Abstract
Neuronal direct cell reprogramming approach allows direct conversion of somatic cells into neurons via forced expression of neuronal cell-lineage transcription factors (TFs). These so-called induced neuronal cells have significant potential as research tools and for therapeutic applications, such as in cell replacement therapy. However, the optimization of TF delivery strategies is crucial to reach clinical practice. In this review, we outlined the currently explored delivery technologies in neuronal direct cell reprogramming and their limitations and advantages. The first employed delivery strategies were mainly integrating viral systems, such as lentiviruses that exert consistently high transgene expression in most cell types. On the other hand, viral systems cause major safety concerns, including the risk for insertional mutagenesis and inflammation. More recently, several safer nonviral delivery systems have been investigated as well; however, these systems generally exert inferior reprogramming efficiency compared with viral systems. Emerging delivery technologies could provide new opportunities in the achievement of safe and effective delivery for neuronal direct cell reprogramming.
Introduction
Direct cell reprogramming allows conversion of differentiated cells into another functional type without passing through pluripotent state (Caiazzo et al., 2011; Davis et al., 1987; Graf, 2011; Vierbuchen et al., 2010). Compared with differentiation from induced pluripotent stem cells (iPSCs), this approach is faster and applicable both ex vivo and in vivo (Qian et al., 2020; Rivetti Di Val Cervo et al., 2017; Wang et al., 2021a).
Neuronal direct cell reprogramming occurs through ectopic expression of specific transcription factors (TFs) (Aversano et al., 2022; Bocchi et al., 2022; Wang et al., 2021a), microRNAs (miRNAs) (Pascale et al., 2022), and small chemical molecules (Hu et al., 2015; Li et al., 2015; Tian et al., 2016; Zhang et al., 2015) and also through the modulation of polypyrimidine tract-binding (PTB)-mediated regulatory circuits (Xue et al., 2013). These elements activate molecular cascades driven by coding and noncoding elements that eventually impose a specific neuronal phenotype (Bocchi et al., 2022; Della Valle et al., 2020; Wang et al., 2021a).
Neuronal direct cell reprogramming can be applied to different starting cell populations, such as astrocytes, oligodendrocytes, fibroblasts, endothelial cells, pericytes, and hepatocytes (Caiazzo et al., 2011; Ghasemi-Kasman et al., 2015; Hu et al., 2014; Karow et al., 2018; Marro et al., 2011; Weinberg et al., 2017). The resulting cell type is determined by the reprogramming factors applied and includes neural progenitors, glutamatergic, GABAergic, or dopaminergic neurons (Rivetti Di Val Cervo et al., 2017; Torper et al., 2015; Yu et al., 2017).
Most neuronal direct cell reprogramming experiments exploit viral vectors to deliver neurogenic TFs (Bocchi et al., 2022; Wang et al., 2021a). Some of them, such as adeno-associated viruses (AAVs), are Food and Drug Administration (FDA) approved with a favorable safety profile (Liu et al., 2024), but still concerns remain regarding immune response and/or insertional mutagenesis (Mátrai et al., 2010; Shirley et al., 2020). Other emerging approaches utilize nonviral delivery systems (Soltani Dehnavi et al., 2022), including physical (i.e., electroporation, microinjection, and ultrasound) and chemical methods (i.e., peptide-based, synthetic cationic polymer-based, inorganic chemical agent-based, and lipid-based delivery systems), which offer several advantages, such as larger gene payloads, easier preparation, and minimal adverse immune reactions. However, nonviral delivery systems are less efficient (Wang et al., 2023) because of roadblocks that include limitations in the resistance to serum nucleases, attachment to cell membranes, entering the cell, endosomal escape, and release of the therapeutic gene. Therefore, nonviral vectors require complex optimization of their physical–chemical properties to improve their efficacy before being considered for biomedical applications (Tasset et al., 2022).
In this review, we discuss the advantages, limitations, and prospects of different gene delivery approaches currently employed for neuronal direct cell reprogramming both in vitro and in vivo (Fig. 1).
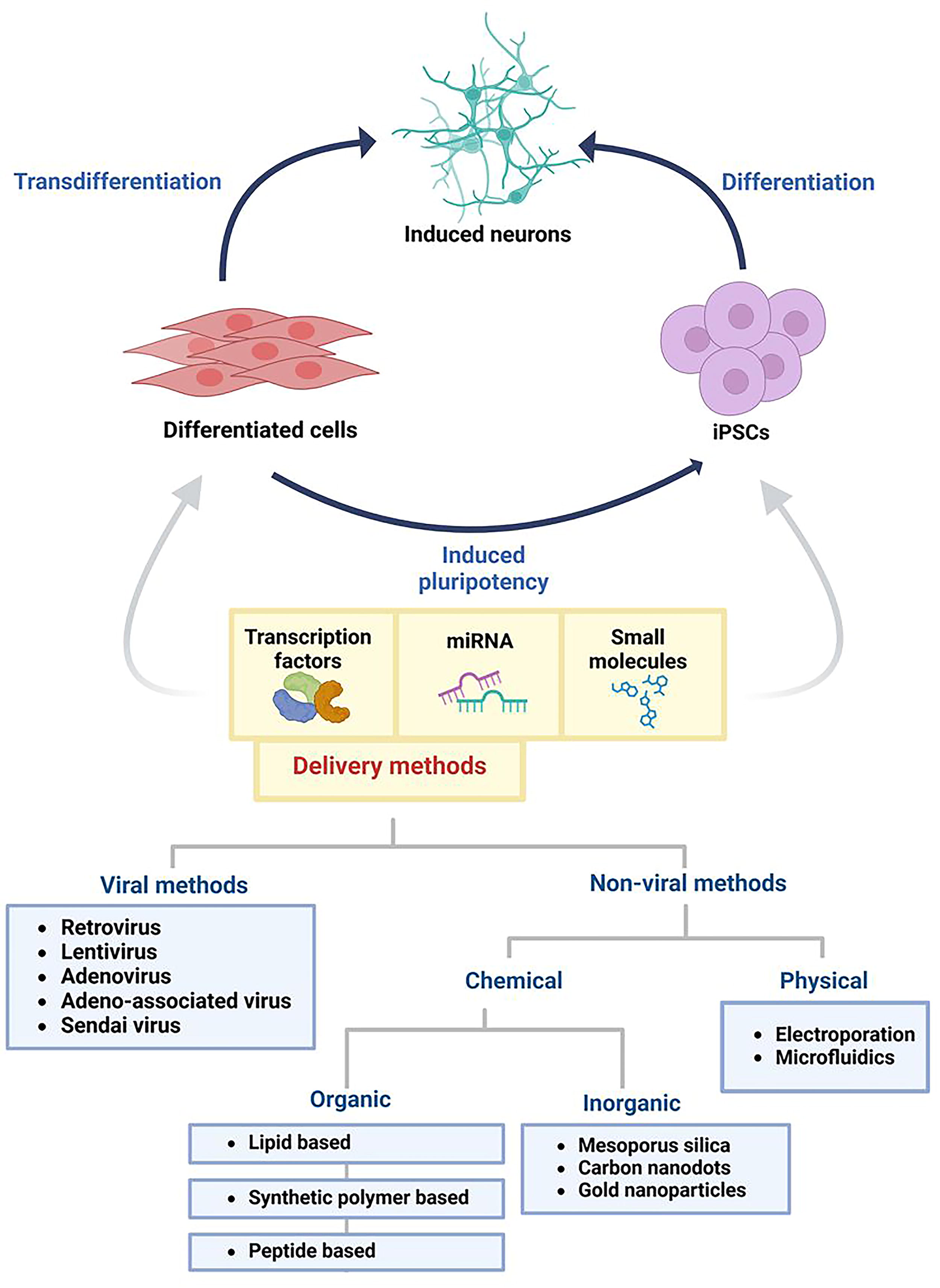
