Abstract
Background:
Chemotherapy based on oxaliplatin (OXA) is the first-line treatment for advanced colorectal cancer (CRC), and acquired resistance to OXA is the main reason for clinical treatment failure in CRC.
Methods:
To search for compounds that can reverse OXA resistance, we screened a small molecule inhibitor drug library and identified a drug, Raddeanin A (RA), that enhanced the anticancer effect of OXA. Using human CRC cell lines, CRC organoid models, and in vivo subcutaneous tumorigenic studies, we determined that RA inhibits the proliferation of CRC cells by promoting apoptosis and inducing cell cycle arrest.
Results:
We constructed OXA-resistant CRC cell lines and demonstrated that RA enhances the sensitivity of these cells to OXA. Further experiments showed that the mechanism by which RA enhanced the anticancer effects of OXA in CRC was by inhibiting the activation of the WNT/β-catenin signaling pathway.
Conclusions:
Because RA has been shown to be biocompatible in animal models, there is a possibility that RA could be developed as a sensitizer for resistant cancer cells or as a novel lead compound to enhance the therapeutic efficacy of OXA in resistant CRCs.
Introduction
Colorectal cancer (CRC) is one of the most common malignant tumors, and it poses a significant threat to human life and health 1,2 It is also the second leading cause of cancer-related deaths globally. 1 –3 While immune therapy is gradually emerging as an effective treatment for CRC, 4 –6 chemotherapy remains an indispensable strategy in the management of the disease. 7,8 For locally advanced CRC, the standard treatment strategy involves neoadjuvant chemotherapy combined with curative resection and adjuvant therapy. 9 –11
Chemotherapy based on oxaliplatin (OXA) is commonly used as first-line treatment for advanced CRC. 12,13 Although chemotherapy is often initially effective, approximately 50% of patients with stage II or III CRC develop resistance to adjuvant therapy based on OXA. 7,14 OXA resistance is thus a major obstacle for the treatment of CRC and a significant cause of chemotherapy failure. Currently, there are no clinically available biomarkers for OXA response. Therefore, studying the mechanisms leading to OXA resistance and searching for compounds that can reverse OXA resistance are key to improving the clinical prognosis of CRC.
A candidate molecule for the treatment of CRC and other malignancies is Raddeanin A (RA), an oleanane class triterpenoid saponin that is isolated from Anemone raddeana Regel. 15,16 Various studies have shown that RA exhibits cytotoxic effects by inducing apoptosis and inhibiting the proliferation and invasion of various types of cancer cells, including breast cancer, hepatocellular carcinoma, gastric cancer, and non-small cell lung cancer. 17 –20 However, the therapeutic role of RA has not yet been tested in the context of OXA-resistant CRC. Thus, further studies are needed to investigate the potential of RA in overcoming OXA resistance in CRC and to elucidate its underlying molecular mechanisms.
Wingless-type MMTV integration site family member 10A (WNT10A) is a member of the WNT family, 21 and WNT10A expression has been found to be upregulated in CRC and esophageal cancer. 22 WNT10A is an upstream protein in a pathway leading to the activation of β-catenin, and increased levels of WNT10A may lead to the upregulation of β-catenin, 23,24 which is associated with tumor invasion and poor prognosis. 25,26 Importantly, the WNT/β-catenin signaling pathway has been found to promote chemotherapy resistance by upregulating the drug transporter P-glycoprotein. 27,28 Therefore, these results suggest that inhibition of WNT10A and the WNT/β-catenin signaling pathway may be a strategy to overcome OXA-induced resistance in CRC.
In this study, in order to identify additional small molecule inhibitors for use in cancer treatment, we screened natural compounds from a library of small molecule inhibitors in an attempt to identify substances that can enhance the sensitivity of CRC cells to OXA. We identified an activity of RA in the enhancing of the anticancer effects of OXA. We then performed in vitro studies in human CRC organoid models and resistant cell lines and in vivo studies in a mouse tumor model to evaluate the antitumor activity of RA and its role in enhancing sensitivity to OXA. Transcriptomic sequencing analysis revealed that RA can reverse OXA resistance in CRC cells by inhibiting the expression of proteins associated with the WNT10A and WNT/β-catenin signaling pathways. In summary, our research findings have revealed the antitumor activity of RA in CRC both in vitro and in vivo, and for the first time its association with the expression of proteins in the WNT10A and WNT/β-catenin signaling pathways.
Materials and Methods
Cell lines and drug treatment
The CRC cell lines HCT116, DLD-1, and LOVO were purchased from American Type Culture Collection (ATCC) and cultured in Dulbecco's modified Eagle medium (Gibco, USA) supplemented with 10% fetal bovine serum (BIOEXPLORER, USA) and 1%penicillin-streptomycin (Gibco, USA) at 37°C. The parental cell lines were subjected to continuous induction with increasing concentrations of OXA, until the induced-resistance strains exhibited normal growth without significant inhibition at an OXA concentration of 2 μg mL−1 for HCT116-R and 1 μg mL−1 for LOVO-R. These OXA concentrations were then included in the media for maintenance of the lines. DLD-R were kindly provided by Zhaoliang Yu of the State Key Laboratory of Oncology in South China Collaborative Innovation Center of Cancer Medicine, Sun Yat-sen University Cancer Center. Routine monthly testing was conducted to check for mycoplasma contamination in cell cultures. The compound library, RA, and OXA were obtained from Topscience (Shanghai, China).
Colony formation assay
Cells were seeded at a density of 2 × 105 per 60 mm culture dish, and the plates were incubated at 37°C in a 5% CO2 atmosphere. The culture medium was refreshed every 3 days for a total of 9 days. Then, the cells were washed with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde, washed again with PBS, and stained with 0.1% crystal violet for 20 min. This process was followed by another PBS wash and drying.
Tumorsphere formation assay
Each well of a 24-well ultralow attachment plate was seeded with 4 × 103 uniformly distributed cells in a total of 500 μL of culture medium with or without test compounds. The cells were replenished with culture medium containing test compounds as appropriate every 3 days. After a 7-d incubation period, the spheroids were photographed.
Examinations of cellular viability
Cells were seeded at a density of 3 × 103 per well of 96-well plates. After 24 h, the cells were treated with different concentrations of drugs and incubated at 37°C for an additional 48 h. The viability of the cells was determined using a Cell-Counting-Kit 8 (CCK-8) assay kit according to the manufacturer’s protocol.
