Abstract
Abstract
Recently, we showed a natural reprogramming process during infection with
Introduction
T
In a mouse model that mimics early ML infection of adult peripheral nerves, we recently showed that Schwann cells from adult peripheral nerves undergo a reprogramming process in response to intracellular ML (iML) and convert infected Schwann cells to highly immature progenitor/stem cell–like cells (pSLCs), which are more suitable for bacterial dissemination (Masaki et al., 2013). In Schwann cells, ML turn off Schwann cell differentiation/myelination-associated genes and reactivate developmental-associated genes/transcription factors, changing cell fate to pSLCs over time. The established methods of cell reprogramming of adult somatic cells, such as fibroblasts to pluripotent stage or cell fate change from one somatic cell type to another by ectopic overexpression of a few defined transcription factors (TFs), are complex processes (Baeyens et al., 2005; Davis et al., 1987; Ieda et al., 2010; Takahashi and Yamanaka 2006; Vierbuchen et al., 2010; Zhou et al., 2008). It is likely that iML-induced reprogramming of Schwann cells is even more complex due to the fact that the ML bacillus is equipped with a plethora of highly biologically active components and each and every bacterial component or their combined effects may have the capacity to activate many biological events in Schwann cells, including cells' defense reactions that may contribute to both reprogramming and to pathological events during early infection. In this regard, it is interesting that innate immune or inflammatory pathways that are triggered by viral vectors used for TF-induced conversion of embryonic fibroblasts to induced pluripotent stem cells (iPSCs) have been linked to effective cell reprogramming (Lee et al., 2012). These findings suggest that host cells' defensive responses to viruses are likely to involve increased transcriptional competence, resulting in the expression of genes that are normally shut down in somatic cells. However, unlike viral vectors, natural infection with whole ML bacilli is likely to produce a full spectrum of highly complex cellular and defensive reactions in Schwann cells to adapt to pathogenic challenges, which in turn may be associated with increased transcriptional competence and subsequent modulation of gene expression driving a wide range of cellular activities, including cell reprogramming. However, such biological processes that precede or accompany Schwann cell reprogramming following ML infection are unknown.
Here, we show that, in addition to downregulating Schwann cell lineage transcripts and reactivating developmental genes, ML rapidly induce a large number of immune-related genes comprising mostly innate immunity and chemokine-associated genes right from the very early stage of Schwann cell infection and peaking in their expression when Schwann cells have changed their cell identity to pSLCs. We also validated some of these immune gene products—chemokines/cytokines at the protein level as secreted soluble factors during the reprogramming process. The functional evidence for the acquisition of such immune-related factors by reprogrammed Schwann cells is shown by their ability to attract macrophages, thus showing a possible role of immune responses and their associated signaling during Schwann cell reprogramming.
Materials and Methods
Preparation of primary Schwann cells from adult peripheral nerves
Schwann cells were separated from adult CD-1 male mice (4–6 weeks old, ICR strain code 022) and purified using a magnetic cell sorting system, as described earlier (Masaki et al., 2013). In brief, peripheral nerve tissues were isolated in minimal essential medium (MEM; Invitrogen), and connective tissues and dorsal root ganglia were carefully removed. The tissues were digested with 0.125% trypsin (Invitrogen)/0.05% EDTA and 0.1 mg/mL collagenase I (Worthington Biochemical) passed through a 100-μm mesh nylon filter (BD Falcon). The cells were collected and seeded on a laminin-coated T25 flask and were passaged and cultured in Schwann cell medium (Masaki et al., 2013) and subjected to purification by a magnetic cell sorting system using anti-p75 antibody (AB1554, Millipore) according to manufacturer's protocol (Miltenyi Biotec). The enriched cells were further purified by FACS Vantage dual-laser flow cytometer (BD Biosciences) using anti-p75 antibody, and clonal cells were prepared from p75+ cells as described (Masaki et al., 2013).
ML infection of mouse Schwann cells and reprogramming
Purified Schwann cells were grown in collagen-coated T75 flasks (BD Biosciences) and infected with ML. Reprogrammed cells were generated according to our previous protocol (Masaki et al., 2013).