Schematic representation of cell reprogramming strategies and delivery methods to generate induced neural cells. Cell reprogramming can be obtained from terminally differentiated cells or iPSCs. Direct reprogramming converts a terminally differentiated cell directly into another functional cell type without passing through an intermediate pluripotent state, making it faster and more efficient and suitable for both ex vivo and in situ applications. Reprogramming, direct or indirect (via iPSCs), is enabled by the ectopic expression of specific transcription factors, microRNAs, or small molecules, which activate pathways that drive differentiation to a specific phenotype. These molecules have been delivered through different approaches based on viral and nonviral systems, which have generated neural cells with different reprogramming efficiencies. iPSCs, induced pluripotent stem cells. Created with biorender.com.
Reprogramming factors delivered by viral vectors in vitro
Retroviral and lentiviral vectors represent key tools for gene delivery in neuronal direct cell reprogramming (Table 1). A significant advantage of these vectors is their long-term stable gene expression without the need for repeated administration (Giorgetti et al., 2012; Han et al., 2012). Particularly, lentiviral vectors have been employed to induce transdifferentiation of different somatic cell types into neurons. Indeed, unlike most retroviruses, they have high transduction efficiency even in nonproliferating cells, becoming particularly attractive for clinical applications (Milone and O’Doherty, 2018).
Viral Vector Delivery
iDAN, induced dopaminergic neuron; iN, induced neuron; iNP, induced neural progenitor; iNSC, induced neural stem cell; MEFs, mouse embryonic fibroblasts.
In 2010, Vierbuchen and colleagues identified the lentiviral cocktail required to obtain induced neurons (iNs) from mouse embryonic fibroblasts (MEFs), consisting of the TFs Ascl1, Brn2, and Myt1l and reaching a conversion efficiency of 19.5% (Vierbuchen et al., 2010). Subsequently, other research groups used lentiviral vectors to induce both neuronal differentiation from iPSCs and transdifferentiation from somatic cells such as hepatocytes, cord blood cells, and fibroblasts, via overexpression of TFs (Caiazzo et al., 2011; Giorgetti et al., 2012; Marro et al., 2011; Pang et al., 2011; Theka et al., 2013), even reaching a nearly 100% conversion of iPSCs to iNs just with the overexpression of the TF Ngn2 (Zhang et al., 2013).
Furthermore, lentiviruses have been also employed for miRNA overexpression and modulation of miRNA circuits by PTB downregulation, thereby promoting the acquisition of the neuronal phenotype (Ghasemi-Kasman et al., 2015; Xue et al., 2013; Yoo et al., 2011). This process is further enhanced by co-expression of specific TFs, which significantly increase the rate of conversion and maturation of somatic cells into neurons (Victor et al., 2014).
Despite retroviral and lentiviral vectors being among the most used tools for gene delivery (Vargas et al., 2016), they have a significant limitation related to their ability to integrate and permanently alter the genome of the host cell (Hong et al., 2013). Particularly for in vivo application, genotoxicity raises serious concerns, as random gene integration could activate oncogenes or disrupt essential genes (Hacein-Bey-Abina et al., 2003; Ott et al., 2006; Varas et al., 2009; Winkler et al., 2010).
To avoid issues related to insertional mutagenesis, nonintegrating viruses, such as adenoviruses (AdVs) and Sendai virus (SeV), have been applied to directly convert cells into neurons (Lu et al., 2013; Meng et al., 2012).
AdVs can carry large DNA fragments (up to 36 kb) and exhibit exceptional infectivity (Bulcha et al., 2021). However, as they do not integrate into the host genome, their gene expression could be insufficient to sustain the protein levels required to initiate and complete transdifferentiation (Meng et al., 2012; Shi et al., 2014). This limitation could contribute to the low efficiency of direct reprogramming. The conversion of fibroblasts into neurons by adenoviral administration of Ascl1, Brn2, and Ngn2 obtained only 3% of neurons after 13 days (Meng et al., 2012). However, this can be improved if AdVs are codelivered with chemical compounds or if more TFs are used at once (Shi et al., 2014).
