Abstract
Background:
The cytotoxic effect of 212Pb on microbial growth was examined, using six microorganisms, at clinically relevant time points, to determine the potency of 212Pb as a self-sterilizing agent in radiopharmaceuticals using [212Pb]Pb-DOTAMTATE.
Materials and Methods:
Vials of radiolabeled [212Pb]Pb-DOTAMTATE, nonradiolabeled DOTAMTATE, and appropriate media were inoculated with Bacillus spizizenii, Candida albicans, Clostridium sporogenes, Pseudomonas aeruginosa, Staphylococcus aureus, or Aspergillus brasiliensis. Samples from each vial type were plated onto tryptic soy agar or Sabouraud dextrose plates and allowed to grow at optimal temperature for each strain to obtain quantifiable colony forming units (CFU).
Results:
Each microbial organism observed at least a 6 log reduction in total CFUs after 6 h of exposure to [212Pb]Pb-DOTAMTATE drug product vials, showing no remaining colonies as compared to the vials containing only media and drug formulation with no radiolabeled material, utilizing a low absorbed dose of no greater than 1.01 kGy. A sterility assessment of the two [212Pb]Pb-DOTAMTATE drug product vials containing the lowest total CFUs per vial displayed no microbial growth upon incubation for 14 d.
Conclusions:
This study suggests that 212Pb in radiopharmaceuticals is a potent microbial cytotoxic agent with self-sterilizing properties.
Introduction
Sterility is a requirement of parenteral radiopharmaceuticals and is defined by the elimination of all microbes present in a drug product, most often achieved by reaching a sterility assurance level (SAL) of a reduction of ≥6 logs of colony forming units (CFU). 1 –3 Sterility of parenteral drugs is often achieved by passing the drug through a 0.2 µm filter, which is proven to retain microorganisms present in drug products. 4,5 However, the required 14-d incubation of samples in media for sterility testing after filtration, combined with the short shelf life of radiolabeled drugs, prohibits the procurement of sterility results before patient drug administration, resulting in drugs being administered “at risk”.
Ionizing radiation is another common form of sterilization and per established pharmacopeias, a dose of 25 kGy is required to ensure SAL in parenteral pharmaceuticals. 6 As such, directly applying this dose has become common practice. 7 Current applications include electron beams, X-rays, and gamma rays that are the most common. 3 γ-radiation is facilitated by a 60Co irradiator and is commonly used in the sterilization of packaged disposables and food products as it can penetrate entire products without leaving a residue. 3,8,9 Here, free radicals are excited upon the loss of an electron as ion pairs are created leading to this ionizing-induced reaction. 6 However, 60Co is not a recommended source when attempting to sterilize parenteral pharmaceuticals, as bacterial spores are less susceptible to this form of radiation. 8
Studies with various radionuclides have been conducted to assess their cytotoxic nature. Technetium-99m (99mTc), a γ-emitter, in doses of 50, 100, 200, and 300 mCi, was observed to have a slight dose-dependent impact on the survival of Pseudomonas aeruginosa and Staphylococcus aureus; however, results were inconclusive to the definitive nature of this radionuclide being self-sterilizing. 10 68Ga and 177Lu, a β+ and β−/γ-emitter, respectively, were analyzed under similar conditions using the micro-organisms Bacillus pumilus and Staphylococcus succinus. 11 This study utilized doses of 54 mCi of 68Ga and 189 mCi of 177Lu, though failed to produce the desired potency on microbial cells. 11 This study reported a theoretical dose < 1% of the established 25 kGy, for each radionuclide, concluding that the beta and positron emissions from these two radionuclides were not significant enough to establish an absence of microbial growth post inoculation. 11 Another study used the α-emitter 213Bi with a monoclonal antibody to target Bacillus anthracis spores. 12 This experiment, while concerned with the radio resistance of the pathogen, observed the nonspecific killing of spores at a dose-dependent rate between 75 µCi and 150 µCi. 12
212Pb, an in vivo α-generator through its decay chain (212Bi to 208Tl and 212Po to 208Pb), has a high linear energy transfer allowing it to be a useful radionuclide in targeted cancer treatment. This study was designed to test Orano Med’s most advanced clinical drug [212Pb]Pb-DOTAMTATE. By directly inoculating the radiolabeled drug product with standard microorganisms, we observed the effect that 212Pb had on their survival. This was observed at t0 and t6hrs post sample inoculation to represent the time immediately after manufacturing (t0) and the minimum amount of time between manufacturing and the earliest possible clinical administration of [212Pb]Pb-DOTAMTATE (t6hrs). This study aimed to provide a better understanding of the cytotoxic and self-sterilizing potential of 212Pb on microbial organisms to help assess the inherent risk of administering these drug products prior to the availability of sterility results, which, for most radiopharmaceuticals, become available two weeks post administration. To alleviate this potential risk, this study sought to demonstrate that short lived in vivo generators of α-particle radiopharmaceuticals can achieve the ≥6 logs of reduction needed to be accepted as sterile products.