Immunofluorescence and microscopy
Immunofluorescence was performed as described (Ng et al., 2000). In brief, cells were fixed with 4% formaldehyde (Sigma) for 10 min at room temperature and 100% methanol (Sigma) for 10 min at −20°C. The samples were washed with phosphate-buffered saline (PBS) three times, blocked with 10% goat serum, and incubated with primary antibody for 1 h. After washing and incubation with a secondary antibody conjugated with Alexa Fluor 488 or 594 (Invitrogen), images were captured with Nikon Eclipse 2100, Zeiss LSM710 confocal, and Zeiss AX10 Observer live imaging microscopies.
Gene expression analyses
Microarrays were performed using Affymetrix mouse Genechips according to the manufacturer's protocol as described previously (Masaki et al., 2013). In brief, total RNA was isolated from control uninfected Schwann cells and ML-infected Schwann cells or pSLCs using RNeasy columns (QIAGEN). Affymetrix Test3 arrays and mouse genome MG-430A2 arrays were probed with the cRNA prepared by reverse transcription of the total RNA. Expression analyses of control primary Schwann cells and infection at days 3, 7, 14, and 28, as well as reprogrammed Schwann cells/pSLCs derived from day-28 post infection were analyzed by applying the Robust Multichip Average (RMA) method (Irizarry et al., 2003). Affymetrix proprietary CEL-format files were read into the R statistical programming environment (v. 2.15.2), complemented with the Bioconductor analysis suite (v. 2.12) (Gentleman et al., 2004). To determine a starting list of immune-related probes, a search was performed, iterating through all probes detailed in the Affymetrix Mouse 430A2 microarray annotations library (version 2.8.1), searching for the immune-related gene ontology (GO) term GO:000695-immune response. Any probes included in this annotation were initially included in the analysis. Gene symbols associated with all probes were provided by this same library. For all time points, mean probe detection values of replicates were used. Individual genes were further verified as immune related on the basis of the available data from published literature. For the analysis of immune-related genes affected by infection with ML, an initial threshold of 1 log2-fold change (at any time point) from the control, uninfected time point, was used. The remaining list of probes for genes with canonical immunological function was then used for further analysis. Data shown in Fig. 1 was analyzed according to Masaki et al. (2013).
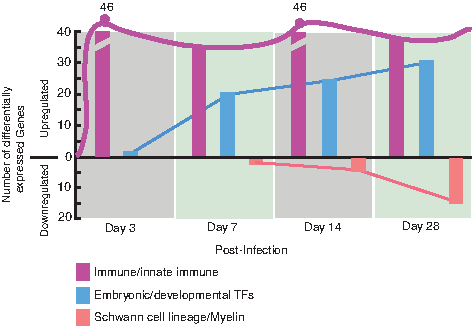
Immune-related genes dominates early
Migration assays and time-lapse and confocal microscopy of macrophage–pSLC co-culture
Isolation of macrophages from adult mouse peritoneal macrophages and co-cultures with green fluorescent protein (GFP)+pSLCs are described in our previous report (Masaki et al., 2013). Briefly, peritoneal macrophages were isolated from adult CD1 mice, centrifuged, and resuspended in RPMI containing 10% fetal calf serum (FCS), 2 mM
Chemokine/cytokine protein expression from infected and reprogrammed cells
Immune factors released from infected Schwann cells at different time points and reprogrammed cells/pSLCs were detected in culture supernatants using cytokine antibody arrays (Proteome Profiler-Mouse Chemokine/Cytokine Array Panel A array Kit, R & D Systems). Both experimental and quantification analyses were performed according to the manufacturer's instructions. Integrated density of each detected protein was calculated using ImageJ.