SeV is another nonintegrating vector, which is characterized by high transduction efficiency, rapid and strong gene expression, and no risk of genomic integration. In addition, temperature-sensitive engineered SeV can be used to remove residual viral components from the reprogrammed cells through a short temperature shift (Ban et al., 2011). However, the reprogramming efficiency into iNs using this vector is only 0.1% (Lu et al., 2013).
Overall, nonintegrating vectors offer significant safety advantages by minimizing the risk of genotoxicity and avoiding permanent changes to the host cell genome. Nevertheless, their efficiency in cellular reprogramming is substantially lower than the one achieved using integrating viruses.
Viral vectors are still challenging for in vivo applications because of their safety issues (Volodina and Smirnikhina, 2025). Nevertheless, they can be used ex vivo to generate neurons before their in vivo transplantation (Caiazzo et al., 2011; Dell’Anno et al., 2014; Lu et al., 2013; Shi et al., 2014). This strategy could offer an alternative for treating neuronal disorders since the autologous patient-derived cells can be reprogrammed into transplantable neurons, thus reducing risks associated with both viral delivery and immune rejection (Gao et al., 2024).
Reprogramming factors delivered by nonviral vectors in vitro
Nonviral gene delivery can be classified into physical and chemical methods (Soltani Dehnavi et al., 2022). Physical methods, such as electroporation, microinjection, and ultrasound, directly introduce naked nucleic acids into target cells. Chemical methods employ organic or inorganic molecules to protect and transport nucleic acids into cells.
Physical delivery methods
Physical methods (Table 2) exploit external forces to facilitate the entry of nucleic acids into the cell. Electroporation is the most used physical delivery method to engineer cells; it employs an electric field to temporarily permeabilize the cell membrane, allowing the entry of genetic material (Fig. 2). Despite its simplicity, this method presents several problems: cell death and phenotype alterations caused by the application of harmful electric fields, stochastic transfection, and low intracellular delivery efficiency (Fang et al., 2020). All this results in low reprogramming efficiency (Capetian et al., 2016; Tanabe et al., 2018; Wang et al., 2013). To address these challenges, Gallego and colleagues designed nanochannel electroporation, a single-cell transfection system, that enables in situ electroporation on cultured cells via a 3D nanochannel array. This system uses localized electric fields, through electrophoresis, to deliver TFs directly into the cytosol using a dosage that leads to minimal cell damage and impact on cell physiology. Although this approach represents an improvement in electroporation strategies, the overall direct neuronal reprogramming efficiency is still only ∼2% (Gallego-Perez et al., 2016). External physical forces can be combined with other delivery systems to improve reprogramming efficiency. For example, mechano-stimulation can be performed via microfluidics that cause a rapid mechanical deformation, resulting in transient membrane breaks or holes (Sharei et al., 2013). These changes, together with the overexpression of neurogenic TFs, improve the efficiency of neuronal induction by about 7-fold compared with the nondeformed control (Song et al., 2022). Similarly, nanogrooved substrates facilitate the direct conversion of MEFs into dopaminergic neurons (Yoo et al., 2015). Also, triboelectric stimulation, for example, the generation of electrical charges resulting from friction between two different materials, accelerates and enhances the conversion of murine fibroblasts, both in vivo and in vitro (Jin et al., 2016). In addition, Jin and colleagues studied the effects of a three-dimensional environment on direct neuronal reprogramming, showing that the decellularized human brain extracellular matrix boosts plasmid-based direct conversion of fibroblasts into iNs (Jin et al., 2018).
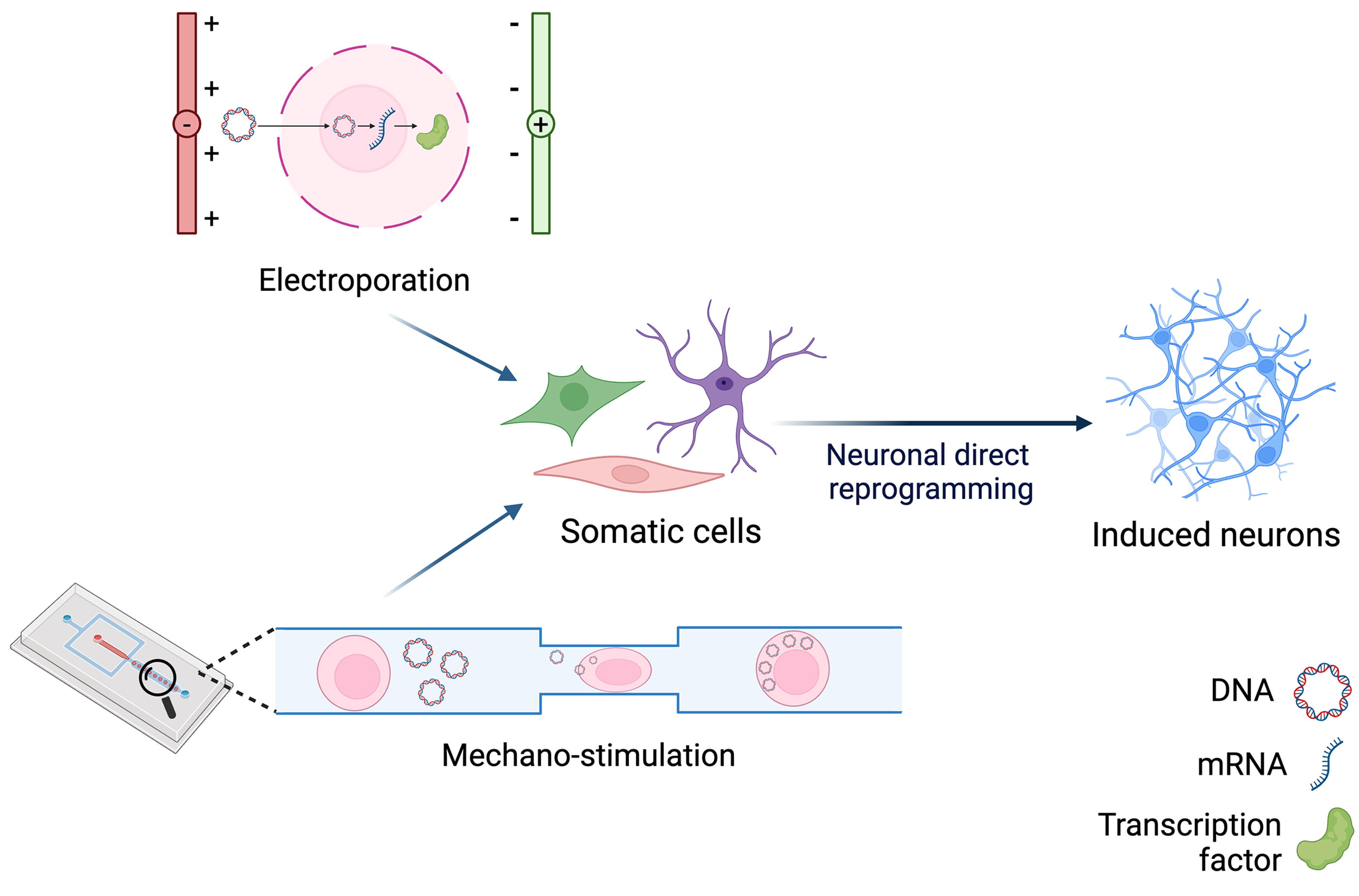
In vitro neural direct reprogramming with physical delivery systems. Electroporation and mechano-stimulation are physical transfection methods that have been used in the field of direct neuronal reprogramming. Electroporation consists in the generation of an electric field that leads to the formation of temporary pores on the cell surface, enabling the delivery of nucleic acids inside the cell. Mechano-stimulation can be performed via microfluidic systems, in which cells pass through microchannels with constrictions smaller than their diameter. This causes mechanical deformation that leads to membrane breaks, allowing the transfection of nucleic acids. Created with biorender.com.
