Abstract
The osteogenic differentiation of mesenchymal stem cells (MSCs) is strongly related with the inflammatory microenvironment. The ability of osteogenic differentiation of MSCs is vital for the bone tissue engineering. Interleukin (IL)-10, a well-known anti-inflammatory factor, plays a key role in tissue repair. Dental pulp stem cells (DPSCs), with the advantage of convenience of extraction, are suitable for the bone tissue engineering. Therefore, it is meaning to explore the effects of IL-10 on the osteogenic differentiation of DPSCs. The proliferation activity of DPSCs were evaluated by MTS assay (CellTiter 96® Aqueous One Solution Cell Proliferation Assay [Promega]) and real-time polymerase chain reaction (RT-PCR). The osteogenic differentiation of DPSCs were determined by Alizarin Red staining, RT-PCR, and alkaline phosphatase activity test. The glucose metabolism was detected by Mito Stress test and glycolysis assay. IL-10 (10 or 20 nM) could enhance the osteogenic differentiation of DPSCs and promoted the metabolic switch from glycolysis to oxidative phosphorylation (OXPHOS), whereas IL-10 (5 and 50 nM) has no obvious effects on the osteogenic differentiation of DPSCs. The OXPHOS inhibitor restrained the promotion of osteogenic differentiation induced by IL-10. These findings show that IL-10 can promote the osteogenesis of DPSCs through the activation of OXPHOS, which provides a potential way for enhancing the osteogenic differentiation of DPSCs in bone tissue engineering.
Introduction
Mesenchymal stem cells are widely present in many tissues, including bone marrow, adipose tissue, and dental pulp, which are considered as potential cells for tissue engineering and stem cell therapy (Mansano et al., 2021; Samsonraj et al., 2017; Zhou et al., 2021). Dental pulp stem cells (DPSCs), as a unique population of mesenchymal stem cells (MSCs), possess multiple differentiation potentials and ease of access, which are widely studied in tissue engineering and clinical applications (Mansano et al., 2021; Zhou et al., 2020). Bone defects are a worldwide and challenging clinical problem which cause a serious burden for health care. It has been reported that DPSCs have obvious therapeutic effect on bone defects (Lee et al., 2019; Lim et al., 2020; Silva et al., 2016).
Consequently, it is meaningful and essential to explore the related factors of osteogenic differentiation of DPSCs and to improve the osteogenic differentiation of DPSCs.
Recently, many studies demonstrate that cell metabolism regulates the function of immune cell and MSCs (Morganti et al., 2020; Salazar-Noratto et al., 2020). Studies show that the metabolic switch from glycolysis to oxidative phosphorylation (OXPHOS) happens when MSCs undergo the process of osteogenic differentiation (Guo et al., 2020; Shares et al., 2018). OXPHOS supplies amounts of energy for the MSCs to exert its function. In addition, the disorder of metabolic switch will alleviate the osteogenic differentiation of MSCs and the decrease of bone formation (Shares et al., 2018). Therefore, cellular metabolism is closely associated with the property of MSCs. However, the relationships between cell metabolism and the osteogenic differentiation of DPSCs is still unknown.
Interleukin (IL)-10 is a cytokine with anti-inflammation immune modulating function, which plays a crucial role in immune defense and many diseases (Fang and Zhu, 2020; Wei et al., 2019). It has been reported that IL-10 delivered by MSCs are capable to attenuate inflammatory and autoimmune diseases (Peruzzaro et al., 2019; Shao et al., 2020). IL-10 could modulate immune cell activity and function. In inflammatory microenvironment, the application of MSCs is able to reduce the amount of leukocyte and neutrophil percentage through IL-10 signaling (Hervas-Salcedo et al., 2021; Lu et al., 2019). However, the effects of IL-10 on the osteogenic differentiation of MSCs have rarely been investigated. Therefore, in this study, we will deeply evaluate the function of IL-10 on the osteogenic differentiation and metabolic function of DPSCs.
