Abstract
Abstract
Background:
Clostridium difficile causing clinical disease of the small bowel is reported rarely in the literature, but may be increasing in incidence as overall Clostridium difficile infection (CDI) rates increase across the country. Common risk factors that have been reported in the literature for C. difficile enteritis include inflammatory bowel disease (IBD), recent antibiotic use, and gastrointestinal surgery. We report a unique case of C. difficile enteritis involving the small bowel with necrosis and perforation in the absence of common risk factors.
Case Presentation:
A 57-year-old African American female with no prior surgical history presented with acute abdominal pain with diarrhea and was found to have CDI. On hospital day eight, the patient de-compensated suddenly. She was found to have a small bowel perforation in the proximal jejunum with pathology demonstrating CDI involving the small bowel.
Conclusion:
Clostridium difficile infection involving the small bowel is being reported increasingly and can lead to perforation, especially in immunocompromised patients. Whereas C. difficile enteritis has been described previously involving the small bowel, this case is unique because the patient did not have any of the strongest risk factors for CDI and presented with acute necrosis and perforation of the jejunum.
Clostridium difficile infection (CDI) is the most common cause of infectious diarrhea in the health care setting and has been observed with increasing incidence and virulence in hospitals across the United States [1]. Clostridium difficile most typically causes an infection of the large bowel, although in rare cases, CDI has been documented to involve the small bowel [2–7]. However, the majority of reports of C. difficile enteritis have occurred in patients with prior recent antibiotic exposure and/or a surgical intervention. We present a case in which C. difficile enteritis presented with small bowel perforation and peritonitis in a patient with no surgical history or recent antibiotic exposure.
Case Report
The patient is a 57-year-old African American female who presented to the emergency department with complaints of two days of generalized fatigue, right-sided abdominal pain, some difficulty breathing, and watery diarrhea. She had a significant past medical history of end-stage renal disease requiring hemodialysis, hypertension, human immunodeficiency virus (HIV; CD4 count of 394), pancreatitis, and methicillin-resistant Staphylococcus aureus (MRSA) endocarditis. She had completed a six-week course of vancomycin for treatment of the MRSA endocarditis more than two months prior to this admission and had not been exposed to antibiotics since completion of treatment. Other than a critical hypophosphatemia, her admission laboratory tests had no major abnormalities. A computed tomography (CT) scan of the abdomen and pelvis was normal but incidentally found a pulmonary embolism in the right lobe of her lung, confirmed by CT angiography of her chest. She was admitted to the hospital and started on therapeutic anticoagulation for her pulmonary embolism. On hospital day three, a stool sample was tested for C. difficile via polymerase chain reaction (PCR) and returned positive. She was started on metronidazole 500 mg by mouth every eight hours for treatment of CDI.
On hospital day seven, she continued to have persistent diarrhea, so the metronidazole was changed from oral to intravenous with the addition of oral vancomycin 125 mg four times per day. Overnight on hospital day seven, she had an acute worsening of abdominal pain necessitating repeat CT scan of her abdomen and pelvis. The CT scan revealed small bowel dilation, pneumatosis, and portal venous gas concerning for small bowel obstruction with ischemia (Fig. 1). The emergency general surgery team was consulted urgently and the patient underwent an exploratory laparotomy.

Computed tomography (CT) scan of the abdomen. Note dilated small bowel with pneumatosis intestinalis (arrow).
At the time of operation, a 50-cm segment of ischemic small bowel with perforation was found, with involvement starting roughly 5 cm distal to the ligament of Treitz (Fig. 2). This area was resected and a hand-sewn end-to-end anastomosis of the small bowel performed. The patient was admitted to the surgical intensive care unit post-operatively and was extubated without complication. Given the intra-operative findings as well as her prior diagnosis of pulmonary embolism, the etiology was initially believed to be mesenteric embolus. However, the final surgical pathology revealed pseudomembranous enteritis with transmural coagulative necrosis (Fig. 3).

Pathologic section of resected segment of small bowel. Note the pseudomembrane (left arrow) amid necrotic small bowel villi (right arrow).
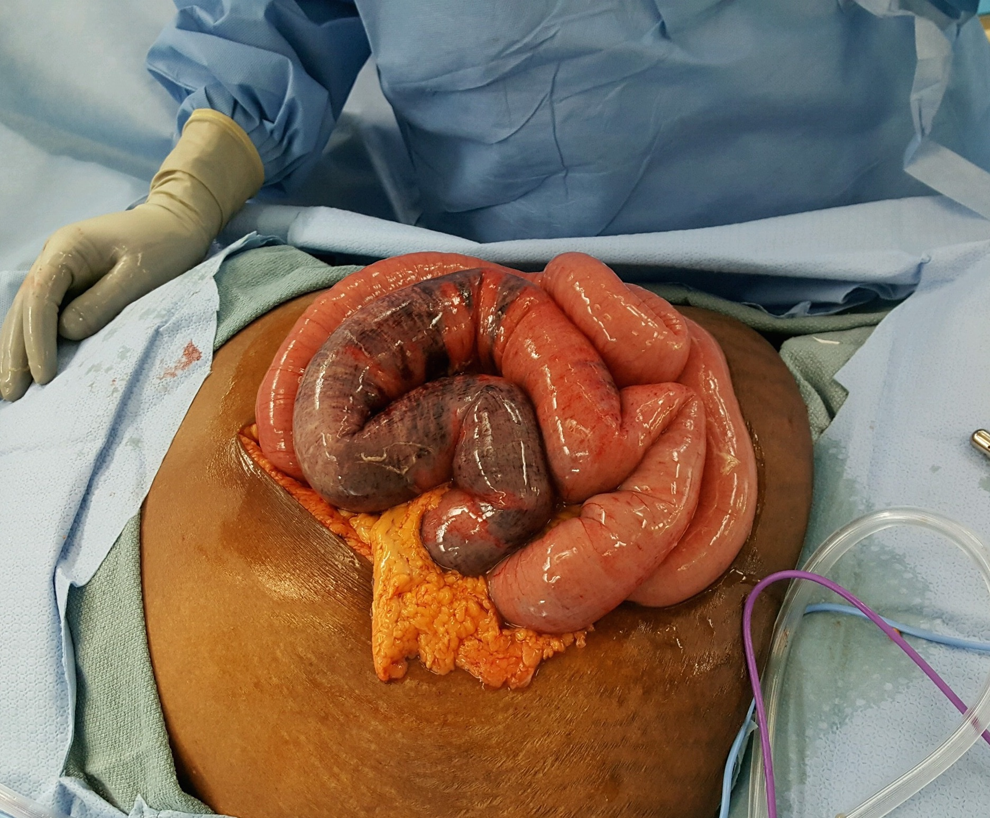
Intra-operative findings during laparotomy. Note necrosis of proximal jejunum.