Physical Delivery
Chemical delivery methods
Chemical vectors provide protection and stability to cargo molecules due to their interaction with the vector. In contrast to physical methods, these vectors also allow targeted cell delivery by exploiting their chemical and physical properties. They can be composed of organic (lipids, amino acids, or synthetic polymers) or inorganic materials (metals or ceramics) as listed in Table 3 and represented in Figure 3.
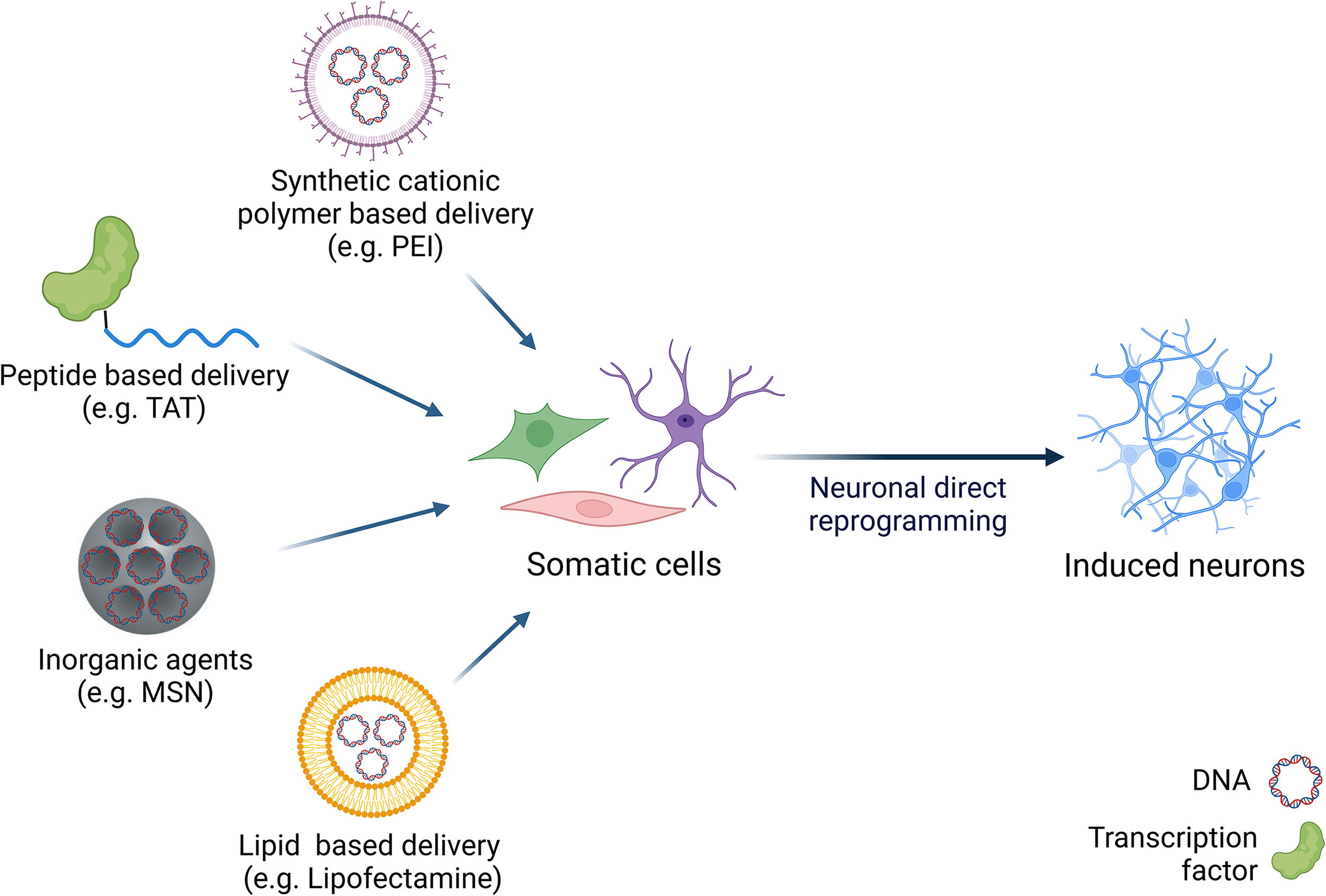
In vitro neuronal direct reprogramming with chemical systems. Several chemical transfection methods have been adopted in direct neuronal reprogramming. In peptide-based approaches, cell-penetrating peptides, such as Transactivating transcriptional activator (TAT), interact with proteins, enabling their transport inside the cells via transient aqueous pores or endocytosis. In synthetic polymer-based delivery systems, cationic functional groups interact with nucleic acid-forming polyplexes that mask the negative charges of DNA by electrostatic interaction, allowing cellular transfection. PEI is an example of a synthetic cationic polymer used for delivering plasmids in direct neuronal reprogramming. Lipid based approach is the most common method for delivering nucleic acids. A representative example is lipofectamine, composed of a cationic lipid that binds nucleic acids, helping its entrance into the cell. Inorganic agents can also be employed in nucleic acids delivery in direct reprogramming. MSNs were used to encapsulate the small molecule ISX-9, while the outer surface was functionalized with plasmid DNA coding for TF. MSN, mesoporous silica nanoparticles; PEI, polyethyleneimine; TF, transcription factor. Created with biorender.com.