Materials and Methods
Cell culture
DPSCs were isolated and obtained from human impacted third molars from six healthy adults (18–26 years) at the Southern University of Science and Technology. The study and procedure were approved by the Ethics Committee of the Southern University of Science and Technology. All patients have signed the informed consent and know about the usage of the teeth. Concisely, the dental pulp was separated from teeth and was digested in a solution of collagenase type I (3 mg/mL; Sigma-Aldrich) and dispase II (4 mg/mL; Sigma-Aldrich) (Anitua et al., 2018, 2019). Then cells were cultured in α-Minimal Essential Medium (Gibco) complemented with 10% fetal bovine serum (Gibco), 100 μg/mL streptomycin (Gibco), and 100 U/mL penicillin (Gibco) at 37°C in 5% CO2.
Cell viability assay
Cells were seeded in 96-well plates at the density of 5 × 103 cells. The cell viability was evaluated by the colorimetric MTS assay (CellTiter 96® Aqueous One Solution Cell Proliferation Assay [Promega]). Cells were treated with IL-10 in different concentrations (0, 5, 10, 20, and 50 nM). After culturing for 2 days, the cells were incubated with 20 μL MTS reagent for 3 hours at 37°C. The optical density of supernatant was recorded at the wavelength of 490 nm by microplate reader (Bio-TEK).
Alizarin Red S staining
DPSCs were cultured with osteogenic induction media (Gibco) for 14 days and fixed with 4% paraformaldehyde. Then cells were washed and stained by Alizarin Red S solution (Sigma). To qualify the mineralized nodule, the mineralization was quantified by 10% (w/v) cetylpyridinium chloride (Sigma-Aldrich). Absorbance of the extracted solution was measured at 570 nm.
Alkaline phosphatase activity assay
After osteogenic differentiation of 7 days, the alkaline phosphatase (ALP) activity of DPSCs was detected by the ALP Activity Kit (Beyotime Biotechnology, China), according to the manufacturer's instructions. Briefly, cells were lysed and incubated with chromogenic substrate at 37°C. Then the absorbance at 405 nm was recorded by microplate reader.
Glycolysis assay
After cells were treated at the supposed time, cell supernatant was collected and analyzed for glycolysis assay. According to the manufacturer's protocol, glucose consumption, ATP (adenosine triphosphate) production, and lactic acid production were detected with assay kits (Nanjing Jiancheng, China).
Real-time polymerase chain reaction
Gene expression was detected by the real-time polymerase chain reaction (RT-PCR) with the RT-PCR Kit (Beyotime Biotechnology), following the manufacturer's protocol. Total RNA was extracted and reversed into cDNA by the RT-PCR Kit. RT-PCR was performed with recommended condition and cycles. Amplification conditions were set as follows: 95°C for 10 minutes, denaturation at 95°C for 15 seconds (45 cycles), annealing at 60°C for 20 seconds, and extension at 72°C for 20 seconds. Primers used in this article are listed in Table 1.
Real-Time Polymerase Chain Reaction Primers
Seahorse Mito Stress test
Cells (3 × 104 cells/well) were plated in XF96 cell culture microplates (Agilent Technologies), according to the manufacturer's recommendations. For oxygen consumption rate (OCR) test, oligomycin, carbonyl cyanide p-(trifluoromethoxy) phenylhydrazone, antimycin A (AA), and rotenone were added. For extracellular acidification rate (ECAR) test, glucose, oligomycin, and 2-deoxy-glucose were used. The parameters were calculated by Seahorse XF96 instrument.
Statistical analysis
The SPSS 20.0 software package (SPSS, Inc., Chicago, IL) was used for the statistical analysis. Data were analyzed using one-way analysis of variance followed by Tukey's post hoc test. A p < 0.05 was considered statistically significant. Data were expressed as the mean ± standard deviation. All data were collected for at least three independent experiments.