Materials and Methods
Safety
Necessary precautions were taken during this study as work involved microorganisms and radioactive materials. All required personal protective equipment was worn by investigating personnel including lab coats, safety glasses, gloves, and dosimeters. All work was performed inside a biological safety cabinet that was decontaminated prior to and upon completion of all work with microorganisms. All personnel obtained extensive radioactive material safety training prior to the initiation of this study.
Organisms
Staphylococcus aureus, Bacillus spizizenii (subspecies of Bacillus subtilis), Pseudomonas aeruginosa, Candida albicans, and Aspergillus brasiliensis were purchased as Epower pellets, while Clostridium sporogenes was purchased as EZ-Accu Shot pellets from Microbiologics. Sabouraud dextrose media, soybean casein digest (SCD) media, tryptic soy agar (TSA) plates, and Sabouraud dextrose plates were purchased from Fisher Scientific. Fluid thioglycolate media (FTM) was purchased from Millipore-Sigma.
Microbial growth
Epower microorganisms come as lyophilized pellets (ranging in CFU from 106 to 107) and once equilibrated at room temperature were added to 1.5 mL of phosphate-buffered saline pH 7.2 and placed in a 20–25°C incubator for 25 min to ensure hydration. The hydrated pellet was vortexed until the suspension was homogenous. Greater than 1 million CFU per vial was targeted in this study.