Chemical Vectors
PolyN, polymer-functionalized nanodot.
Peptide-based delivery methods
The peptide-based approach enables the delivery of proteins into cells, bypassing the need for transcription and translation. Cell-penetrating peptides (CPPs), consisting of 5–30 amino acids, facilitate the intracellular transport of conjugated molecules that would otherwise be unable to cross the cell membrane (Dowaidar, 2024). This process typically involves transient aqueous pores or endocytosis (Dowaidar, 2024). Several types of CPPs, such as TAT or 30Kc19-derived peptides, have been employed to deliver TFs into cells to induce a neuronal phenotype (Table 4). However, the success of protein transduction could be limited by the low stability of recombinant factors in culture medium and their inefficient endosomal release, requiring many administrations and often resulting in reprogramming efficiency of less than 10% (Hu et al., 2014; Mirakhori et al., 2015; Robinson et al., 2018; Ryu et al., 2020; Seo et al., 2017).
Peptide-Based Delivery
iDPC: induced dopaminergic neural progenitor-like cell.
Synthetic cationic polymer-based delivery methods
Synthetic cationic polymer-based drug delivery systems are appreciated for their biocompatibility, low toxicity, and biodegradability. Their functional groups (hydroxyls, amines, and carboxylic acids) allow fine tuning of several features such as: 1) forming polyplex with nucleic acids by electrostatic interactions, 2) chemical modifications with cross-linking moieties providing stability to the nanoparticle core, 3) reduction or delay of clearance by the mononuclear phagocyte system, 4) enhanced endosomal escape (van den Berg et al., 2021).
In 2012, Adler and colleagues demonstrated the low toxicity and high transfection efficiency of poly(amidoamine) p(CBA-ABOL). This gene carrier allowed for repeated dosing to maintain high expression levels of the transgenes necessary for generation of iNs (Adler et al., 2012).
Afterward, Yu and coworkers developed a vector based on ethylenediamine-modified polymer derived from Porphyra yezoensis (Ed-PYP) for the co-delivery of plasmids encoding the TFs Ascl1, Brn4, and Tcf3 into MEFs, reaching 80% of cells positive for NeuN staining even though with unproven neuronal functional properties (Yu et al., 2017).
Because of the electrostatic interaction with plasmid DNA and efficient endosomal release via the “proton sponge” effect (Behr, 1997), cationic polymer-based vectors demonstrated a higher transfection efficiency compared with other nonviral delivery agents such as lipofectamine and polyethyleneimine (PEI) and a lower toxicity, allowing repeated administration (Adler et al., 2012; Yu et al., 2017).
Inorganic chemical agent-based methods
Inorganic agents have also been successfully employed in neuronal direct cell reprogramming. Mesoporous silica nanoparticles were used to encapsulate the small molecule ISX-9, while the outer surface was functionalized with plasmid DNA coding for TFs. This system ensured high transduction efficiency in vitro and remarkable endosomal escape capacity, resulting in the ∼80% conversion of MEFs into dopaminergic neurons, albeit with an immature morphological and electrophysiological phenotype (Chang et al., 2018). Similarly, polymer-modified nano-reagent (polymer-functionalized nanodot, PolyN) complexed with plasmid DNA encoding for TFs showed reduced cytotoxicity and increased gene expression compared with conventional transfection agents such as PEI. This is attributed to the neutral surface charge of PolyN, which increases transfection efficiency and transgene overexpression, reaching an efficiency of 15% of induced neural stem cells and minimal cell toxicity (Park et al., 2021).
Inorganic agents can also be combined with physical methods and viral vectors to achieve neuronal direct cell reprogramming. For example, gold nanoparticles, when exposed to specific ranges of electromagnetic fields, facilitate the conversion of fibroblasts into dopaminergic neuronal cells via lentiviral overexpression of TFs. This process upregulates genes encoding histone acetylation complex proteins, enhancing H3K27 acetylation and the activation dopaminergic genes (Yoo et al., 2017).
Lipid-based delivery methods
Lipids are amphiphilic molecules with both polar (hydrophilic) and nonpolar (hydrophobic) components, which makes them ideal for transporting different molecules, including nucleic acids.
Cationic lipids are among the most used types of lipids in gene delivery. Their positive charge allows them to bind to the phosphate groups of nucleic acids, forming stable complexes known as lipoplexes. A representative example of a lipid-based gene delivery system is lipofectamine (Felgner et al., 1987). Lipofectamine is mainly composed of two types of lipids: 1) 1,2-dioleoyl-sn-glycerophosphoethanolamine (cationic lipid): essential for encapsulation and protection of nucleic acids, facilitating their transport into the target cell; 2) 1,2-dioleoylphosphatidylethanolamine (neutral lipid): plays a key role in interacting with the cell membrane and in the process of endosomal escape. Its conical shape leads to the formation of a structure called nonbilayer hexagonal phase (H_II), which facilitates the release of genetic material into the cytoplasm (Cullis and Felgner, 2024). Lipofectamine is commonly used as a nucleic acid transfectant in vitro and has also been successfully employed for the generation of iNs to deliver synthetic mRNA (Connor et al., 2018; Goparaju et al., 2017). However, the use of lipofectamine in vivo has significant limitations, mainly because of the toxicity associated with cationic lipids. Indeed, the permanent positive charges of cationic lipids can cause high cytotoxicity and activate the complement system, leading to their rapid elimination from the blood circulation after intravenous administration (Chonn et al., 1991; van etten et al., 1998).