Results
Innate and inflammatory gene activation in early reprogramming process during ML infection
Infection of lineage-committed Schwann cells with leprosy bacteria results in reactivation of developmental/embryonic transcription factors and downregulation of Schwann cell lineage marker genes, resulting in the reprogramming of infected cells to pSLCs (Masaki et al., 2013). Although these transcriptional changes occur progressively over the course of infection, further transcriptome analysis revealed that numerous immune-related genes were differentially expressed as early as 3 days after infection, and some of these genes continued this expression during reprogramming process (Fig. 1). A heatmap of log2-fold changes relative to the uninfected control time point are presented in Figure 2A, with the genes clustered by Euclidean distance and average linkage for clarity and to facilitate highlighting of genes with similar patterns in change of expression across the time points. The majority of these selected immune gene profiles are related to the innate immune response, and others are members of the chemokine and cytokine families. Selected genes with individual expression levels from the heat map are depicted in Figure 2B. The heatmap shows the progression of the innate immunity changes across the time course. Although several immune genes were upregulated in Schwann cells as early as day 3 and day 7 in response to intracellular ML, they were quickly downregulated over time with a strong downregulation in reprogrammed cells/pSLCs. However, other immune genes were upregulated right from day 3 postinfection and maintained or increased their expression up to the reprogrammed stage (Fig. 2A), suggesting complexity and nonlinearity of this innate immune response signal upon infection on the road to reprogramming.
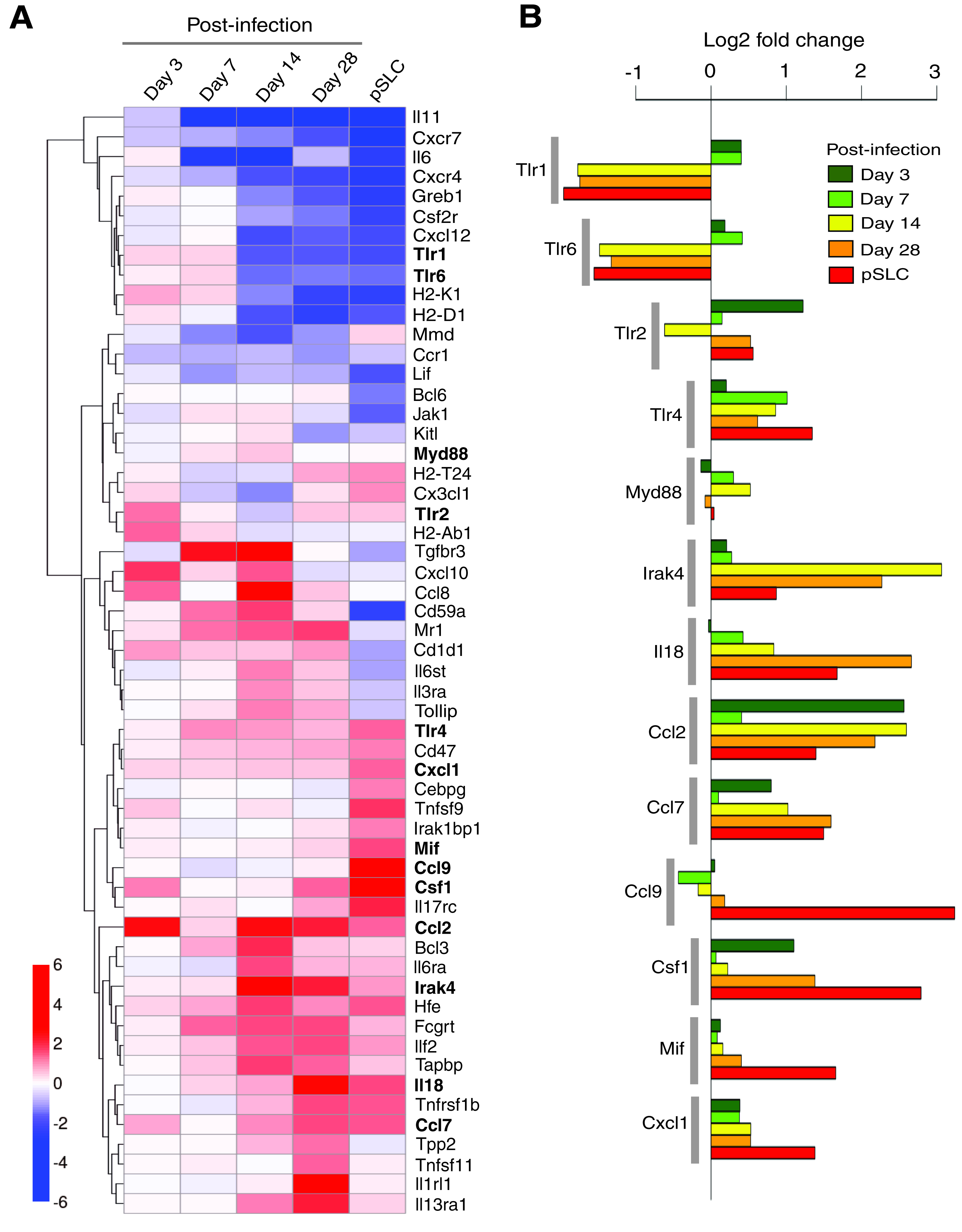
Differentially expressed individual immune-related genes in
Of particular interest is the modulation of genes encoding the Toll-like receptors (TLRs), which are receptors of the innate immune system and recognize pathogen-associated molecules. TLRs are known to sense distinct pathogen-associated molecular patterns (PAMPs) (Iwasaki and Medzhitov, 2010). The recognition of PAMPs by TLRs leads to the recruitment of the adaptor molecules, such as Myd88, and subsequent activation of kinase-driven signaling pathways, leading