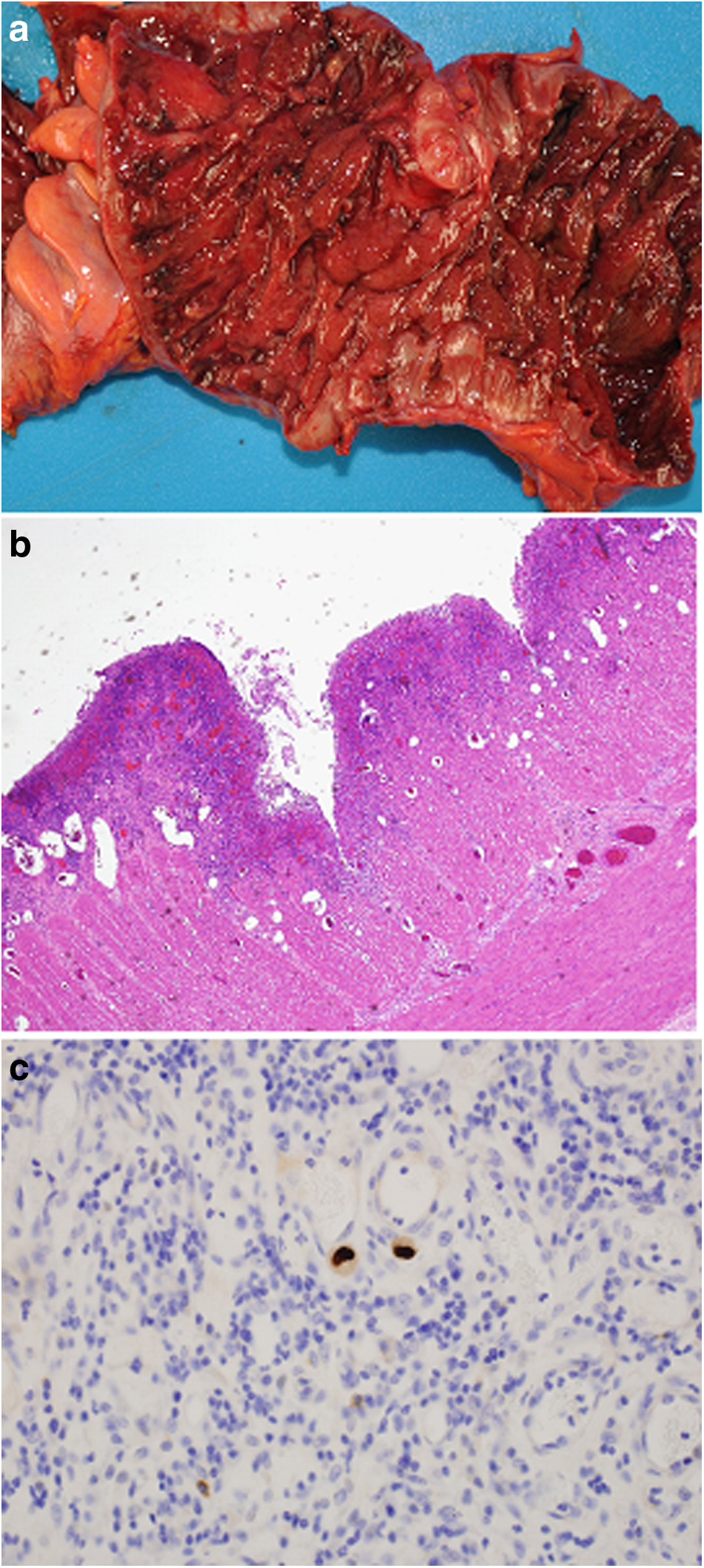
On admission, the patient had a tender distended abdomen. His white blood cell (WBC) count was 17.8 cells per milliliter with a hematocrit of 23%. He was continued on antibiotic therapy including metronidazole, oral vancomycin, and doripenem. Records from the outside hospital showed that the patient had positive sputum cultures for Candida krusei as well as Aspergillus fumigatus and therefore micafungin was started. Clostridium difficile toxin assay was negative as was HIV antibody testing. Computed tomography scan revealed bilateral pneumonia with pleural effusions, a distended colon with significant wall thickening (Fig. 1), and a sub-total infarction of the spleen. The patient had several bloody bowel movements; a colonoscopy seemed to be associated with substantial risk for perforation, and therefore, it was decided to perform emergency laparotomy after correction of the coagulopathy. The left colon was found to be severely inflamed and the sigmoid colon showed maximum pathology and was resected (Fig. 2a); an end descendostomy was created and the rectal stump was stapled. Histopathology showed severe pseudomembranous colitis but a diagnosis of inflammatory bowel disease (IBD) was also discussed. No specific signs of CMV colitis were found on routine hematoxylin and eosin (H&E) staining and immunohistochemistry was also negative for CMV (Fig. 2b). Cytomegalovirus serology showed positive IgG but negative IgM antibodies. The patient improved slightly after surgery, however, there was still substantial bleeding from the ostomy and the patient required further transfusion of red blood cells. Therefore, a completion sub-total colectomy with end ileostomy was performed. The patient required massive additional blood, platelet, and fresh frozen plasma transfusions, fluid resuscitation, as well as high-dose vasopressor therapy. His course was complicated further by acute cardiac failure and he was started on milrinone 475 mg/h. Because of the undiagnosed cause of the disease, pathology was requested to perform in vitro hybridization of the specimen for potential pathogens. This revealed the diagnosis of CMV colitis (Fig. 2c) although a co-diagnosis of IBD was also discussed. Intravenous ganciclovir at a dose of 10 mg/kg was started. Also CMV PCR testing was positive with 3,171 copies per milliliter; repeat testing four days and three weeks after initiation of antiviral therapy was negative. Although the patient improved overall, he developed Pseudomonas aeruginosa pneumonia, which was treated with doripenem in combination with gentamicin. Immunologic workup showed that the patient had a low concentration of IgM and IgG and elevated IgA and IgE concentrations; in addition his NK cell count was found to be low (<1%); his CD4 and CD8 counts were normal. The patient underwent tracheostomy because he could not be weaned off the ventilator; total parenteral nutrition (TPN) was started and later switched to enteral feeding. Gradually his condition improved and the patient could be transferred to a rehabilitation clinic; his tracheostomy was closed and a sacral decubitus was treated surgically. The patient finally recovered and was discharged with a plan for reversal of his ileostomy.

Abdominal computed tomography (CT) scan showing a distended transverse colon (