Results
IL-10 in 10 nM promotes the proliferation of DPSCs
DPSCs were isolated and cultured (Supplementary Fig. S1). According to the MTS assay, it exhibited that IL-10 (10 and 20 nM) could enhance the proliferation of DPSCs (Fig. 1A), while IL-10 (5 and 50 nM) has no obvious effects on the proliferation of DPSCs. qPCR results showed that IL-10 in 10 nM promoted the expression of c-Myc, Sox2, and Nanog, whereas IL-10 in 50 nM inhibited Nanog expression (Fig. 1B).
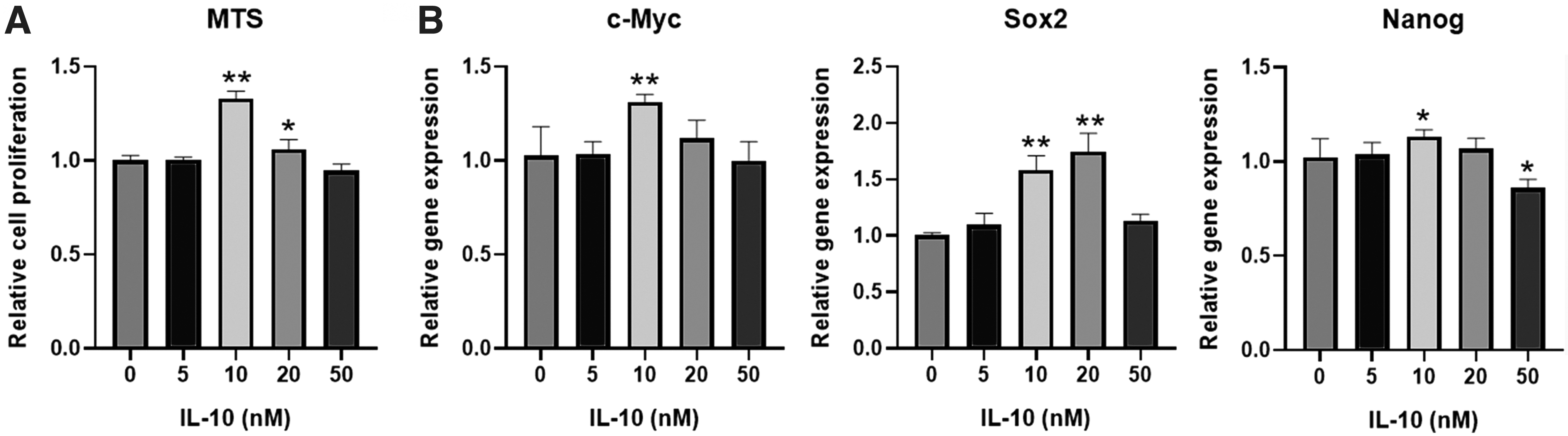
The effects of IL-10 on the viability of DPSCs. The cell proliferation of DPSCs under the IL-10 treatment was evaluated by MTS assay
IL-10 activates the OXPHOS of DPSCs
The effects of IL-10 on the glucose metabolism of DPSCs were evaluated. OCR test manifested IL-10 in 10 and 20 nM improved the ratio of OXPHOS in DPSCs (Fig. 2A). Compared with other groups, IL-10 