Abstract
Abstract
Pluripotent stem cells would have great potential in cell therapies and drug development when genetically matched with the patient; thus, histocompatible cells could be used in transplantation therapy or as a source of patient-specific cells for drug testing. Pluripotent embryonic stem cells (ESCs)—generated via somatic cell nuclear transfer (SCNT) or parthenogenesis (pESC)—are potential sources of histocompatible cells and tissues for transplantation. Earlier studies used the piezoelectric microinjection (PEM) technique for nuclear transfer (NT) in mouse. No specific studies examined zona-free (ZF) NT as an alternative NT method to generate genetically matched ESCs of a nuclear donor. In this study, we compared the efficiency of nuclear transfer-derived ESC (ntESC) line establishment from ZF-NT, ZF-parthenogenetic (PGA), and ZF-fertilized embryos with that of the PEM-NT method. Different nuclei donor cells [cumulus, ESC, and mouse embryonic fibroblast (MEF)] were used and the efficiency of ntESC derivation was investigated, along with their in vitro characterization. The ZF-NT method's efficiency was higher than that of the PEM-NT using cumulus cells. When ESCs and cumulus cells were used as nuclear donor cells, they resulted in significantly higher ZF-NT-derived ntESC line establishment rates compared to MEF cells. In conclusion, the nuclear donor cell type significantly affected the efficiency of ntESC line establishment, and the ZF-NT method was efficient to establish pluripotent ntESC lines.
Introduction
Previous studies in mouse described that nuclear transfer-derived ESCs (ntESCs) possess the same characteristics for self-renewal and differentiation as those derived from fertilized blastocysts (Kawase et al., 2000; Wakayama et al., 2001). Furthermore, molecular biology studies have shown that the transcriptional-, DNA methylation-, and DNA microarray profiles of mouse ntESCs are almost identical to fertilized embryo-derived ESCs (Brambrink et al., 2006; Wakayama et al., 2006). In the mouse model, ntESCs have been shown the potential to differentiate in vitro to neural or to myogenic cells (Munsie et al., 2000) and have also the capacity to contribute to all the three germ layers besides the germ line in vivo, similarly to fertilized embryo-derived counterparts (Wakayama et al., 2001).
ESCs derived from parthenogenetically activated embryos (PGA), namely pESCs, could provide histocompatible cells for transplantation therapy purposes (Kim et al., 2007). Serial NT of pESC revealed significantly better differentiation capability of the NT-pESCs in vitro and in vivo compared to the original pESC (Hikichi et al., 2007). Recent establishment of human pESCs (Revazova et al., 2008) brought the attention to its potential in human transplantation therapy.
There are three micromanipulation methods for creating mouse blastocyst-stage NT embryos: cell fusion (CF) (Ogura et al., 2000), piezoelectric microinjection (PEM) (Wakayama et al., 1998), and zona-free (ZF) (Ribas et al., 2005) NT methods. Most ntESCs were established via PEM-NT (Wakayama et al., 2005, 2006).
The ZF-NT method was developed first for bovine cloning (Oback et al., 2003). This method is relatively simple and cost-effective by reducing the need for extensive micromanipulation training and by eliminating the need for zona-piercing by using special capillary design, piezoelectric, or laser devices. The ZF-NT technique had been investigated in farm animals (cattle, sheep, pig, and horse) and the results favored the output of NT blastocysts over the conventional zona-enclosed system (see review by Lagutina et al., 2007). For human NT, the methods are still under development.
In the mouse, the ZF-NT resulted in NT blastocysts without application of a piezoelectric equipment (Ribas et al., 2005). In these mouse experiments, cumulus and ESCs were used as nuclear donors, but only embryos generated from ESCs resulted in live offspring. However, several important aspects of the ZF-NT method were not evaluated including establishing ntESC lines using different types of nuclear donor somatic cells.
Our goal was to investigate the efficiency of ESC derivation from blastocyst-stage embryos generated by ZF-NT, parthenogenetic activation (PGA), and fertilization in mouse model, and to compare with the efficiency of ESC derivation from PEM-NT blastocysts. The effect of nuclear donor cell type [mouse embryonic fibroblast (MEF), ESCs or cumulus cells] on ZF-NT was also studied.
Materials and Methods
Chemicals for embryo culture and manipulation, unless specified otherwise, were purchased from Sigma-Aldrich Chem. Inc. (St. Louis, MO, USA; www.sigmaaldrich.com). All other materials, unless specified otherwise, were purchased from Invitrogen (Carlsbad, CA, USA; www.invitrogen.com).