In vivo neuronal direct cell reprogramming delivery systems
Neuronal direct reprogramming could represent a turning point for central nervous system (CNS) disorder therapies (Man et al., 2018; Yavarpour-Bali et al., 2020). Indeed, several studies showed that neurons obtained from in vitro reprogramming can be successfully transplanted in vivo (Caiazzo et al., 2011; Colasante et al., 2015; Lu et al., 2013; Shi et al., 2014; Theka et al., 2013; Wang et al., 2013).
Intriguingly, over the last decade, in vivo neuronal direct cell reprogramming has gained great attention since it could reduce the risks associated with conventional cell therapy. This approach could avoid drawbacks associated with transplantation (Wan and Ding, 2023). For this purpose, glial cells are considered prime targets because of their wide distribution throughout the CNS and their origin from the same neural precursor cells (Lei et al., 2019). In vivo reprogramming of resident glial cells into neurons has been explored in several disorders (Fig. 4), including ischemic injury, traumatic brain injury, epilepsy, Huntington’s disease, Alzheimer’s disease, and Parkinson’s disease (Chen et al., 2020; Guo et al., 2014; Lentini et al., 2021; Rivetti Di Val Cervo et al., 2017; Wu et al., 2020).
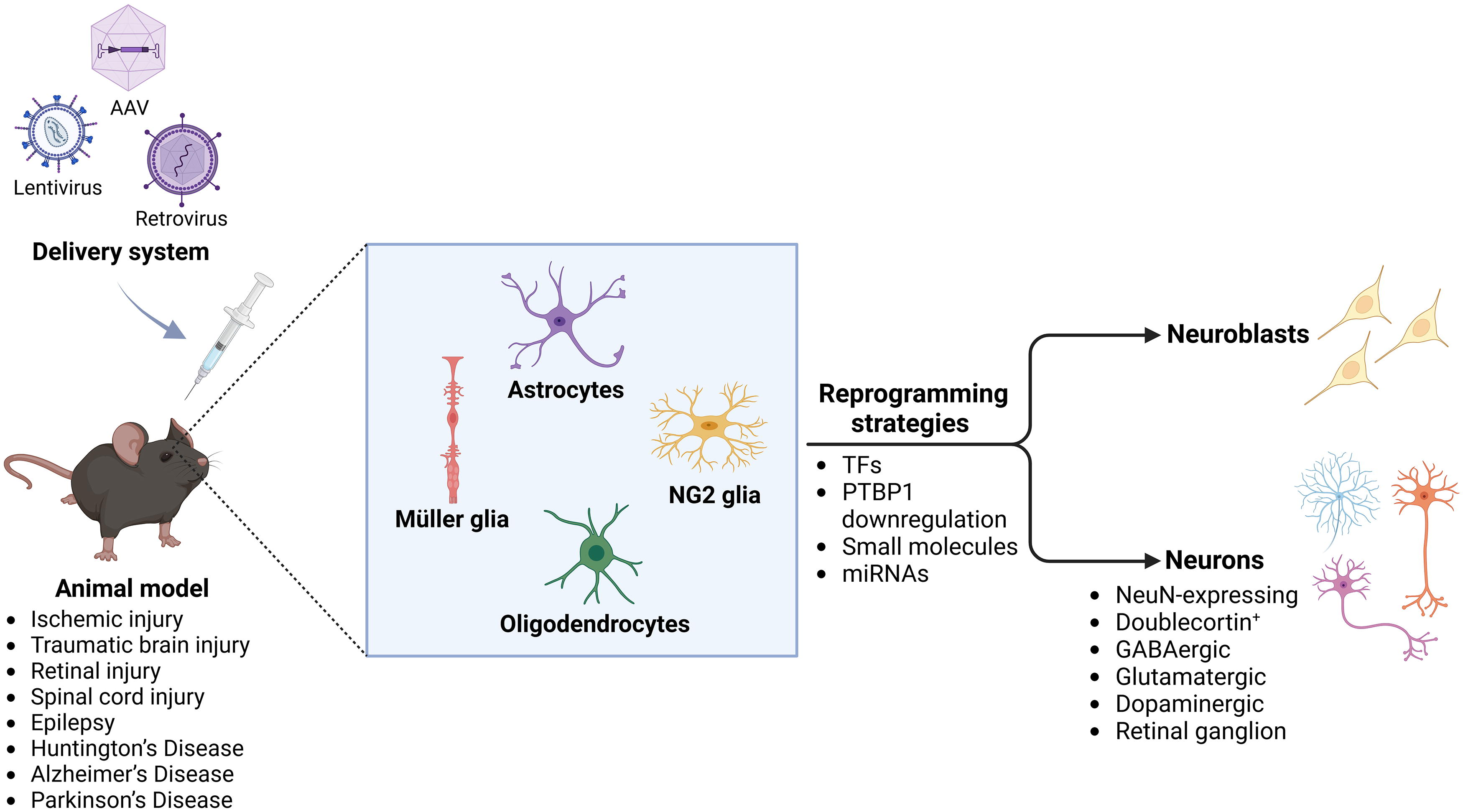
Delivery systems and strategies for in vivo neuronal reprogramming. In vivo neuronal reprogramming has been investigated in various CNS disorders, including ischemic injury, traumatic brain injury, retinal injury, spinal cord injury, epilepsy, Huntington’s disease, Alzheimer’s disease, and Parkinson’s disease. This approach enables the generation of neuroblasts and neurons (i.e., NeuN expressing, Doublecortin+, retinal ganglion, GABAergic, glutamatergic, and dopaminergic neurons) from resident glial cells, such as astrocytes, oligodendrocytes, NG2 glia cells, and Müller glia cells. The most used delivery system is viral transduction based on the use of AAV, lentivirus, and retrovirus. The reprogramming strategies are based on: a) overexpression of TFs alone or in combination, b) downregulation of the protein PTBP, c) use of small molecules, and d) use of miRNAs. AAV, adeno-associated virus; miRNAs, microRNAs; PTBP, polypyrimidine tract-binding protein 1; TFs, transcription factors. Created with biorender.com.