The patient's post-operative recovery was complicated by an episode of Escherichia coli pneumonia with bacteremia and septic shock that responded to appropriate antibiotic therapy. Her C. difficile infection was treated with intravenous metronidazole 500 mg every eight hours and oral vancomycin 500 mg every six hours for an additional 10 d after the antibiotics for pneumonia were completed. On hospital day 21 she was transferred from the intensive care unit and subsequently discharged to a rehabilitation facility.
Discussion
The incidence of C. difficile enteritis appears to be increasing as the overall burden of CDI increases [3]. Numerous studies have described the growing problem of CDI across the United States, the vast majority of which involve the colon only [1]. A systematic review by Dineen et al. [3] in 2013 identified 83 total published cases of C. difficile enteritis; 90% of these cases had been published after 2000 suggesting that enteritis could be an emerging issue. The pathobiology leading to an apparent increase in C. difficile enteritis remains unclear but is speculated to be related to the increasing prevalence of highly virulent NAP1/BI/027 strain of C. difficile. Patients infected with this strain typically have more severe disease compared with patients infected with other strains. Whether this case was caused by the NAP1/BI/027 strain is unknown because our hospital does not have the capability to identify the specific strain of C. difficile.
Clostridium difficile enteritis is nearly uniformly preceded by certain risk factors for CDI. Antibiotic exposure is the most consistently reported risk factor for CDI, with 71% of C. difficile enteritis cases having had antibiotic exposure within four weeks of presentation [3,8]. Clostridium difficile enteritis has also been associated with inflammatory bowel disease (IBD), recent surgical procedure, and colostomy or ileostomy [3,6]. The case presented in this report is unique in that none of the risk factors were present for C. difficile enteritis.
Two other recent cases have been presented in which the patients presented with CDI enteritis associated with small bowel perforation [4,5]. The first was in an 83-y-old female who presented to the hospital for confusion and was found to have a urinary tract infection. She was placed on trimethoprim-sulfamethoxazole. On hospital day eight, she developed an acute abdomen. Computed tomography revealed dilated small bowel with thickened, inflamed small bowel wall caused by C. difficile enteritis. She underwent laparotomy with colon and small bowel resection. Histopathologic examination of the small and large bowel was consistent with CDI [5]. The second case is another 83-y-old female with critical aortic stenosis and high-grade stenosis of her coronary arteries who presented to the hospital with community-acquired pneumonia. She was prescribed levofloxacin and developed CDI on hospital day five. The levofloxacin was discontinued and metronidazole was initiated. After improvement of the CDI, the patient underwent elective aortic valve replacement and coronary artery bypass grafting. On post-operative day 10, the patient was found to have inflammation of the colon and perforation of the distal ileum [4]. These cases demonstrate the potential for C. difficile enteritis to cause severe small bowel disease as both cases required surgical exploration for perforations. However, these two cases differ from our case in that both had had recent antibiotic exposure and one had recent surgery.
Despite the lack of some of the most common risk factors for CDI, our patient did have some other known risk factors for CDI. She had a recent health care exposure in which had been hospitalized for pancreatitis approximately one month prior to her diagnosis of CDI. In addition, the patient had been treated with a six-week course of vancomycin for MRSA endocarditis and had finished treatment more than two months before the CDI diagnosis. The risk associated with CDI from antimicrobial exposure can persist for many weeks after the antimicrobial agents have been discontinued [9]. Another possible risk factor is her history of chronic kidney disease requiring dialysis. There is limited evidence supporting chronic kidney disease as a risk factor for CDI, but this may have contributed to her risk of overall infection [10]. Perhaps the strongest risk factor for the development of her CDI was her immunocompromised state resulting from HIV infection. Immunocompromised patients, such as those undergoing cancer chemotherapy and those with HIV, have been well documented as having increased incidence of CDI [1]. It is unknown whether these factors increased the risk of having C. difficile enteritis.
Whereas C. difficile enteritis is rare, this case illustrates that it can cause severe small bowel disease and perforation, even without clearly identified risk factors. With its increasing incidence, CDI enteritis should be considered in patients with any health care exposure, particularly in the presence of other known risk factors. Initial reports suggested mortality as high as 66%, however, recent reports have demonstrated a mortality around 25% [3]. Early recognition and management could be essential for treatment success. Optimal treatment and risk identification still needs to be determined, although treatment regimens described in the literature have been consistent with the treatment recommendations for CDI colitis. Surgical indications for CDI enteritis remain the same as in CDI colitis.
Footnotes
Author Disclosure Statement
The authors had no conflicts of interest regarding publication of this article, and this case report did not receive any funding.