The C. sporogenes EZ-Accu Shot pellet came in an allotted 103 CFU. A single C. sporogenes pellet was resuspended in associated hydrating fluid, added to 9 mL of FTM media, and grown at 33°C ± 2°C until an OD600 of 0.45 was reached. This bacterium was plated at multiple dilutions for quantification and determined that the 9 mL vial of media held ∼120 million CFU.
A schematic detailing the experimental design is shown in Figure 1.
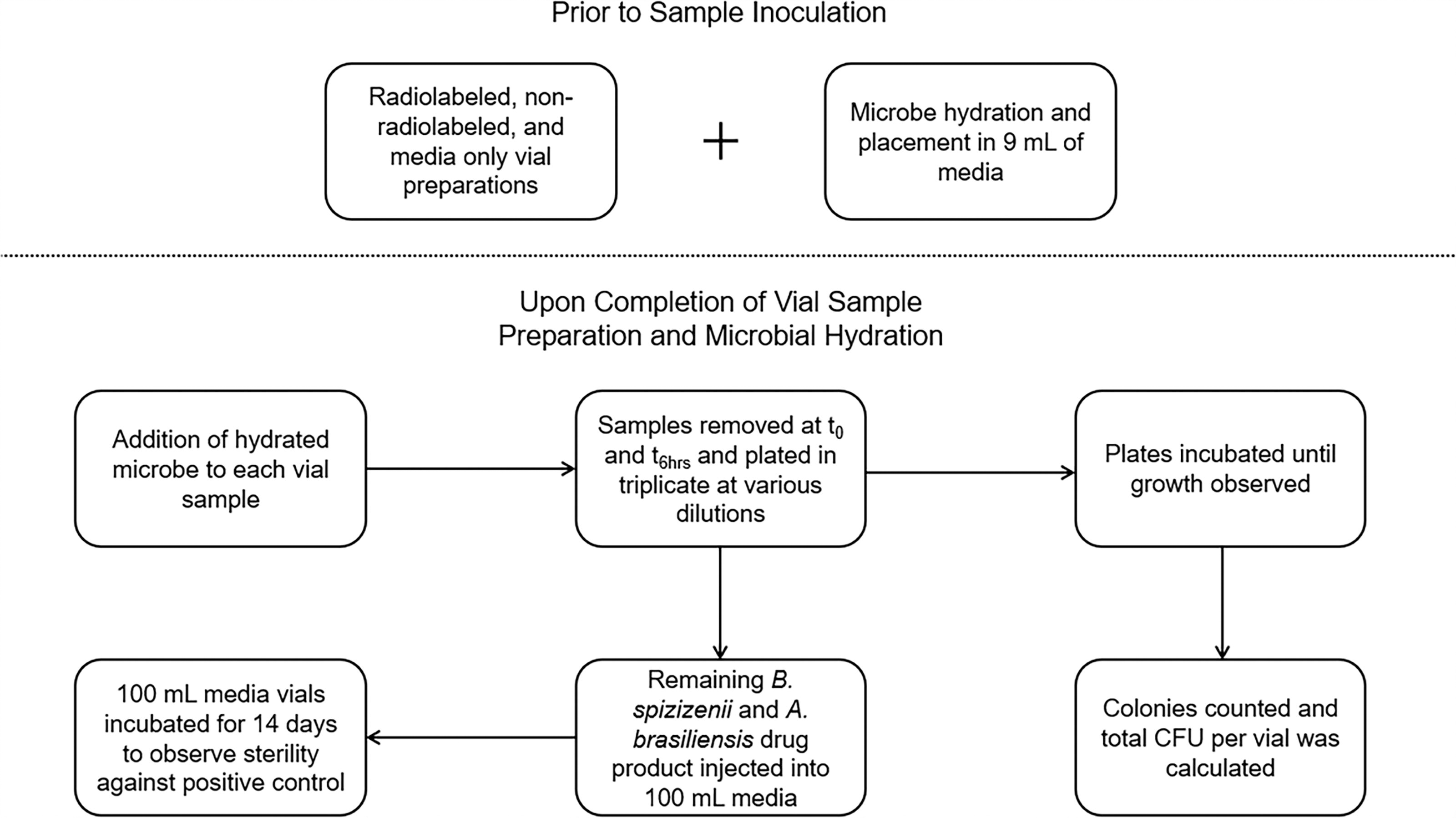
Process overview of the experiment details. Process overview depicting the high-level steps of the experimental design.
Drug product and control preparation
[212Pb]Pb-DOTAMTATE in formulation solution was prepared by sterile filtering 3.9 mL of formulation solution (50 µg of DOTAMTATE, 6.9% ethanol, 28 mM ascorbic acid, 0.028% Tween 80 in metal-free water) into a 10 mL glass vial followed by 5.2–9.1 mCi of 212Pb in 1.5 mL of 0.4M ammonium acetate, pH 6.5–7. The final formulation contains 50 µg DOTAMTATE, 111 mM ammonium acetate, 5% ethanol, 20 mM ascorbic acid, and 0.2% Tween 80. The vials were incubated for 10 min at room temperature before confirming that chelation was ≥95% by iTLC. The vials containing nonradiolabeled DOTAMTATE in formulation solution were prepared by sterile filtering 3.9 mL of formulation solution into a 10 mL glass vial followed by 1.5 mL of 0.4M ammonium acetate bringing it to the same final concentrations as the radiolabeled vials. The vials were prepared and kept at 4°C until they were brought to room temperature the afternoon prior to chelation. The media-only vials contained 5.4 mL of filtered media; SCD, FTM, or Sabouraud dextrose.
Microbial inoculation of sample vials
About 500 µL of each rehydrated microbial suspension ranging from 106 to 107 CFUs was added to each vial type.