In vivo neuronal direct cell reprogramming can be achieved via different strategies (Table 5). Among those, lentivirus injection in the CNS has initially been used to deliver TFs and miRNAs (Ghasemi-Kasman et al., 2015; Su et al., 2014; Torper et al., 2013). However, the risk associated with virus integration is an obstacle for therapeutic application. To this end, studies in the field of in vivo neuronal direct reprogramming are implementing AAVs because they rarely integrate into the genome (Soltani Dehnavi et al., 2022; Wang et al., 2024) and they have a low pathogenic profile in humans, exerting lower inflammation and high transduction rate (Chen et al., 2020; Mattugini et al., 2019; Nayerossadat et al., 2012; Soltani Dehnavi et al., 2022). Moreover, AAVs offer the advantage of having broad capsid diversity, which allows for cell type-specific tropism, thereby increasing gene delivery to the desired target and also enabling them to cross the blood–brain barrier (BBB) (Wang et al., 2024). Moreover, they have already been approved for the treatment of some genetic diseases, further validating their safety and efficacy (Liu et al., 2024).
In Vivo Neuronal Reprogramming Strategies
AD, Alzheimer’s disease; PD, Parkinson’s disease.
On the other hand, AAVs have a rather limited carrying capacity (∼4.7 Kb) and can cause serious adverse effects if administered at high doses because of possible preexisting immunity (Hinderer et al., 2018; Khabou et al., 2018). In addition, AAVs can produce false-positive results because of local viral leakage into pre-existing neural cells other than glial cells. Thus, the apparent astrocyte-converted neurons could not be originated from the resident astrocytes but from the infected endogenous neurons. In fact, Wang et al. (2021b) reported that AAV-mediated NeuroD1 expression was evident in neurons in vivo, but these could not be traced back to astrocytes. Similarly, they showed that despite efficient knockdown of PTBP1 in vivo, genetically traced resident astrocytes did not convert into neurons. Accordingly, Chen et al. (2022) reported that PTBP1 knockdown did not convert astrocytes into neurons, but they observed AAV leakage into nearby neurons (Chen et al., 2022).
Thus, although in vivo neuronal reprogramming has a promising future, the safe and efficient delivery of reprogramming factors remains a critical challenge.
Future Directions
Currently, about 56% of advanced therapy medicinal products in clinical trials use viral vectors, including retroviruses, lentiviruses, AdVs, and AAVs (Ginn et al., 2024). Recent developments also permit targeted therapy for a specific cell type that reaches more efficiently the CNS and could be implemented in neuronal reprogramming approaches. For example, new AAV serotypes with the capacity to target brain cells, among which glial cells, have been recently identified and could be adapted to neuronal direct cell reprogramming (Campos et al., 2023; Giacomoni et al., 2024). The targeting of specific brain cells could be improved by employing cell type-specific promoters (Mseis-Jackson et al., 2024) or by implementing miRNA detargeting approaches as already shown by Bido and colleagues (Bido et al., 2024).
However, viral clinical safety is a matter of debate because of some inherent limitations, such as the tendency to stimulate immunogenic responses and the risk of insertional mutagenesis, inefficient biodistribution, and the difficulty of repeated administration (Butt et al., 2022).
Therefore, in order to facilitate in vivo direct neuronal cell reprogramming toward clinical practice, nonviral systems are likely to be increasingly implemented (Bocchi et al., 2022). In this context, lipid nanoparticles (LNPs, Fig. 5) are emerging as an innovative and promising solution for nonviral delivery of drugs and gene therapy, also serving as an efficient carrier for RNA delivery (Cullis and Felgner, 2024). LNPs offer numerous advantages as nonviral vectors for gene delivery: they avoid significant immune responses, allowing repeated treatments, and are easy to produce on a large scale (Wang et al., 2023). They also could have less genetic loading restrictions than lentivirus and AAVs (Cullis and Felgner, 2024). The number of mRNA molecules carried by a single LNP depends on the size of both the mRNA and the LNP with a theoretical packaging potential higher than 10 Kbp (Bepperling and Richter, 2023). This feature could enable the potential co-delivery of different transgenes within the same LNP (Ball et al., 2018).