Animals, oocyte, and embryo collection
The animal experiments were established in full compliance with European and Hungarian laws and regulations, and were approved by the Animal Experimentation Committee of Agricultural Biotechnology Center, Gödöllő, Six- to 8-week-old B6D2F1 (Charles River Hungary Ltd., Hungary) female mice were superovulated with five IU pregnant mare serum gonadotropin (PMSG; Folligon®, Intervet, B.V., Boxneer, The Netherlands) followed by 5 IU human chorionic gonadotropin (hCG; Choragon®, Richter Gedeon Rt., Budapest, Hungary) injection 48 h later. Oocytes were collected 14 h after hCG administration. The cumulus cells were removed from the oocytes by 0.1% hyaluronidase. C57Bl/6J × DBA/2J (F1) zygotes were collected from the ampulla of the oviduct 20 h following hCG injection by flushing with M2 medium. Zygotes were cultured in KSOM medium (Chemicon-Millipore, Temecula, CA, USA) for 4 days prior reaching the blastocyst-stage.
Piezoelectric microinjection nuclear transfer
Freshly isolated B6D2F1 cumulus cells were used as nuclear donors. Nuclear transfer was performed as described earlier (Kishigami et al., 2006; Wakayama et al., 1998). In brief, the oocytes were enucleated using piezoelectric microcapillary (PMM-150FU; PrimeTech, Orange County, CA, USA) in HEPES-supplemented CZB (Chatot Ziomek Bavister) embryo culture medium (Chatot et al., 1989) containing 5 μg/mL cytochalasin B (CB) at room temperature (RT). The metaphase II chromosome-spindle complex was drawn into the pipette with a small amount of accompanying ooplasm and gently pulled away from the oocyte until a stretched cytoplasmic bridge was pinched off. The enucleated oocytes were transferred into CB-free CZB medium and kept there for up to 2 h at 37°C. For donor nuclei injection, the cumulus cell was drawn in and out of the injection pipette until the cell membrane was broken. Then the cumulus nuclei were injected into enucleated oocytes at RT. After NT, the reconstructed oocytes were activated by 5 nM TSA and 1 mM SrCl2 in Ca2+-free CZB medium in the presence of 5 μg/mL CB and 1% dimethyl sulfoxide (DMSO) for 6 h and placed into 5 nM TSA for another 2 h. The embryos were cultured in vitro until blastocyst stage for 4 days in KSOM medium at 37°C, in a humidified atmosphere of 5% CO2 in an incubator.
Zona-free nuclear transfer
Freshly isolated B6D2F1 cumulus cells, cultured C57Bl/6J × DBA/2J MEF cells or HM1 ESCs (passage 22–26; 129/Ola genotype) were used as nuclear donors (HM1 ESC line was kindly provided by Dr. Jim McWhir; Roslin Institute, Roslin, UK) (Selfridge et al., 1992). NT was performed according to the protocol as described earlier (Ribas et al., 2005). The zona pellucida was removed with 0.5% Pronase dissolved in CZB-HEPES 1 h after hyaluronidase treatment at 37°C. Metaphase II chromosome spindle complexes were removed by micromanipulation using an enucleation pipette with 18–20 μm outer diameter in CZB-HEPES medium containing 5 μg/mL CB on RT. Five to 10 PHA lectin-treated cytoplasts were attached to the donor cells in CZB-HEPES containing 0.1 mg/mL polyvinyl alcohol (PVA). The constructs were fused applying two pulses of 2 kV/cm DC for 12–14 μsec using a BLS B-150 fusion machine (BLS, Hungary) in 0.28 M Mannitol medium. Following 60 min incubation in CZBG medium (Gao et al., 2003) the fusion was assessed. The fused NT constructs were activated in 1 mM SrCl2 in Ca2+-free CZB medium supplemented with 5 μg/mL CB and 15 mg/mL bovine serum albumin (BSA) for 5–6 h. Following activation, embryos were cultured in vitro in groups of 10 in 30 μL CZBG droplets under mineral oil at 37°C and 5% CO2.
Parthenogenetic activation
The B6D2F1 oocytes were activated in Ca2+-free CZB medium supplemented with 10 mM SrCl2 and 5 μg/mL of CB. Six hours later they were washed three times and finally cultured in the regular CZBG medium (Otaegui et al., 1999).
Derivation and culture of ESC lines
The conventional method (Evans and Kaufman, 1981) was used with few modifications to establish mouse ESCs. Control fertilized blastocyst-stage embryos were processed directly onto feeder cells (controls of PEM experiments) or treated with acid Tyrode's solution to remove the zona pellucida (controls of ZF experiments) and plated individually on a 24-well dish precoated with mitomycin-C inactivated primary MEF cell layer. MEF cells were obtained from 13.5 d.p.c. mouse embryos as described elsewhere (Robertson, 1987). Embryos were cultured in ESC establishment medium: high glucose Dulbecco-modified Eagle medium (DMEM), supplemented with 2000 IU/mL recombinant mouse leukemia inhibitory factor (ESGRO-LIF, Chemicon-Millipore), 20% fetal bovine serum (FBS, HyClone, Logan, UT, USA), 1× nonessential amino acids (NEAA), 0.2 mM 2-mercaptoethanol, 50 IU penicillin/mL, 50 mg streptomycin/mL, nucleoside mixture (Robertson, 1987), and insulin-transferrin-selenium supplementation (ITS). Five to 6 days after initial plating, the inner cell mass (ICM) outgrowths were picked mechanically with a fine pipette, the clumps were disaggregated further by gentle pipetting and replated on fresh feeder. A few days later, the growing ESC-like colonies were disaggregated with trypsin–EDTA and the cells were replaced on fresh feeder layer until a stable cell line was established. The culture dishes were kept at 37°C in a humidified atmosphere of 5% CO2 in air. The ESCs were fed daily with regular ESC medium (high glucose DMEM, supplemented with 1000 IU/mL ESGRO-LIF 15% FBS, 1× NEAA, 0.2 mM 2-mercaptoethanol, 50 IU/mL penicillin, 50 mg/mL streptomycin), and passaged every 2–3 days onto fresh feeder.