Analyses of cell cycle distribution and apoptosis
A flow cytometry approach was used to evaluate the cell cycle distribution of treated cells. After treatment of with a drug for a specified time period, the cells were released from the plate with trypsin, centrifuged, fixed in 70% ethanol, and stained with propidium iodide. The DNA content of the stained cells was analyzed using a Beckman flow cytometer.
For cell apoptosis analyses, treated cells were released from plates with trypsin and resuspended in PBS, and then an appropriate number of cells were centrifuged and incubated with Fluorescein isothiocyanate isomer (FITC)-Annexin V binding solution. Further staining was performed using annexin V and propidium iodide. The apoptotic status of the cells was analyzed using a Beckman flow cytometer.
Immunohistochemistry
Immunohistochemical staining was performed essentially as described. 29 Briefly, mouse colon sections were deparaffinized in xylene and rehydrated in ethanol and then incubated with 3% hydrogen peroxide to block endogenous peroxidase activity. To retrieve antigens, the sections were immersed in citrate buffer (pH 6.0) and then heated in a microwave for 15 min. After blocking with 20% goat serum, the sections were incubated overnight at 4°C with primary antibodies against WNT10A or β-catenin (PROTEINTECH, Wuhan, China). The sections were washed and then incubated with the enVision Plus system-HRP (DAB; DAKO, Denmark) and counterstained with Mayer’s hematoxylin. The area containing positive staining and the percentage of positive nuclei were quantified using ImageJ software (NIH).
Western blot analysis
The nuclear and cytoplasm proteins of two cell lines were extracted by using the nuclear and cytoplasmic protein extraction kit (KeyGen Biotech, China). Protein concentrations were determined using the BCA protein analysis kit (KeyGen Biotech, China). Equal amounts of protein were separated by sodium dodecyl-polyacrylamide gel electrophoresis and transferred onto a polyvinylidene difluoride membrane (EMD Millipore, MA, USA). The membrane was blocked with 5% skim milk at room temperature for 1 h, followed by incubation with primary antibodies at the appropriate dilutions overnight at 4°C. β-Actin was used as a loading control.
RNA extraction and qPCR
Total RNA was extracted using Trizol reagent (Invitrogen, USA). An reverse transcription polymerase chain reaction (RT-PCR) kit (Takara, China) was used for cDNA synthesis, using 1 μg of total RNA as the template. The synthesized cDNA was subjected to real-time quantitative PCR (qPCR) using a SYBR Green PCR reagent kit with primers specific to the target gene (WNT10A forward primer: 5′-GGT CAG CAC CCA ATG ACA TTC, reverse primer: 5′-GGA TGG CGA TCT GGA TGC). The amplification program consisted of incubation at 95°C for 15 s and 60°C for 60 s. GAPDH was used as an internal control. All cycle threshold (Ct) values were determined using the CFX96 PCR Real-time™ Detection System (CA).
siRNA-mediated knockdown
Experimental siRNA and nontargeting control constructs were purchased from IGE BIOTECHNOLOGY LTD, Guangzhou, China. Cells were transfected with siRNA duplexes at a final concentration of 10 nM using Lipofectamine RNAiMAX, according to the manufacturer’s instructions. The sequences of the WNT10A-siRNA constructs: sense strand, 5′-CCA CGA AUG CCA ACA CCA AdTdT; antisense strand, UUG GUG UUG GCA UUC GUG GdTdT.
Organoid culture
CRC tumor tissues were obtained from The Sixth Affiliated Hospital of Sun Yat-sen University with informed consent, and the study was approved by the relevant ethical committee. CRC organoid culture was performed using previously described methods. 29
Mice experiments
Female nude mice (4 weeks old) were obtained from GemPharmatech (Jiangsu, China) and housed in a high-quality animal laboratory with SPF standards. They received a subcutaneous injection of 3 × 106 HCT116 cells on the right side. The tumor widths (W) and lengths (L) were measured with a caliper, and the volumes (V) were calculated with the formula V = W2 × L/2. When tumor volumes reached 50 mm3, the mice with similar tumor burdens were randomly assigned to different groups. During the pharmaceutical tests with RA, HCT116-xenograft mice were administered the following treatments by injection into the peritoneum once every 3 days: Dimethyl sulfoxide (DMSO) (n = 4), OXA (n = 4, 2 mg/kg), RA (n = 4, 4 mg/kg), and a mixture of both substances at the same dosages (n = 4).
Tumor progression was monitored every 2 or 3 days based on the body weight and tumor size for approximately 2 weeks. No experimental samples were excluded, and no mouse deaths were observed during the observation period. The tumors were excised when they reach 1500 mm3 or at the end of the study. All animal studies were conducted in accordance with the animal protocol that was approved by the Animal Care and Ethics Committee of Sun Yat-sen University, and they followed national guidelines for animal care and use.
Statistical analysis
Statistical analyses of the data were performed using GraphPad Prism 9.0. Student’s t-tests were used to ascertain the disparities between two groups. Two-way analysis of variance tests were used to determine the statistical differences between dose–response curves. The data are reported as the mean ± standard deviation of three separate trials. Statistical tests were considered to indicate statistical significance if the p-value was less than 0.05, unless otherwise specified. The statistical significance is indicated in figures as follows: *p < 0.05; **p < 0.01; ***p < 0.001.
Results
Construction of drug-resistant cell lines
In order to identify novel compounds that could overcome the OXA resistance that is often observed during chemotherapy, we first constructed two OXA-resistant CRC cell lines (HCT116-R and LOVO-R). They were obtained by long-term induction in culture media with a gradient of OXA concentrations, resulting in the acquisition of stable drug resistance. As seen in cell proliferation inhibition curves (Fig. 1A), the IC50 value for oxa in the resistant cell line HCT116-R (15.15 μM, 95%CI: 14.17–16.22 μM) was 7.5-fold higher than that of wild-type HCT116 (2.08 μM, 95%CI: 1.10–4.27 μM), and the IC50 value for OXA in the resistant cell line LOVO-R (9.03 μM, 95%CI: 8.20–9.95 μM) was fivefold higher than that of wild-type LOVO (1.76 μM, 95%CI: 1.47–2.11 μM). The resistance index for both resistant cell lines was greater than fivefold, indicating successful induction of drug resistance. Resistance in both cell lines was further confirmed upon the performing of clonogenic assays in the presence of increasing concentrations of OXA (Fig. 1B).