to the activation of global regulator nuclear factor-κβ (NF-κβ and thereby expression of host immune genes (Dev et al., 2011). Interestingly, TLR1 and TLR2 were upregulated in Schwann cells early in ML infection and then turned off, whereas TLR2, TLR4, and their common adaptor molecule Myd88 remained activated mostly over the course of reprogramming. Although various TLRs exhibit different patterns of expression in response to different PAMP recognition patterns, ML-induced initial and continuous upregulation of various TLRs suggests that Schwann cells, which are typically nonimmune cells, possess the capacity to elicit innate immune response to a greater extent by expressing a large number of innate immune genes in response to ML. However, the functional relevance of TLR gene modulation in relation to reprogramming or nerve pathogenesis associated with ML infection remains to be elucidated.
The signalling cascade coupled to TLR activation is also very similar to that of interleukin-1 receptors (Loiarro et al., 2010). The interleukin-1 receptor–associated kinase 4 (Irak4) gene encodes a kinase that activates NF-κβ in both TLR and T lymphocyte receptor signaling pathways and is a serine/threonine-protein kinase that plays a critical role in initiating innate immune response against foreign pathogens (Loiarro et al., 2010). Interestingly, ML uprgulated the expression of Irak4 in Schwann cells from day 3 and peaked at days 14 and 28 postinfection and maintained its expression in pSLCs, albeit, at a slightly lower level (Fig. 1A, B). It is known that the IRAK4-mediated innate immune response is orchestrated by TLRs and interleukin (IL)-receptor family members, including IL-18 receptor (IL-18R) (Loiarro et al., 2010). Like Irak4, ML also continuously upregulated the IL-18 transcript, which encodes the ligand for IL-18R (Gillespie and Horwood, 1998). Because Irak4 activation is mediated by Myd88 (Loiarro et al., 2010), a common signaling adaptor protein downstream of TLRs and IL-18R, and ML activate key innate immune genes encoding Irak4, TLRs, and Myd88 as well as IL-18R ligand (Fig. 2), it is likely that intracellular ML orchestrate a complex innate immune signaling cascade that might influence other nonimmune gene transcription associated with reprogramming of Schwann cells or begin the inflammatory response that plays a key role in neurodegeneration during infection.