Characterization of ESCs
The pluripotency potential of the established ESC lines was characterized by conventional in vitro studies. ESC colonies were fixed in 4% paraformaldehyde (PFA) for 15 min at RT, washed three times with phosphate-buffered saline (PBS), and treated in blocking solution (PBS supplemented with 1% BSA, 5% FBS, and 0.1% Triton-X) for 1 h at RT. ESCs were incubated with the selected primary antibody overnight at 4°C, whereas cells with the selected fluorescent secondary antibody were incubated 1 h at RT. The nuclei of ESCs were counterstained with DAPI (Vector Laboratories, Burlingame, CA, USA) and visualized by fluorescent microscopy. Primary antibodies used were as follows: mouse monoclonal stage-specific embryonic antigen (SSEA-1; MC-480, Developmental Studies Hybridoma Bank; 1:300), mouse polyclonal anti-Pou5f1 IgG (Santa Cruz Biotechnology, Santa Cruz, CA, USA; 1:400), and goat polyclonal antinanog IgG (Santa Cruz Biotechnology; 1:400). Fluorescent-labeled secondary antibodies (Jackson ImmunoResearch Europe Ltd., Newmarket, Suffolk, UK): antimouse IgM-Cy3 (1:200), antimouse IgG-FITC (1:200), and antigoat IgG-Cy3 (1:200) were applied. Alkaline phosphatase (ALP) staining and chromosome analysis of ESCs lines were prepared according to the protocol of Nagy (2003). Chromosomes were stained with Giemsa or DAPI stain on slides. At least 50 metaphases of each cell lines were examined for chromosome numbers.
In vitro differentiation
Embryoid body (EB) formation was used to test the in vitro differentiation potential of the newly established ESC lines. ESCs were treated with 0.25% trypsin–EDTA, single cell suspension (600 cells/20 μL drop) was cultured by hanging drop method (Doetschman et al., 1985) for 2 days in regular ESC medium with LIF withdrawal. The resulting ESC aggregates were transferred into a bacteriological dish (Greiner Bio-One GmbH, Germany) and cultured in suspension continuously up to 4 weeks. The ability of EBs to form simple EB (SEB) and cystic EB (CEB) were observed. At least 20 embryoid bodies per dish were examined to determine the appearance of SEB and of CEB on a given day.
For further differentiation tests after 2 days of culture (day 2) in hanging drops, the formed EBs were collected and placed into suspension culture in 10-cm bacterial Petri dishes with daily medium changes. After day 8, the EBs were transferred to gelatin-coated 24-well dishes individually, and cultured for further analysis.
Immunohistochemistry
Differentiated EB samples, grown on round cover slips, were fixed in 4% PFA fixative for 15 min, followed by three washing steps in PBS containing 1% BSA and 0.1% Triton X-100 for 3 × 10 min. For blocking, washing solution containing 10% FBS was used for 1 h.
Primary antibodies were incubated in blocking solution overnight at 4°C, in the indicated dilution: nestin (IgG, Hybridoma Bank, Iowa City, IA, USA), 1:100; neurofilament (IgG, Hybridoma Bank), 1:100; troponin-T (IgG, Hybridoma Bank), 1:100; GATA-4 (IgG, Sigma-Aldrich), 1:200; and vimentin (IgM, Hybridoma Bank) 1:100. Samples were incubated at RT with the isotope and host matching secondary antibody (goat antimouse IgG-Alexa 488, 1:200; goat antimouse IgG-Cy3, 1:500; goat antirabbit IgG-Alexa 488, 1:200; goat antimouse IgM-Cy3, 1:200; all from Jackson ImmunoResearch Europe, Suffolk, UK) for 1 h the following day. Samples were mounted with Vectashield-DAPI mounting media (Vector Laboratories); thus, the nuclei were labeled with DAPI (blue). The immunostainings were visualized with an AxioObserver Z.1 inverse fluorescent microscope and ApoTome slider system and AxioCam MRm camera system (Carl Zeiss GmbH, Oberkochen, Germany).
Statistical analysis
Statistical analysis was performed by the chi-square test. Values of p < 0.05 were considered statistically significant. Immunoassay results were confirmed in at least three independent experiments.