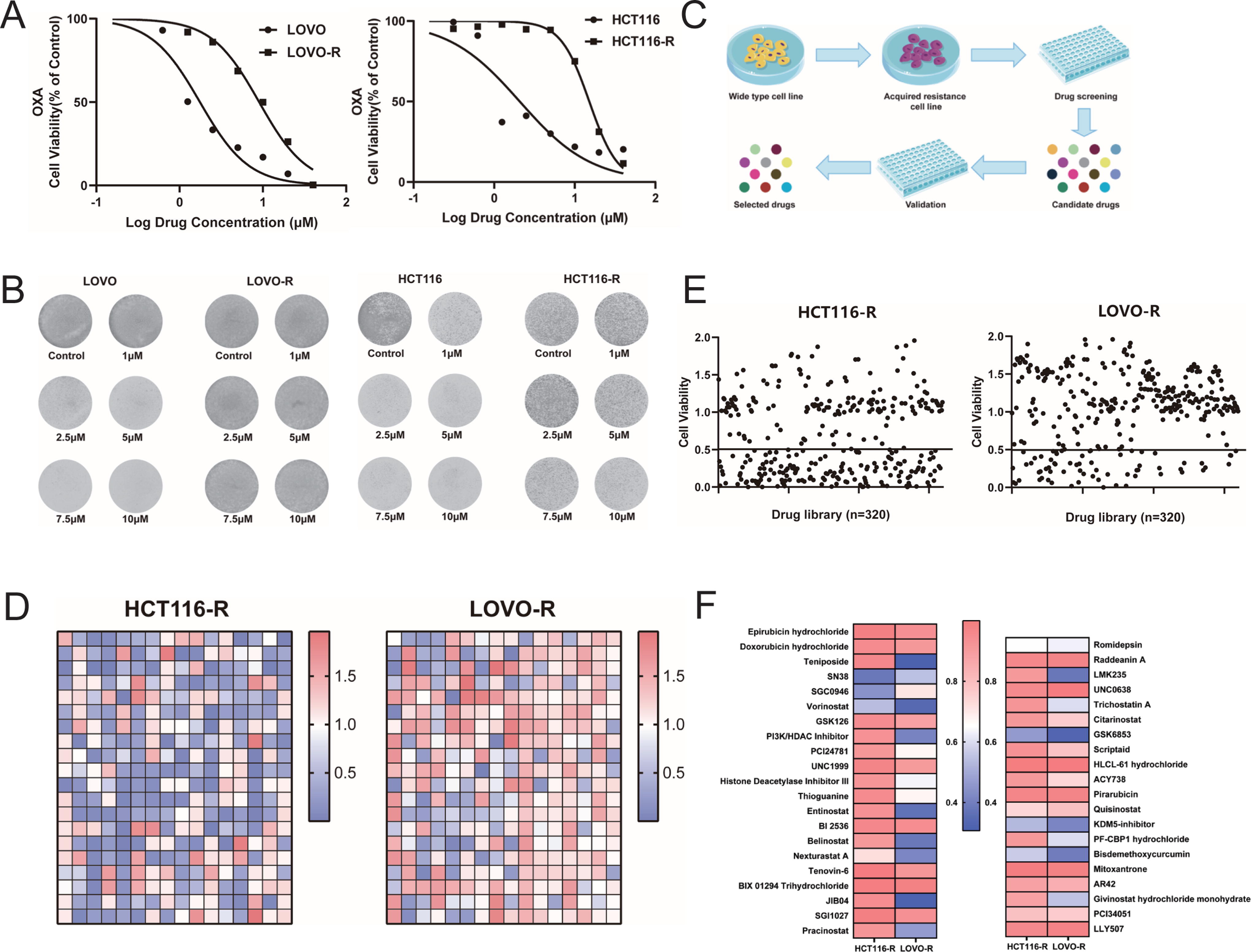
Construction of drug-resistant cell lines and drug screening.
Identification of small molecules with anti-CRC activity
We screened a small molecule drug library using the resistant cell lines HCT116-R and LOVO-R, and we identified and validated small molecule drugs capable of overcoming resistance (Fig. 1C). The drug library used in the study included small molecule inhibitors that primarily targeted pathways related to cell cycle, epigenetic regulation, metabolism, and apoptosis. After 72 h of drug treatment, the viability of the cells was tested using the CCK-8 assay, and drugs leading to a cell viability index below 0.5 were considered to be potentially capable of overcoming OXA resistance in CRC.
The preliminary screening results demonstrated that 41 small molecule inhibitors exhibited the ability to overcome drug resistance in both resistance models (Fig. 1D and E). These 41 potential drugs were further validated in a secondary screen in both resistant cell lines (Fig. 1F), using as a cutoff value an inhibition rate in the resistant cells that was 10-fold higher than that in control cells. We ultimately identified 13 small molecule inhibitors (Table 1) that were capable of reversing the resistant phenotypes of both HCT116-R and LOVO-R. Of these 13 candidates, we selected RA for the subsequent experiments based on a literature review and preliminary experimental results.
13 Small Molecule Inhibitors
RA inhibits the growth of colorectal cancer cell lines
To determine the inhibitory effect of RA on three wild-type CRC cell lines (HCT116, DLD-1, and LOVO), CCK-8 and plate colony formation assays were conducted on cells treated with different concentrations of RA. The results demonstrated that RA inhibited the proliferation of CRC cells in a dose-dependent manner (Fig. 2A and B).