C. sporogenes was grown to an OD600 of 0.45, and the total volume was concentrated into 900 µL. This was split into three, 300 µL samples, and added to the three vials containing different drug products.
At t0, a sample was removed from each vial, and 400 µL of diluted material, ranging from 1/1,000 to 1/50,000, was plated in triplicate. At t6hrs, samples were removed, and 400 µL of material (ranging from no dilution to 1/50,000) was plated in triplicate. Bacteria were grown on TSA plates at 30–35°C, while fungus was grown on Sabouraud dextrose plates at 20–25°C and incubated until colonies were present.
CFU quantification and calculations
Colonies on each triplicate set of plates were counted, and the average CFUs were calculated for each vial type along with the fold and log reductions of the vials with and without the radionuclide. Below are the calculations used:
Average CFU/Vial:
The dilution factor corresponds to the dilution at which the microorganism was plated following t0 and t6hrs incubation that achieved the required CFU per plate to be counted. While 400 µL of each dilution was plated, this corresponds to 1/13.5 of the total sample volume, 5.4 mL, contained in each vial. Each CFU replicate was multiplied by the appropriate dilution factor and 13.5 to determine the total CFU in the sample vial.
When determining which CFU replicates to consider for this calculation, the recommendation per the plate count method of USP < 61 > was used. The highest CFU per triplicate set of plates that did not exceed 250 CFU/plate on average was used for bacteria, while the highest CFU per triplicate set of plates that did not exceed 50 CFU/plate on average was used for fungus samples.
Fold Reduction Due to Formulation at t6hrs
:
Fold Reduction Due to Radionuclide at t6hrs:
Log Reduction:
This equation assumes 1 as the denominator as this cannot process with 0. As this results in values being nonexact, the log reduction is stated as greater than (>) the value calculated with 1 as the denominator.
Sterility Testing Inoculation of B. spizizenii and A. brasiliensis for 14 d.
Upon the conclusion of the t6hrs sample plating for B. spizizenii and A. brasiliensis, the remaining ∼5 mL of drug product from the [212Pb]Pb-DOTAMTATE inoculated samples were injected into a 100 mL SCD media bottle. Positive control samples were prepared by diluting the initial, rehydrated Epower pellet to a total of 75 CFU/mL, and injecting this into a separate 100 mL SCD media bottle. These SCD media bottles containing B. spizizenii, A. brasiliensis, or positive controls were observed for growth by evaluation of turbidity following 14 d of incubation at 20–25°C.
Dosimetry
Absorbed dose determination description
The energy deposited within the vial over the 6 h following production was calculated based on the number of atom decays for 212Pb and its daughters over that time span. For this purpose, it was assumed that 212Pb was pure at the time of elution and ingrowth of the daughters started at t0.
Using the Bateman equation, the amount of 212Pb and its daughters remaining at the end of the 6 h was calculated. About 68% of the 212Pb atoms have not yet decayed, while 6.95% are 212 Bi, < 0.001% are 212Po, 0.13% are 208Tl, and the remaining 25.45% are 208Pb. 13 The number of atoms of 212Pb at t0, remaining 212Pb at t6, 212Bi at t6, 212Po at t6, 208Tl at t6, and newly formed 208Pb were calculated.
The sum of average α and β− eV emissions were calculated by multiplying the number of atoms at each step of the decay chain by the energy released by all parents assuming 101.3 keV for 212Pb, 2,174 (to 208Tl) and 493.7 (to 212Po) keV for 212Bi, 9,785.2 keV for 212Po, and 559 keV for 208Tl. 14 The branching ratio of 64.07% for 212Po and 35.93% for 208Tl was applied.
The sum of the eV was converted to Joules and then divided by the mass of the final formulation to obtain kGy.