In addition to the essential role played by Irak4 in mediating TLR and IL-receptor signaling, recent studies have also shown that Irak4 is responsible for the production of cytokines, chemokines, and other proinflammatory mediators (Li, 2008). In this regard, there is evidence that plasmacytoid dendritic cell response in rheumatoid arthritis can be repressed by Irak4 kinase inhibition (Chiang et al., 2011), suggesting a therapeutic potential of Irak4 inhibitors for the treatment of autoimmune diseases (Cohen, 2009; Li, 2008). Interestingly, our findings showed that ML induced genes encoding a number of cytokines and chemokines, including those involved in monocytes/macrophage and neutrophil recruitment or activation. These cytokines/chemokines include Ccl2, Ccl7, Ccl9, Csf1, Mif Tnfsf11, and Cxcl1 (Fig. 2A, B). Furthermore, it is worth noting that genes associated with Th2 response (Il13ra, Il1rl1, Il18; Joshi et al., 2010; Kroeger et al., 2009) were also upregulated, suggesting the possibility that ML may induce Th2 response in the microenvironment of infected Schwann cells, which might lead to the initiation of Th2-associated inflammatory responses in the infected peripheral nerves under
Reprogramming process accompanies secretion of immune factor proteins in infected Schwann cells
We next examined if the immune-related gene expression in infected and reprogrammed Schwann cells could be translated to immune factor proteins released by these infected cells. To test this, we collected serum/supplement-free conditioned medium (CM) from Schwann cells at different time points after infection and also from pSLCs. CM from uninfected Schwann cells was used as a control. Soluble proteins released into the medium within 24 h were analyzed and quantified using mouse protein arrays that cover a range of chemokines, cytokines, and immunomodulatory/growth factors. Figure 3 shows the quantitative analysis of mean protein expression profiles of CM from Schwann cells without and with infection at days 14 and 28 as well as reprogrammed Schwann cells/pSLCs derived from day 28 postinfection. Although infected Schwann cells at day 14 show limited release of immune factor protein at low levels, upregulation of MCP1 was significant. MCP1/CCL2 belongs to the CXC subfamily of cytokines and displays chemotactic activity for monocytes and basophils but not for neutrophils or eosinophils (Sica et al., 2000). Unlike mRNA expression of immune genes, their corresponding protein secretion to extracellular medium is a complex process; although proteins may be expressed intracellularly, they may not necessarily be secreted from the cells at a detectable level. Nevertheless, at day 28, infected cells produced a range of chemokines (MCP1/CCL2, MCP5/CCL5, CXCl1, CXCL10, CXCL11, MIG), cytokines/growth factors [IL-6, tumor necrosis factor-α (TNF-α), IL-7, IL-2, macrophage colony-stimulating factor (M-CSF)] to varying degrees, and this trend progressed to reprogrammed cells, which showed the highest number of released immune factors (Fig. 3). Many chemokines and cytokines released from either infected or reprogrammed Schwann cells are of interest because they play major roles in recruitment and activation of macrophages/monocytes and other immune cells as well as initiating a cascade of immune/inflammatory reactions (White et al., 2013). Importantly, many of the released chemokines and cytokines are downstream targets of innate immune signaling exerted by TLRs and their adaptor proteins following detection of pathogens (Qian and Cao, 2013). In this regard, a recent study that shows the role of viral vector–induced innate immune signaling in reprogramming fibroblasts to iPSCs is of particular interest (Lee et al., 2012). It is possible that bacteria-induced TLR responses and subsequent production of chemokines and cytokines and their related signaling pathways could potentially influence ML-induced reprogramming of Schwann cells to pSLCs as well as initiating inflammation within the peripheral nerves during infection.