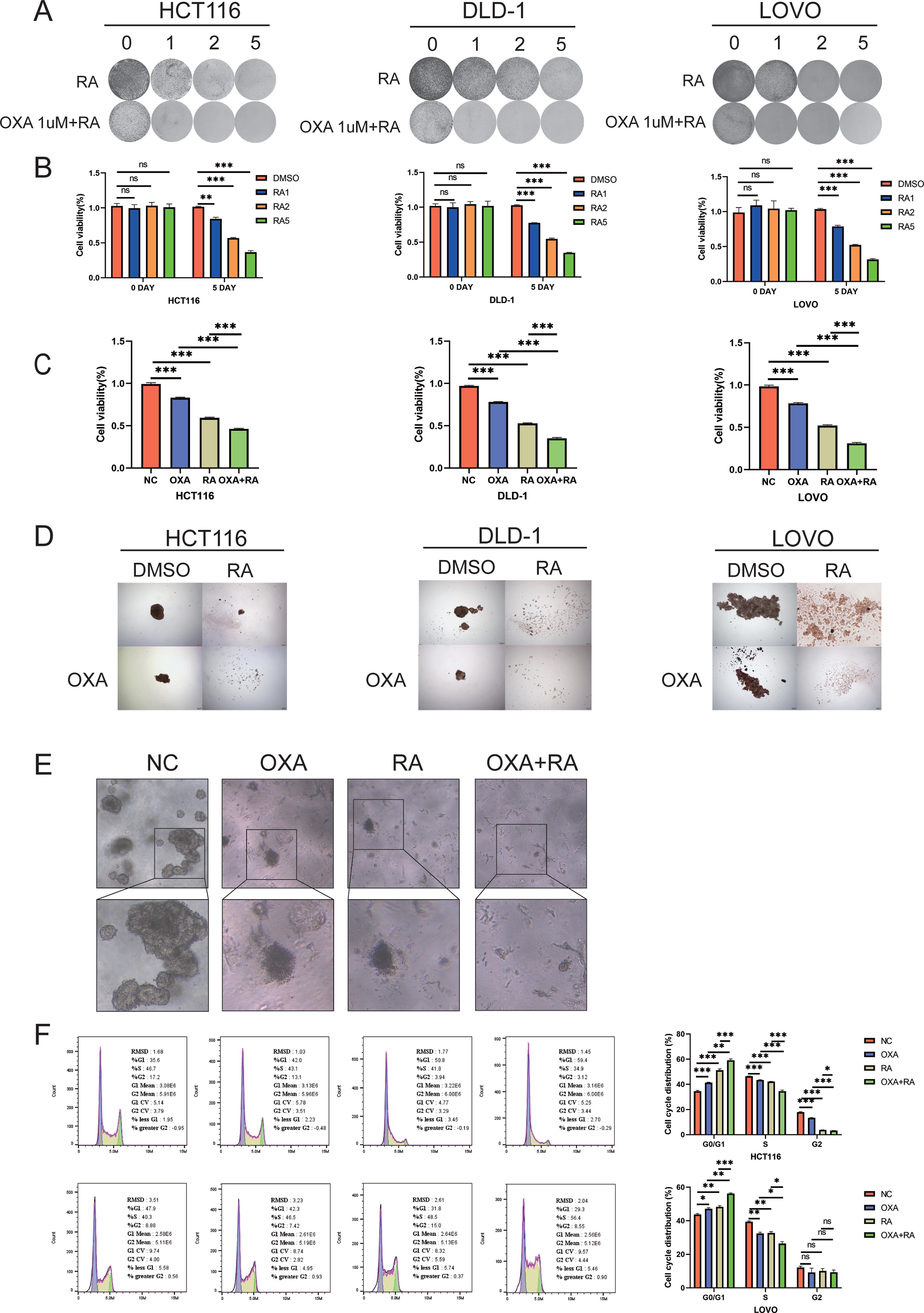
RA inhibits proliferation and induces apoptosis of CRC cells.
(Continued.)
To determine whether RA can enhance the anticancer effect of OXA, we treated the three wild-type CRC cell lines with OXA and RA and then evaluated cellular activity with CCK-8, plate colony formation, tumor sphere formation assays, and CRC organoid models (Fig. 2A,C–E). Here, RA was found to enhance the sensitivity of the three wild-type CRC cell lines to OXA.
We performed additional experiments to investigate the mechanism by which RA sensitizes cells to OXA. The molecular mechanisms underlying OXA resistance in tumors are known to involve apoptosis evasion and cell cycle arrest, 28,30 and RA has been shown to promote apoptosis in CRC cells in a dose-dependent manner. 31 Therefore, to further investigate the mechanism behind the growth inhibition of wild-type CRC cell lines by the combination of RA and OXA, we examined the cell cycle distribution of treated HCT116 cells using flow cytometry (Fig. 2F). Compared to the control group, treatment of HCT116 cells with both OXA had statistically significant effects on the distribution of cells in G0/G1, S, and G2/M phases (all p < 0.001). Moreover, compared with treatment with either drug alone, the treatment with the drug combination led to a significant increase in cells in the G0/G1 phase (p < 0.05) and a significant decrease in cells in the S phase (p < 0.05) (Table 2). These results indicate that, in addition to the increase in apoptosis, treatment with the combination of OXA and RA leads to cell cycle arrest at the G1/S phase. Similar results were observed in the wild-type LOVO cell lines (Fig. 2F).
Cell Cycle Distribution
NC,normal control; OXA, oxaliplatin; RA, Raddeanin A.