Results
Comparison of initial outgrowth formation and ESC line establishment rates of PEM-NT and ZF-NT blastocysts
The differences in the attachment and ESC line establishment potentials of embryos produced by the two NT methods were investigated. In the case of ZF-NT, the embryos are more sensitive to mechanical stress, and the activation also differs from PEM-NT (Ribas et al., 2005).
Establishment rates of ESC lines (the ratio of the number of established cell lines and of the number of cultured embryos) harvested from NT (using cumulus cells as nuclear donor cells), PGA and fertilized embryos are summarized in Table 1. As the controls of the two NT methods were different, both type (zona-enclosed and zona-free) of fertilized and PGA groups are presented in Table 1.
Different superscripts indicate statistically significant difference within a column (p < 0.05).
As expected, fertilized blastocysts showed the highest rate of attachment with or without zona pellucida (87.5 and 83.3%, respectively). In the zona-intact experimental group, the attachment of embryos to the feeder layer was observed from day 2 of culturing. Attachment occurred as a gradual flattening of blastocysts followed by spreading out of trophoblast cells. In contrast, attachment of ZF-embryos was observed from day 1 through day 4 of blastocyst culture. ZF-embryos, which attached within 48 h, showed more developed ICMs than those that could attach only between 48 and 96 h. These “late” ZF-blastocysts often possessed only trophectoderm (TE) type cells.
The zona-intact and ZF-PGA and the PEM-NT blastocysts all showed low percentage of attachment compared to the fertilized embryos while the ZF-NT embryos were not impaired (Table 1).
In the zona-intact group of embryos, at day 5 of culturing, the outgrowth, mostly from fertilized blastocysts, contained a central core of putative ICM-derived cells, large enough for mechanical and enzymatic isolation for further subculture. In the case of the outgrowth from cloned blastocysts of both methods, the putative ICM-derived cells appeared to be smaller compared to the outgrowth from the fertilized blastocysts (no comparative cell counting was performed). Probably due to this relatively small size of ICMs observed, our modified mechanical isolation of ICM clumps was more successful than trypsinization during ESC line establishment. Three out of four PEM-NT ESC lines and both ZF-NT ESC lines were generated by this modification.
Following the first trypsinization of ICM outgrowth, ESC-like colonies were observed mostly in the fertilized group. In the NT groups, typically mixed populations of ESC-like colonies and differentiated colonies were observed. The derivation rate of PEM-NT ESC lines was significantly lower (15.4%) compared to the PGA (32.1%) and to the fertilized (62.5%) groups, respectively (Table 1). In the ZF-group, the ESC line establishment was significantly reduced in the fertilized controls compared to their zona-intact counterparts. Surprisingly, ESC line establishment in the ZF-NT group was not different from that of the ZF-fertilized controls, whereas it was significantly lower in the ZF-PGA group (Table 1).
Effect of donor nuclear cell type on ZF-NT
Previously published data (Wakayama et al., 2005) in PEM-NT experiments demonstrated the effect of nuclei donor cell type on the efficiency of NT, thus the efficiency of ESC line establishment. Here, the effect of different nuclear donor cell types on ZF-NT, using ESC, MEF, or fresh cumulus cells were compared (Table 2). The attachment rates of embryos differed significantly among the three nuclear donor groups. The attachment rates of the cumulus- or ESC-derived (85.7 and 76.5%, respectively) NT embryos were significantly different from that of the MEF group (47.6%). Due to the low efficiency rate of ntESC line derivation from MEF a high number of MEF-derived NT blastocysts were used compared to the other two nuclear donor cell types (Table 2). The ESC- and cumulus-derived NT embryos produced significantly higher rates of ESC lines (20.6 and 28.6%, respectively) compared to the MEF donor cell group (1.2%).
Different superscripts indicate statistically significant difference within a column (p < 0.05).
Characterization of ZF-ESC lines
Investigating the morphology of the cell lines, colonies of stable ESCs showed a smooth outline with round or slightly oval shape. At the molecular level, all the examined established ntESC lines were positive for the pluripotency markers, including ALP, SSEA-1, Pou5f1, and Nanog as shown in Figure 1.