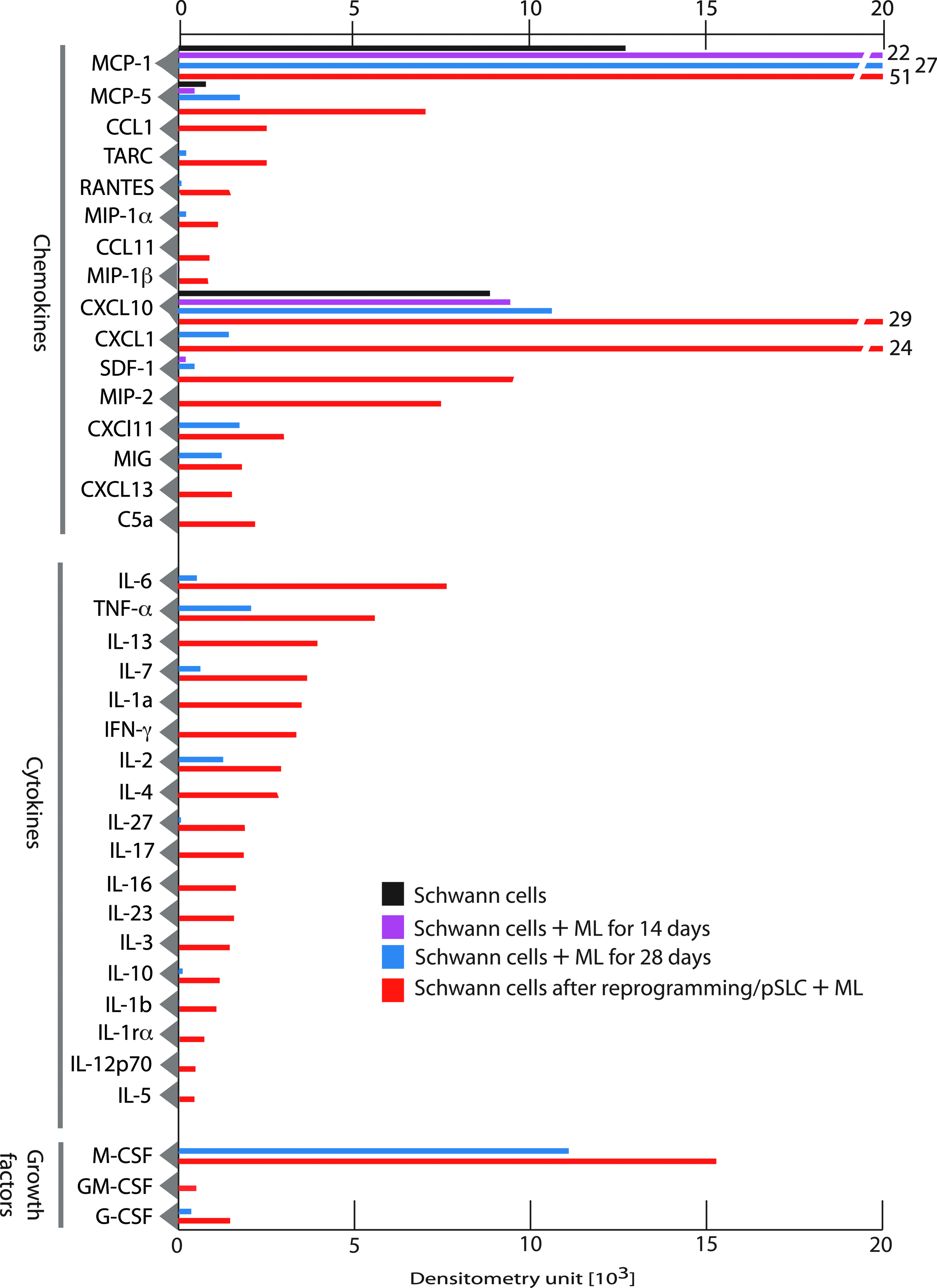
Immune factor proteins are secreted by infected and reprogrammed Schwann cells. Shown is a protein array analysis of conditioned medium from control Schwann cells, infected Schwann cells at day 14 and day 28, and reprogrammed cells/pSLCs derived from day 28-infected Schwann cells. Relative levels of secretory proteins detected by dot blot chemiluminescence were quantified by densitometry following subtraction from signals from the medium alone. Data were presented as mean values from two to three arrays. Note the progressive secretion of chemokines, cytokines, and growth factor proteins during reprogramming. Color images available online at www.liebertpub.com/cell
Released immune factors from reprogrammed cells influence macrophage attraction
During the reprogramming process, pSLCs derived from infected Schwann cells at day 28 postinfection released the highest number of secretory factors (Fig. 3). Because most of these immune factors are chemokines and cytokines that exert immunomodulatory functions and recruit macrophages in inflammatory processes (White et al., 2013), we used both Transwell macrophage migration assays and time-lapse microscopy with co-cultured GFP+ pSLCs with primary macrophages to evaluate their chemotactic activity in response to secreted factors from infected pSLCs. Our live cell imaging data showed that after addition of macrophages to pSLCs individual macrophages displayed a movement toward clustered GFP+ pSLCs in real time. Immunolabeling of fixed co-cultures with macrophage markers CD11b and F4/80 at different points also revealed the accumulation of numerous macrophages around pSLC clusters and macrophage migration directed toward reprogrammed cells. Double labeling with antibodies to ML (anti-PGL1 that detect ML-specific phenolic glycolipid-1) (Ng et al., 2000) and macrophage markers also revealed that migrated macrophages take up ML from infected pSLCs. Because added macrophages were not infected initially and only the GFP+ pSLCs were infected with ML, bacterial transfer to macrophages in high numbers is likely due to macrophage infiltration toward pSLC clusters. Together, these data provide evidence that released immune factors from reprogrammed cells is functional based on their ability to influence macrophage recruitment. In agreement with the chemokine/cytokine production, supernatant from pSLCs attracted a high number of macrophages when a cell migration assay was performed with a Transwell system. In this system, cell supernatant or medium alone was placed in the lower chamber and macrophages were added to upper chamber; migrated macrophages after 24 h were labeled with Crystal Violet dye staining. Figure 4 shows the markedly increased number of migrated macrophages, as demonstrated by Crystal Violet dye staining, under the influence of pSLC medium. It appears that multiple immune factors released from pSLCs are involved in this macrophage migration because a broader CC-chemokine inhibitor alone was insufficient to ablate macrophage migration in response to pSLC conditioned medium (Masaki et al., 2013).