Stability study
[212Pb]Pb-DOTAMTATE, prepared as described above, was sampled at t0 and stored at room temperature for 24 h before another sampling occurred. t0 and t24hrs samples were analyzed for stability by reverse phase radio high-performance liquid chromatography (Radio-HPLC) using an Agilent 1220 HPLC with a C18 column (Restek) and an acetonitrile gradient of 17.5%−37.5% over 8 min. Fractions were collected every 6 s and analyzed by auto γ-counter (Perkin Elmer).
Results
[212Pb]Pb-DOTAMTATE, nonradiolabeled DOTAMTATE, and media-only vials were inoculated with six microorganisms to assess the bactericidal and fungicidal efficacy of the drug, both nonradiolabeled and radiolabeled with 212Pb. After inoculation, samples were taken from each vial and plated in triplicate.
The aerobic bacteria, S. aureus (Table 1), P. aeruginosa (Table 2), and B. Spizizenii (Table 3), all exhibited >6 logs of reduction with the first two bacteria exhibiting >7 logs of reduction, suggesting a potent sterilization effect of [212Pb]Pb-DOTAMTATE. This is particularly noticeable with S. aureus, which was more resistant to the formulation solution of the nonradiolabeled DOTAMTATE vial; however, zero colonies were present after incubation with [212Pb]Pb-DOTAMTATE for 6 h.
S. aureus Average CFU per Vial with Dilution Correction and Fold Reduction of Samples
CFU, colony forming units.
P. aeruginosa Average CFU per Vial with Dilution Correction and Fold Reduction of Samples
CFU, colony forming units.
B. spizizenii Average CFU per Vial with Dilution Correction and Fold Reduction of Samples
CFU, colony forming units; d, days.
Anaerobic bacteria, C. sporogenes, also showed >7 logs of reduction from over 16 million bacteria present in the media-only sample at t0 and zero colonies present in the t6hrs [212Pb]Pb-DOTAMTATE samples (Table 4).
C. Sporogenes Average CFU per Vial with Dilution Correction and Fold Reduction of Samples
CFU, colony forming units.
The fungi, C. albicans (Table 5) and A. brasiliensis (Table 6), both displayed > 6 logs of reduction. C. albicans displayed > 7 logs of reduction as the high number of CFUs used to inoculate the drug product vials allowed for a pronounced cytotoxic effect of 212Pb to be observed. Neither fungus displayed a high sensitivity to the formulation solution as A. brasiliensis had a 1.35-fold reduction while C. albicans had no fold reduction as this fungus increased in CFUs when incubated in the formulation solution. Though, similar to S. aureus (Table 1), zero colonies were present after incubation with [212Pb]Pb-DOTAMTATE for 6 h.
C. albicans Average CFU per Vial with Dilution Correction and Fold Reduction of Samples
CFU, colony forming units.
A. Brasiliensis Average CFU per Vial with Dilution Correction and Fold Reduction of Samples
CFU, colony forming units; d, days.
The microorganisms, B. spizizenii and A. brasiliensis, only had single digit millions of bacteria per vial. To ensure that no bacterial growth occurred after the t6hrs incubation, the vials containing ∼5 mL of drug product were inoculated into 100 mL of SCD media and incubated at 20–25°C for 14 d. No growth was observed upon the completion of these 14 d (Table 3 and Table 6).
The absorbed dose to each vial was determined (Table 7) and the data shows that a cytotoxic effect was observed despite an absorbed dose 25–45 times less than the recommended dose for sterility of 25 kGy, highlighting the potency of 212Pb.
Absorbed Dose per Vial Containing [212Pb]Pb-DOTAMTATE and Various Microorganisms
Despite the cytotoxic effect of the 212Pb, which causes 6–7 logs of reduction of microbial growth, [212Pb]Pb-DOTAMTATE stability is not affected. The drug remains stable up to 24 h, the expiration time point of this drug product, when stored at room temperature and examined by radio-HPLC (Fig. 2).