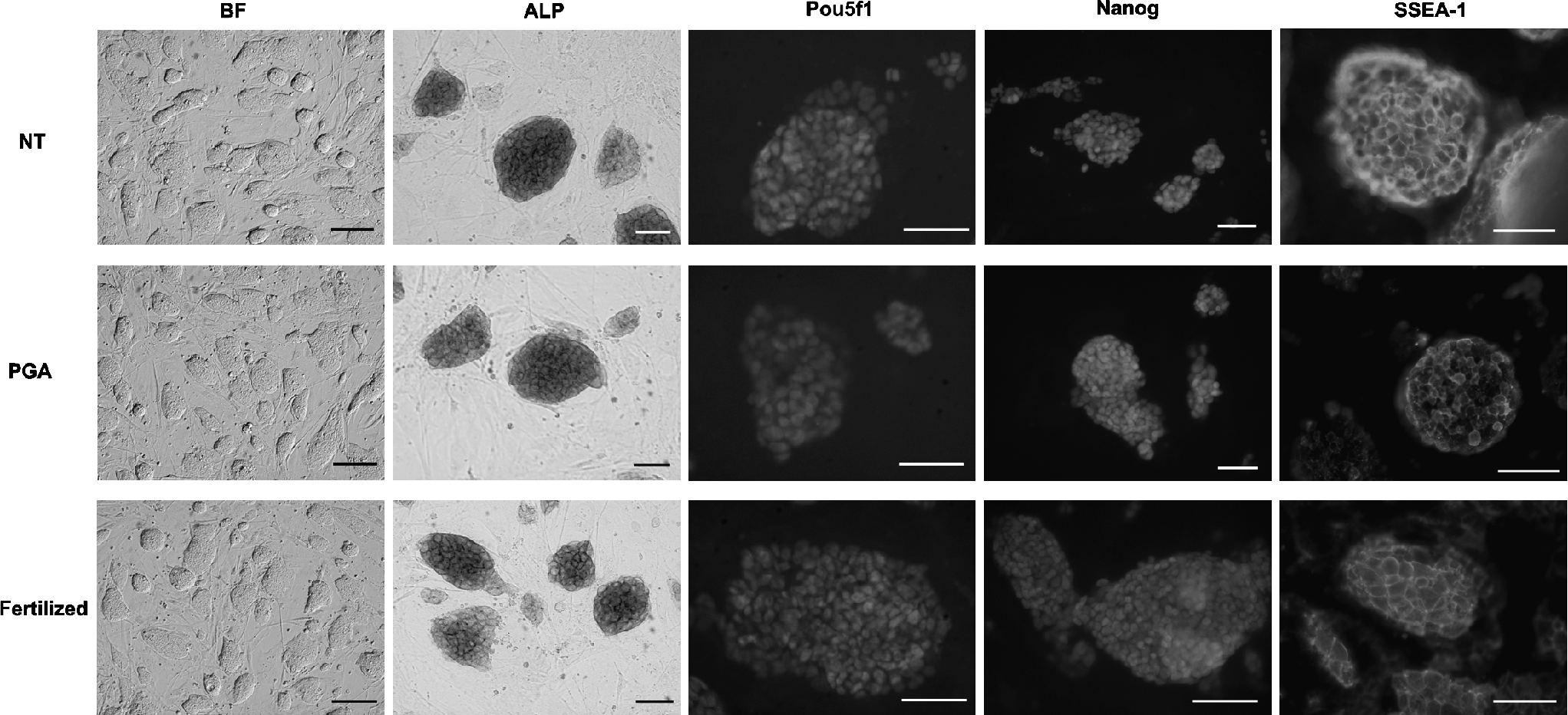
In vitro pluripotency analysis of zona-free method derived ESCs established from nuclear transfer (NT), parthenogenetically activated (PGA), and fertilized embryos. Representative pictures show the bright field (BF, scale bar = 200 μm) view of colonies and the expression of alkaline phosphatase (ALP), Pou5f1, Nanog, and stage-specific antigen 1 (SSEA-1) in each cell lines chosen from all groups of zona-free experiment (scale bar = 100 μm).
Chromosome numbers of ntESCs were counted after Giemsa staining and euploidy percentage was calculated. All the examined cell lines showed more than 60% euploidy (data not shown).
To prove the pluripotency and monitor the in vitro differentiation capacity of our ntESC lines an in vitro differentiation study was performed using the hanging drop method (Doetschman et al., 1985). The aggregated cells derived from ntESCs exhibited a proper morphology of EBs following 2 days culturing in hanging drop. Following the same period, there were no differences in the formation of EBs compared to the fertilized embryo-derived ESCs.
After transferring EBs to suspension culture and culturing them for an additional 4 days, simple aggregates (SEB) were formed (Fig. 2). Following 7–10 days culturing in suspension, ntESC lines were capable to form cystic EB (CEB) as indicated in Figure 2. Furthermore, when EBs were transferred to gelatinized dish and attached, the in vitro differentiation of the three germ layers were observed and demonstrated with immunohistochemistry (Fig. 3). These results indicate that all of the established cell lines have the potential to differentiate into different tissues in vitro, which is one of the criteria of pluripotency.