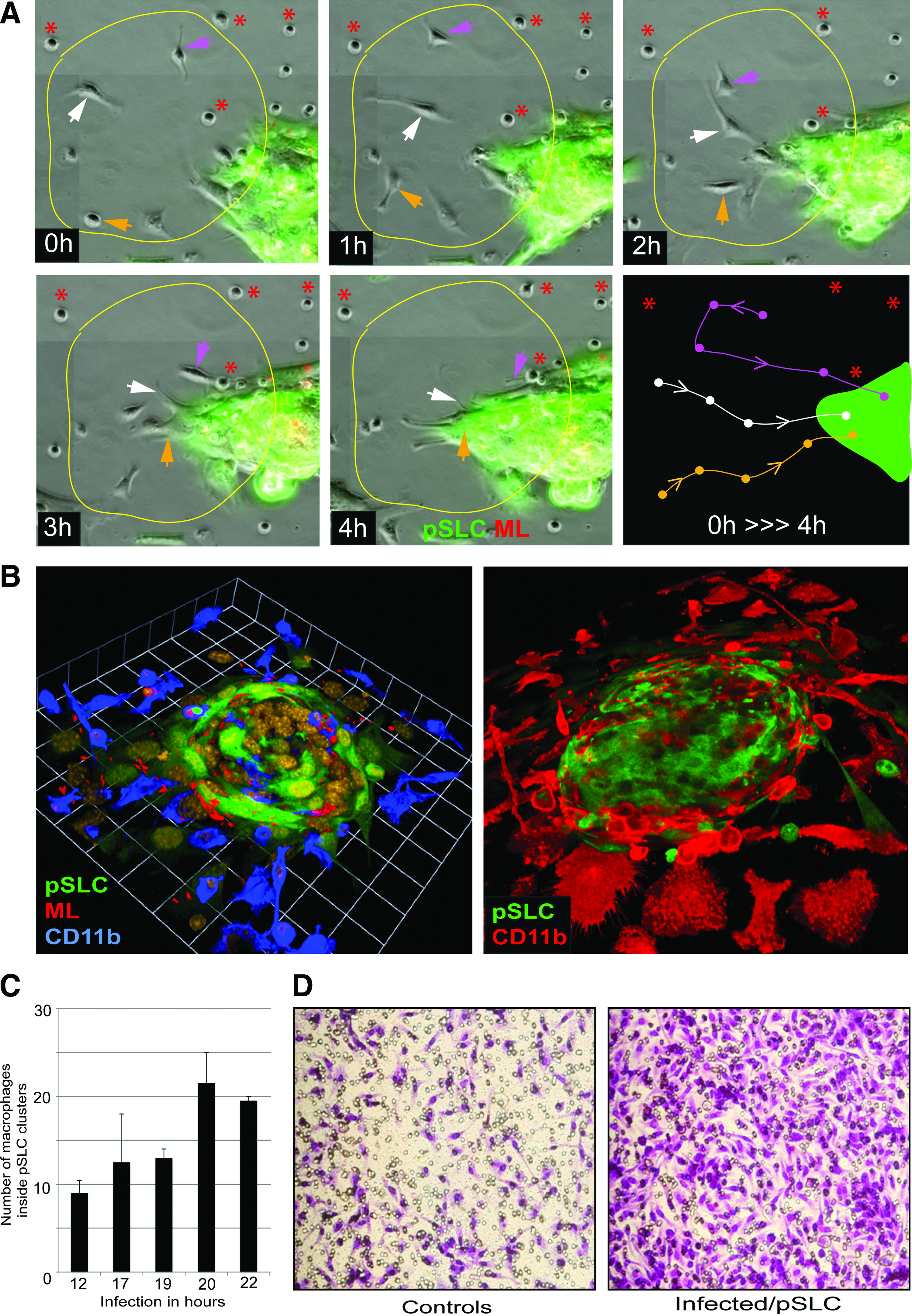
Immune factors secreted from infected reprogrammed Schwann cells/pSLC as chemoattractants for macrophages.