in 50 nM increased the ECAR value and the production of lactic acid (Fig. 2B). In addition, the glucose consumption in IL-10 treatment (10, 20, and 50 nM) was higher than other groups, and the ATP production is increased in IL-10-treated groups (5, 10, 20, and 50 nM; Fig. 2C–E)
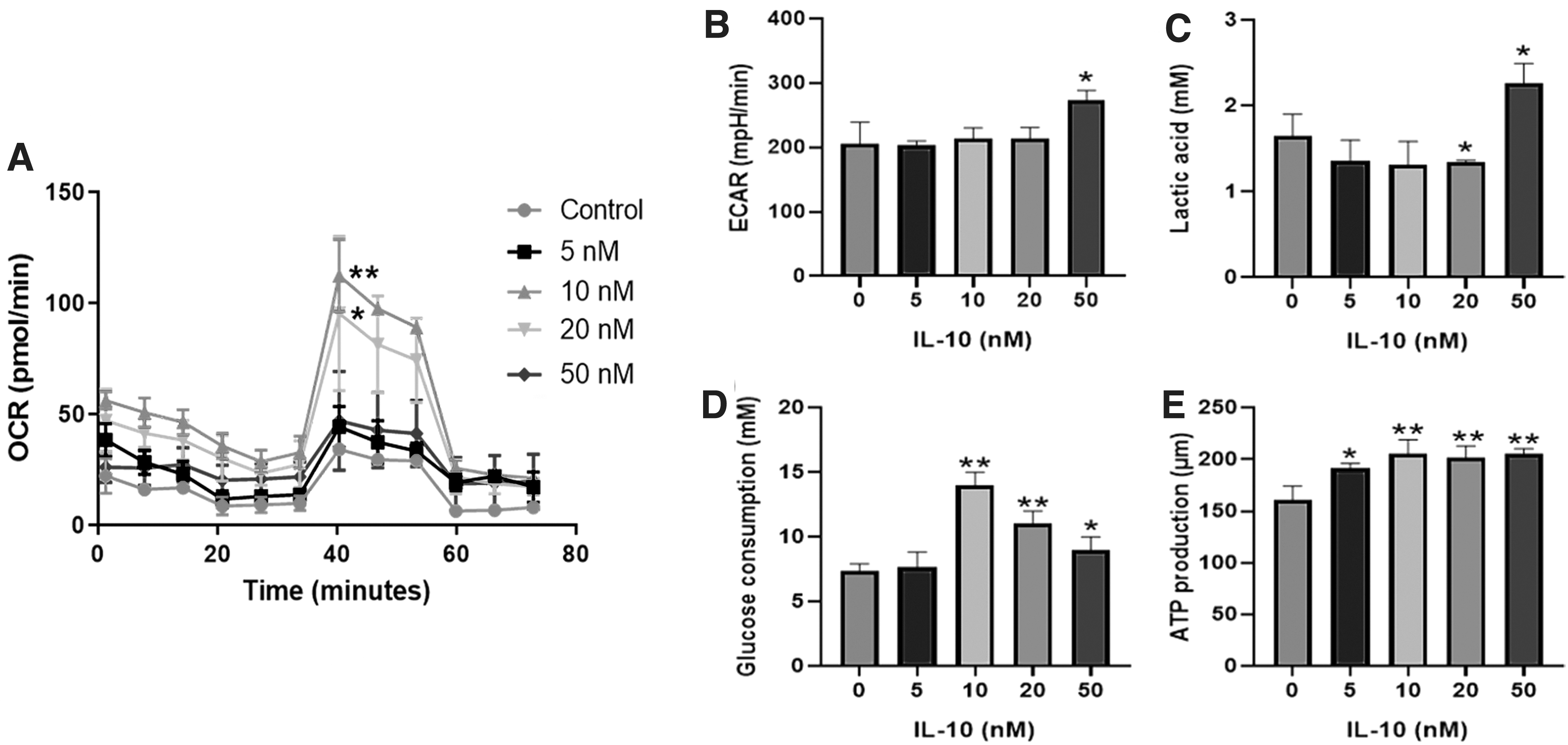
The effects of IL-10 on the glucose metabolism of DPSCs. The OCR and ECAR value of DPSCs were tested by seahorse analyzer
IL-10 improves the osteogenic differentiation of DPSCs
From the results of Alizarin Red staining and ALP staining, IL-10 in 10 nM could significantly promote the osteogenic differentiation of DPSCs, while IL-10 (5, 20, 50 nM) has not obvious effects on the osteogenic differentiation (Fig. 3A, B). In addition, RT-PCR test showed that IL-10 in 10 or 20 nM increased the expression of osteogenic genes (BSP, RUNX2, and BMP4; Fig. 3C).