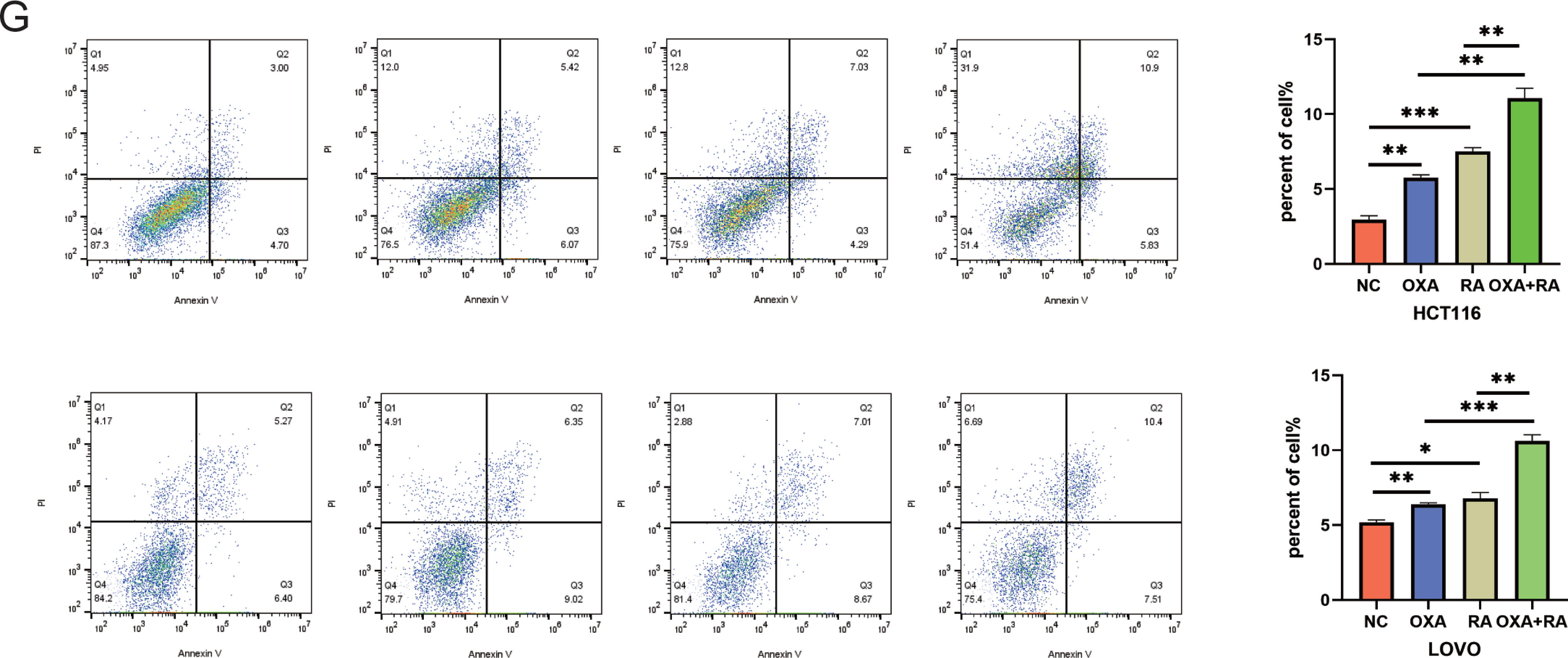
In addition, we hypothesized that RA might induce sensitivity to CRC by enhancing cellular apoptosis. To investigate the effects of both RA and OXA on the rate of apoptosis of CRC cells, we performed flow cytometric assays following Annexin V-FITC/Propidium Iodine (PI) double staining in HCT116 cells treated with vehicle, OXA alone, RA alone, or both OXA and RA. The apoptosis rates determined for each treatment group were as follows: vehicle-treated cells, 2.94 ± 0.49; cells treated with OXA alone, 5.76 ± 0.34; cells treated with RA alone, 7.51 ± 0.45; and cells treated with both OXA and RA, 11.07 ± 1.16(Fig. 2G). Thus, relative to control cells, both OXA and RA treatments promoted apoptosis in HCT116 cells. Furthermore, the rate of apoptosis observed in cells treated with the combination therapy was significantly higher than the rates observed in the monotherapy groups (p < 0.001), indicating that the combination of the two drugs significantly increased apoptosis in a CRC cell line. Similar observations were made in the other CRC cell lines, LOVO (Fig. 2G).
RA inhibits the proliferation of oxaliplatin-resistant colorectal cancer cells
In order to address potential confounding variables leading to the additive effects of the two chemical drugs, we further investigated the effects of RA on the proliferation and apoptosis of OXA-resistant CRC cells (HCT116-R, DLD-R, and LOVO-R). First, we used the CCK-8 assay and plate colony formation assay to observe the inhibitory effects of RA on OXA-resistant CRC cells. As shown in Figure 3A and B, RA exhibited a significant and dose-dependent inhibitory effect on the viability of OXA-resistant CRC cells.
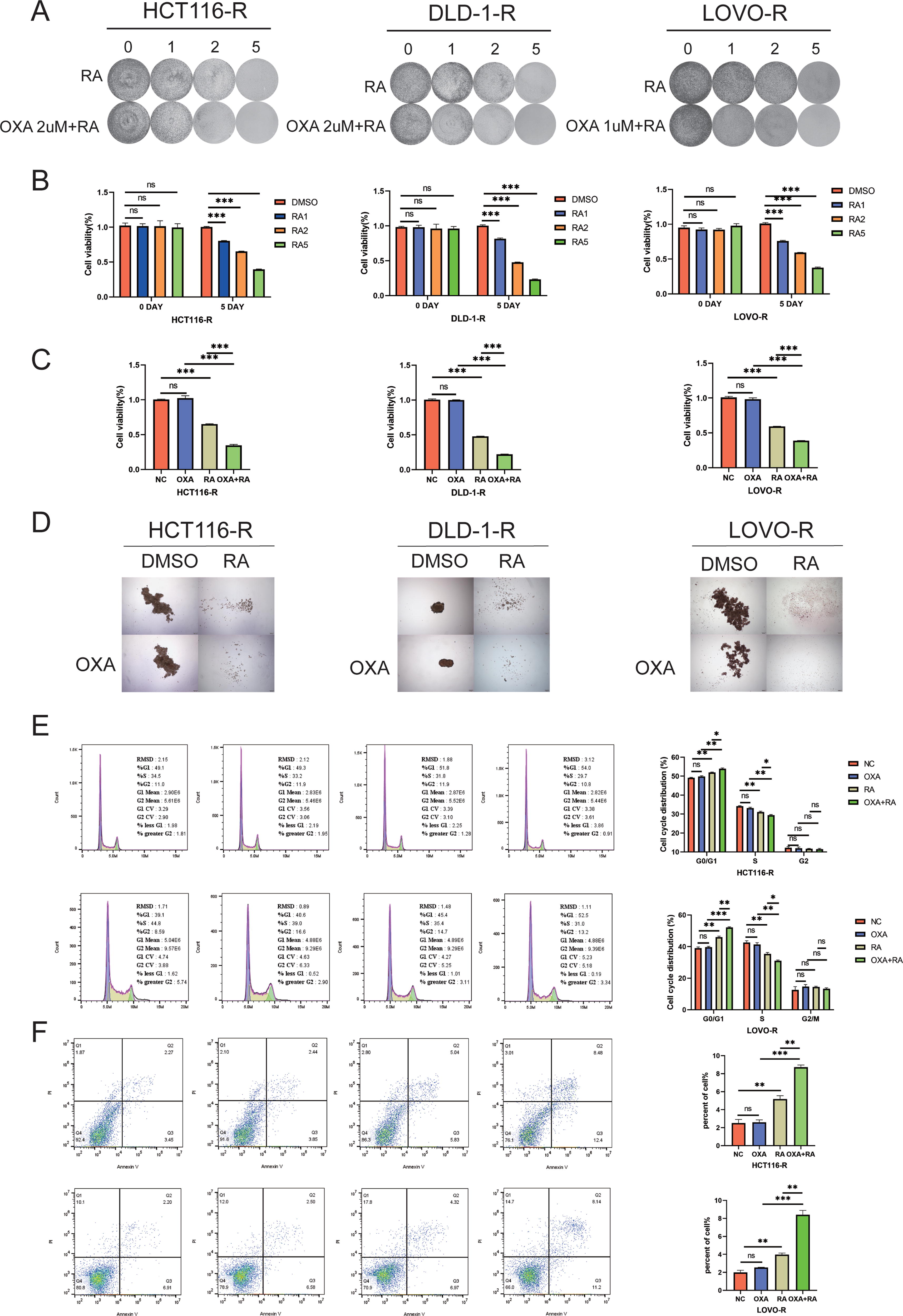
Effects of RA on OXA-resistant CRC cells.