Discussion
Reprogramming lineage-committed tissue cells back to an embryonic stem cell–like stage or to another lineage is a highly complex and long process, involving multiple phases of molecular, biochemical, epigenetic, and structural remodeling events (Buganim et al., 2013). Despite the use of only three to four well-characterized transcription factors to convert fibroblasts to pluripotent iPSCs (Takahashi and Yamanaka, 2006) and the massive accumulation of literature related to iPSC cell research, how exactly these defined three to four transcription factors reprogram parent cells to iPSCs still remains unknown. Therefore, it is expected that reprogramming of primary adult Schwann cells to a stem cell–like state by infection with intact leprosy bacteria, resembling
In natural settings, when host cells are infected with pathogens, the first defensive mechanism of cells, innate immunity, comes into action and it, in turn, generates a flexibility for host cells to express more genes and proteins to resist the infection (Iwasaki and Medzhitov, 2010). Schwann cell infection by ML may also follow the same natural process, with the exception that prolonged infection converts the parent Schwann cells to the pSLC stage. It is possible that the data presented in this report—the initial induction of multiple immune genes and proteins in Schwann cells during ML infection—might be linked to the subsequent reprogramming process. However, detailed analyses are necessary to understand how this multifunctional immune signaling network connects to highly complex bacteria-induced reprogramming. In this regard, it should be noted that an intriguing innate immune/inflammatory link has already been made recently during reprogramming embryonic fibroblasts to iPSCs by a relatively less complicated (as compared to entire intact bacteria) four defined factors–induced reprogramming system (Lee et al., 2012). These studies have shown that activation of TLR3 signaling by viral vectors used for ectopic expression causes fibroblasts to express more genes by loosening the chromatin, thereby facilitating reprogramming in the presence of the four defined transcription factors (Lee et al., 2012). Interestingly, Schwann cells upregulate many TLRs and their adaptor proteins and downstream targets, including chemokines/cytokines during ML infection. How these complex innate immune signaling networks might operate during ML-induced reprogramming needs careful further investigation.
It is also well established that innate immunity is an early response to injury. This also suggests that innate immunity is also very much associated with the regeneration process, which occurs at varying degrees in different adult tissues following injury. Adult peripheral nerves possess a remarkable capacity to regenerate following injury, and dedifferentiated Schwann cells generated during this injury process are largely attributed to this regenerative property (Chen et al., 2007; Fancy et al., 2011; Jessen and Mirsky 2008). Intriguingly, leprosy bacteria appear to hijack this process and generate dedifferentiated Schwann cells by causing initial demyelination to establish the infection, colonize the cells, and subsequently reprogram them to a pSLC-like stage to spread the infection (Masaki et al., 2013; Rambukkana 2010; Rambukkana et al., 2002; Tapinos et al., 2006). In peripheral nerve injury, immune-related genes are also activated in dedifferentiated adult Schwann cells, which serve as repair cells for regeneration process (Nagarajan et al., 2002; Napoli et al., 2012). These include immune factors that activate and attract macrophages and other immune cells. Our studies have revealed that there is a significant overlap between immune-related genes activated during peripheral nerve injury and those activated during ML infection, such as Ccl2, Ccl7, Kitl, and Cxcl10, suggesting that ML may hijack, at least partly, the immune-related signaling network activated after denervation for bacterial advantage. Taken together, studies presented in this report provide valuable information for laying the foundation for further studies concerning the role of the innate immune signaling network in both cellular reprogramming and pathogenesis associated with neurodegenerative diseases.
Footnotes
Acknowledgments
We thank Richard Truman and Ramnuj Lahiri for the provision of
Author Disclosure Statement
The authors declare that no competing financial interests exist.