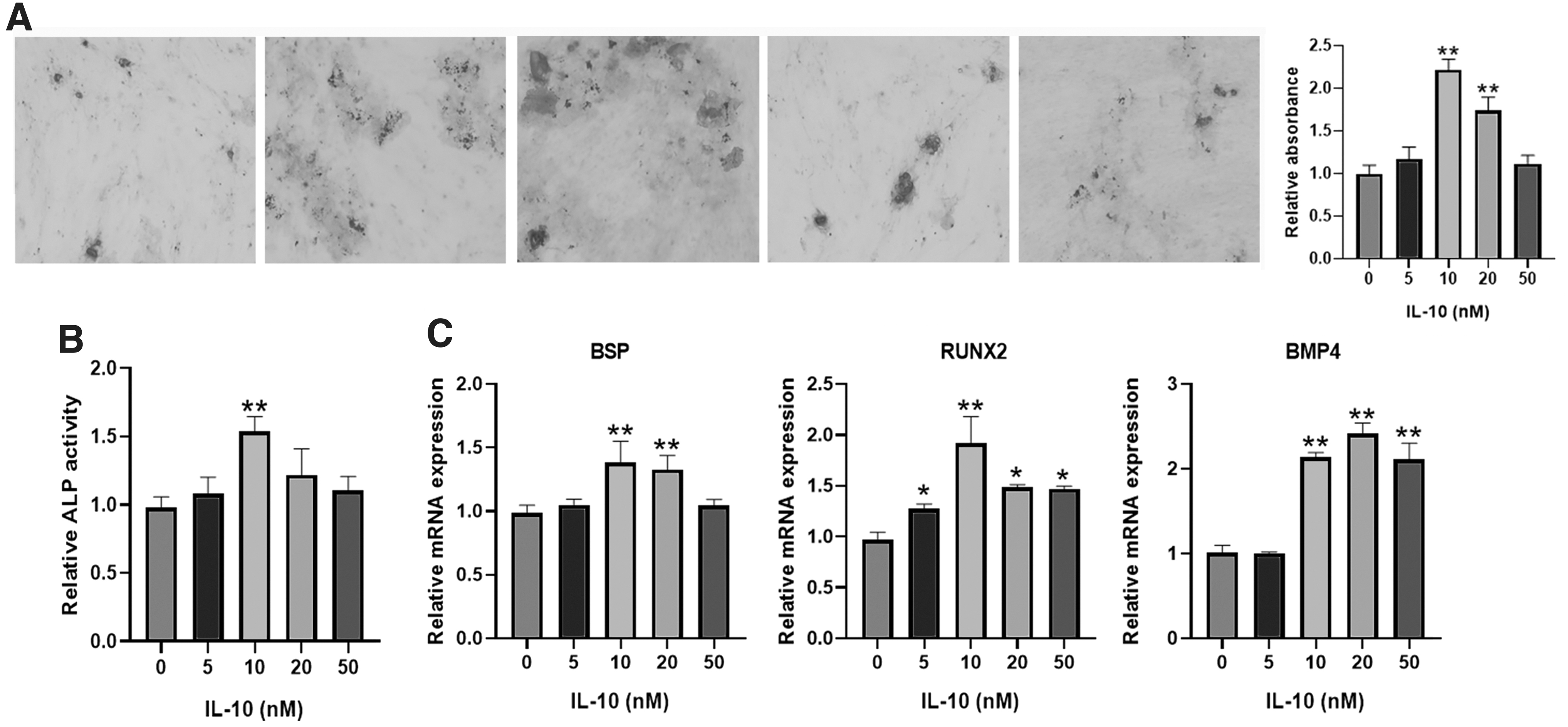
The impact of IL-10 on the osteogenic differentiation. The Alizarin Red staining
AA inhibited the osteogenic differentiation promoted by IL-10
To further assess the function of IL-10 (10 nM) on the OXPHOS and osteogenic differentiation, AA was used. The application of AA inhibited the promotion of OXPHOS by IL-10 (Fig. 4A). AA restrained the promotion of osteogenic differentiation induced by IL-10 (Fig. 4B, C). In addition, AA suppressed the expression of osteogenic genes (Fig. 4D). Western blotting results showed that IL-10 increased the ratio of P-ERK/ERK (phosphorylated extracellular signal-regulated kinase/extracellular regulated protein kinase) and NF-κB/β-actin (nuclear factor kappa B/β-actin), whereas the addition of AA alleviated the activation by IL-10 (Fig. 4E, F).