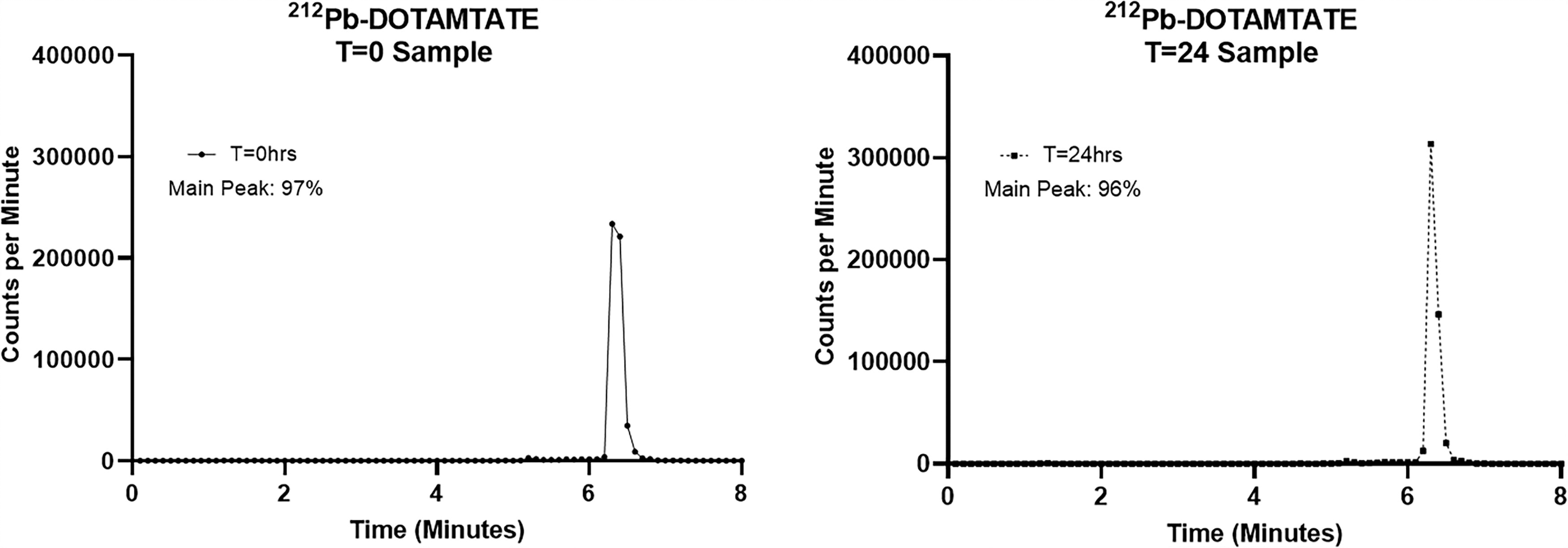
Stability of [212Pb]Pb-DOTAMTATE over 24 h by Radio-HPLC. Radiolabeled 212Pb-DOTAMTATE at 25°C was tested for stability over 24 h. At t0 and t24hrs, 2 µCi/50 µL samples of the [212Pb]Pb-DOTAMTATE were injected into a C18 column on an Agilent 1220 HPLC using an acetonitrile gradient. Fractions are collected every 6 s over an 8-min method and then run on a Perkin Elmer Auto Gamma Counter to obtain the counts per minute. The main peak is determined by dividing the counts making up the main peak of the [212Pb]Pb-DOTAMTATE sample (two fractions to the left of the apex, the fraction of the apex, and eight fractions to the right of the apex) by the total counts of all fractions. Radio-HPLC, radio high-performance liquid chromatography.
Discussion
This study investigated the cytotoxic effect of 212Pb on six different microbes, which were selected based on their recommended usage for sterility testing per USP < 61 > and < 71 >. Vials containing [212Pb]Pb-DOTAMTATE drug product, nonradiolabeled DOTAMTATE in formulation solution, or media alone were inoculated with ≥1 million CFUs to assess the cytotoxic efficacy of the drug alone.