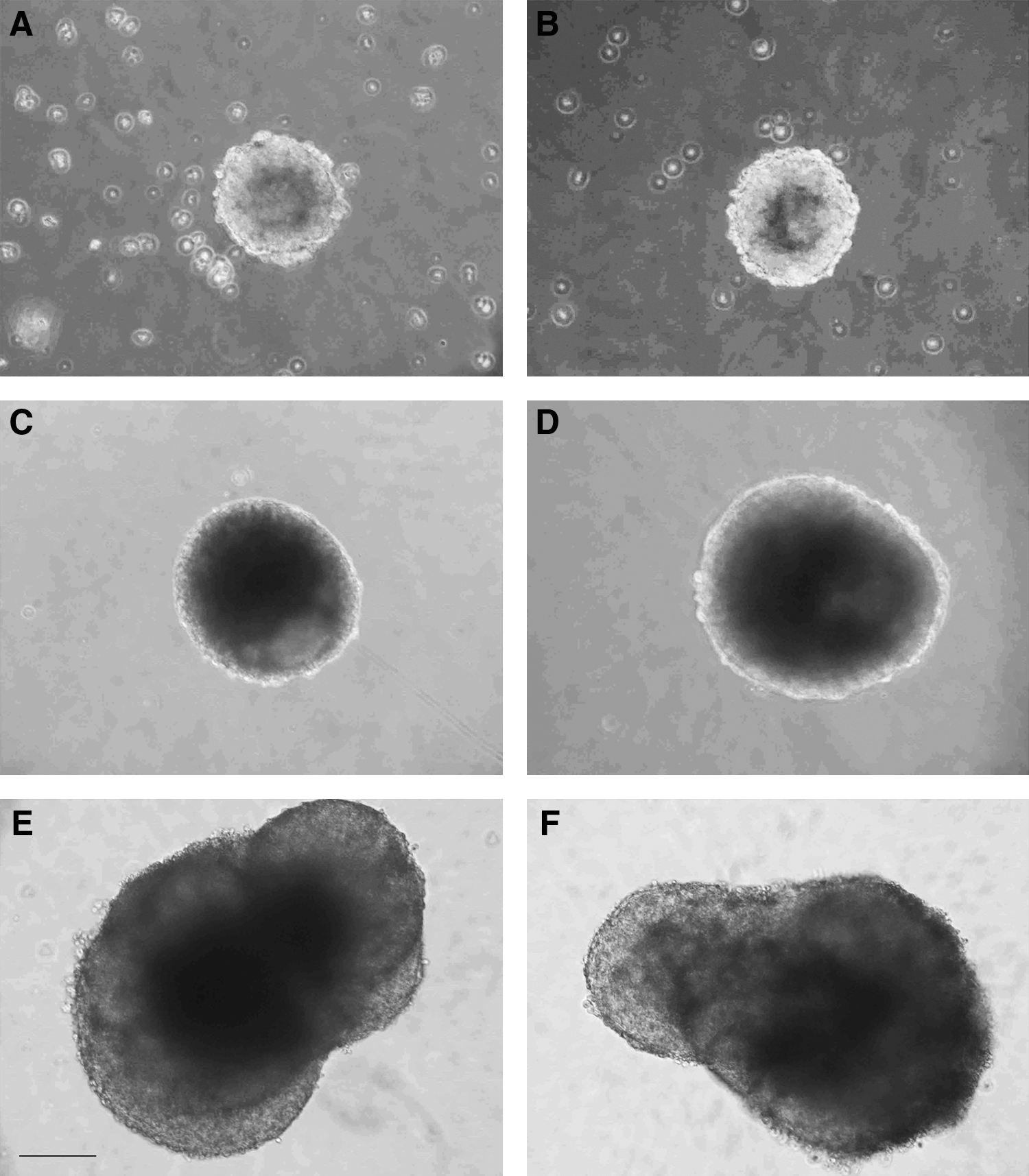
In vitro differentiation of cumulus-derived zona-free ntESCs and fertilized embryo-derived ESC. Representative pictures show the bright-field (BF) view of day 2 (
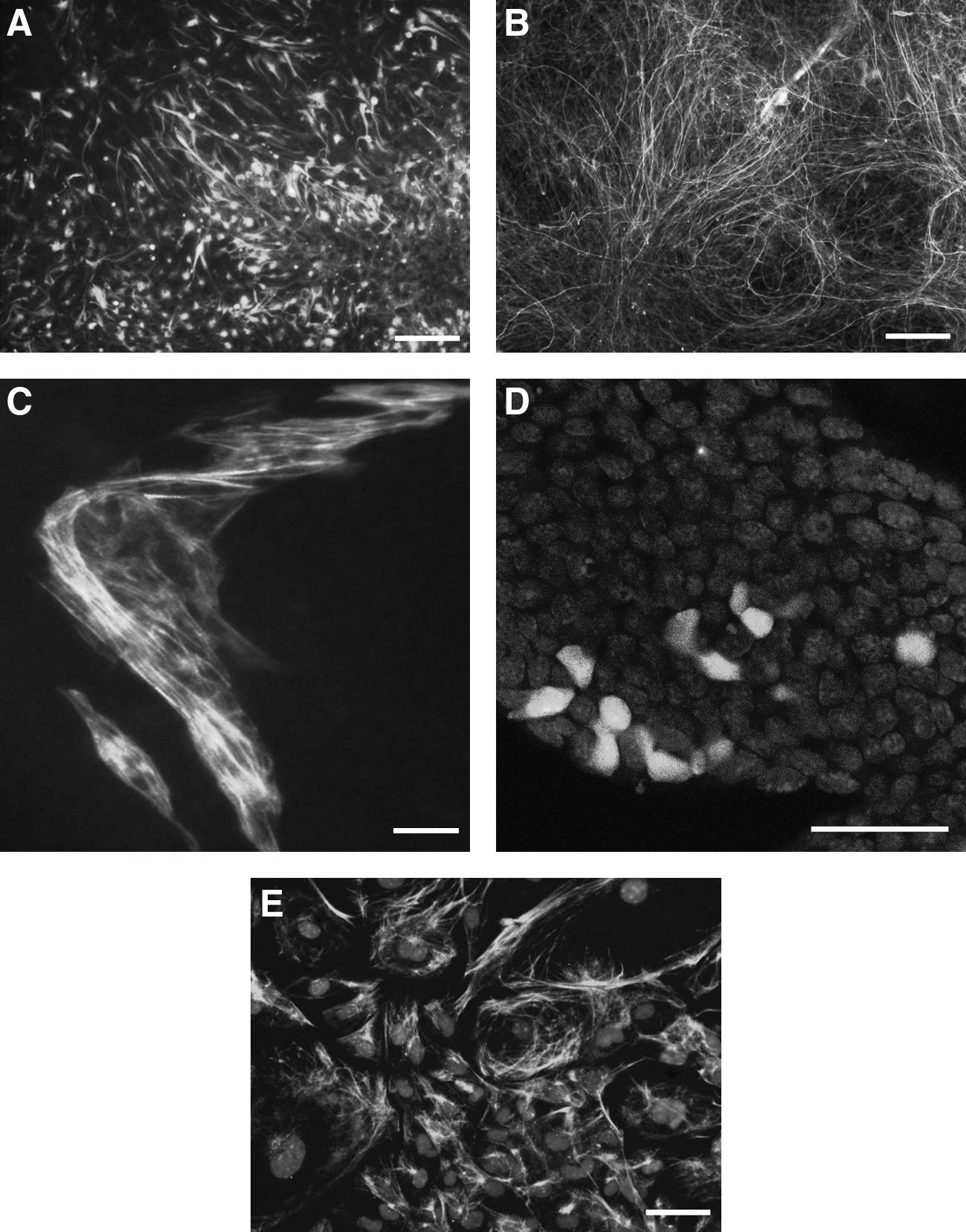
Representative immunohistochemistry of a ZF-cumulus ntESC line during spontaneous in vitro differentiation. The markers of the three germ layers were used to test the differentiation potential of the ntESCs after LIF withdrawal. Two ectoderm markers (
Parthenogenetic ESCs lines—either zona-free or zona-intact—were isolated and characterized for their pluripotency. There were no significant differences in the differentiation potential of ntESCs compared to pESCs. Both cell types were capable to form SEB and CEB with the chosen differentiation method, and furthermore, to differentiate into cell types of the three germ layers (data not shown).
Discussion
The objective of this study was to evaluate the feasibility of ntESC line establishment obtained from cloned blastocysts produced by ZF-NT. This alternative technique of NT has been successfully applied for the production of live offspring (Ribas et al., 2005) using ESCs as donor cells in mouse. Here we evaluated systematically the efficiency of each steps of the ntESC derivation process, by comparing zona-free and zona-intact NT methods.
In the first step, when the attachment rates of blastocysts-stage embryos of ZF- and PEM-NT methods were compared, no significant difference was observed. Fertilized embryos gave the best results in attachment rates in accordance with our expectations. PGA and PEM-NT embryo showed significantly lower