To determine whether RA can enhance the anticancer effect of OXA in OXA-resistant CRC cells, we conducted CCK-8, plate colony formation, and tumor sphere formation assays on the three OXA-resistant cell lines following treatment with RA and OXA (Fig. 3A, C, and D). The results demonstrated that treatment with RA enhanced the sensitivity of the three OXA-resistant CRC cell lines to OXA.
To further investigate the mechanism underlying the growth inhibition of OXA-resistant CRC cell lines by the combination of RA and OXA, we examined the distribution of treated cells throughout the cell cycle using flow cytometry (Fig. 3E). Compared to the control treatment, treatment of the resistant cells with OXA did not have a statistically significant effect on the distribution of cells in G0/G1 phase (p = 0.1386), S phase (p = 0.1497), or G2/M phase (p = 0.7882) (Table 2). However, relative to single drug, the combination treatment involving OXA and RA led to a significant increase in cells in G0/G1 phase (p < 0.05) and a significant decrease in cells in S phase (p < 0.05), indicating that along with increasing apoptosis, the cotreatment leads to cell cycle arrest at the G1/S phase transition. Furthermore, similar results were observed using the LOVO-R cell line (Fig. 3E).
To further investigate whether the inhibitory effect of RA in combination with OXA on cell proliferation is associated with apoptosis, we utilized flow cytometry to examine the proapoptotic effects of RA on OXA-resistant CRC cell lines. According to flow cytometric analyses (Fig. 3F), the rate of apoptosis in cells treated with OXA alone (2.59 ± 0.49) was not significantly higher than the rate in control cells (2.51 ± 0.71, p = 0.8794). However, treatment with the combination of RA and OXA led to a significantly higher rate of apoptosis (8.72 ± 0.43) as compared to treatment with OXA alone or treatment with RA alone (5.18 ± 0.65, both p < 0.001).
RA enhances the anticancer effects of oxaliplatin by inhibiting the activation of the WNT/β-catenin signaling pathway
We performed RNA sequencing (RNA-seq) to provide more detailed insights into the mechanism by which RA enhances the anticancer effect of OXA. Specifically, we treated HCT116-R cells with OXA, or both OXA and RA for 48 h, then we investigated changes to gene expression by RNA-seq analyses. Of these 71 candidates, we selected WNT10A for the subsequent experiments based on a literature review and preliminary experimental results (Fig. 4A–C). Relative to the control group, we observed a significant increase in the expression of WNT10A in cells treated with OXA, while there was no significant change in WNT10A expression in cells treated with both OXA and RA relative to the expression in control cells. Subsequent, RT-qPCR and Western blot analyses using HCT116 wild-type cells to confirm the effects of OXA and RA treatments on the expression of this gene at the mRNA and protein levels (Fig. 4D and E). As shown in Figure 4E, RA reduced the expression of β-catenin in nuclear and cytoplasm and notably increased p-β-catenin levels in cytoplasm.
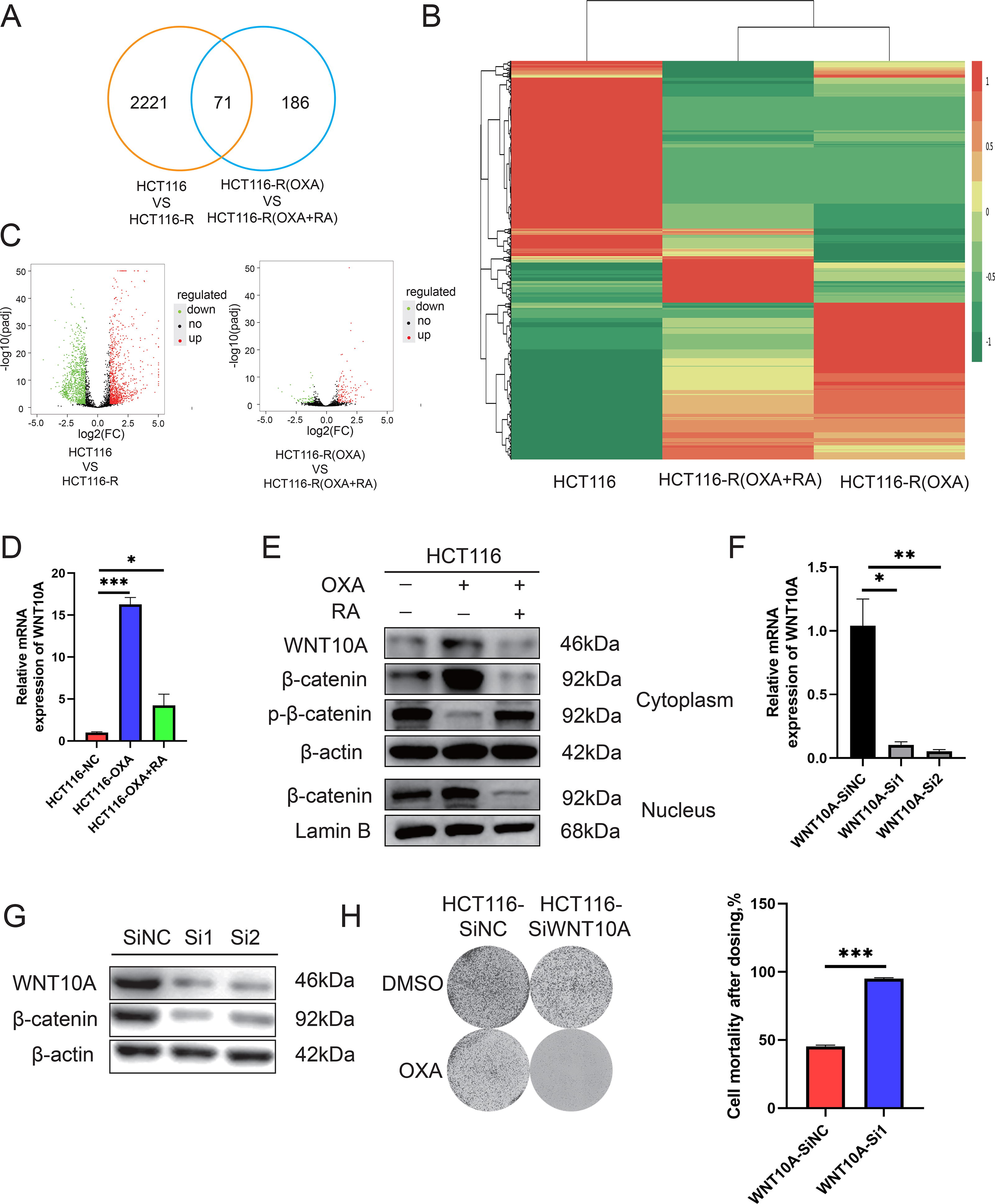
RA targets the WNT/β-catenin pathway.