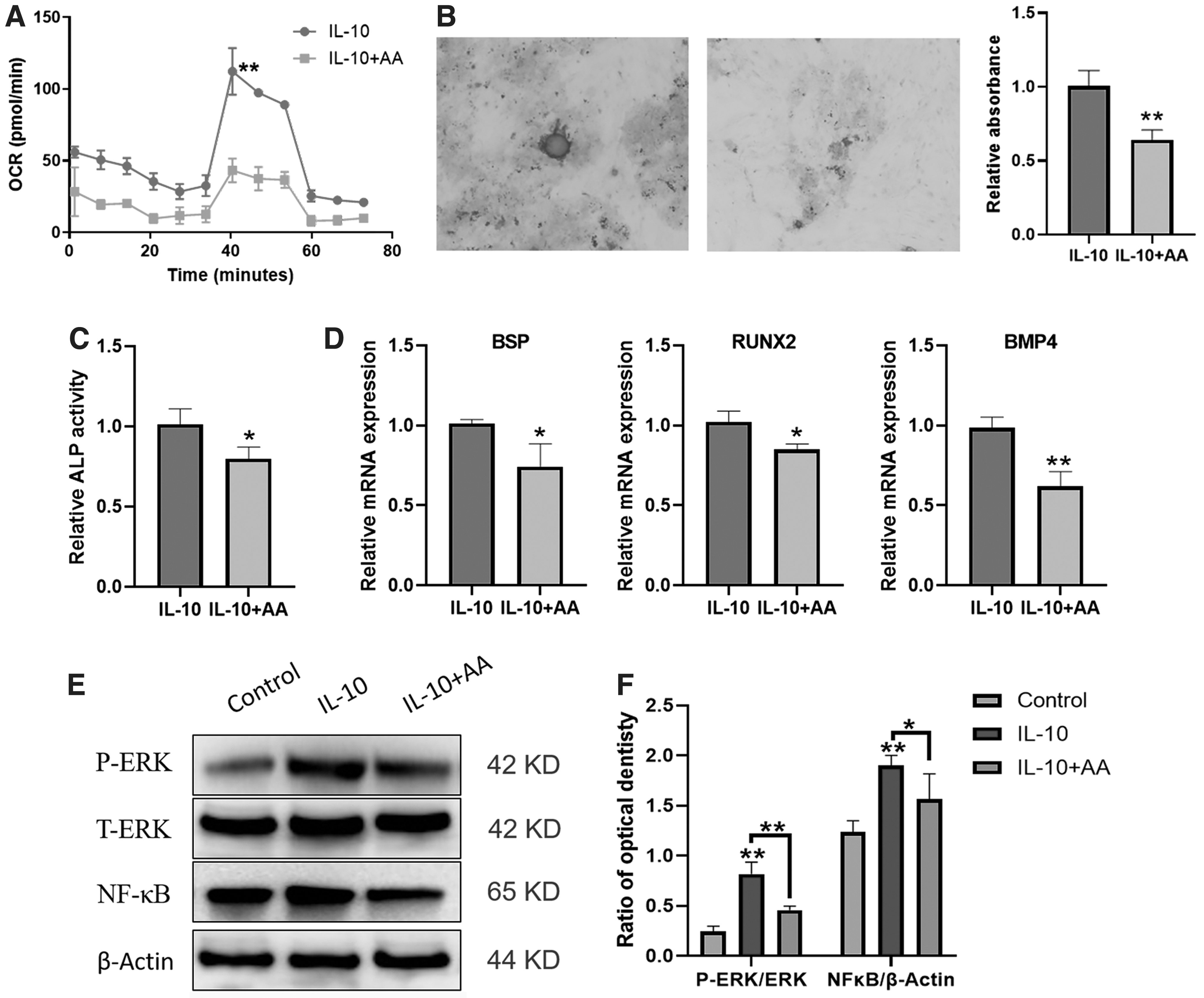
AA reduces the promotion induced by IL-10. The OCR value of DPSCs treated by IL-10 and AA
Discussion
Currently, due to the higher proliferative potential than BMSCs, DPSCs have been explored and used in the bone regeneration and tissue engineering widely (Hosmani et al., 2020; Huo et al., 2021). DPSCs exhibit greatly, excellent biocompatibility in clinical therapy. Therefore, the regulation and improvement of DPSCs is necessary for the application of bone regeneration. In our study, we found that IL-10 improves the osteogenic differentiation of DPSCs through enhancing the OXPHOS.