attachment rates compared to the fertilized embryos. These observations are in accordance with the published results of PEM-derived ntESC establishment (Wakayama et al., 2006). However, the ZF-NT embryos have had a significantly higher rate of attachment, which is a promising observation for this method. Maybe the lack of zona pellucida helped the ICMs to attach early to the surface in case of NT embryos, although such benefits were not clear in the other ZF groups.
When the numbers of isolated ICM outgrowths were compared from PGA embryos, using the ZF method the success was significantly lower compared to the zona-intact group. The observed difference may be accounted for the differences in the parthenogenetic activation of the two groups as in the ZF-PGA group the zona pellucida of the oocytes was removed at the beginning of the activation process. The mechanical stress due to zona removal might cause the difference between the two groups of PGA embryos (Fulka et al., 2004; Sayaka et al., 2008), and subsequently, the establishment of ESC lines was also very low in the ZF-PGA group. However, our observations on the high isolation rate of ICM outgrowths and high rate of ESC line establishment from ZF-NT embryos contradict this explanation.
Indeed, comparing the rate of established ESCs lines, a significant difference was observed between PEM-NT and ZF-NT methods, when cumulus cells were used as donor cells. Furthermore, the observed ZF-NT ntESCs establishment rate with ECS nuclear donors was also similar to that with cumulus cells, demonstrating that the ZF-NT method indeed is capable to produce ESC lines efficiently.