WNT10A, as a member of the WNT gene family, plays important roles in the development of cancer by activating the WNT/β-catenin signaling pathway. Overexpression of WNT10A has been reported to lead to increased activity of β-catenin, which has been associated with tumor invasion and poor prognosis. 23,24 Previous studies have reported that the WNT/β-catenin signaling pathway is suppressed in CRC cells treated with RA. This suppression leads to growth inhibition and induction of apoptosis in these cells. 15,31
Based on these previous findings, we further investigated whether treatment with RA alone could suppress the WNT/β-catenin signaling pathway by reducing WNT10A expression. Western blot analyses showed a decrease in β-catenin expression along with a reduction of WNT10A protein levels upon treatment of HCT116 cells with RA. To further study the role of this effect in the synergy between RA and OXA, we constructed HCT116 cells that exhibited siRNA-mediated knockdown of WNT10A expression. A decreased expression of WNT10A mRNA in these cells was verified by RT-qPCR analyses (Fig. 4F); in addition, we observed by Western blot analyses that knockdown of WNT10A is correlated with a decrease in β-catenin protein levels (Fig. 4G).
When colony formation assays were performed in these cells, we found that the HCT116 cells in which WNT10A was knocked down were more sensitive to treatment with OXA as compared to control HCT116 cells (Fig. 4H). Taken together, these experimental findings suggest that RA may enhance the anticancer effect of OXA by inhibiting the WNT/β-catenin signaling pathway.
RA inhibits HCT116 tumor growth in vivo
To evaluate the anticancer effect of RA in vivo, we established a subcutaneous tumor model by injecting HCT116 cells into nude mice. Upon model establishment, the mice were divided into four treatment groups: a control group treated with DMSO, a group treated with 2 mg/kg OXA, a group treated with 4 mg/kg RA, and a group treated with both 2 mg/kg OXA and 4 mg/kg RA (Fig. 5A). With regard to the safety profile of RA, when compared to the control group, there was no significant decrease in the body weight of mice after treatment with RA or OXA, indicating good safety of RA in this animal model (Fig. 5B). When the effects of these treatments on tumor growth were investigated, the results showed that compared to the control treatment, RA treatment reduced both tumor weights and tumor volumes (Fig. 5C and D). Furthermore, the combination therapy of OXA and RA demonstrated significantly better therapeutic efficacy than did either monotherapy.
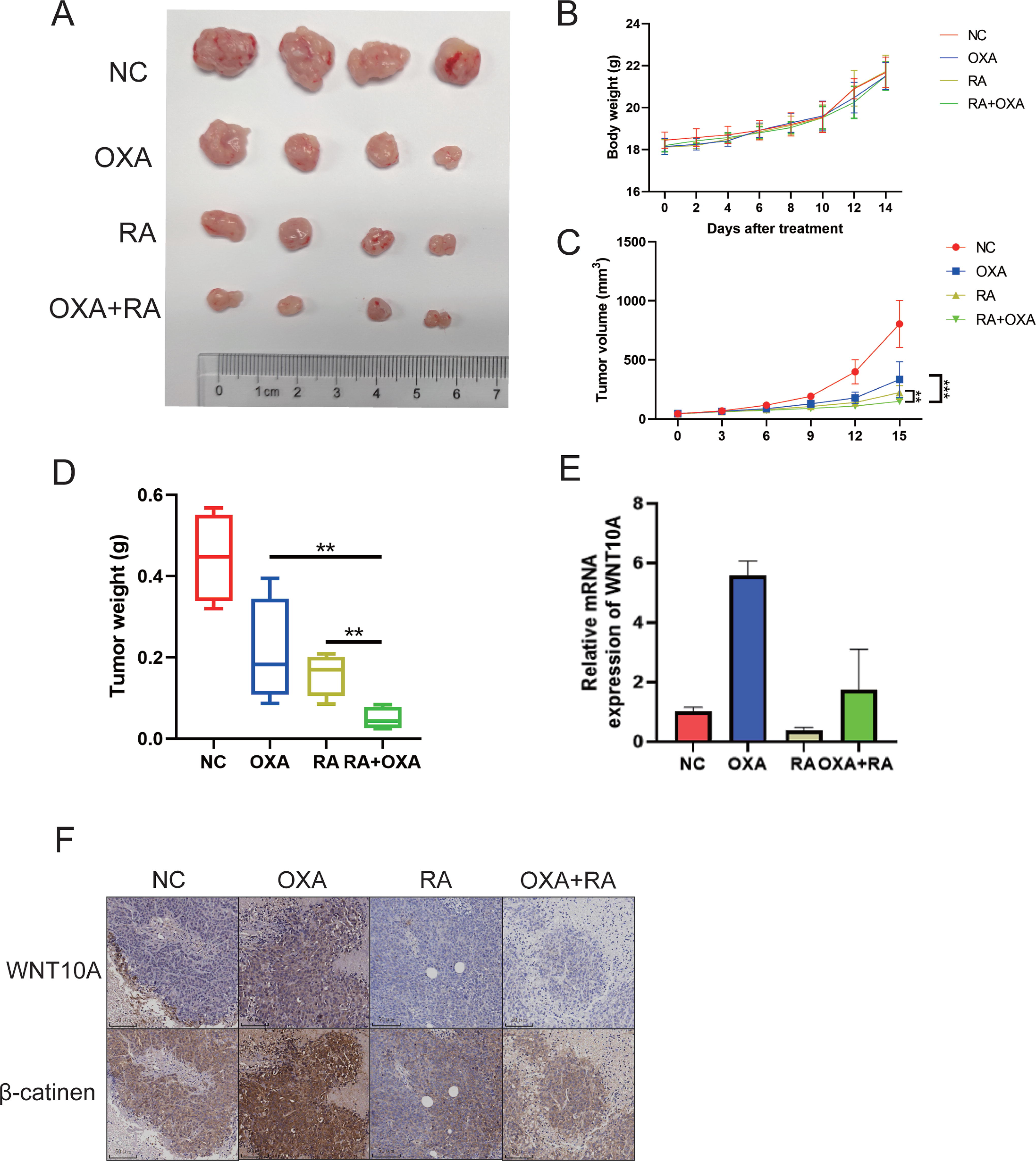
RA inhibits tumor growth in an HCT116 CRC mouse model. Model: After successfully establishing the HCT116 xenograft model in nude mice, DMSO, RA (4 mg/kg), OXA (2 mg/kg), or their combination were administered intraperitoneally.