IL-10, an anti-inflammatory factor, is vitally important for inflammatory repair and bone homeostasis. Studies show that IL-10 knockout mice show bone abnormalities, including the decrease of bone mineral density and bone resilience (Holgersen et al., 2015; Sasaki et al., 2019). It has been reported that IL-10 dampens periodontitis and bone loss, which is caused by the activation of IL-17 (Sun et al., 2020).
In addition, IL-10 influences the osteogenic differentiation of BMSCs in a concentration-dependent manner (Chen et al., 2018). Low physiologic concentration of IL-10 enhances the osteogenesis of BMSCs through the activation of p38/MAPK signaling pathway (Chen et al., 2018). High dose of IL-10 inhibits osteogenesis of BMSCs through the activation of NF-κB pathway. Similarly, in our study, results showed that IL-10 (10 or 20 nM) could enhance the osteogenic differentiation of DPSCs with more mineralized nodule.
During the proliferation and differentiation process, BMSCs demand lots of energy to maintain the function. Therefore, the glucose metabolism is strongly associated with the proliferation and differentiation potential of BMSCs. In psoriasis disease, the abnormal glucose metabolism inhibits the proliferation and vascular differentiation of MSCs (Zhao et al., 2021). Study shows that MIR-34a suppresses osteogenic differentiation of BMSCs through glycolysis inhibition by targeting lactate dehydrogenase-A (Hong et al., 2020). The activation of glycolysis can facilitate adipogenic differentiation of BMSCs by regulating β-catenin signaling and mitochondrial fusion and fission (Guo et al., 2020). In this research, data demonstrated that IL-10 activates the OXPHOS of DPSCs, which can promote the osteogenic differentiation of DPSCs.
Therefore, the function of IL-10 on the MSCs and immune cell may be through metabolic regulation. The osteogenic differentiation of MSCs is mediated by multiple pathways (Diomede et al., 2019; Marycz et al., 2020). Some studies demonstrate that ERK and NF-κB pathway are related to the osteogenic differentiation of DPSCs (Diomede et al., 2020; Sinjari et al., 2019). In our study, we also found that IL-10 could activate ERK/P-ERK/NF-κB pathway in DPSCs, whereas the addition of AA could inhibit the activation.
In addition, the activation of immunosuppression of BMSCs is accompanied by the metabolic reconfiguration of BMSCs (Jitschin et al., 2019; Liu et al., 2019). In inflammatory environment, BMSCs enhances glycolysis and fatty acid oxidation through STAT1 glycosylation to inhibit the activation of T cells (Jitschin et al., 2019). IFNβ increased glycolytic capacity in MSCs and ATP production, which directly promote MSCs' immunomodulatory function and therapeutic efficiency (Vigo et al., 2019). IL-10 plays a vital role in inflammatory environment, which accelerates the alleviation of inflammation and tissue repair (Wang et al., 2020). Therefore, IL-10 also modulates the immunoregulatory ability of DPSCs through metabolism switch.
Conclusion
IL-10 improves the osteogenic differentiation of DPSCs through the activation of OXPHOS. AA inhibited the OXPHOS of BMSCs, which weakens the role of IL-10 in promoting the osteogenesis of DPSCs.
Footnotes
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Disclosure Statement
The authors declare they have no conflicting financial interests.
Funding Information
This work was supported by the Natural Science Foundation of Shenzhen Science and Technology Innovation Commission (CYJ20190807145815129).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