In our study, we demonstrated that ntESC lines can be established from ZF-NT blastocysts with a variable efficiency (1.2–28.6%), and this process is strongly dependent on the donor cell type: the use of cumulus and ESC as nuclear donor cells resulted significantly higher ntESC ratio than using MEF cells. It may be due to the inadequate reprogramming of fibroblast cells during the NT process contrary to the ESC and cumulus cells, where the quality of NT blastocysts, originated from more completed reprogrammed NT constructs, supports subsequent ntESC establishment more efficiently than the fetus-derived fibroblast cells (Wakayama et al., 2006). It is still unknown why MEF cells gave rise to less ntESC lines than cumulus cells, although both cell types are somatic cells. One of the differences, besides the cell origin is that MEF cells were extensively treated prior to manipulation, whereas cumulus cells were not. For instance, MEF cells were treated with repeated trypsin treatments and kept in culture longer prior to manipulation than cumulus cells (Dinnyes et al., 2004). In accordance with our results, a previous study in the donor cell type of ntESC establishment reported a significantly better rate when freshly isolated cumulus cells were used as donor nuclei compared to cultured tail-tip fibroblast (Wakayama et al., 2005). Our study confirms these results using a fibroblast (MEF) of different origin and a different NT technique (ZF-NT).
The success rate of cloned mice born from ESC nuclei is reported to be also higher than the ones from somatic cells (Rideout et al., 2000; Wakayama et al., 1999), suggesting that the undifferentiated stage of ESC nuclei may favor reprogramming and increase NT efficiency. In some species, using noncultured cumulus cells as nuclear donor gave significantly higher progeny rates compared to cultured fibroblast (Meng et al., 2009).
Characterization of the newly generated cell lines demonstrated that at molecular and chromosomal level, the examined ntESCs were similar to regular ESCs. They were positive for the main ESC markers (ALP, SSEA-1, Pou5f1, and Nanog), and showed at least 60% of normal euploidy. ESCs having more than 50% of cells with normal chromosome numbers were suggested to be efficient for germ-line transmission due to successful segregation of chromosomes throughout meiosis (Longo et al., 1997; Suzuki et al., 1997). During the in vitro culture, these ntESCs are able to maintain their undifferentiated status.
In the differentiation studies, they were able to form EB aggregates using hanging drops methods then form embryoid bodies (both simple and cystic) as well as differentiate into the three germ layers in vitro. The ability of ESCs to form simple and cystic EBs is one of the most admired methods to differentiate ESCs in vitro. It is considered that ESCs, which are able to form only simple EBs, have restricted pluripotency (Sukoyan et al., 2002). The developmental potential of ESCs could be lost due to the accumulation of profound epigenetic modifications by long-term in vitro cultivation (Amano et al., 2006).
These data suggest that these ntESCs were pluripotent, resembling the characteristics of ones derived from fertilized blastocysts. Furthermore, differentiation into cell types of the three germ layers, suggest that our established ntESC lines share a similar differentiation potential as their fertilized embryo derived equivalents. Similar findings had been earlier reported with PEM-NT ESC lines (Wakayama et al., 2001; 2006).
As an additional observation, parthenogenetic ESCs lines—either zona-free or zona-intact—were isolated and characterized for their pluripotency. In accordance with earlier reports (Hikichi et al., 2007; Kim et al., 2007), we found the differentiation potential of the pESCs into cell types of all three germ layers, demonstrating that they might serve as a potential source of histocompatible cells for transplantation.
In summary, in this study ntESCs were successfully established from NT mouse blastocysts. We demonstrated that the nuclear donor cell type of NT embryos influenced the efficiency of ntESC establishment. Our study is the first to demonstrate that ntESC lines can be derived efficiently from the nuclei of somatic and embryonic cells by zona-free nuclear transfer technique, which provides a technological model for regenerative medicine.
Footnotes
Acknowledgments
The authors thank Dr. Melinda Pirity for the critical reading and discussion of the article. We are grateful to Mrs. Györgyi Kungl for her excellent technical assistance during the cell culture experiment. This work was supported by grants from the Wellcome Trust (Grant No. 070246), EU FP6 (MEXT-CT-2003-509582; MEDRAT- LSHG-CT-2005-518240; “Clonet” MRTN-CT-2006-035468), EU FP7 (“PartnErS” PIAP-GA-2008-218205; “Plurisys” HEALTH-F4-2009-223485), OTKA T046171, NKFP_07_1-ES2HEART-HU (OM-00202-2007), the Thai Research Fund (Royal Golden Jubilee Ph.D. program; PHD 0111 12545 and CHE-TRF senior scholarship; RTA 5080010), and the National Research Council of Thailand (Grant No. GRB 03503001).
Author Disclosure Statement
The authors declare that no conflicting financial interests exist.
J. Kobolak and Sz. Bodo contributed equally to this work.