(
Discussion
Cytomegalovirus colitis is a rare infection in immunocompetent individuals [6,11,19]. However, as this case illustrates, a delay in diagnosis has devastating consequences for the patient. It should also be considered that the low NK cell count in our patient may have been the reason why he re-activated the virus and the infection could not be controlled [20,21]. A de novo infection was excluded by serology and would have been unusual at the age of 50 y in a non-transplant patient. Our patient had had diarrhea for three months and experienced severe weight loss and superinfections with unusual pathogens including atypical fungi [22]. Nevertheless, neither immunologic nor thorough infectious workup was carried out. Clostridium difficile-associated colitis has become a problem in the United States and was suspected despite several negative assays [23]. Negative assays do not exclude C. difficile infection, however, one should always consider other enteric infections even if pseudomembranous colitis is observed on colonoscopy [24]. Cytomegalovirus has the potential to mimic CDAC; also co-infection with multiple pathogens both in immunosuppressed and immunocompetent patients has been observed [3,25]. Finally, non-infectious causes and a multitude of pathogens such as rotavirus, Mycobacterium tuberculosis, or Cryptosporidia among others may cause megacolon and/or pseudomembranous colitis.
The severe course of our patient's illness with multiple superinfections also outlines the indirect effects that CMV can exhibit [26]. In the transplant population it is well established that in addition to CMV disease, the virus causes paralysis of the immune system giving way to other infections such as other viruses, rare bacteria, and fungi such as Pneumocystis jiroveci, Candida spp., and filamentous fungi. It is difficult to determine whether our patient's NK cell deficiency had been present at onset of disease because baseline values were unavailable [27]. He had a history of alcohol abuse and low NK cell count has been reported in alcoholism [28]. On the other hand, the condition may have been a result of the CMV disease.
Cytomegalovirus PCR can readily establish the diagnosis of CMV disease, however, patients may occasionally test negative on PCR from blood despite gastrointestinal disease [29]. Therefore, colonoscopy with biopsy and specific staining is the test of choice if CMV colitis is suspected. Once diagnosed, treatment with ganciclovir is successful in most patients. Cytomegalovirus colitis should respond to therapy with ganciclovir and only in rare cases surgical intervention is necessary [4,7]. This becomes necessary mostly in cases of delayed diagnosis, co-infection with other pathogens, or in immunocompromised individuals who cannot clear the virus despite antiviral therapy.
To summarize, we herein report the case of a patient with undiagnosed CMV colitis, which led to colectomy. Awareness regarding this disease, testing using CMV PCR and/or colonoscopy with biopsy, and specific CMV staining at an early stage followed by treatment with ganciclovir could have resulted in complete remission and could have avoided the permanent disability, the multiple complications, and excessive costs in this patient.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