At t0, the media-only vials exhibited CFU counts ranging from 1 × 106–2 × 107 CFU/vial indicating the presence of viable microorganisms in substantial numbers immediately after drug inoculation. A growth reduction of > 2 logarithmic orders was observed between t0 and t6hrs in the vials containing nonradiolabeled DOTAMTATE. These findings suggest that the drug product’s formulation exerts a cytotoxic effect on bacteria, with varying susceptibility among different microorganisms. This outcome aligns with previous research that demonstrated variations in minimum inhibitory concentrations (MIC) of bacteria when exposed to ethanol. 15 For instance, P. aeruginosa exhibited an MIC of 1.56% compared to S. aureus with an MIC of 6.25%. Our observations regarding sensitivity to nonradiolabeled DOTAMTATE were consistent with these previous findings, with P. aeruginosa and B. spizizenii displaying greater susceptibility compared to S. aureus. Similarly, ascorbic acid (vitamin C) has been shown to inhibit growth of bacteria including S. aureus, B. subtillis, and P. aeruginosa although at higher concentrations than used in this study. 16
For vials containing [212Pb]Pb-DOTAMTATE drug product, between 1.2 × 106–2.3 × 107 CFUs of the microorganisms were detected at t0, indicating that instantaneous microbial death did not occur. After 6 h, however, no CFUs were found on the plates inoculated with any of the tested microorganisms. This suggests a substantial 6-log reduction or greater in microbial growth in the presence of 212Pb compared to both the media-only samples and the [212Pb]Pb-DOTAMTATE samples at t0. As the 212Pb activities used in this study are lower than the activity present at the time of manufacturing for the [212Pb]Pb-DOTAMTATE phase 2 clinical doses, the log reduction in a clinical manufacturing setting would therefore be at least the 6 logs observed in this study. These results corroborate expectations, suggesting that 212Pb works as a self-sterilizing agent against microbial growth.
The significance of this data lies in the demonstration of all [212Pb]Pb-DOTAMTATE vials being free of viable microorganisms 6 h post inoculation. While no microorganisms are present upon the completion of radiopharmaceutical production under Good Manufacturing Practices (GMP), if trace amounts were to be present in the drug product (t0), they would no longer be viable, as demonstrated, at the time of patient administration (t6hrs). Though this observed the effects of [212Pb]Pb-DOTAMTATE as the primary drug product, there is a clear indication that the 212Pb itself is the primary cytotoxic agent resulting in 6–7 logs of reduction more than just the formulation solution. This has broad implications beyond this specific drug product, as any drug product prepared with 212Pb can also carry out this self-sterilizing ability. This is significant as the > 6 logs of reduction in microbes of each [212Pb]Pb-DOTAMTATE drug product vial reach the SAL of a sterile product, while being far below the accepted dose of 25 kGy.
Conclusion
This study sought to demonstrate that in vivo generators of α-particle radiopharmaceuticals can achieve at least 6 logs of reduction and can be accepted as sterile products. The outcome of this experiment displays the potency of 212Pb and gives credit to the notion that therapeutic clinical doses utilizing such α-generators with relatively short half-lives, such as 212Pb, can sterilize products due to the inherent nature and cytotoxicity of the radionuclide itself. This potency of 212Pb to kill any microbial growth that might be present in a GMP produced drug product gives confidence in a clinical setting that otherwise would not be assured due to the long time points of traditional sterility testing. This is important, as clinical doses are administered before the allowable time that sterility tests would ensure a sterile drug product. The observation that 212Pb serves as a self-sterilizing agent lessens the risk that these radiopharmaceuticals are administered and later found to produce positive sterility results. The data presented here warrant further research that aims to reduce future sterility testing in GMP environments when using an in vivo α-generator like 212Pb.
Footnotes
Authors’ Contributions
A.T.S.: Validation, formal analysis, investigation, writing—original draft, visualization, supervision, and project administration. T.A.S.: Methodology, formal analysis, resources, writing—review and editing, visualization, supervision, and project administration. R.R.: Investigation. Q.P.N.: Investigation. R.D.: Resources. J.J.T.: Conceptualization, formal analysis, writing—review and editing, and supervision.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article (and its supplementary information).
Author Disclosure Statement
All authors are Orano Med employees and declare no potential conflict of interest.
Funding Information
This study was funded by Orano Med.