We also extracted RNA from subcutaneous tumor tissues of mice and performed qPCR analyses. The results demonstrated a significant increase in WNT10A expression in the subcutaneous tumor tissues of mice treated with OXA alone, while the expression of WNT10A in the subcutaneous tumors of mice treated with RA alone or with both drugs was similar to that of the control group (Fig. 5E). Similarly, the results of immunohistochemical analyses showed that compared to the control group, the levels of WNT10A and β-catenin proteins were significantly increased in mice treated with OXA alone, while the levels of these proteins were not significantly different from control in mice treated with RA or with both OXA and RA (Fig. 5F). These results further support the notion that RA may enhance the anticancer effect of OXA on CRC by inhibiting the activation of the WNT/β-catenin signaling pathway in vivo.
Discussion
First-line chemotherapy based on OXA improves overall survival in CRC. 32 However, acquired resistance to OXA is the main cause of clinical treatment failure in CRC cases. 28 Therefore, finding a new treatment strategy to overcome OXA resistance is important to improving treatment options for CRC. Here, we attempted to rapidly identify active compounds from a small molecule inhibitor drug library that can enhance the effects of OXA. To achieve this, we established CRC cell lines that are resistant to OXA, which were used to screen a drug library, and we identified 13 natural compounds that kill OXA-resistant CRC cells. Based on literature review and preliminary experimental results, we selected RA for further experiments.
RA, an oleanane class triterpenoid saponin, can be isolated from Anemone raddeana Regel. Recent studies have shown that RA initiates apoptosis and disrupts the cell cycle, leading to the exertion of cytotoxic effects in various tumor cell lines. 31,33 In our study, we observed a significant reduction in cell viability and colony formation ability in three CRC cell lines after treatment with RA. We discovered that RA sensitizes tumor cell lines to OXA treatment and promotes apoptosis in CRC cell lines. We also found that the effect of combined treatment of these cells with RA and OXA is associated with factors related to the cell cycle and apoptosis. Our results thus suggest that the anti-CRC effect of RA may be attributed to its regulation of cellular proliferation and apoptosis through a series of mechanisms. In addition, to eliminate the potential confounding effects of drug combination, we also conducted experiments using the three OXA-resistant CRC cell lines that we developed. The results of these analyses further suggested that RA sensitizes OXA-resistant CRC cell lines to OXA treatment and enhances the anticancer effects of OXA.
OXA is often used as a first-line treatment for advanced CRC. 16,17 To explore the mechanism of sensitization of OXA-resistant CRC cell lines to OXA by RA, we conducted transcriptome sequencing. We found that compared to the control group, there was a significant increase in the expression of WNT10A in HCT116-R. However, in HCT116-R treated with a combination of OXA and RA, the expression of WNT10A was similar to that observed in wild-type HCT116 cells. The link between OXA resistance and WNT signaling is in agreement with the findings of Liu et al., who discovered that aberrant activation of the WNT/β-catenin signaling pathway is an important factor contributing to OXA resistance in CRC. 34 Similarly, Wang et al. also found that the aberrant activation of the WNT/β-catenin signaling pathway leads to OXA resistance in HCT116 cells. 28 In addition, other studies have reported that in CRC cells treated with RA, the WNT/β-catenin signaling pathway is inhibited, leading to growth arrest and induction of apoptosis. 15,31
WNT10A is an upstream protein in a pathway leading to activation of β-catenin, and its overexpression may lead to increased β-catenin activity, which is associated with tumor invasion and poor prognosis. 19,20 Our research findings also demonstrated a positive correlation between WNT10A and β-catenin levels. Both treatment with RA and genetic knockdown of WNT10A expression resulted in increased sensitivity of HCT116 cells to OXA.
In vivo experiments also confirmed the sensitization of CRC to OXA, as a combined treatment with OXA and RA showed better therapeutic efficacy compared to the use of OXA alone. Furthermore, the combination therapy resulted in reduced expression of WNT10A and the levels of WNT10A and β-catenin proteins. Therefore, we conclude that RA can enhance the anticancer efficacy of OXA, and this effect is achieved through its ability to reduce the expression of WNT10A.
The present study also has limitations, including the absence of constructed cell lines overexpressing WNT10A and the lack of clinical samples sensitive or resistant to OXA for relevant experimental validation. These limitations will be the focus of our future research.
In summary, this study has revealed for the first time that RA enhances the anticancer effect of OXA on CRC both in vitro and in vivo by inhibiting the activation of the WNT/β-catenin signaling pathway. Notably, RA has exhibited a good safety profile in animal models. Therefore, RA holds the potential to be developed as a sensitizer for cancer-resistant cells or as a novel lead compound to improve the therapeutic efficacy of OXA-resistant CRC.
Footnotes
Authors’ Contributions
J.G.C., X.J.C., Z.L.Y., X.S.H., J.N.H., and L.L.: conceived and designed the study; J.G.C., Y.H.Z., D.D.L., and X.J.C.: performed in vitro experiments; J.G.C., Y.H.Z., Z.L.Y., and Y.Y.L.: performed animal studies; J.G.C. and Y.H.Z.: were involved in acquisition, analysis, and interpretation of data; J.G.C. and Y.H.Z.: wrote and drafted the article, X.J.C., X.S.H., J.N.H., and L.L.: were involved in critical revision of the article for important intellectual content; and J.N.H. and L.L.: supervised this study and provided funding. All authors approved the final version of the article.
Disclosure Statement
The authors have declared that no conflict of interest exists.
Funding Information
This work was supported by the National Natural Science Foundation of China (81970482, 82172561, and 82070684), the Natural Science Foundation of Guangdong (2019A1515011313), the Guangdong Special Young Talent Plan of Scientific and Technological Innovation (2022B1515020022 and 2020B151502005), the Complete Peeriod Talent Project of the Sixth Affiliated Hospital (R20210217202501976), the program of Guangdong Provincial Clinical Research Center for Digestive Diseases (2020B1111170004), and the National Key Clinical Discipline.