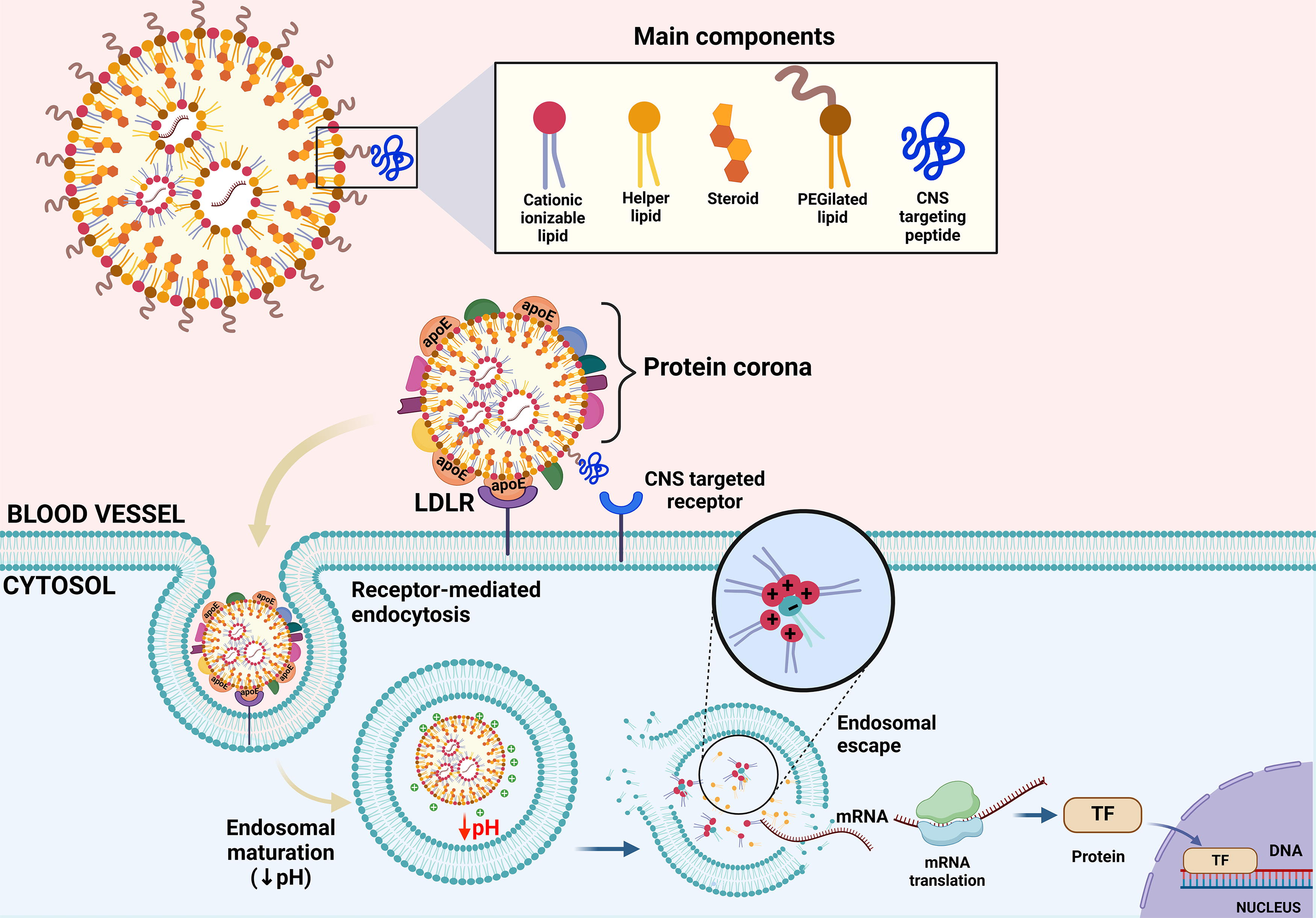
Representation of LNP structure and mechanism. LNPs are composed of (1) ionizable cationic lipids, which remain neutral at physiological pH and acquire a positive charge in the acidic endosomal environment (pH 5.5–6.5), promoting endosomal escape and mRNA release into the cytoplasm by electrostatic interactions between positively charged lipids and negatively charged phospholipids in the endosomal membrane; (2) phospholipids and cholesterol, which enhance LNP stability and facilitate interaction with target cells; and (3) PEGylated lipids, which form a protective surface coating to evade immune detection and extend circulatory retention. In addition, peptides conferring tissue-specific targeting can be conjugated to the nanoparticle. Upon intravenous administration, plasma proteins form a protein corona on the LNP surface, influencing biodistribution, cellular uptake, and intracellular trafficking. For example, apolipoprotein E is one of the most abundant proteins in the corona, interacting with LDLR and promoting endocytosis. LDLR, low-density lipoprotein receptor; LNP, lipid nanoparticles; TF, transcription factor. Created with biorender.com.
Another advantage of LNPs is their modifiable composition, which allows, theoretically, precise targeting of organs or tissues, improving therapeutic efficacy and reducing systemic toxicity (Cullis and Felgner, 2024; Tenchov et al., 2021). Functionalization of LNPs with targeting ligands is a promising strategy to achieve therapeutically relevant amounts of the drug in the brain tissue even after systemic administration (Khare et al., 2023). Within this range of possibilities, neuronal reprogramming strategies may involve LNPs conjugated with targeting peptides that bind CNS-specific receptors, also facilitating BBB crossing (Han et al., 2025). Thus, LNPs could play a key role in developing new delivery systems for therapeutic RNA targeting the CNS (Han et al., 2025; Tsakiri et al., 2022).
Nonviral delivery approaches for RNA therapy have already made major advances in safe and efficient delivery (Hoy, 2018; Patel et al., 2022); however, a recurrent limitation is the need for multiple dosing. To address this, recent progress in sustained RNA expression using self-amplifying or circular RNAs offers a potential solution (Chen et al., 2023; Lundstrom, 2025).
Overall, neuronal direct cell reprogramming shows promising biomedical potential, but it is still in the process of identifying the optimal delivery systems for clinical applications. Nonviral vectors could address current challenges faced in neuronal reprogramming. Unraveling the requirements for safe and successful delivery, both in vitro and in vivo, is crucial to envision a future in which neuronal reprogramming could be used for treating neurodegenerative and neurological disorders.
Footnotes
Authors’ Contributions
G.R.: Writing—original draft, writing—review and editing. F.D.P.: Writing—original draft, writing—review and editing. S.C.: Writing—review and editing. M.P.: Writing—review and editing. A.d.B.: Writing—original draft. M.H.A.M.F.: Writing—review and editing. M.C.: Conceptualization, writing—review and editing, funding acquisition.
Author Disclosure Statement
The authors declare that no conflicting financial interests exist.
Funding Information
We acknowledge financial support by European’s Union’s Horizon 2020 research and innovation programme under grant agreement n. 964497 (ENLIGHT) and under the National Recovery and Resilience Plan (NRRP), Mission 4, Component 2, Investment 1.1, Call for tender No. 1409 published on September 14, 2022, by the Italian Ministry of University and Research (MUR), funded by the European Union’s NextGenerationEU Project Title: “Modulation of dopaminergic transmission through microRNAs as a measure to counteract dopamine neuronal degeneration”—CUP: E53D23018200001, Grant Assignment Decree No. 1065 adopted on July 18, 2023, by the Italian Ministry of Ministry of University and Research (MUR).
